Thu, Apr 25, 2024
[Archive]
Volume 18, Issue 8 (August 2020)
IJRM 2020, 18(8): 667-682 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Mehdikhani H, Shariati M, Forouzanfar M, Hosseini S E. Protective effects of Ceratonia siliqua extract on protamine gene expression, testicular function and testicular histology in doxorubicin-treated adult rats. IJRM 2020; 18 (8) :667-682
URL: http://ijrm.ir/article-1-1650-en.html
URL: http://ijrm.ir/article-1-1650-en.html
1- Department of Biology, Shiraz Branch, Islamic Azad University, Shiraz, Iran
2- Department of Biology, Kazerun Branch, Islamic Azad University, Kazerun, Iran. , mehrdadshariati@hotmail.com
3- Department of Biology, Marvdasht Branch, Islamic Azad University, Marvdasht, Iran.
4- Department of Biology, Shiraz Branch, Islamic Azad University, Shiraz, Iran.
2- Department of Biology, Kazerun Branch, Islamic Azad University, Kazerun, Iran. , mehrdadshariati@hotmail.com
3- Department of Biology, Marvdasht Branch, Islamic Azad University, Marvdasht, Iran.
4- Department of Biology, Shiraz Branch, Islamic Azad University, Shiraz, Iran.
Full-Text [PDF 1723 kb]
(802 Downloads)
| Abstract (HTML) (1953 Views)
1. Introduction
Sperm production and testosterone secretion are the two basic functions of the testes (1). Spermatogenesis is a complex process in mammals involving the division and differentiation of spermatogonial stem cells into adult sperm (2). Spermatogenesis occurs in the testis under the control of the secreted testosterone hormone, and the testicular secretory activity is controlled by the hypothalamus-pituitary-testis axis (3). The male reproductive system is responsible for the production, maturation, and transmission of sperm, and any structural and molecular abnormalities in this system may result in infertility (4).
Genomic DNA is heavily compressed in adult sperm, and this DNA compression and packaging is accomplished by the nuclear matrix and protamine (5). Protamines are the main and the smallest protein in the sperm nucleus, and their structures are constant in different species (6). These proteins play key roles in sperm chromatin, and their deficiency has negative effects on male morphology and fertility (7). The most significant role that protamine plays is condensing chromatin in the sperm head (8), which is critical in the proper packaging of paternal DNA (9). Protamine genes are grouped or clustered together and are specifically involved in the process of spermatogenesis, that is, the production of mature spermatozoa (10).
Doxorubicin is an anthracycline drug that is mostly prescribed for solid tumors and has deleterious effects on fertility (11). Doxorubicin can attenuate testicular fat and testicular fatty acids by apoptosis in rat, thereby reducing spermatogenesis and primary spermatogony and spermatid counts (12). It damages spermatogonia in immature mice, and causes irreversible damage to spermatogenesis in adults (13).
The Ceratonia siliqua (C. siliqua) tree belongs to the Fabaceae family (14). It is native to the Mediterranean region, and is found in southern Syria, India and many parts of the Eastern Mediterranean area including Iran (15). C. siliqua seed is a great source of bioactive compounds such as fiber, polyphenols, cyclitols, and low levels of fresh fat (16), and its fruit contains flavonoids, like quercetin, luteolin, and kaempferol (17). Beside, gallic acid has been isolated from the leaves and pods of the C. siliqua (18), and lupeol, genistein, and liquiritigenin have also been found in the C. siliqua extract (19). Also, its seed stem cells contain fatty acids including myristic acid, palmitic acid, oleic acid, linoleic acid, and arachidonic acid (20). Its pulp contains sugars, fats, proteins, amino acids, gallic acids, and phenols (21). Clinical trials related to C. siliqua have used its combination in the treatment of elevated cholesterol level (22). A limited number of studies address its importance in the treatment of diseases such as cancer and diabetes (23, 24). The compounds present in C. siliqua pods have potential protective effects in patients with hepatotoxicity due to inflammation and oxidative stress against dextran sulfate sodium (25). In addition, its leaf and pod extracts are able to induce apoptosis in hepatocellular carcinoma cell line (T1) after 24-hr treatment with maximal dose through direct activation of caspase3 pathway (26). Moreover, C. siliqua extract is effective against DNA damage in L1210 murine leukemia cell nucleus by its anti-toxic capacity (27). Ata and colleagues found that C. siliqua extract can have beneficial effects on sperm concentration in rabbits (28). In a study by Mahdiani and coworkers, it was reported that the sperm concentration and motility were increased in the C. siliqua recipient groups and decreased oxidative stress and inflammatory factors (29).
Due to the antioxidant properties of C. siliqua, we try to investigate the possible protective effects of C. siliqua fruit and pod hydroalcoholic extract on protamine gene expression, testicular function, and testicular histology in doxorubicin-treated rats. If it was found to be effective and can be used as a dietary supplement to reduce side effects of chemotherapeutic drugs including doxorubicin on male sex cells and aid in their recovery process.
2. Materials and Methods
2.1. Preparation of hydroalcoholic extract
C. siliqua fruits were grown in and imported from Turkey. The dried fruit of C. siliqua was milled into powder form, 100 gr of the powder was soaked in 500 ml 70% ethanol for 72 hr in a percolator. The mixture was filtered and the solution was placed in a rotary apparatus to evaporate the solvent. The honey-like material left was placed in a desiccator attached to a vacuum pump to dry completely. Then, the desired doses of the extract were prepared by dissolving the appropriate amount of dried extract (150, 300, and 600 mg) in 1 ml of normal saline to be administered according to the animal weight (30).
2.2. Animal treatment
This experimental study was performed in April, 2018 at the Namazi Hospital of Shiraz. In this experimental study, 56 adult male Wistar rats (2.5 to 3 months old) weighing 210 ± 10 gr were used. They were randomly housed in standard cages under identical conditions at 20-22°C in 12-hr light/dark cycle. They were provided with sufficient water and dried food, and all ethical considerations regarding laboratory animals were met.
Animals were divided into seven groups (n = 8/each) as follows: the control group was left untreated; the sham group received 0.3 ml distilled water, intraperitoneally; the negative control group received 3 mg/kg doxorubicin, intraperitoneally, once a week for 28 days; the positive control group received the maximum dose (600 mg/kg) of C. siliqua extract, orally, for 48 days by gavage; the experimental groups 1, 2, and 3 received 150, 300, and 600 mg/kg of extract for 48 days by gavage, respectively + 3 mg/kg doxorubicin once a week for 28 days (31-33).
At the end of the experiment, animals were anesthetized with ketamine and xylazine (Rotexmedica GmbH, Germany), blood samples were taken from the left ventricle of their hearts, and their testes were removed and weighed separately by digital scales.
2.3. Histological studies
Testes were fixed in 10% neutral formalin. Alcohol dehydration was performed at different concentrations (from low to high). The clearing step was performed by placing the tissues in two containers containing xylene. In the replacement phase, tissues were placed in three containers of molten paraffin (65ºC) for 1 hr each. The fixed testes were sectioned at 5-4 μm thickness, and the tissue sections were stained using hematoxylin-eosin (H&E) dye. In order to count the cells from each group, transverse sections of the seminiferous tubules were examined by light microscopy. At least five seminiferous tubules were selected from each slide. The numbers of spermatogonia, spermatocytes, spermatids, Sertoli, and Leydig cells present in lumen were compared between the different groups. All histological studies were performed under supervision of a pathologist (34).
2.4. Testicular volume measurement
Testicular volume was measured by the amount of water displaced when the testicles were immersed in a cylinder of warm water (37ºC) filled to the brim. Testicles were placed in a 2-L graduated cylinder with a known volume of water. The water displaced by testicles was then read to the nearest 5 ml to determine testicular volume (35).
2.5. Testosterone measurement
Blood samples were kept in vitro for 20 min and centrifuged at 5000g for 15 min. Their sera were removed from the clot with a sampler. Testosterone level was measured by enzyme-linked immunosorbent assay (ELISA) method (ELISA kit DR6, Germany).
Protamine gene expression
In general, the following four steps were carried out to evaluate protamine gene expression:
Using the extracted RNA, cDNA synthesis was performed according to the kit instructions (Cat # RR037Q) based on reverse transcriptase enzyme. The synthesized cDNA was stored at -20°C to be used in real time PCR (39).
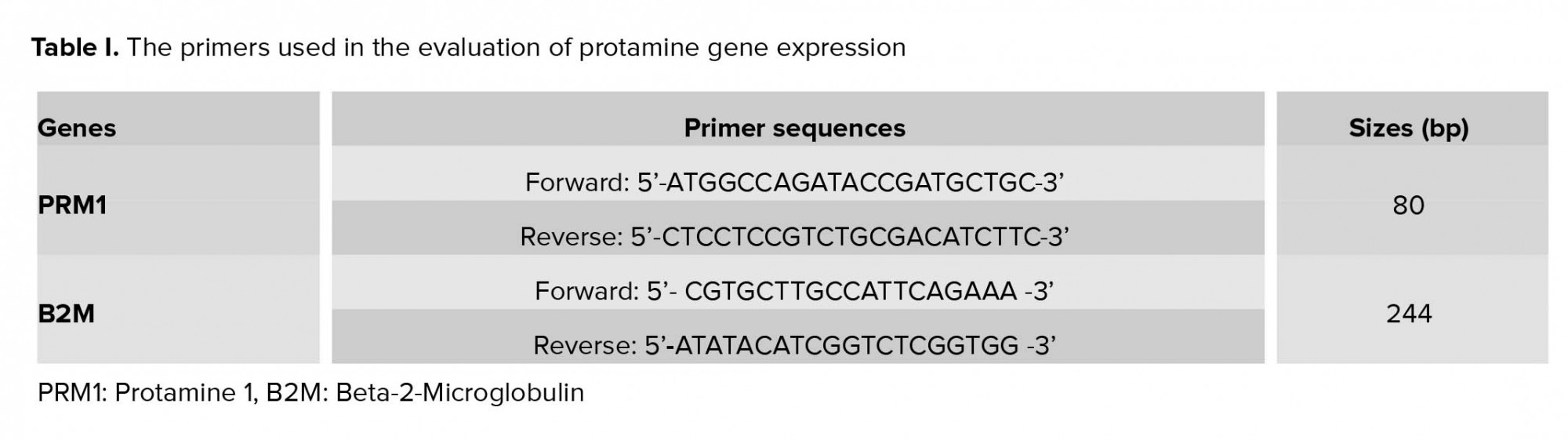
To measure the possible changes in the protamine gene expression, relative quantitative real-time PCR was performed using a kit (Takara Cat. # RR081A). The PCR process for each cDNA sample was carried out using both forward and reverse primers, and Master Mix (SYBR® Premix). The reaction was initiated with temperature and time settings according to the kit protocol. Since the quality and design of primers is the most important factor affecting PCR efficiency, all primers were designed by Allele IDv7.8 software (Table I), and Beta-2-Microglobulin (B2M) gene was used as the internal control.
The basic requirements for gene expression evaluation were: (1) determining the PCR efficiency and standard curve to obtain identical duplication of fragments per cycle for the gene in question compared to the internal control, (2) determination of template concentration (cDNA) for subsequent experiments, and (3) to obtain a threshold number that shows the most appropriate PCR efficiency (40).
Real-time PCR efficiency is calculated using linear gradient obtained from CTs (cycle of threshold) differences.
After completing the thermocycler activity, viewing the graphs showing increased number of desired fragments and the rate of fluorescence emission, we measured the rate of change in gene expression relative to B2M gene and the control (lacking differentiating environments) by calculating ΔΔCt.
The lessΔΔCt = The more gene expression
2.6. Ethical consideration
All interventions in rats were done according to the protocol provided by the ethics committee of the Islamic Azad University Shiraz Branch (IR.IAU.SHIRAZ.REC.1398.028).
2.7. Statistical analysis
Histological, hormonal, and gene expression data were analyzed using the SPSS software version 22 and One-way ANOVA and Tukey test. Real-time PCR data were analyzed using either the ΔΔCT-2 or Livak method. P ≤ 0.05 was considered as statistically significance.
3. Results
The mean body weight, testicular weight , and volume and number of spermatogonia, spermatocytes, spermatids, and Leydig cells, testosterone level, and protamine gene expression (p≤0.001) and Sertoli cells (p ≤ 0.05) showed a significant decrease in the negative control (receiving doxorubicin alone) group compared to the control and sham groups (Tables II, III).
Also, the mean body weight, testicular weight volume, number of spermatogonia, Leydig cells, testosterone concentration(p ≤ 0.001) and spermatocytes (p ≤ 0.05) in the experimental group 1 decreased significantly compared to the control and sham groups (Tables II, III).
The mean number of spermatid cells (p ≤ 0.001) was significantly increased in the experimental group 1 compared to the negative control group, however, it was significantly lower than the control and sham groups (Table II). No significant differences were observed in the mean of the Sertoli cell count and protamine gene expression level in the experimental group 1 compared to the control, sham, and negative control groups (Table III).
Mean testicular weight, testicular volume, and the number of spermatogonia, spermatocyte in the experimental group 2 significantly increased compared to the negative control group, while showing a significant decrease compared to the control and sham groups (Tables II, III). The mean number of spermatid cells was significantly increased in the experimental group 2 compared to the negative control group, however, it was significantly lower than the control and sham groups (Table III). The mean body weight, Leydig cell count, testosterone level, and protamine gene expression (p=0.008) were significantly increased in the experimental group 2 compared to the negative control group (Tables II, III). The mean number of Sertoli cells in the experimental group 2 remained unchanged compared to the negative control, sham, and control groups (Table III).
The mean body weight, testicular weight and volume, number of spermatogonia, spermatocytes, spermatids and Leydig cells, testosterone concentration (p ≤ 0.001), and spermatocytes (p ≤ 0.05) Also protamine gene expression (p = 0.008) in the experimental group 3 (receiving doxorubicin and 600 mg/kg extract) decreased significantly in comparison with the sham and control groups (Tables II and III). The mean number of Sertoli cells in experimental group 3 did not change significantly compared to the negative control, sham, and control groups (Table III).
Finally, the mean body weight, testicular weight and volume, number of spermatogonia, spermatocytes, spermatids and Leydig cells, and testosterone level in the positive control group (receiving 600 mg/kg extract) rose significantly compared to the negative control group (Tables II and III). The mean protamine gene expression (p ≤ 0.001) in the positive control group (receiving 600 mg/kg extract) rose significantly compared to the negative control group. The mean protamine gene expression (p ≤ 0.001) in the positive control group elevated significantly compared to the control and sham groups (Tables II, III). The mean number of Sertoli cells in the positive control group did not change significantly compared to the sham and control groups (Table III).
3.1. Interpretation of protamine gene expression results
The protamine gene amplification curve was interpreted in different samples (experimental group 2 and treatment with doxorubicin and positive control) in real-time PCR reaction. The blue sample had a higher pattern of the protamine gene than the purple sample (negative control) at the start of the reaction. So, it enters the exponential phase sooner. The blue sample in cycle 12 (positive control), the bold blue sample (experimental group 2) in cycle 14, and the purple sample in cycle 16 enter the exponential phase. Since the blue (positive control) sample reached the exponential phase earlier and had a higher pattern, it could be said that the gene expression in the blue sample was higher (Figures 1, 2).
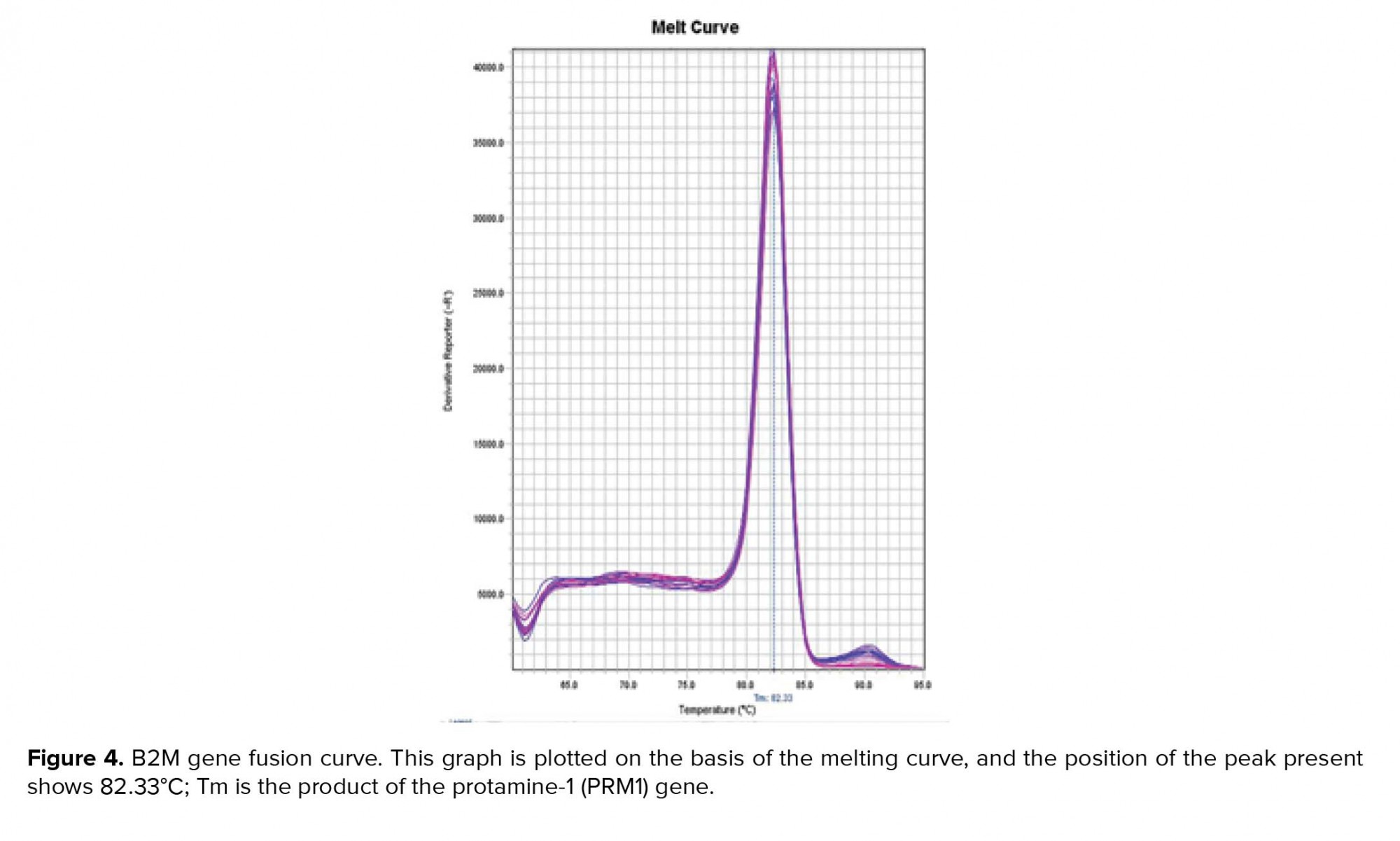
Examine the melt curve of the samples: If you use fluorescent dyes such as SYBR Green in real-time, you can test the melting curve for each sample. Since each gene has its own melting curve, therefore, the curves of a single gene in all samples must match. All curves must also be single-peak. Note that all peaks of a gene must be at the same temperature but the height of the peaks need not be the same (Figures 3, 4).
To draw a standard curve and calculate the amplification reaction efficiency after the end of the experiment, in most software the standard curve for each of the genes is plotted on the X-Y axis. In this logarithmic diagram, the sample pattern is on the X-axis and the cycle threshold (Ct) on the Y-axis. In standard curve diagrams, the slope of the line should be between 3.1-3.6 (Figures 5, 6).
3.2. Histological results
There were no pathological changes in testicular photomicrographs of the control and sham groups, and testicular tubules and germ cells appeared normal (Figures 7A, B). Similar to the control and sham groups, tubules and germ cells were normal in testicular photomicrographs of the positive control group and tubules were filled with spermatogonia, spermatocytes, spermatids, and spermatozoa. Also, no pathological changes were observed in the testicular tissues of this group (Figure 7C). In contrast, wrinkled tubules, germinal epithelial fusion, and severe decrease in germ cell count were observed in testicular photomicrographs of the negative control group (receiving doxorubicin), and the presence of vacuoles inside the tubules indicated the deleterious effects of doxorubicin (Figure 7D).
In testicular photomicrographs of the experimental group 1, the deleterious effects of doxorubicin were still seen and the number of germ cells were decreased (Figure 7E). Conversely, greater reduction in wrinkled tubules were seen in testicular photomicrographs of the experimental group 2 compared to other experimental groups receiving higher and lower doses of the extract, indicating the positive effects of this dose on testicular regeneration (Figure 7F). The decline in the number of germ cells and the presence of vacuoles inside the tubules in testicular photomicrographs of experimental group 3 shows that high dose of C. siliqua hydroalcoholic extract (CSHAE) has no positive effect on testes (Figure 7G).







4. Discussion
According to the results of the present study, the mean body weight, testicular weight, volume, the number of spermatogonia, spermatocytes, spermatids, Sertoli and Leydig cells, serum testosterone level, and protamine gene expression declined significantly in the negative control group (receiving doxorubicin) compared to the control and sham groups, which is consistent with previous studies. It has been shown that doxorubicin induces spermatogenic damage in immature mice, and irreversibly impairs spermatogenesis in adults. These impairments are caused by injury to the blood-testicular barrier mediated by free radical generation and peroxidation (41). An administration of this drug to pre-adolescent rats also results in morphological and functional damage to Sertoli cells. It was demonstrated that after 21 and 28 days of treatment with 7.5 mg/kg doxorubicin, a significant decrease in testicular germ cells was observed, and semen producing segments, seminal tube height, and seminal tube diameter declined, and an increase in intra-tube segments was observed during the study period (42). These effects are not specific to cancer cells and can cause damage to healthy cells as well. Other studies have shown that the administration of doxorubicin to immature mouse leads to irreversible damage to Sertoli cells and induces severe disorder in spermatogenesis (43, 44). Such changes also occur in the testicular tissues of patients treated with doxorubicin (45, 46).
Based on our findings, the body weight, testicular weight, testicular volume, Leydig cell number, spermatogonia, spermatocytes, spermatid, testosterone level, and protamine gene expression were significantly higher in the experimental group 2 (receiving doxorubicin and 300 mg/kg extract) compared to the negative control group (receiving doxorubicin alone), but there was no significant difference in the number of Sertoli cells. The improvement observed in the testicular histology and the spermiogenesis of the experimental group 2 might be as a result of the antioxidant and antiapoptotic properties of C. siliqua plant (31, 47).
Although the mean body weight, all studied testicular parameters, and protamine gene expression in the positive control group (receiving 600 mg/kg extract) rose significantly compared to the negative control group, there was no such differences compared to the control and sham groups. However, protamine gene expression in the positive control group was significantly higher than the control group. It has been reported that the administration of hydroalcoholic C. siliqua seed extract increases sex hormone levels and sperm density in seminal tubes (28). Moreover, Soleimanzadeh and colleagues have found that C. siliqua fruit extract alleviated lead-induced reproductive toxicity in male mice. In this study, it was reported that a co-administration of CSHAE and Lead (Pb) significantly improved sperm parameters, glutathione content, antioxidant enzyme activity, and decreased the downregulation of testicular expression of nuclear factor erythroid 2-related factor 2 (Nrf2) and inducible nitric oxide synthase (iNOS) genes relative to the Pb control group (48).
Similarly, Vafaie and co-workers) showed that the consumption of 800 mg/kg C. siliqua extract for 35 days improves the sperm quality, biochemical parameters, thickening of testicular epithelium, and testosterone levels in busulfan-induced infertile mice (49). Also, malonaldehyde levels in the groups receiving extract were significantly lower than those of busulfan (a chemotherapeutic drug) group, while superoxide dismutase enzyme was significantly increased in these groups (49).
C. siliqua seed contains gamma-Linoleic acid and alpha-Linolenic acid, which can be converted to di-homogamma fatty acid and then to arachidonic acid (the precursor of all prostaglandins such as prostaglandin E2). It seems that arachidonic acid plays a major role in the testicular activities (50). Hossein and colleagues showed that arachidonic, oleic, and linoleic acids significantly enhance the motility of spermatozoa (51). These fatty acids also improve sperm acrosomal reaction at all incubation periods (51).
Furthermore, the increase in testosterone level can be justified by the direct effect of C. siliqua extract on Leydig cells, which play a key role in testicular hormone secretion (52). An important compound found in C. siliqua extract is genistein. Numerous studies have shown that genistein present in C. siliqua seed has protective effects on spermatogenesis (50-54). Verma and co-workers Found that genistein improves spermatogenesis by increasing lactate formation and antioxidant enzymes induced by insulin (53). That is why this compound can adjust the testicular dysfunction induced by type 2 diabetes in mice (53). In addition, Jalili and colleagues have shown that the administration of genistein can enhance spermatozoa quality and prevent morphine-induced side effects on sperm parameters (54). It has also been found that this compound relieves oxidative stress and spermatogenic injury induced by reperfusion and testicular ischemia by reducing the activity of testicular matrix metalloprotease (55). Chi and coworkers Showed that genistein effectively lowered intra-testicular testosterone level and stimulated spermatogenesis in rats treated with busulfan (56). Finally, it has been reported that genistein reduces gamma-ray-induced testicular injuries by antiapoptotic activity and remodeling spermatogenesis (57).
Another compound present in the extract of C. siliqua is gallic acid. Several studies have shown that gallic acid has protective effects on spermatogenesis (58, 59). Mehraban and colleagues showed that gallic acid can amend cyclophosphamide-induced reproductive toxicity in adult NMRI mice and increase the antioxidant capacity of testicular tissue (58). In another study, they also found that gallic acid has protective effects against sperm cell death as well as in vitro fertilization in adult male rats treated with cyclophosphamide (59).
C. siliqua extract also contains quercetin and luteolin, which have protective effects on spermatogenesis. For example, Ma and coworkers found that luteolin can amend testicular injuries and dysfunction through the Nrf2 signaling pathway by increasing regulation of Connexin 43 (Cx43) (60), and Khorsandi and colleagues reported that quercetin has some beneficial effects on spermatogenesis defects induced by titanium dioxide nanoparticles (61). Pretreatment with quercetin may increase testosterone concentration, decrease cell proliferation and oxidative stress in testicular tissue, and improve sperm quality as well as testicular tissue regeneration (61). Baltaci and coworkers (2016) showed that quercetin prevents arsenic-induced testicular injury through its antioxidant and antitumor effects (62). Similarly, Abd Ellah and colleagues found that this compound amends testicular injuries induced by di-(2-ethylhexyl) phthalate (63), and it can modify lambda cyhalothrin-induced reproductive toxicity in adult rats (64). Finally, Jahan and coworkers found that quercetin can possess potent therapeutic effects against bisphenol A-induced testicular toxicity in Sprague Dawley rats (65).
Based on the antiapoptotic and strong antioxidant (66) properties of C. siliqua, we conclude that this plant possesses the potential to protect subjects against doxorubicin toxicity through boosting activities of enzymes such as superoxide dismutase (SOX), Glutathione peroxidase (GPX), and catalase to protect against free radicals with cell toxicity (67). Since the moderate use of this plant has no apparent toxic effects, its beneficial potential in protecting against various toxicities is tempting.
5. Conclusion
Based on our findings, it appears that CSHAE at 300 mg/kg dose has a protective effect on doxorubicin-induced spermatogenesis toxicity, whereas its minimum (150 mg/kg) and maximum (600 mg/kg) doses have no significant influence on the number of germ cells, testicular tissue regeneration, and protamine gene expression. These results also indicate that the protective effects of C. siliqua extract are not dose-dependent, and even a dose as low as 300 mg/kg significantly increases the number of cells and regenerates testicular tissue injuries. Such protective effects are related to the antioxidant and antiapoptotic properties of various compounds present in the C. siliqua herb. Hence, this dose of CSHAE can be used as an effective substance against doxorubicin-induced testicular dysfunctions.
Acknowledgements
The authors hereby express their deep appreciation for the sincere cooperation of the research deputy of the Shiraz Azad University to conduct this research. The results described in this article are part of a Ph.D. thesis by Hengameh Mehdi Khani.
Conflict of Interest
The authors declare that they have no conflict of interests.
Full-Text: (617 Views)
1. Introduction
Sperm production and testosterone secretion are the two basic functions of the testes (1). Spermatogenesis is a complex process in mammals involving the division and differentiation of spermatogonial stem cells into adult sperm (2). Spermatogenesis occurs in the testis under the control of the secreted testosterone hormone, and the testicular secretory activity is controlled by the hypothalamus-pituitary-testis axis (3). The male reproductive system is responsible for the production, maturation, and transmission of sperm, and any structural and molecular abnormalities in this system may result in infertility (4).
Genomic DNA is heavily compressed in adult sperm, and this DNA compression and packaging is accomplished by the nuclear matrix and protamine (5). Protamines are the main and the smallest protein in the sperm nucleus, and their structures are constant in different species (6). These proteins play key roles in sperm chromatin, and their deficiency has negative effects on male morphology and fertility (7). The most significant role that protamine plays is condensing chromatin in the sperm head (8), which is critical in the proper packaging of paternal DNA (9). Protamine genes are grouped or clustered together and are specifically involved in the process of spermatogenesis, that is, the production of mature spermatozoa (10).
Doxorubicin is an anthracycline drug that is mostly prescribed for solid tumors and has deleterious effects on fertility (11). Doxorubicin can attenuate testicular fat and testicular fatty acids by apoptosis in rat, thereby reducing spermatogenesis and primary spermatogony and spermatid counts (12). It damages spermatogonia in immature mice, and causes irreversible damage to spermatogenesis in adults (13).
The Ceratonia siliqua (C. siliqua) tree belongs to the Fabaceae family (14). It is native to the Mediterranean region, and is found in southern Syria, India and many parts of the Eastern Mediterranean area including Iran (15). C. siliqua seed is a great source of bioactive compounds such as fiber, polyphenols, cyclitols, and low levels of fresh fat (16), and its fruit contains flavonoids, like quercetin, luteolin, and kaempferol (17). Beside, gallic acid has been isolated from the leaves and pods of the C. siliqua (18), and lupeol, genistein, and liquiritigenin have also been found in the C. siliqua extract (19). Also, its seed stem cells contain fatty acids including myristic acid, palmitic acid, oleic acid, linoleic acid, and arachidonic acid (20). Its pulp contains sugars, fats, proteins, amino acids, gallic acids, and phenols (21). Clinical trials related to C. siliqua have used its combination in the treatment of elevated cholesterol level (22). A limited number of studies address its importance in the treatment of diseases such as cancer and diabetes (23, 24). The compounds present in C. siliqua pods have potential protective effects in patients with hepatotoxicity due to inflammation and oxidative stress against dextran sulfate sodium (25). In addition, its leaf and pod extracts are able to induce apoptosis in hepatocellular carcinoma cell line (T1) after 24-hr treatment with maximal dose through direct activation of caspase3 pathway (26). Moreover, C. siliqua extract is effective against DNA damage in L1210 murine leukemia cell nucleus by its anti-toxic capacity (27). Ata and colleagues found that C. siliqua extract can have beneficial effects on sperm concentration in rabbits (28). In a study by Mahdiani and coworkers, it was reported that the sperm concentration and motility were increased in the C. siliqua recipient groups and decreased oxidative stress and inflammatory factors (29).
Due to the antioxidant properties of C. siliqua, we try to investigate the possible protective effects of C. siliqua fruit and pod hydroalcoholic extract on protamine gene expression, testicular function, and testicular histology in doxorubicin-treated rats. If it was found to be effective and can be used as a dietary supplement to reduce side effects of chemotherapeutic drugs including doxorubicin on male sex cells and aid in their recovery process.
2. Materials and Methods
2.1. Preparation of hydroalcoholic extract
C. siliqua fruits were grown in and imported from Turkey. The dried fruit of C. siliqua was milled into powder form, 100 gr of the powder was soaked in 500 ml 70% ethanol for 72 hr in a percolator. The mixture was filtered and the solution was placed in a rotary apparatus to evaporate the solvent. The honey-like material left was placed in a desiccator attached to a vacuum pump to dry completely. Then, the desired doses of the extract were prepared by dissolving the appropriate amount of dried extract (150, 300, and 600 mg) in 1 ml of normal saline to be administered according to the animal weight (30).
2.2. Animal treatment
This experimental study was performed in April, 2018 at the Namazi Hospital of Shiraz. In this experimental study, 56 adult male Wistar rats (2.5 to 3 months old) weighing 210 ± 10 gr were used. They were randomly housed in standard cages under identical conditions at 20-22°C in 12-hr light/dark cycle. They were provided with sufficient water and dried food, and all ethical considerations regarding laboratory animals were met.
Animals were divided into seven groups (n = 8/each) as follows: the control group was left untreated; the sham group received 0.3 ml distilled water, intraperitoneally; the negative control group received 3 mg/kg doxorubicin, intraperitoneally, once a week for 28 days; the positive control group received the maximum dose (600 mg/kg) of C. siliqua extract, orally, for 48 days by gavage; the experimental groups 1, 2, and 3 received 150, 300, and 600 mg/kg of extract for 48 days by gavage, respectively + 3 mg/kg doxorubicin once a week for 28 days (31-33).
At the end of the experiment, animals were anesthetized with ketamine and xylazine (Rotexmedica GmbH, Germany), blood samples were taken from the left ventricle of their hearts, and their testes were removed and weighed separately by digital scales.
2.3. Histological studies
Testes were fixed in 10% neutral formalin. Alcohol dehydration was performed at different concentrations (from low to high). The clearing step was performed by placing the tissues in two containers containing xylene. In the replacement phase, tissues were placed in three containers of molten paraffin (65ºC) for 1 hr each. The fixed testes were sectioned at 5-4 μm thickness, and the tissue sections were stained using hematoxylin-eosin (H&E) dye. In order to count the cells from each group, transverse sections of the seminiferous tubules were examined by light microscopy. At least five seminiferous tubules were selected from each slide. The numbers of spermatogonia, spermatocytes, spermatids, Sertoli, and Leydig cells present in lumen were compared between the different groups. All histological studies were performed under supervision of a pathologist (34).
2.4. Testicular volume measurement
Testicular volume was measured by the amount of water displaced when the testicles were immersed in a cylinder of warm water (37ºC) filled to the brim. Testicles were placed in a 2-L graduated cylinder with a known volume of water. The water displaced by testicles was then read to the nearest 5 ml to determine testicular volume (35).
2.5. Testosterone measurement
Blood samples were kept in vitro for 20 min and centrifuged at 5000g for 15 min. Their sera were removed from the clot with a sampler. Testosterone level was measured by enzyme-linked immunosorbent assay (ELISA) method (ELISA kit DR6, Germany).
Protamine gene expression
In general, the following four steps were carried out to evaluate protamine gene expression:
- RNA extraction by Biobasic Manufacturing Kit (EZ-10 Spin Column Total RNA Mini-Preps Kit.
- DNase I treatment by Takara Manufacturing Kit (Cat. # 2215A).
- Complementary DNA (cDNA) synthesis by Takara Company Kit (# RR037Q (Cat. (PrimeScript; RT Reagent Kit)).
- Real-time polymerase chain reaction (PCR) by Takara Machine Bucket (Cat. # RR081A) Syber green 1 (36).
Using the extracted RNA, cDNA synthesis was performed according to the kit instructions (Cat # RR037Q) based on reverse transcriptase enzyme. The synthesized cDNA was stored at -20°C to be used in real time PCR (39).
To measure the possible changes in the protamine gene expression, relative quantitative real-time PCR was performed using a kit (Takara Cat. # RR081A). The PCR process for each cDNA sample was carried out using both forward and reverse primers, and Master Mix (SYBR® Premix). The reaction was initiated with temperature and time settings according to the kit protocol. Since the quality and design of primers is the most important factor affecting PCR efficiency, all primers were designed by Allele IDv7.8 software (Table I), and Beta-2-Microglobulin (B2M) gene was used as the internal control.
The basic requirements for gene expression evaluation were: (1) determining the PCR efficiency and standard curve to obtain identical duplication of fragments per cycle for the gene in question compared to the internal control, (2) determination of template concentration (cDNA) for subsequent experiments, and (3) to obtain a threshold number that shows the most appropriate PCR efficiency (40).
Real-time PCR efficiency is calculated using linear gradient obtained from CTs (cycle of threshold) differences.
After completing the thermocycler activity, viewing the graphs showing increased number of desired fragments and the rate of fluorescence emission, we measured the rate of change in gene expression relative to B2M gene and the control (lacking differentiating environments) by calculating ΔΔCt.
The lessΔΔCt = The more gene expression

2.6. Ethical consideration
All interventions in rats were done according to the protocol provided by the ethics committee of the Islamic Azad University Shiraz Branch (IR.IAU.SHIRAZ.REC.1398.028).
2.7. Statistical analysis
Histological, hormonal, and gene expression data were analyzed using the SPSS software version 22 and One-way ANOVA and Tukey test. Real-time PCR data were analyzed using either the ΔΔCT-2 or Livak method. P ≤ 0.05 was considered as statistically significance.
3. Results
The mean body weight, testicular weight , and volume and number of spermatogonia, spermatocytes, spermatids, and Leydig cells, testosterone level, and protamine gene expression (p≤0.001) and Sertoli cells (p ≤ 0.05) showed a significant decrease in the negative control (receiving doxorubicin alone) group compared to the control and sham groups (Tables II, III).
Also, the mean body weight, testicular weight volume, number of spermatogonia, Leydig cells, testosterone concentration(p ≤ 0.001) and spermatocytes (p ≤ 0.05) in the experimental group 1 decreased significantly compared to the control and sham groups (Tables II, III).
The mean number of spermatid cells (p ≤ 0.001) was significantly increased in the experimental group 1 compared to the negative control group, however, it was significantly lower than the control and sham groups (Table II). No significant differences were observed in the mean of the Sertoli cell count and protamine gene expression level in the experimental group 1 compared to the control, sham, and negative control groups (Table III).
Mean testicular weight, testicular volume, and the number of spermatogonia, spermatocyte in the experimental group 2 significantly increased compared to the negative control group, while showing a significant decrease compared to the control and sham groups (Tables II, III). The mean number of spermatid cells was significantly increased in the experimental group 2 compared to the negative control group, however, it was significantly lower than the control and sham groups (Table III). The mean body weight, Leydig cell count, testosterone level, and protamine gene expression (p=0.008) were significantly increased in the experimental group 2 compared to the negative control group (Tables II, III). The mean number of Sertoli cells in the experimental group 2 remained unchanged compared to the negative control, sham, and control groups (Table III).
The mean body weight, testicular weight and volume, number of spermatogonia, spermatocytes, spermatids and Leydig cells, testosterone concentration (p ≤ 0.001), and spermatocytes (p ≤ 0.05) Also protamine gene expression (p = 0.008) in the experimental group 3 (receiving doxorubicin and 600 mg/kg extract) decreased significantly in comparison with the sham and control groups (Tables II and III). The mean number of Sertoli cells in experimental group 3 did not change significantly compared to the negative control, sham, and control groups (Table III).
Finally, the mean body weight, testicular weight and volume, number of spermatogonia, spermatocytes, spermatids and Leydig cells, and testosterone level in the positive control group (receiving 600 mg/kg extract) rose significantly compared to the negative control group (Tables II and III). The mean protamine gene expression (p ≤ 0.001) in the positive control group (receiving 600 mg/kg extract) rose significantly compared to the negative control group. The mean protamine gene expression (p ≤ 0.001) in the positive control group elevated significantly compared to the control and sham groups (Tables II, III). The mean number of Sertoli cells in the positive control group did not change significantly compared to the sham and control groups (Table III).
3.1. Interpretation of protamine gene expression results
The protamine gene amplification curve was interpreted in different samples (experimental group 2 and treatment with doxorubicin and positive control) in real-time PCR reaction. The blue sample had a higher pattern of the protamine gene than the purple sample (negative control) at the start of the reaction. So, it enters the exponential phase sooner. The blue sample in cycle 12 (positive control), the bold blue sample (experimental group 2) in cycle 14, and the purple sample in cycle 16 enter the exponential phase. Since the blue (positive control) sample reached the exponential phase earlier and had a higher pattern, it could be said that the gene expression in the blue sample was higher (Figures 1, 2).
Examine the melt curve of the samples: If you use fluorescent dyes such as SYBR Green in real-time, you can test the melting curve for each sample. Since each gene has its own melting curve, therefore, the curves of a single gene in all samples must match. All curves must also be single-peak. Note that all peaks of a gene must be at the same temperature but the height of the peaks need not be the same (Figures 3, 4).
To draw a standard curve and calculate the amplification reaction efficiency after the end of the experiment, in most software the standard curve for each of the genes is plotted on the X-Y axis. In this logarithmic diagram, the sample pattern is on the X-axis and the cycle threshold (Ct) on the Y-axis. In standard curve diagrams, the slope of the line should be between 3.1-3.6 (Figures 5, 6).
3.2. Histological results
There were no pathological changes in testicular photomicrographs of the control and sham groups, and testicular tubules and germ cells appeared normal (Figures 7A, B). Similar to the control and sham groups, tubules and germ cells were normal in testicular photomicrographs of the positive control group and tubules were filled with spermatogonia, spermatocytes, spermatids, and spermatozoa. Also, no pathological changes were observed in the testicular tissues of this group (Figure 7C). In contrast, wrinkled tubules, germinal epithelial fusion, and severe decrease in germ cell count were observed in testicular photomicrographs of the negative control group (receiving doxorubicin), and the presence of vacuoles inside the tubules indicated the deleterious effects of doxorubicin (Figure 7D).
In testicular photomicrographs of the experimental group 1, the deleterious effects of doxorubicin were still seen and the number of germ cells were decreased (Figure 7E). Conversely, greater reduction in wrinkled tubules were seen in testicular photomicrographs of the experimental group 2 compared to other experimental groups receiving higher and lower doses of the extract, indicating the positive effects of this dose on testicular regeneration (Figure 7F). The decline in the number of germ cells and the presence of vacuoles inside the tubules in testicular photomicrographs of experimental group 3 shows that high dose of C. siliqua hydroalcoholic extract (CSHAE) has no positive effect on testes (Figure 7G).









4. Discussion
According to the results of the present study, the mean body weight, testicular weight, volume, the number of spermatogonia, spermatocytes, spermatids, Sertoli and Leydig cells, serum testosterone level, and protamine gene expression declined significantly in the negative control group (receiving doxorubicin) compared to the control and sham groups, which is consistent with previous studies. It has been shown that doxorubicin induces spermatogenic damage in immature mice, and irreversibly impairs spermatogenesis in adults. These impairments are caused by injury to the blood-testicular barrier mediated by free radical generation and peroxidation (41). An administration of this drug to pre-adolescent rats also results in morphological and functional damage to Sertoli cells. It was demonstrated that after 21 and 28 days of treatment with 7.5 mg/kg doxorubicin, a significant decrease in testicular germ cells was observed, and semen producing segments, seminal tube height, and seminal tube diameter declined, and an increase in intra-tube segments was observed during the study period (42). These effects are not specific to cancer cells and can cause damage to healthy cells as well. Other studies have shown that the administration of doxorubicin to immature mouse leads to irreversible damage to Sertoli cells and induces severe disorder in spermatogenesis (43, 44). Such changes also occur in the testicular tissues of patients treated with doxorubicin (45, 46).
Based on our findings, the body weight, testicular weight, testicular volume, Leydig cell number, spermatogonia, spermatocytes, spermatid, testosterone level, and protamine gene expression were significantly higher in the experimental group 2 (receiving doxorubicin and 300 mg/kg extract) compared to the negative control group (receiving doxorubicin alone), but there was no significant difference in the number of Sertoli cells. The improvement observed in the testicular histology and the spermiogenesis of the experimental group 2 might be as a result of the antioxidant and antiapoptotic properties of C. siliqua plant (31, 47).
Although the mean body weight, all studied testicular parameters, and protamine gene expression in the positive control group (receiving 600 mg/kg extract) rose significantly compared to the negative control group, there was no such differences compared to the control and sham groups. However, protamine gene expression in the positive control group was significantly higher than the control group. It has been reported that the administration of hydroalcoholic C. siliqua seed extract increases sex hormone levels and sperm density in seminal tubes (28). Moreover, Soleimanzadeh and colleagues have found that C. siliqua fruit extract alleviated lead-induced reproductive toxicity in male mice. In this study, it was reported that a co-administration of CSHAE and Lead (Pb) significantly improved sperm parameters, glutathione content, antioxidant enzyme activity, and decreased the downregulation of testicular expression of nuclear factor erythroid 2-related factor 2 (Nrf2) and inducible nitric oxide synthase (iNOS) genes relative to the Pb control group (48).
Similarly, Vafaie and co-workers) showed that the consumption of 800 mg/kg C. siliqua extract for 35 days improves the sperm quality, biochemical parameters, thickening of testicular epithelium, and testosterone levels in busulfan-induced infertile mice (49). Also, malonaldehyde levels in the groups receiving extract were significantly lower than those of busulfan (a chemotherapeutic drug) group, while superoxide dismutase enzyme was significantly increased in these groups (49).
C. siliqua seed contains gamma-Linoleic acid and alpha-Linolenic acid, which can be converted to di-homogamma fatty acid and then to arachidonic acid (the precursor of all prostaglandins such as prostaglandin E2). It seems that arachidonic acid plays a major role in the testicular activities (50). Hossein and colleagues showed that arachidonic, oleic, and linoleic acids significantly enhance the motility of spermatozoa (51). These fatty acids also improve sperm acrosomal reaction at all incubation periods (51).
Furthermore, the increase in testosterone level can be justified by the direct effect of C. siliqua extract on Leydig cells, which play a key role in testicular hormone secretion (52). An important compound found in C. siliqua extract is genistein. Numerous studies have shown that genistein present in C. siliqua seed has protective effects on spermatogenesis (50-54). Verma and co-workers Found that genistein improves spermatogenesis by increasing lactate formation and antioxidant enzymes induced by insulin (53). That is why this compound can adjust the testicular dysfunction induced by type 2 diabetes in mice (53). In addition, Jalili and colleagues have shown that the administration of genistein can enhance spermatozoa quality and prevent morphine-induced side effects on sperm parameters (54). It has also been found that this compound relieves oxidative stress and spermatogenic injury induced by reperfusion and testicular ischemia by reducing the activity of testicular matrix metalloprotease (55). Chi and coworkers Showed that genistein effectively lowered intra-testicular testosterone level and stimulated spermatogenesis in rats treated with busulfan (56). Finally, it has been reported that genistein reduces gamma-ray-induced testicular injuries by antiapoptotic activity and remodeling spermatogenesis (57).
Another compound present in the extract of C. siliqua is gallic acid. Several studies have shown that gallic acid has protective effects on spermatogenesis (58, 59). Mehraban and colleagues showed that gallic acid can amend cyclophosphamide-induced reproductive toxicity in adult NMRI mice and increase the antioxidant capacity of testicular tissue (58). In another study, they also found that gallic acid has protective effects against sperm cell death as well as in vitro fertilization in adult male rats treated with cyclophosphamide (59).
C. siliqua extract also contains quercetin and luteolin, which have protective effects on spermatogenesis. For example, Ma and coworkers found that luteolin can amend testicular injuries and dysfunction through the Nrf2 signaling pathway by increasing regulation of Connexin 43 (Cx43) (60), and Khorsandi and colleagues reported that quercetin has some beneficial effects on spermatogenesis defects induced by titanium dioxide nanoparticles (61). Pretreatment with quercetin may increase testosterone concentration, decrease cell proliferation and oxidative stress in testicular tissue, and improve sperm quality as well as testicular tissue regeneration (61). Baltaci and coworkers (2016) showed that quercetin prevents arsenic-induced testicular injury through its antioxidant and antitumor effects (62). Similarly, Abd Ellah and colleagues found that this compound amends testicular injuries induced by di-(2-ethylhexyl) phthalate (63), and it can modify lambda cyhalothrin-induced reproductive toxicity in adult rats (64). Finally, Jahan and coworkers found that quercetin can possess potent therapeutic effects against bisphenol A-induced testicular toxicity in Sprague Dawley rats (65).
Based on the antiapoptotic and strong antioxidant (66) properties of C. siliqua, we conclude that this plant possesses the potential to protect subjects against doxorubicin toxicity through boosting activities of enzymes such as superoxide dismutase (SOX), Glutathione peroxidase (GPX), and catalase to protect against free radicals with cell toxicity (67). Since the moderate use of this plant has no apparent toxic effects, its beneficial potential in protecting against various toxicities is tempting.
5. Conclusion
Based on our findings, it appears that CSHAE at 300 mg/kg dose has a protective effect on doxorubicin-induced spermatogenesis toxicity, whereas its minimum (150 mg/kg) and maximum (600 mg/kg) doses have no significant influence on the number of germ cells, testicular tissue regeneration, and protamine gene expression. These results also indicate that the protective effects of C. siliqua extract are not dose-dependent, and even a dose as low as 300 mg/kg significantly increases the number of cells and regenerates testicular tissue injuries. Such protective effects are related to the antioxidant and antiapoptotic properties of various compounds present in the C. siliqua herb. Hence, this dose of CSHAE can be used as an effective substance against doxorubicin-induced testicular dysfunctions.
Acknowledgements
The authors hereby express their deep appreciation for the sincere cooperation of the research deputy of the Shiraz Azad University to conduct this research. The results described in this article are part of a Ph.D. thesis by Hengameh Mehdi Khani.
Conflict of Interest
The authors declare that they have no conflict of interests.
Type of Study: Original Article |
Subject:
Reproductive Andrology
References
1. Shalet SM. Normal testicular function and spermatogenesis. Pediat Blood Cancer 2009; 53: 285-288. [DOI:10.1002/pbc.22000] [PMID]
2. He Z, Kokkinaki M, Pant D, Gallicano GI, Dym M. Small RNA molecules in the regulation of spermatogenesis. Reproduction 2009; 137: 901-911. [DOI:10.1530/REP-08-0494] [PMID]
3. Ramaswamy S, Weinbauer GF. Endocrine control of spermatogenesis: Role of FSH and LH/testosterone. Spermatogenesis 2015; 4: e996025-1- e996025-15. [DOI:10.1080/21565562.2014.996025] [PMID] [PMCID]
4. Schulte RT, Ohl DA, Sigman M, Smith GD. Sperm DNA damage in male infertility: etiologies, assays, and outcomes. J Assist Reprod Genet 2010; 27: 3-12. [DOI:10.1007/s10815-009-9359-x] [PMID] [PMCID]
5. O'Flynn O'Brien KL, Varghese AC, Agarwal A. The genetic causes of male factor infertility: A review. Fertil Steril 2010; 93: 1-12. [DOI:10.1016/j.fertnstert.2009.10.045] [PMID]
6. Imken L, Rouba H, El Houate B, Louanjli N, Barakat A, Chafik A, et al. Mutations in the protamine locus: association with spermatogenic failure? Mol Hum Reprod 2009; 15: 733-738. [DOI:10.1093/molehr/gap056] [PMID]
7. Lüke L, Vicens A, Tourmente M, Roldan ERS. Evolution of protamine genes and changes in sperm head phenotype in rodents. Biol Reprod 2014; 90: 67-74. [DOI:10.1095/biolreprod.113.115956] [PMID]
8. Ravel C, Chantot-Bastaraud S, El Houate B, Berthaut I, Verstraete L, De Larouziere V, et al. Mutations in the protamine 1 gene associated with male infertility. Mol Hum Reprod 2007; 13: 461-464. [DOI:10.1093/molehr/gam031] [PMID]
9. Jodar M, Oliva R. Protamine alterations in human spermatozoa. Adv Exp Med Biol 2014; 791: 83-102. [DOI:10.1007/978-1-4614-7783-9_6] [PMID]
10. Donkin I, Barrès R. Sperm epigenetics and influence of environmental factors. Mol Metab 2018; 14: 1-11. [DOI:10.1016/j.molmet.2018.02.006] [PMID] [PMCID]
11. Taskin E, Dursun N. Recovery of adriamycin induced mitochondrial dysfunction in liver by selenium. Cytotechnology 2015; 67: 977-986. [DOI:10.1007/s10616-014-9736-x] [PMID] [PMCID]
12. Zanetti SR, Maldonado EN, Aveldano MI. Doxorubicin affects testicular lipids with long-chain (C18-C22) and very long-chain (C24-C32) polyunsaturated fatty acids. Cancer Res 2007; 67: 6973-6980. [DOI:10.1158/0008-5472.CAN-07-0376] [PMID]
13. Brilhante O, Okada FK, Sasso-Cerri E, Stumpp T, Miraglia SM. Late morfofunctional alterations of the Sertoli cell caused by doxorubicin administered to prepubertal rats. Reprod Biol Endocrinol 2012; 10: 79-94. [DOI:10.1186/1477-7827-10-79] [PMID] [PMCID]
14. Ekinci K, Yılmaz D, Ertekin C. Effects of moisture content and compression positions on mechanical properties of carob pod (Ceratonia siliqua L.). Afr J Agric Res 2010; 5: 1015-1021.
15. Bureš P, Pavlíček T, Horová L, Nevo E. Microgeographic genome size differentiation of the carob tree, Ceratonia siliqua, at 'Evolution Canyon', Israel. Ann Bot 2004; 93: 529-535. [DOI:10.1093/aob/mch074] [PMID] [PMCID]
16. Durazzo A, Turfani V, Narducci V, Azzini E, Maiani G, Carcea M. Nutritional characterisation and bioactive components of commercial carobs flours. Food Chem 2014; 153: 109-113. [DOI:10.1016/j.foodchem.2013.12.045] [PMID]
17. Goulas V, Stylos E, Chatziathanasiadou MV, Mavromoustakos T, Tzakos AG. Functional components of carob fruit: Linking the chemical and biological space. Int J Mol Sci 2016; 17: 1875-1894. [DOI:10.3390/ijms17111875] [PMID] [PMCID]
18. Eldahshan OA. Isolation and structure elucidation of phenolic compounds of carob leaves grown in Egypt. Curr Res J Biol Sci 2011; 3: 52-55.
19. Azab A. Carob (Ceratonia siliqua): Health, medicine and chemistry. Eur Chem Bull 2017; 6: 456-469. [DOI:10.17628/ecb.2017.6.456-469]
20. Dakia PA, Wathelet B, Paquot M. Isolation and chemical evaluation of carob (Ceratonia siliqua L.) seed germ. Food Chemistry 2007; 102: 1368-1374. [DOI:10.1016/j.foodchem.2006.05.059]
21. Ayaz FA, Torun H, Ayaz S, Correia PJ, Alaiz M, Sanz C, et al. Determination of chemical composition of anatolian carob pod (Ceratonia siliqua L.): sugars, amino and organic acids, minerals and phenolic compounds. Journal of Food Quality 2007; 30: 1040-1055. [DOI:10.1111/j.1745-4557.2007.00176.x]
22. Macho-González A, Garcimartín A, Naes F, López-Oliva ME, Amores-Arrojo A, González-Muñoz MJ, et al. Effects of fiber purified extract of carob fruit on fat digestion and postprandial lipemia in healthy rats. J Agric Food Chem 2018; 66: 6734-6741. [DOI:10.1021/acs.jafc.8b01476] [PMID]
23. Zhu BJ, Zayed MZ, Zhu HX, Zhao J, Li SP. Functional polysaccharides of carob fruit: a review. Chin Med 2019; 14: 40-49. [DOI:10.1186/s13020-019-0261-x] [PMID] [PMCID]
24. El-Haskoury R, Al-Waili N, El-Hilaly J, Al-Waili W, Lyoussi B. Antioxidant, hypoglycemic, and hepatoprotective effect of aqueous and ethyl acetate extract of carob honey in streptozotocin-induced diabetic rats. Vet World 2019; 12: 1916-1923. [DOI:10.14202/vetworld.2019.1916-1923] [PMID] [PMCID]
25. Rtibi K, Selmi S, Jabri MA, El-Benna J, Amri M, Marzouki L, et al. Protective effect of Ceratonia siliqua L. against a dextran sulfate sodium-induced alterations in liver and kidney in rat. J Med Food 2016; 19: 882-889. [DOI:10.1089/jmf.2016.0020] [PMID]
26. Corsi L, Avallone R, Cosenza F, Farina F, Baraldi C, Baraldi M. Antiproliferative effects of Ceratonia siliqua L. on mouse hepatocellular carcinoma cell line. Fitoterapia 2002; 73: 674-684. [DOI:10.1016/S0367-326X(02)00227-7]
27. Sassi A, Bouhlel I, Mustapha N, Mokdad-Bzeouich I, Chaabane F, Ghedira K, et al. Assessment in vitro of the genotoxicity, antigenotoxicity and antioxidant of Ceratonia siliqua L. extracts in murine leukaemia cells L1210 by comet assay. Regul Toxicol Pharmacol 2016; 77: 117-124. [DOI:10.1016/j.yrtph.2016.02.009] [PMID]
28. Ata A, Yildiz-Gulay O, Güngör S, Balic A, Gulay MS. The effect of carob (Ceratonia siliqua) bean extract on male New Zealand White rabbit semen. World Rabbit Sci 2018; 26: 209-215. [DOI:10.4995/wrs.2018.10154]
29. Mahdiani E, Khadem Haghighian H, Javadi M, Karami AA, Kavianpour M. Effect of Carob (Ceratonia siliqua L.) oral supplementation on changes of semen parameters, oxidative stress, inflammatory biomarkers and reproductive hormones in infertile men. Sci J Kurdistan Univ Med Sci 2018; 23: 56-66.
30. Jain H, Parial SD, Jarald E, Daud AS, Ahmad S. Extraction of Ashwagandha by conventional extraction methods and evaluation of its anti-stress activity. International Journal of Green Pharmacy (IJGP) 2010; 4: 183-185. [DOI:10.4103/0973-8258.69178]
31. Mokhtari M, Sharifi E, Azadian Sh. The effects of hydro alcoholic extract of Ceratonia siliqua L. seeds on pituitary--testis hormones and spermatogenesis in rat. Advances in Environmental Biology 2012; 2778-2784.
32. Bazrafkan M, Sobhani A. Study of spermatogenesis in wistar adult rats administrated to long term of ruta graveolens. Jentashapir Journal of Health Research 2014; 5: e21870. [DOI:10.17795/jjhr-21870]
33. Boussada M, Ali RB, Said AB, Bokri K, Akacha AB, Dziri C, et al. Selenium and a newly synthesized Thiocyanoacetamide reduce Doxorubicin gonadotoxicity in male rat. Biomed Pharmacother 2017; 89: 1005-1017. [DOI:10.1016/j.biopha.2017.03.002] [PMID]
34. Badkoobeh P, Parivar K, Kalantar SM, Hosseini SD, Salabat A. Protective effect of nano-zinc oxide on histological parameters of testis following doxorubicin treatment. Journal of Cell & Tissue 2013; 4: 159-167.
35. Faraji Z, Nikzad H, Parivar K, Nikzad M. The effect of aqueous extract of Salep Tubers on the structure of testis and sexual hormones in male mice. J Jahrom Univ Med Sci 2013; 11: 61-66. [DOI:10.29252/jmj.11.1.10]
36. Bustin SA. Real-time reverse transcription PCR. Encyclopedia of diagnostic genomics and proteomics 2005; 1131-1135. [DOI:10.3109/9780203997352.226]
37. Popal W, Nagy ZP. Laboratory processing and intracytoplasmic sperm injection using epididymal and testicular spermatozoa: what can be done to improve outcomes? Clinics 2013; 68: 125-130. [DOI:10.6061/clinics/2013(Sup01)14]
38. Lim NYN, Roco CA, Frostegård Å. Transparent DNA/RNA co-extraction workflow protocol suitable for inhibitor-rich environmental samples that focuses on complete DNA removal for transcriptomic analyses. Front Microbiol 2016; 7: 1588-1602. [DOI:10.3389/fmicb.2016.01588] [PMID] [PMCID]
39. Mo Y, Wan R, Zhang Q. Application of reverse transcription-PCR and real-time PCR in nanotoxicity research. Methods Mol Biol 2012; 926: 99-112. [DOI:10.1007/978-1-62703-002-1_7] [PMID] [PMCID]
40. Singh J, Birbian N, Sinha S, Goswami A. A critical review on PCR, its types and applications. Int J Adv Res Biol Sci 2014; 1: 65-80.
41. Vendramini V, Sasso-Cerri E, Miraglia SM. Amifostine reduces the seminiferous epithelium damage in doxorubicin-treated prepubertal rats without improving the fertility status. Reprod Biol Endocrinol 2010; 8: 3-15. [DOI:10.1186/1477-7827-8-3] [PMID] [PMCID]
42. Silva RC, Coelho Britto DM, de Fátima Pereira W, Brito-Melo GEA, Machado CT, Pedreira MM. Effect of short-and medium-term toxicity of doxorubicin on spermatogenesis in adult Wistar rats. Reprod Biol 2018; 18: 169-176. [DOI:10.1016/j.repbio.2018.03.002] [PMID]
43. Stumpp T, Freymüller E, Miraglia SM. Sertoli cell function in albino rats treated with etoposide during prepubertal phase. Histochem Cell Biol 2006; 126: 353-361. [DOI:10.1007/s00418-006-0168-3] [PMID]
44. Yeh YC, Lai HC, Ting CT, Lee WL, Wang LC, Wang KY, et al. Protection by doxycycline against doxorubicin-induced oxidative stress and apoptosis in mouse testes. Biochem Pharmacol 2007; 74: 969-980. [DOI:10.1016/j.bcp.2007.06.031] [PMID]
45. Uyeturk U, Uyeturk U, Firat T, Cetinkaya A, Tekce BK, Cakir S. Protective effects of rosmarinic acid on doxorubicin-induced testicular damage. Chemotherapy 2014; 60: 7-12. [DOI:10.1159/000365727] [PMID]
46. Cabral REL, Okada FK, Stumpp T, Vendramini V, Miraglia SM. Carnitine partially protects the rat testis against the late damage produced by doxorubicin administered during pre‐puberty. Andrology 2014; 2: 931-942. [DOI:10.1111/andr.279] [PMID]
47. Lachkar N, Al-Sobarry M, El Hajaji H, Lamkinsi T, Lachkar M, Cherrah Y, et al. Anti-inflammatory and antioxidant effect of Ceratonia siliqua L. methanol barks extract. J Chem Pharm Res 2016; 8: 202-210.
48. Soleimanzadeh A, Kian M, Moradi S, Mahmoudi S. Carob (Ceratonia siliqua L.) fruit hydro-alcoholic extract alleviates reproductive toxicity of lead in male mice: Evidence on sperm parameters, sex hormones, oxidative stress biomarkers and expression of Nrf2 and iNOS. Avicenna J Phytomed 2019; 10: 35-49.
49. Vafaei A, Mohammadi S, Fazel A, Soukhtanloo M, Mohammadipour A, Beheshti F. Effects of carob (Ceratonia siliqua) on sperm quality, testicular structure, testosterone level and oxidative stress in busulfan-induced infertile mice. Pharm Sci 2018; 24: 104-111. [DOI:10.15171/PS.2018.16]
50. Bengoechea C, Puppo MC, Romero A, Cordobes F, Guerrero A. Linear and non-linear viscoelasticity of emulsions containing carob protein as emulsifier. Journal of Food Engineering 2008; 87: 124-135. [DOI:10.1016/j.jfoodeng.2007.11.024]
51. Hossein MS, Tareq KMA, Hammano KI, Tsujii H. Effect of fatty acids on boar sperm motility, viability and acrosome reaction. Reprod Med Biol 2007; 6: 235-239. [DOI:10.1111/j.1447-0578.2007.00191.x] [PMID] [PMCID]
52. Melmed S, Polonsky KS, Larsen PR, Kronenberg HM. Chapter 38: Gastrointestinal hormones and gut endocrine tumors. Williams text book of endocrinology. 13th ed. Philadelphia: Elsevier 2016: 1701-1722.
53. Verma R, Samanta R, Krishna A. Comparative effects of estrogen and phytoestrogen, genistein on testicular activities of streptozotocin-induced type 2 diabetic mice. Reprod Sci 2019; 26: 1294-1306. [DOI:10.1177/1933719118815576] [PMID]
54. Jalili C, Ahmadi S, Roshankhah S, Salahshoor MR. Effect of genistein on reproductive parameter and serum nitric oxide levels in morphine-treated mice. Int J Reprod Biomed 2016; 14: 95-102. [DOI:10.29252/ijrm.14.2.95]
55. Al-Maghrebi M, Renno WM. Genistein alleviates testicular ischemia and reperfusion injury-induced spermatogenic damage and oxidative stress by suppressing abnormal testicular matrix metalloproteinase system via the Notch 2/Jagged 1/Hes-1 and caspase-8 pathways. J Physiol Pharmacol 2016; 67: 129-137.
56. Chi H, Chun K, Son H, Kim J, Kim G, Roh S. Effect of genistein administration on the recovery of spermatogenesis in the busulfan-treated rat testis. Clin Exp Reprod Med 2013; 40: 60-66. [DOI:10.5653/cerm.2013.40.2.60] [PMID] [PMCID]
57. Kim JS, Heo K, Yi JM, Gong EJ, Yang K, Moon C, et al. Genistein mitigates radiation‐induced testicular injury. Phytother Res 2012; 26: 1119-1125. [DOI:10.1002/ptr.3689] [PMID]
58. Mehraban Z, Ghaffari Novin M, Golmohammadi MG, Sagha M, Ziai SA, Abdollahifar MA, et al. Protective effect of gallic acid on testicular tissue, sperm parameters, and DNA fragmentation against toxicity induced by cyclophosphamide in Adult NMRI mice. Urol J 2020; 17: 78-85.
59. Mehraban Z, Ghaffari Novin M, Golmohammadi MG, Sagha M, Pouriran K, Nazarian H. Protective effect of gallic acid on apoptosis of sperm and in vitro fertilization in adult male mice treated with cyclophosphamide. J Cell Biochem 2019; 120: 17250-17257. [DOI:10.1002/jcb.28987] [PMID]
60. Ma B, Zhang J, Zhu Z, Zhao A, Zhou Y, Ying H, et al. Luteolin ameliorates testis injury and blood-testis barrier disruption through the Nrf2 signaling pathway and by upregulating Cx43. Mol Nutr Food Res 2019; 63: e1800843. [DOI:10.1002/mnfr.201800843] [PMID]
61. Khorsandi L, Orazizadeh M, Moradi-Gharibvand N, Hemadi M, Mansouri E. Beneficial effects of quercetin on titanium dioxide nanoparticles induced spermatogenesis defects in mice. Environ Sci Pollut Res 2017; 24: 5595-5606. [DOI:10.1007/s11356-016-8325-2] [PMID]
62. Baltaci BB, Uygur R, Caglar V, Aktas C, Aydin M, Ozen OA. Protective effects of quercetin against arsenic‐induced testicular damage in rats. Andrologia 2016; 48: 1202-1213. [DOI:10.1111/and.12561] [PMID]
63. Abd-Ellah MF, Aly HA, Mokhlis HA, Abdel-Aziz AH. Quercetin attenuates di-(2-ethylhexyl) phthalate-induced testicular toxicity in adult rats. Hum Exp Toxicol 2016; 35: 232-243. [DOI:10.1177/0960327115580602] [PMID]
64. Ben Abdallah F, Fetoui H, Zribi N, Fakhfakh F, Keskes L. Quercetin attenuates lambda cyhalothrin‐induced reproductive toxicity in male rats. Environ Toxicol 2013; 28: 673-680. [DOI:10.1002/tox.20762] [PMID]
65. Jahan S, Ain QU, Ullah H. Therapeutic effects of quercetin against bisphenol A induced testicular damage in male Sprague Dawley rats. Syst Biol Reprod Med 2016; 62: 114-124. [DOI:10.3109/19396368.2015.1115139] [PMID]
66. Custódio L, Fernandes E, Escapa AL, López-Avilés S, Fajardo A, Aligué R et al. Antioxidant activity and in vitro inhibition of tumor cell growth by leaf extracts from the carob tree (Ceratonia siliqua). Pharmaceutical Biology 2009; 47: 721-728. [DOI:10.1080/13880200902936891]
67. Sadeghnia HR, Kamkar M, Assadpour E, Boroushaki MT, Ghorbani A. Protective effect of safranal, a constituent of Crocus sativus, on quinolinic acid-induced oxidative damage in rat hippocampus. Iran J Basic Med Sci 2013; 16: 73-82.
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |








