Sun, May 5, 2024
[Archive]
Volume 6, Issue 4 (7-2008)
IJRM 2008, 6(4): 125-0 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Peyghambari F, Salehnia M, Forouzandeh Moghadam M, Rezazadeh Valujerdi M, Hajizadeh E. The changes in morphology and morphometrical indices of endometrium of ovariectomized mice in response to exogenous ovarian hormones. IJRM 2008; 6 (4) :125-0
URL: http://ijrm.ir/article-1-120-en.html
URL: http://ijrm.ir/article-1-120-en.html
Fatemeh Peyghambari1 
 , Mojdeh Salehnia *
, Mojdeh Salehnia * 
 2, Mehdi Forouzandeh Moghadam3
2, Mehdi Forouzandeh Moghadam3 
 , Mojtaba Rezazadeh Valujerdi1
, Mojtaba Rezazadeh Valujerdi1 
 , Ebrahim Hajizadeh4
, Ebrahim Hajizadeh4 


 , Mojdeh Salehnia *
, Mojdeh Salehnia * 
 2, Mehdi Forouzandeh Moghadam3
2, Mehdi Forouzandeh Moghadam3 
 , Mojtaba Rezazadeh Valujerdi1
, Mojtaba Rezazadeh Valujerdi1 
 , Ebrahim Hajizadeh4
, Ebrahim Hajizadeh4 

1- Department of Anatomy, Tarbiat Modares University, Iran
2- Department of Anatomy, Tarbiat Modares University, Iran , mogdeh@dr.com
3- Department of Biotechnology, Tarbiat Modares University, Iran
4- Department of Biostatistics, Tarbiat Modares University, Iran
2- Department of Anatomy, Tarbiat Modares University, Iran , mogdeh@dr.com
3- Department of Biotechnology, Tarbiat Modares University, Iran
4- Department of Biostatistics, Tarbiat Modares University, Iran
Full-Text [PDF 203 kb]
(449 Downloads)
| Abstract (HTML) (2258 Views)
Full-Text: (341 Views)
Introduction
The endometrium is the site of embryo implantation (1) and its preparation for embryo reception is dependent on the estrogen and progesterone (2, 3). These hormones cause some molecular and cellular events during uterine receptivity (2-4) and the balance between them is important for cyclical changes of endometrium(1).
The actions of estrogen and progesterone are mediated by their receptors (5-8). Previous researches showed the effect of estrogen and progesterone with different doses on the proliferation of endometrial cells, angiogenesis and stromal cells functions (7-10). The estrogen promotes endometrial proliferation and stimulates expression of estrogen and progesterone receptors in all cell types (6). The progesterone is needed to create typical luteal changes and the secretary stage of the endometrium during the decidual reaction (10, 11). Results of Walter et al. indicated that estrogen priming is not required for progesterone to stimulate endometrial endothelial cell proliferation. The progesterone induced endometrial angiogenesis and estrogen is inhibitory to this process (10). Brinsfield and Hawk investigated the ultrastructure of ovariectomized sheep endometrial stromal cells after hormonal treatment. They suggested that an increase in rough endoplasmic reticulum probably reflects increase in protein synthesis by endometrial stromal cells (12). Morphometric studies of ovariectomized rat uterus in the response of exogenous hormones showed that, progesterone acts on luminal epithelium by a different mechanism than estrogen (13).
Murray evaluated the effect of estrogen and progesterone on the ultrastructure of the uterine gland in ovariectomized sheep. It was shown that the uterine epithelial cells of the ovariectomized sheep undergo morphological alteration in protein-synthesizing organelles (14).The effects of estradiol treatment on growth of uterus in ovariectomized ewes indicated that both progesterone and estrogen could affect on uterine size, and the uterine size was increased by tissue hypertrophy rather than tissue hyperplasia (7).
Heryanto and Rogers showed that administration of progesterone with low dose estrogen inhibits endometrial endothelial cell proliferation but subsequently, in combination with high dose estrogen, significantly increases endometrial endothelial cell proliferation (15). As mentioned before in the literature some researches showed the similar results and the others need additional investigations to explain some controversially in this subject. To gain more detail about the effects of ovarian hormones on the endometrium it is better to use ovariectomized animal model.
Thus this study was undertaken to evaluate the effects of exogenous estrogen and progesterone hormones during five days after mouse ovariectomy on different parameters of uterus such as surface epithelial thickness, the diameter and the number of gland in order to introduce the suitable day for selecting these animals as a models for molecular and gene expression analysis.
Materials and methods
Preparation of ovariectomized mice
Female virgin NMRI (National Medical Research Institute) mice (n=60) aged 8-10 weeks were cared for and used according to the Tarbiat Modares University Guide for the Care and Use of Laboratory Animals. The mice were ovariectomized via a dorsal incision under anesthesia using ketamin (100 mg/kg) and xylazine (10 mg/kg). Two weeks later, they were randomly divided into five groups (16).
Experimental design
Group A (Control group): Ovariectomized mice without any injection (n=10).
Group B (Sham group): Ovariectomized mice, which were injected daily 0.1 ml /mouse of solvent oil through intra peritoneal for five days (n=10).
Group C: Estrogen treated ovariectomized mice (n=25), which received daily dosage of 0.5 ml/mouse of 17β-estradiol hormone through intra peritoneal injection for five days.
Group D: Progesterone treated ovariectomized mice (n=25), which received daily dosage of 0.2 ml/mouse of progesterone hormone through intra peritoneal injection for five days.
Group E: Estrogen-progesterone treated mice (n=25) in which the animals received 17 β-estradiol injection on the first day of treatment (0.5 ml/mouse) and four progesterone injections (0.2 ml/mouse) from the second day to the fifth day of treatment (17).
Tissue preparation
The sampling were done (n=5 for every day) one day after injection in every day up to five days (the day of first injection was considered as day 0). All the mice received 17β-estradiol or progesterone hormones (according to the experimental design) once a day for five days. In the other words, on the first day, the mice received only one injection, consequently on the fifth day the mice had received five injections before sampling. They were sacrificed by cervical dislocation and the tissues were obtained from the middle 1/3 part of their uterine horns immediately and processed for further studies.
Morphological and morphometrical studies
The tissues were fixed in Bouin’s solution, embedded in paraffin wax, sectioned at 6 micrometer and stained using routine hematoxylin and eosin technique.
Then the sections were examined under light microscope, 3 slides were chosen randomly from each sample and at least four fields of view were measured from each slide. The following endometrial parameters were measured in each field of view: the surface epithelial cell thickness (µ) from the luminal border to its basement membrane, the gland diameter (µ), and the number of glands per field of study.
The measurement on each slide were made using the 40 times objective of a Ziess microscope with a calibrated eyepiece (18).
Statistical analysis
Data was collected from each group and mean ± SD was calculated. Groups were compared using one-way ANOVA and then LSD (least significant difference) post hoc test.
Results
The morphology of the uterus
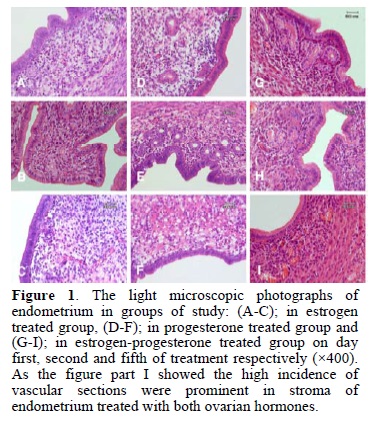
The morphology of endometrium in experimental, control and sham groups are demonstrated in fig. 1 and fig. 2. As shown in estrogen (figure 1C) and progesterone (figure 1F) treated groups, there was stromal edema on day 5 of treatment, whereas in estrogen-progesterone treated groups (figure 1I) the stroma had normal density. Although some sign of degeneration were apparent in the stroma of both estrogen and progesterone treated groups on day 5 of sampling.
In progesterone treated group some eosinophilic extra cellular matrix and vacuolation were observed (figure 1F).
The distribution of blood vessels and the infiltration of lymphocyte were apparent and more numerous in estrogen-progesterone treated group than other groups (estrogen or progesterone treated alone groups) and these phenomenon was prominent on day 5 of sampling (figure 1I).
Surface epithelial cell thickness
The heights of surface epithelial of endometrium in the control group on the first and fifth day of treatment were 12.38±1.42 and 11.25±3.25 and in sham group were 11.63±1.97 and 11.08 ± 1.84 μm respectively (Table I). There was no significant difference in the height of endometrium in two days of evaluation in both groups.
The height of surface epithelial of endometrium in estrogen treated groups from the first day to fifth day of treatment were 30.17±3.67, 39.58±3.37, 38.83±3.17, 35.59±4.01 and 24.42±3.84 μm respectively (Table I). The epithelial cell thickness was significantly different on day two than the other days (p=0.01).
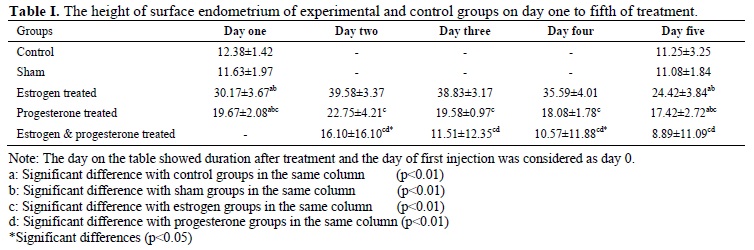

These data in the progesterone treated groups from day 1 to 5 were 19.67±2.08, 22.75±4.21, 19.58±0.97, 18.08±1.78 and 17.42±2.72 µm respectively (Table I); the height of endometrium on the second day was significantly higher than the other days of treatment (p=0.01).
The thickness of surface epithelial of endometrium in estrogen-progesterone treated group from day 2 to 5 were 16.10±16.10, 11.51±12.35, 10.57±11.88 and 8.89±11.09 μm respectively (Table I). The data on the second day was significantly different with other days of treatment (p=0.05). The highest thickness of surface epithelial of endometrium was observed in the estrogen treated group on the second day groups (p=0.01).
The number of glands
The mean number of gland per sections of uterine (at × 10 magnification) in all groups were determined and summarized in Table II.
This data in the control group on the first and fifth day of sampling was 23.96±6.44 and 22.71±5.88 and in sham groups was 26.67±8.55 and 25.42±7.37 respectively. There were no significant differences between two days of evaluation in both groups.
The mean numbers of glands in estrogen treated group from first day to fifth day of treatment were 23.17±10.68, 76.25±17.37, 61.67±12.67, 48.54±16.84 and 36.67±12.03 respectively. The data on second day was significantly different with other days (p = 0.01).
In the progesterone treated group, the mean number of glands from the first day to the fifth day of treatment were 42.08±9.10, 60.00±20.20, 50.63±16.83, 42.50±15.74 and 26.25±8.08 respectively; the data on the second day was significantly different with the other days of treatment (p=0.01).
The mean number of gland in estrogen-progesterone treated group were 31.54±7.81, 43.58±11.30, 49.00±11.08 and 36.42±13.83 from day two to fifth day of injection respectively. The number of glands on the forth day was significantly different from other days (p=0.01).
The gland diameter
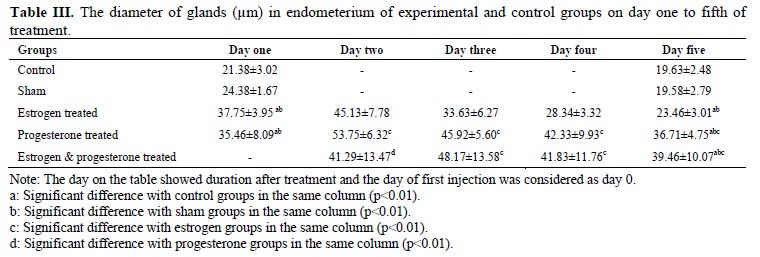
The mean diameter of gland in the control groups on the first and fifth day of treatment were 21.38±3.02 and 19.63±2.48 μm and in sham groups were 24.38±1.67 and 19.58±2.79 μm respectively (Table III). There were significant differences in the mean number of glands between two days of study in both groups (p = 0.01).
The mean diameters of glands in estrogen treated group from first day to fifth day of treatment were 37.75±3.95, 45.13±7.78, 33.63±6.27, 28.34±3.32 and 23.46±3.01 µm respectively (Table III). There were significant differences between the second day with the other days (p=0.01).In the progesterone treated group, the mean diameter of glands from the first day to the fifth day of treatment were 35.46±8.0 9, 53.75±6.32, 45.92±5.60, 42.33±9.93 and 36.71±4.75 μm (Table III) in which the mean diameter of glands on the second day proved to be more significant compared to the other days of treatment (p = 0.01). The mean diameter of glands in estrogen-progesterone treated group from day 2 to 5 were 41.29±13.47, 48.17±13.58, 41.83±11.76 and 39.46±10.07 μm respectively (Table III); the data on the third day was significantly different with other day. The highest mean diameter of glands was observed on the second day of progesterone treated group (0.01).
Discussion
The uterine cellular changes during the cycle and pregnancy are regulated by the circulating levels of ovarian sex steroids progesterone and estrogen (1-4). Our observation showed that the height of surface epithelium of endometrium was increased in the estrogen treated group on second day of treatment in comparison with the other groups. These may be due to increase in the cell number and size on the other hand increase in cell proliferation.
Proliferation of epithelial cells in response to estrogen was demonstrated in mice and rats (19, 20), suggesting that proliferation of epithelial cells in response to estrogen is a common phenomenon in the uterus. Pollard and Finn showed estrogen stimulate rapid turnover of the luminal and glandular epithelium cells, and in the absence of the hormone, the rate of division of the endometrial cells is very low (21).
Johnson et al. in ewe model showed that the major effect of estrogen is on uterine size. He showed estrogen increase tissue hypertrophy (cell size as reflected by RNA; DNA and protein; DNA ratios) rather than tissue hyperplasia (changes in DNA content) (7). Heryanto and Rogers showed in the uterus of adult ovariectomized, under the influence of estrogen, proliferation of endometrial cells commences within 24 h and may precede endometrial tissue growth (15).
Estrogen influence endometrium with activation of cell genome through its receptor in the nucleus of a target cell (5, 6, 11, 13). Murray showed that the uterine epithelial cells of the ovariectomized sheep undergo morphological alternations in protein-synthesizing organelles (14).
Furthermore, we emphasized that the epithelium showed a significance increased in its height on second day of injection of estrogen and after that with several injections of estrogen the height of epithelium was decreased and sign of tissue degeneration was seen. It means that the high concentrations (over dosage) of estrogen had harmful effects on the uterine epithelium and stroma.
In agreement with our results, the study of Heryanto and Rogers, clearly demonstrated that the initial proliferation induced within 24 h of estrogen administration to ovariectomized mice and was no longer evident four days later (15).
In contrast, Martin et al in ovariectomized mouse model showed repeated injections of estrogen prevented premature cell death and produced as much uterine hypertrophy and hyperplasia (16).
Using ovariectomized mouse model in our study showed progesterone injection alone or with estrogen prime mice had not affected on increase of height of surface epithelium. It seems that the cells of the luminal epithelium of the uterus have very different patterns of morphological response to progesterone and to estradiol and this data confirmed the previous reports in this regards (23-25).
The progesterone strongly opposes the proliferative effects of estrogen in the luminal epithelium (25, 26). Uterine luminal and glandular cell proliferation is stimulated by 17ß-oestradiol. Progesterone, both inhibits this proliferation and induces the epithelial cells to differentiate in preparation for implantation (27).Our observations showed in estrogen and progesterone treated groups there were stromal edema on day 5 of treatment, whereas in estrogen-progesterone treated groups the stroma had normal density.
Although some signs of degeneration were apparent in the stroma of both estrogen and progesterone treated groups on day 5 of sampling. Animal studies have shown that treatment of 17ß-estradiol alone to ovariectomized mice stimulated the uterine epithelial cell proliferation and treatment with progesterone alone induced stromal cell proliferation (19). However, treatment of progesterone-primed animals with estrogen mainly maintained the proliferation in stromal cells but does not stimulate proliferation in epithelial cells (19, 28).
In contrast to our results, it was shown that administration of progesterone, either alone or following estrogen, does not cause tissue edema. The stromal cell density in the mice treated with progesterone either with or without estrogen priming, was not significantly different from that observed in the mice treated with the vehicle alone (29). These differences may be due to difference in regime of hormone administration however it needs additional study.
On the second day of treatment, the gland diameter and the number of glands sections were observed to be the highest in the progesterone treated group. The effects of progesterone on the proliferation of the glandular cells were shown before (1). Also the secretion of glands was increased during secretory phase of uterine in response to high level of progesterone which produced by growing follicles. It seems that on the second day of progesterone injections in ovariectomized mice the uterus had the morphology and structure similar to secretory phase in estrous cycle.
As we mentioned in our observation, the distribution of blood vessels were apparent and more numerous in estrogen-progesterone treated group than other groups (estrogen or progesterone treated alone groups) and these phenomenon was prominent on day 5 of sampling. Other function of progesterone on the endometrium is increase of angiogenesis (10, 29).
In this regards, Walter et al. reported that progesterone stimulates endometrial endothelial cell proliferation and this proliferation is mediated in part by vascular endothelial growth factor (10). In contrast to our observation, Heryanto and Rogers by using ovariectomized mice model showed the highest amount of endothelial cell proliferation was observed in the animals that were treated with progesterone alone. Estrogen priming of the endometrium inhibited this progesterone-induced proliferation. It seems that it needs additional study to understand more about mechanism of steroid hormones on uterine angiogenesis.
In conclusion, estrogen and progesterone affect differently on the endometrium of ovariectomized mouse model. Progesterone affects on the gland, whereas, estrogen causes an increase in height of surface epithelial of endometrium.
Acknowledgements
The authors are grateful to Mr. PourByranvand and Mrs Ebrahimi for technical assistance and photographic artwork.
This work was supported by a grant from Tarbiat Modares University.
The endometrium is the site of embryo implantation (1) and its preparation for embryo reception is dependent on the estrogen and progesterone (2, 3). These hormones cause some molecular and cellular events during uterine receptivity (2-4) and the balance between them is important for cyclical changes of endometrium(1).
The actions of estrogen and progesterone are mediated by their receptors (5-8). Previous researches showed the effect of estrogen and progesterone with different doses on the proliferation of endometrial cells, angiogenesis and stromal cells functions (7-10). The estrogen promotes endometrial proliferation and stimulates expression of estrogen and progesterone receptors in all cell types (6). The progesterone is needed to create typical luteal changes and the secretary stage of the endometrium during the decidual reaction (10, 11). Results of Walter et al. indicated that estrogen priming is not required for progesterone to stimulate endometrial endothelial cell proliferation. The progesterone induced endometrial angiogenesis and estrogen is inhibitory to this process (10). Brinsfield and Hawk investigated the ultrastructure of ovariectomized sheep endometrial stromal cells after hormonal treatment. They suggested that an increase in rough endoplasmic reticulum probably reflects increase in protein synthesis by endometrial stromal cells (12). Morphometric studies of ovariectomized rat uterus in the response of exogenous hormones showed that, progesterone acts on luminal epithelium by a different mechanism than estrogen (13).
Murray evaluated the effect of estrogen and progesterone on the ultrastructure of the uterine gland in ovariectomized sheep. It was shown that the uterine epithelial cells of the ovariectomized sheep undergo morphological alteration in protein-synthesizing organelles (14).The effects of estradiol treatment on growth of uterus in ovariectomized ewes indicated that both progesterone and estrogen could affect on uterine size, and the uterine size was increased by tissue hypertrophy rather than tissue hyperplasia (7).
Heryanto and Rogers showed that administration of progesterone with low dose estrogen inhibits endometrial endothelial cell proliferation but subsequently, in combination with high dose estrogen, significantly increases endometrial endothelial cell proliferation (15). As mentioned before in the literature some researches showed the similar results and the others need additional investigations to explain some controversially in this subject. To gain more detail about the effects of ovarian hormones on the endometrium it is better to use ovariectomized animal model.
Thus this study was undertaken to evaluate the effects of exogenous estrogen and progesterone hormones during five days after mouse ovariectomy on different parameters of uterus such as surface epithelial thickness, the diameter and the number of gland in order to introduce the suitable day for selecting these animals as a models for molecular and gene expression analysis.
Materials and methods
Preparation of ovariectomized mice
Female virgin NMRI (National Medical Research Institute) mice (n=60) aged 8-10 weeks were cared for and used according to the Tarbiat Modares University Guide for the Care and Use of Laboratory Animals. The mice were ovariectomized via a dorsal incision under anesthesia using ketamin (100 mg/kg) and xylazine (10 mg/kg). Two weeks later, they were randomly divided into five groups (16).
Experimental design
Group A (Control group): Ovariectomized mice without any injection (n=10).
Group B (Sham group): Ovariectomized mice, which were injected daily 0.1 ml /mouse of solvent oil through intra peritoneal for five days (n=10).
Group C: Estrogen treated ovariectomized mice (n=25), which received daily dosage of 0.5 ml/mouse of 17β-estradiol hormone through intra peritoneal injection for five days.
Group D: Progesterone treated ovariectomized mice (n=25), which received daily dosage of 0.2 ml/mouse of progesterone hormone through intra peritoneal injection for five days.
Group E: Estrogen-progesterone treated mice (n=25) in which the animals received 17 β-estradiol injection on the first day of treatment (0.5 ml/mouse) and four progesterone injections (0.2 ml/mouse) from the second day to the fifth day of treatment (17).
Tissue preparation
The sampling were done (n=5 for every day) one day after injection in every day up to five days (the day of first injection was considered as day 0). All the mice received 17β-estradiol or progesterone hormones (according to the experimental design) once a day for five days. In the other words, on the first day, the mice received only one injection, consequently on the fifth day the mice had received five injections before sampling. They were sacrificed by cervical dislocation and the tissues were obtained from the middle 1/3 part of their uterine horns immediately and processed for further studies.
Morphological and morphometrical studies
The tissues were fixed in Bouin’s solution, embedded in paraffin wax, sectioned at 6 micrometer and stained using routine hematoxylin and eosin technique.
Then the sections were examined under light microscope, 3 slides were chosen randomly from each sample and at least four fields of view were measured from each slide. The following endometrial parameters were measured in each field of view: the surface epithelial cell thickness (µ) from the luminal border to its basement membrane, the gland diameter (µ), and the number of glands per field of study.
The measurement on each slide were made using the 40 times objective of a Ziess microscope with a calibrated eyepiece (18).
Statistical analysis
Data was collected from each group and mean ± SD was calculated. Groups were compared using one-way ANOVA and then LSD (least significant difference) post hoc test.
Results
The morphology of the uterus
The morphology of endometrium in experimental, control and sham groups are demonstrated in fig. 1 and fig. 2. As shown in estrogen (figure 1C) and progesterone (figure 1F) treated groups, there was stromal edema on day 5 of treatment, whereas in estrogen-progesterone treated groups (figure 1I) the stroma had normal density. Although some sign of degeneration were apparent in the stroma of both estrogen and progesterone treated groups on day 5 of sampling.
In progesterone treated group some eosinophilic extra cellular matrix and vacuolation were observed (figure 1F).
The distribution of blood vessels and the infiltration of lymphocyte were apparent and more numerous in estrogen-progesterone treated group than other groups (estrogen or progesterone treated alone groups) and these phenomenon was prominent on day 5 of sampling (figure 1I).
Surface epithelial cell thickness
The heights of surface epithelial of endometrium in the control group on the first and fifth day of treatment were 12.38±1.42 and 11.25±3.25 and in sham group were 11.63±1.97 and 11.08 ± 1.84 μm respectively (Table I). There was no significant difference in the height of endometrium in two days of evaluation in both groups.
The height of surface epithelial of endometrium in estrogen treated groups from the first day to fifth day of treatment were 30.17±3.67, 39.58±3.37, 38.83±3.17, 35.59±4.01 and 24.42±3.84 μm respectively (Table I). The epithelial cell thickness was significantly different on day two than the other days (p=0.01).



These data in the progesterone treated groups from day 1 to 5 were 19.67±2.08, 22.75±4.21, 19.58±0.97, 18.08±1.78 and 17.42±2.72 µm respectively (Table I); the height of endometrium on the second day was significantly higher than the other days of treatment (p=0.01).
The thickness of surface epithelial of endometrium in estrogen-progesterone treated group from day 2 to 5 were 16.10±16.10, 11.51±12.35, 10.57±11.88 and 8.89±11.09 μm respectively (Table I). The data on the second day was significantly different with other days of treatment (p=0.05). The highest thickness of surface epithelial of endometrium was observed in the estrogen treated group on the second day groups (p=0.01).
The number of glands
The mean number of gland per sections of uterine (at × 10 magnification) in all groups were determined and summarized in Table II.
This data in the control group on the first and fifth day of sampling was 23.96±6.44 and 22.71±5.88 and in sham groups was 26.67±8.55 and 25.42±7.37 respectively. There were no significant differences between two days of evaluation in both groups.
The mean numbers of glands in estrogen treated group from first day to fifth day of treatment were 23.17±10.68, 76.25±17.37, 61.67±12.67, 48.54±16.84 and 36.67±12.03 respectively. The data on second day was significantly different with other days (p = 0.01).
In the progesterone treated group, the mean number of glands from the first day to the fifth day of treatment were 42.08±9.10, 60.00±20.20, 50.63±16.83, 42.50±15.74 and 26.25±8.08 respectively; the data on the second day was significantly different with the other days of treatment (p=0.01).
The mean number of gland in estrogen-progesterone treated group were 31.54±7.81, 43.58±11.30, 49.00±11.08 and 36.42±13.83 from day two to fifth day of injection respectively. The number of glands on the forth day was significantly different from other days (p=0.01).
The gland diameter
The mean diameter of gland in the control groups on the first and fifth day of treatment were 21.38±3.02 and 19.63±2.48 μm and in sham groups were 24.38±1.67 and 19.58±2.79 μm respectively (Table III). There were significant differences in the mean number of glands between two days of study in both groups (p = 0.01).
The mean diameters of glands in estrogen treated group from first day to fifth day of treatment were 37.75±3.95, 45.13±7.78, 33.63±6.27, 28.34±3.32 and 23.46±3.01 µm respectively (Table III). There were significant differences between the second day with the other days (p=0.01).In the progesterone treated group, the mean diameter of glands from the first day to the fifth day of treatment were 35.46±8.0 9, 53.75±6.32, 45.92±5.60, 42.33±9.93 and 36.71±4.75 μm (Table III) in which the mean diameter of glands on the second day proved to be more significant compared to the other days of treatment (p = 0.01). The mean diameter of glands in estrogen-progesterone treated group from day 2 to 5 were 41.29±13.47, 48.17±13.58, 41.83±11.76 and 39.46±10.07 μm respectively (Table III); the data on the third day was significantly different with other day. The highest mean diameter of glands was observed on the second day of progesterone treated group (0.01).


Discussion
The uterine cellular changes during the cycle and pregnancy are regulated by the circulating levels of ovarian sex steroids progesterone and estrogen (1-4). Our observation showed that the height of surface epithelium of endometrium was increased in the estrogen treated group on second day of treatment in comparison with the other groups. These may be due to increase in the cell number and size on the other hand increase in cell proliferation.
Proliferation of epithelial cells in response to estrogen was demonstrated in mice and rats (19, 20), suggesting that proliferation of epithelial cells in response to estrogen is a common phenomenon in the uterus. Pollard and Finn showed estrogen stimulate rapid turnover of the luminal and glandular epithelium cells, and in the absence of the hormone, the rate of division of the endometrial cells is very low (21).
Johnson et al. in ewe model showed that the major effect of estrogen is on uterine size. He showed estrogen increase tissue hypertrophy (cell size as reflected by RNA; DNA and protein; DNA ratios) rather than tissue hyperplasia (changes in DNA content) (7). Heryanto and Rogers showed in the uterus of adult ovariectomized, under the influence of estrogen, proliferation of endometrial cells commences within 24 h and may precede endometrial tissue growth (15).
Estrogen influence endometrium with activation of cell genome through its receptor in the nucleus of a target cell (5, 6, 11, 13). Murray showed that the uterine epithelial cells of the ovariectomized sheep undergo morphological alternations in protein-synthesizing organelles (14).
Furthermore, we emphasized that the epithelium showed a significance increased in its height on second day of injection of estrogen and after that with several injections of estrogen the height of epithelium was decreased and sign of tissue degeneration was seen. It means that the high concentrations (over dosage) of estrogen had harmful effects on the uterine epithelium and stroma.
In agreement with our results, the study of Heryanto and Rogers, clearly demonstrated that the initial proliferation induced within 24 h of estrogen administration to ovariectomized mice and was no longer evident four days later (15).
In contrast, Martin et al in ovariectomized mouse model showed repeated injections of estrogen prevented premature cell death and produced as much uterine hypertrophy and hyperplasia (16).
Using ovariectomized mouse model in our study showed progesterone injection alone or with estrogen prime mice had not affected on increase of height of surface epithelium. It seems that the cells of the luminal epithelium of the uterus have very different patterns of morphological response to progesterone and to estradiol and this data confirmed the previous reports in this regards (23-25).
The progesterone strongly opposes the proliferative effects of estrogen in the luminal epithelium (25, 26). Uterine luminal and glandular cell proliferation is stimulated by 17ß-oestradiol. Progesterone, both inhibits this proliferation and induces the epithelial cells to differentiate in preparation for implantation (27).Our observations showed in estrogen and progesterone treated groups there were stromal edema on day 5 of treatment, whereas in estrogen-progesterone treated groups the stroma had normal density.
Although some signs of degeneration were apparent in the stroma of both estrogen and progesterone treated groups on day 5 of sampling. Animal studies have shown that treatment of 17ß-estradiol alone to ovariectomized mice stimulated the uterine epithelial cell proliferation and treatment with progesterone alone induced stromal cell proliferation (19). However, treatment of progesterone-primed animals with estrogen mainly maintained the proliferation in stromal cells but does not stimulate proliferation in epithelial cells (19, 28).
In contrast to our results, it was shown that administration of progesterone, either alone or following estrogen, does not cause tissue edema. The stromal cell density in the mice treated with progesterone either with or without estrogen priming, was not significantly different from that observed in the mice treated with the vehicle alone (29). These differences may be due to difference in regime of hormone administration however it needs additional study.
On the second day of treatment, the gland diameter and the number of glands sections were observed to be the highest in the progesterone treated group. The effects of progesterone on the proliferation of the glandular cells were shown before (1). Also the secretion of glands was increased during secretory phase of uterine in response to high level of progesterone which produced by growing follicles. It seems that on the second day of progesterone injections in ovariectomized mice the uterus had the morphology and structure similar to secretory phase in estrous cycle.
As we mentioned in our observation, the distribution of blood vessels were apparent and more numerous in estrogen-progesterone treated group than other groups (estrogen or progesterone treated alone groups) and these phenomenon was prominent on day 5 of sampling. Other function of progesterone on the endometrium is increase of angiogenesis (10, 29).
In this regards, Walter et al. reported that progesterone stimulates endometrial endothelial cell proliferation and this proliferation is mediated in part by vascular endothelial growth factor (10). In contrast to our observation, Heryanto and Rogers by using ovariectomized mice model showed the highest amount of endothelial cell proliferation was observed in the animals that were treated with progesterone alone. Estrogen priming of the endometrium inhibited this progesterone-induced proliferation. It seems that it needs additional study to understand more about mechanism of steroid hormones on uterine angiogenesis.
In conclusion, estrogen and progesterone affect differently on the endometrium of ovariectomized mouse model. Progesterone affects on the gland, whereas, estrogen causes an increase in height of surface epithelial of endometrium.
Acknowledgements
The authors are grateful to Mr. PourByranvand and Mrs Ebrahimi for technical assistance and photographic artwork.
This work was supported by a grant from Tarbiat Modares University.
Type of Study: Original Article |
References
1. Lessy BA. Two pathways of progesterone action in the human endometrium: implications for implantation and contraception. Steroids 2003; 68: 809-815. [DOI:10.1016/j.steroids.2003.09.004]
2. Song H, Han K, Lim H. Progesterone supplementation extends uterine receptivity for blastocyst implantation in mice. Reproduction 2007; 133: 487-493. [DOI:10.1530/REP-06-0330]
3. Ma WG, Song H, Das SK, Paria BC, Dey SK. Estrogen is a critical determinant that specifies the duration of the window of uterine receptivity for implantation. Proc Natl Acad Sci USA 2003; 100: 2963-2968. [DOI:10.1073/pnas.0530162100]
4. Dey SK. Implantation. In: Adashi EY, Rock JA,Rosenwaks Z, eds. Reproductive endocrinology, surgery and technology. 1996 New York: Lippincott-Raven; 421-434.
5. Das SK, Chakraborty I, Paria BC, Wang XN, Plowman G, Dey SK. Amphiregulin is an implantation-specific and progesterone-regulated gene in the mouse uterus. Mol Endocrinol 1995; 9: 691-705. [DOI:10.1210/mend.9.6.8592515]
6. Horne FM, Blithe DL. Progesterone receptor modulators and the endometrium: changes and consequences. Hum Reprod Update 2007; 13: 567-580. [DOI:10.1093/humupd/dmm023]
7. Johnson ML, Redmer DA, Reynolds LP. Effects of ovarian steroids on uterine growth, morphology and cell proliferation in ovariectomized, steroid-treated ewes. Biol Reprod 1997; 57:588-596. [DOI:10.1095/biolreprod57.3.588]
8. Cheon YP, Xu X, Bagchi MK, Bagchi IC. Immune-responsive gene 1 is a novel target of progesterone receptor and plays a critical role during implantation in the mouse. Endocrinol 2003; 144:5623-5630. [DOI:10.1210/en.2003-0585]
9. Hewitt SC, Korach KS. Progesterone action and responses in the alphaERKO mouse. Steroids 2000; 65: 551-557. [DOI:10.1016/S0039-128X(00)00113-6]
10. Walter LM, Rogers PA, Girling JE. The role of progesterone in endometrial angiogenesis in pregnant and ovariectomised mice. Reproduction 2005; 129:765-777. [DOI:10.1530/rep.1.00625]
11. Sengupta J, Ghosh D. Role of progesterone on peri-implantation stage endometium-emberyo interaction in the primate. Steroids 2000; 65:753-762. [DOI:10.1016/S0039-128X(00)00191-4]
12. Brinsfield TH, Hawk HW. Ultrastructure of sheep endometrial. stromal cells after ovariectomy and hormone treatment Biol Reprod 1974;10:98-102. [DOI:10.1095/biolreprod10.1.98]
13. Williams T, Rogers AW. Morphometric studies of the luminal epithelium in the rat uterus to exogenous hormones. Anat 1980; 130: 867-881.
14. Murray MK. The effect of estrogen and progesterone on structural changes in the uterine glandular epithelium of the ovariectomized sheep. Biol Reprod 1992; 47:408-417. [DOI:10.1095/biolreprod47.3.408]
15. Heryanto B, Rogers PA. Regulation of endometrial cell proliferation by oestrogen and progesterone in the ovariectomized mouse. Reproduction 2002; 123:107-113. [DOI:10.1530/rep.0.1230107]
16. Martin L, Pollard JW, Fagg B. Oestriol, oestradiol- 17beta and the proliferation and death of uterine cells. Endocrinol 1976; 69:103-115. [DOI:10.1677/joe.0.0690103]
17. Miller BG. Delayed interactions between progesterone and low dose of 17beta-estradiol in the mouse uterus. Endocrinol 1979; 104:26-33. [DOI:10.1210/endo-104-1-26]
18. Stein B, Kramer B. The effect of exogenous gonadotropic hormones on the endometrium of the rat. Anat 1989; 164:123-130.
19. Huet-Hudson YM, Andrews GK, Dey SK. Cell type-specific localization of c-myc protein in the mouse uterus: modulation by steroid hormones and analysis of the periimplantation period. Endocrinol 1989; 125:1683-1690. [DOI:10.1210/endo-125-3-1683]
20. Finn CA, Porter DG . The uterus. In: Reproductive biology handbooks. Acton, MA: Publishing Sciences Group 1975; 1:42-56.
21. Pollard RM,Finn CA. The effect of ovariectomy of puberty on cell proliferation and differentiation in the endometrium of the aged mouse. Biol Reprod 1974; 10:74-77. [DOI:10.1095/biolreprod10.1.74]
22. Zhang Q, Paria BC. Importance of uterine cell death, renewal, and their hormonal regulation in hamsters that show progesterone-dependent implantation. Endocrinol 2006; 147:2215-2227. [DOI:10.1210/en.2005-1555]
23. Chu PY, Lee CS, Moore PF, Wright PJ. Oestrogen and progesterone treated ovariectomized bitches: a model for the study of uterine function. Reprod Fertil 2001; 57:45-54.
24. Evans GS, Gibson DF, Roberts SA, Hind TM, Potten CS. Proliferative changes in the genital tissue of female mice during the oestrous cycle. Cell Tissue Kinet 1990; 23:619-35. [DOI:10.1111/j.1365-2184.1990.tb01350.x]
25. Martin L, Finn CA, Trinder G. Hypertrophy and hyperplasia in the mouse uterus after oestrogen treatment: an autoradiographic study. J Endocrinol 1973; 56:133-144. [DOI:10.1677/joe.0.0560133]
26. Finn CA, Martin L. The role of the oestrogen secreted before oestrus in the preparation of the uterus for implantation in the mouse. J Endocrinol 1970; 47:431-438. [DOI:10.1677/joe.0.0470431]
27. Tong W, Pollard JW. Progesterone inhibits estrogen-induced cyclin D1 and cdk4 nuclear translocation, cyclin E- and cyclin A-cdk2 kinase activation, and cell proliferation in uterine epithelial cells in mice. Mol Cell Biol 1999; 19:2251-2264. [DOI:10.1128/MCB.19.3.2251]
28. Terada N, Yamamoto R, Takada T, Miyake T, Terakawa N, Wakimoto H, Taniguchi H, et al. Inhibitory effects of progesterone on cell death of mouse uterine epithelium . J Steroid Biochem 1989; 33:1091-1096. [DOI:10.1016/0022-4731(89)90414-7]
29. Girling JE, Lederman FL, Walter LM, Rogers PA. Progesterone, but not estrogen, stimulates vessel maturation in the mouse endometrium. Endocrinol 2007; 148:5433-5441. [DOI:10.1210/en.2007-0856]
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |


