Thu, Apr 25, 2024
[Archive]
Volume 3, Issue 1 (7-2005)
IJRM 2005, 3(1): 36-41 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Rezaei N, Chian R. Effects of essential and non-essential amino acids on in-vitro maturation, fertilization and development of immature bovine oocytes. IJRM 2005; 3 (1) :36-41
URL: http://ijrm.ir/article-1-30-en.html
URL: http://ijrm.ir/article-1-30-en.html
Full-Text [PDF 106 kb]
(655 Downloads)
| Abstract (HTML) (2875 Views)
Full-Text: (389 Views)
Introduction
Essential and/or non-essential amino acids are commonly added to serum-supplemented or serum-free culture media used for mammalian embryo development in vitro. In many species, it has been known that addition of amino acids to the culture medium is beneficial for embryonic development (1-3). Apart from amino acids used for protein synthesis, they play important role as osmolytes (4), intracellular buffers (5), heavy metal chelators and energy sources as well as precursors for versatile physiological regulators, such as nitric oxide and polyamines (6). It has also been shown that the culture medium with amino acids affect glucose metabolism in mouse blastocysts in vitro (2). Although it has been shown that amino acids support rabbit (7), hamster (8), porcine (9) and bovine (10) oocyte maturation, amino acid requirements for oocyte maturation in culture is not fully understood. The objective of this study was to investigate the effects of essential and non-essential amino acids on in-vitro maturation, subsequent fertilization and embryo development of immature bovine oocyte.
Materials and Methods
After washing, COCs were randomly assigned to following 4 groups of maturation medium respectively: (1) Basic medium alone (Control); (2) Basic medium supplemented with 2% MEM essential amino acids solution (GIBCO; 50X); (3) Basic medium supplemented with 1% MEM non-essential amino acids solution (GIBCO; 100X); (4) Basic medium supplemented with 2% MEM essential amino acids solution + 1% MEM non-essential amino acids solution. COCs were cultured in 1 ml maturation medium in an Organ culture dish (Falcon; 60x15 mm) at 38.5°C in an atmosphere of 5% CO2 with high humidity. After maturation for 24 h, some oocytes (n=372) were fixed and stained, then evaluated for stage of nuclear maturation by bright field microscopy (magnification: 400X) to examine maturation rate. The stages of nuclear maturation were assessed as germinal vesicle (GV: oocytes arrested at prophase I of meiosis are characterized at the light microscope level, as having a visible nucleus), metaphase I (MI: when meiosis resumes, the oocyte undergoes dissolution of the nuclear envelope, subsequently chromatin condenses into discrete bivalents that align on the meiotic spindle at metaphase I) and metaphase II (MII: the separation is complete at MII, which is recognizable at the light microscope level by the presence of the first polar body) (11). The remaining oocytes (n=840) were used for in vitro fertilization (IVF).
Results
As shown in Table II when the immature bovine oocytes were cultured in basic IVM- medium supplement with 2% essential and 1% non-essential amino acids, there were no significant differences in maturation rates among the four groups.
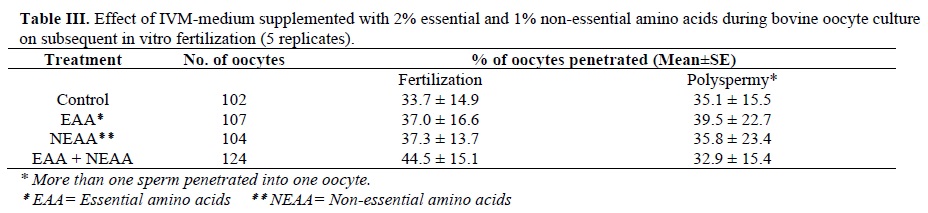
Table III shows the effect of IVM-medium supplement with 2% essential and 1% non-essential amino acids during bovine oocyte culture on subsequent in-vitro fertilization. There were no significant differences in penetration rates among the four groups.
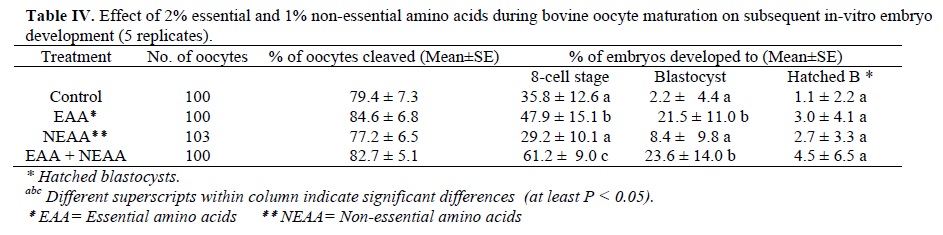
Table IV shows the effect of 2% essential and 1% non-essential amino acids during bovine oocyte maturation on subsequent embryo development. The oocyte cleavage rates were not different in the four groups.
The embryo development to the 8-cell stage were significantly (p<0.05) higher in groups 2 (47.9±15.1) and 4 (61.2± 9) than in the control (35.8±12.6) and group 3 (29.2±10.1).
The blastocyst rates in groups 2 (21.5±11.0) and 4 (23.6±14.0) were significantly (p<0.05) higher than in the control (2.2±4.4) and group 3 (8.4±9.8). There were no differences in hatched blastocyst rates among the four groups.

Discussion
One of the important factors regulating the number and quality of oocytes maturing in vitro is the culture system used for IVM. Culture media components and culture conditions can affect and even modulate the meiotic regulation of mammalian oocytes (12-14). It is therefore necessary to devise and optimize culture systems that take into account all the factors essential for the completion of oocyte maturation in vitro.
The present study showed that there were not significant differences in oocyte maturation rates (table II) and penetration rates (table III) among the four groups. These results were similar to the previous results of by lim et al. (1999), who have been studied the effects of carbohydrates and amino acids on the maturation and fertilization of bovine oocytes. They have shown that the addition of glucose to simply defined medium significantly enhanced oocyte maturation to the metaphase–II stage, but the addition of EAA and NEAA to basic medium supplement with glucose did not further improve in vitro maturation or in vitro fertilization of bovine oocytes. Also, they have suggested that the exogenous carbohydrates and amino acids are prerequisites for the maturation and fertilization of bovine oocytes in vitro, glucose alone promotes the nuclear maturation of oocytes, whereas amino acids aid the pronuclear formation of fertilized oocytes (15). Because in our study, glucose and pyruvate were the components of the basic medium in all four groups, probably due to their presences, maturation rates were not different after the addition of EAA or NEAA or both. Downs and Hudson (2000) have shown when glucose was added to pyruvate-containing cultures in mouse oocyte, the combination of 1mM pyruvate/5.5mM glucose was most effective in supporting maturation. The positive effect of glucose was in part attributed to stimulation of glycolysis and increased production of pyruvate (16). Kerisher and Bavister (17) study in cattle and Zheng et al. (18) study in rhesus monkeys have shown that the addition of glucose to maturation media improves the resumption of meiosis, embryo cleavage, morulae and blastocyst rates. Also, our findings support a previous report of effects of amino acids on pig oocytes maturation in vitro (19).
Studies on several mammalians species, including the rabbit (20), hamster (21, 22), mouse (23), sheep (24), cattle (25) and rhesus monkey (18) have revealed that amino acids can stimulate both oocyte maturation and embryo development in culture.
In vivo, the mammalian embryo is exposed to significant levels of amino acids (EAA and NEAA) in oviduct and uterine fluids (6,26). It has been known that amino acids are transferred to the oocytes by the action of gap junction between cumulus cells and oocytes (9). Specific amino acid transporters are present on the membranes of oocytes and embryos and a supply of amino acids for protein synthesis is essential for normal embryo growth (27). It is generally accepted that mRNA and protein molecules synthesized during oocyte growth, maturation and early embryo development is driven by mRNA and protein stored in the oocytes (28). The embryonic genome turns on during the 2–cell stage in mice (29), the 4–cell stage in rabbits (28), and the 8–16–cell stage in bovine (30) embryos, leading to quantitative and qualitative changes in protein synthesis, an increase in metabolic activity and the uptake of carbohydrates. Amino acid supplementation of oocyte maturation media was associated with enhanced developmental frequencies, increased blastocyst cell number, and elevated oocyte maternal mRNA levels compared with defined media without amino acids (31). The uptake and incorporation of amino acids by embryos increased from the zygote to the blastocyst stages. Liu et al. (1996) have demonstrated that protein synthesis must occur prior to the morula stage for bovine embryos to develop normally into blastocysts (28). Embryos synthesize a considerable amount of protein, particularly as they reach the blastocyst stage (28).Therefore, the results of present study suggest that presence of EAA and NEAA in maturation medium by the increase endogenous amino acid pool sizes and/or de novo protein synthesis may be essential for maturation of bovine oocytes.
In the current experiment, the percentage of embryos that reached cleavage rate was similar for the four treatment groups, but embryo development to the 8–cell stage (68 hr post-insemination) and blastocyst were significantly higher in groups 2 (contained EAA) and 4 (contained EAA and NEAA) than these in the Groups 3 (NEAA) and 1 (control), (table IV). This is in agreement with the work of Liu et al. (1999), who found that when the amino acids were excluded during the first 24 hr of culture, rabbit embryo development proceeded compartment to the controls (28), this indicated that the supply of endogenous amino acids available to the embryo is sufficient during very early development (32). This is in contrast to previous report for mouse. Amino acids have been shown to increase the cleavage rate of mouse, producing blastocyst with higher viability (33, 36). Gardner et al. (1994) have reported, that amino acids reduced the percentage of embryos arrested during culture and stimulated both cleavage and hatching (26). Kim et al. (1993) have been demonstrated that the uptake of amino acids increases from 8-cell to blastocyst stage of mouse embryos (35). Lane and Gardner (1997) have reported that mouse embryo changed its requirements for amino acids as it developed from the zygote to the blastocyst, development of the early cleavage stages was stimulated by the non-essential amino acids and glutamine, but was not effected by the essential amino acids (34). Steeves and Gardner (1994) have revealed not only that the bovine embryos has a requirement for amino acids, but also that amino acids have both a temporal and differential effect during development from the 1-to-2-cell zygote to the blastocyst. They suggest that the requirement for amino acids changes according to the developmental stage of embryos and the metabolic requirements are different in different developmental stage (30). Koo et al. (1997) reported that the addition of amino acids to NCSU 23 enhanced in vitro development of 1-to 2–cell stage porcine embryos to the hatching stage, because events related to protein synthesis in the hatching process that occur prior to morula formation may be dependent upon the availability of certain amino acids (1).
In our experiments EAA alone tended to increase blastocyst development, while when it combined with NEAA, blastocyst production and hatching were superior to the results using other treatments (table IV). Our results confirm Rosenkrans et al. findings (37). This is in contrast to previous reports for hamster embryo (38), which shows that EAA alone tended to depress blastocyst development, but similar with reports on addition of EAA and NEAA in culture medium for mouse (23). Gardner et al. (1994) have shown that all Eagle's amino acids significantly increase blastocyst formation, hatching and cell number in sheep (24). Eagle's essential amino acids were inhibitory when present before the 8-cell stage, but promoted blastocyst development and cell number when present after the 8-cell stage (36). During development from the 8-cell stage to the blastocyst, the non-essential amino acids and glutamine stimulated blastocyst formation and hatching, while the essential amino acids increased blastocyst cell number and differentiation of cells into the inner cell mass (30). Gardner and lane (1993) have demonstrated that the inclusion of EAA in the medium had no effect on mouse blastocyst hatching, in contrast, when all of Eagle's amino acids were present (23), hatching was significantly increased and further increases in the hatching rates were observed when only the non-essential amino acids, with or without glutamine, were present. This discrepancy might be due to a species-specific or stage-specific requirement for amino acids during oocyte maturation and embryo development.
Conclusion
In conclusion, bovine oocytes can be successfully developed in a chemically defined, protein free medium supplemented with EAA and NEAA, from maturation through fertilization and culture. These data indicate that the presence of amino acids, especially essential amino acids, in the maturation medium is beneficial to oocyte cytoplasmic maturation and subsequent early embryonic development in vitro. However, additional research is needed to compare protein synthesis in oocyte cytoplasm among each group and to compare the changes in poly (A) tail length of maternal transcripts in each group.
Acknowledgement
The authors would like to thank Jin-tea Chung and Ahmad-Kamal Abduljalil for their technical assistance.
Essential and/or non-essential amino acids are commonly added to serum-supplemented or serum-free culture media used for mammalian embryo development in vitro. In many species, it has been known that addition of amino acids to the culture medium is beneficial for embryonic development (1-3). Apart from amino acids used for protein synthesis, they play important role as osmolytes (4), intracellular buffers (5), heavy metal chelators and energy sources as well as precursors for versatile physiological regulators, such as nitric oxide and polyamines (6). It has also been shown that the culture medium with amino acids affect glucose metabolism in mouse blastocysts in vitro (2). Although it has been shown that amino acids support rabbit (7), hamster (8), porcine (9) and bovine (10) oocyte maturation, amino acid requirements for oocyte maturation in culture is not fully understood. The objective of this study was to investigate the effects of essential and non-essential amino acids on in-vitro maturation, subsequent fertilization and embryo development of immature bovine oocyte.
Materials and Methods
Maturation of Oocytes in Vitro
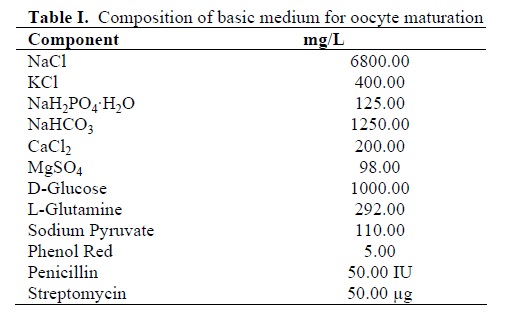
This study was an experimental type. Ovaries from Holstein heifers and cows were collected at local abattoir shortly after slaughter and transported to the laboratory within 3 h in 0.9% NaCl aqueous solution containing 100 IU/ml penicillin, and 100µg/ml streptomycin at approximately 35°C. Cumulus-oocyte complexes (COCs; n=1212) were aspirated from 2 to 8 mm follicles with an 18 G needle connected to a 10ml disposable syringe. Oocytes with unexpanded cumulus mass, having more than two layers of cumulus cells and with homogeneous granular ooplasm were selected, as described previously (11). The COCs were rapidly washed 4 times in HEPES buffered Tyrode’s medium (TLH) supplemented with 0.3% polyvinylpyrrolidone (PVP), 0.25mM pyruvic acid (Sigma) and 50 µg/ml gentamycin (Sigma). The basic medium for oocyte maturation is a chemically defined protein-free medium (Table I) supplemented with 0.3% PVP (Sigma), 75mIU/ml FSH and LH (Humegon; Organon, Scarborough, ON, Canada).
After washing, COCs were randomly assigned to following 4 groups of maturation medium respectively: (1) Basic medium alone (Control); (2) Basic medium supplemented with 2% MEM essential amino acids solution (GIBCO; 50X); (3) Basic medium supplemented with 1% MEM non-essential amino acids solution (GIBCO; 100X); (4) Basic medium supplemented with 2% MEM essential amino acids solution + 1% MEM non-essential amino acids solution. COCs were cultured in 1 ml maturation medium in an Organ culture dish (Falcon; 60x15 mm) at 38.5°C in an atmosphere of 5% CO2 with high humidity. After maturation for 24 h, some oocytes (n=372) were fixed and stained, then evaluated for stage of nuclear maturation by bright field microscopy (magnification: 400X) to examine maturation rate. The stages of nuclear maturation were assessed as germinal vesicle (GV: oocytes arrested at prophase I of meiosis are characterized at the light microscope level, as having a visible nucleus), metaphase I (MI: when meiosis resumes, the oocyte undergoes dissolution of the nuclear envelope, subsequently chromatin condenses into discrete bivalents that align on the meiotic spindle at metaphase I) and metaphase II (MII: the separation is complete at MII, which is recognizable at the light microscope level by the presence of the first polar body) (11). The remaining oocytes (n=840) were used for in vitro fertilization (IVF).
Sperm Preparation and IVF
Frozen semen was used for IVF. Straws of semen were thawed in a water bath (35°C) for 30 seconds and processed by swim-up as described before (11). The sperm were then washed twice in modified Tyrode’s albumin lactate pyruvate medium (Sp-TALP) used for sperm culture containing 6 mg/ml fatty acid-free BSA (Sigma), 10mM pyruvic acid, and 50µg/ml gentamycin. Following maturation, COCs were washed three times with TLH and then sperm/oocytes were incubated in 50µl droplets of the fertilization medium, modified Tyrode’s medium (mTALP), and contained 2µg/ml heparin under mineral oil at 38.5°C in 5% CO2 with high humidity. The final sperm concentration of 1 x 106 sperm/ml was used for oocyte insemination and five oocytes were used for each 50µl droplet.Embryo developmental culture
Following 18 h of insemination, some oocytes (n=437) were fixed for examining fertilization rate and the remaining oocytes (n=403) were washed three times with TLH and then transferred to 50µl droplets of development medium (BECM: Bovine Embryo Culture Medium) supplemented with 3 mg/ml BSA (fatty acid-free, Sigma) and 1µg/ml gentamycin under mineral oil. The culture medium was changed at 24 h intervals until 120 h after insemination and then the embryos were transferred to 50µl droplets of BECM supplemented with 10% FBS and 0.25mM pyruvic acid for further developmental culture (8days).Fixation of oocytes
At 24 h of maturation and at 18 h of insemination, the oocytes were mounted on slides with coverslips and fixed with acetic acid/ethanol (1:3) solution for at least 24 h. The oocytes were then stained with 1% orcein dissolved in 45% acetic acid solution and examined for evidence of fertilization. Fertilization was identified by observing two pronuclei with an accompanying sperm tail in the cytoplasm. Oocytes with two pronuclei and a clear second polar body but without a sperm tail were also considered to have been fertilized. Oocytes with a female pronucleus and a decondensed sperm head were considered abnormal fertilization. Oocytes with three pronuclei (two sperm) or more were considered polyspermy.Statistical analysis
The numbers of immature oocytes, maturation, fertilization and embryo cleavage rates as well as blastocyst formation rate from each group were analyzed by one-way analysis of variance. When analysis revealed significance, the groups were compared using the Student-Newman-Keuls’ test.Results
As shown in Table II when the immature bovine oocytes were cultured in basic IVM- medium supplement with 2% essential and 1% non-essential amino acids, there were no significant differences in maturation rates among the four groups.
Table III shows the effect of IVM-medium supplement with 2% essential and 1% non-essential amino acids during bovine oocyte culture on subsequent in-vitro fertilization. There were no significant differences in penetration rates among the four groups.
Table IV shows the effect of 2% essential and 1% non-essential amino acids during bovine oocyte maturation on subsequent embryo development. The oocyte cleavage rates were not different in the four groups.
The embryo development to the 8-cell stage were significantly (p<0.05) higher in groups 2 (47.9±15.1) and 4 (61.2± 9) than in the control (35.8±12.6) and group 3 (29.2±10.1).
The blastocyst rates in groups 2 (21.5±11.0) and 4 (23.6±14.0) were significantly (p<0.05) higher than in the control (2.2±4.4) and group 3 (8.4±9.8). There were no differences in hatched blastocyst rates among the four groups.



Discussion
One of the important factors regulating the number and quality of oocytes maturing in vitro is the culture system used for IVM. Culture media components and culture conditions can affect and even modulate the meiotic regulation of mammalian oocytes (12-14). It is therefore necessary to devise and optimize culture systems that take into account all the factors essential for the completion of oocyte maturation in vitro.
The present study showed that there were not significant differences in oocyte maturation rates (table II) and penetration rates (table III) among the four groups. These results were similar to the previous results of by lim et al. (1999), who have been studied the effects of carbohydrates and amino acids on the maturation and fertilization of bovine oocytes. They have shown that the addition of glucose to simply defined medium significantly enhanced oocyte maturation to the metaphase–II stage, but the addition of EAA and NEAA to basic medium supplement with glucose did not further improve in vitro maturation or in vitro fertilization of bovine oocytes. Also, they have suggested that the exogenous carbohydrates and amino acids are prerequisites for the maturation and fertilization of bovine oocytes in vitro, glucose alone promotes the nuclear maturation of oocytes, whereas amino acids aid the pronuclear formation of fertilized oocytes (15). Because in our study, glucose and pyruvate were the components of the basic medium in all four groups, probably due to their presences, maturation rates were not different after the addition of EAA or NEAA or both. Downs and Hudson (2000) have shown when glucose was added to pyruvate-containing cultures in mouse oocyte, the combination of 1mM pyruvate/5.5mM glucose was most effective in supporting maturation. The positive effect of glucose was in part attributed to stimulation of glycolysis and increased production of pyruvate (16). Kerisher and Bavister (17) study in cattle and Zheng et al. (18) study in rhesus monkeys have shown that the addition of glucose to maturation media improves the resumption of meiosis, embryo cleavage, morulae and blastocyst rates. Also, our findings support a previous report of effects of amino acids on pig oocytes maturation in vitro (19).
Studies on several mammalians species, including the rabbit (20), hamster (21, 22), mouse (23), sheep (24), cattle (25) and rhesus monkey (18) have revealed that amino acids can stimulate both oocyte maturation and embryo development in culture.
In vivo, the mammalian embryo is exposed to significant levels of amino acids (EAA and NEAA) in oviduct and uterine fluids (6,26). It has been known that amino acids are transferred to the oocytes by the action of gap junction between cumulus cells and oocytes (9). Specific amino acid transporters are present on the membranes of oocytes and embryos and a supply of amino acids for protein synthesis is essential for normal embryo growth (27). It is generally accepted that mRNA and protein molecules synthesized during oocyte growth, maturation and early embryo development is driven by mRNA and protein stored in the oocytes (28). The embryonic genome turns on during the 2–cell stage in mice (29), the 4–cell stage in rabbits (28), and the 8–16–cell stage in bovine (30) embryos, leading to quantitative and qualitative changes in protein synthesis, an increase in metabolic activity and the uptake of carbohydrates. Amino acid supplementation of oocyte maturation media was associated with enhanced developmental frequencies, increased blastocyst cell number, and elevated oocyte maternal mRNA levels compared with defined media without amino acids (31). The uptake and incorporation of amino acids by embryos increased from the zygote to the blastocyst stages. Liu et al. (1996) have demonstrated that protein synthesis must occur prior to the morula stage for bovine embryos to develop normally into blastocysts (28). Embryos synthesize a considerable amount of protein, particularly as they reach the blastocyst stage (28).Therefore, the results of present study suggest that presence of EAA and NEAA in maturation medium by the increase endogenous amino acid pool sizes and/or de novo protein synthesis may be essential for maturation of bovine oocytes.
In the current experiment, the percentage of embryos that reached cleavage rate was similar for the four treatment groups, but embryo development to the 8–cell stage (68 hr post-insemination) and blastocyst were significantly higher in groups 2 (contained EAA) and 4 (contained EAA and NEAA) than these in the Groups 3 (NEAA) and 1 (control), (table IV). This is in agreement with the work of Liu et al. (1999), who found that when the amino acids were excluded during the first 24 hr of culture, rabbit embryo development proceeded compartment to the controls (28), this indicated that the supply of endogenous amino acids available to the embryo is sufficient during very early development (32). This is in contrast to previous report for mouse. Amino acids have been shown to increase the cleavage rate of mouse, producing blastocyst with higher viability (33, 36). Gardner et al. (1994) have reported, that amino acids reduced the percentage of embryos arrested during culture and stimulated both cleavage and hatching (26). Kim et al. (1993) have been demonstrated that the uptake of amino acids increases from 8-cell to blastocyst stage of mouse embryos (35). Lane and Gardner (1997) have reported that mouse embryo changed its requirements for amino acids as it developed from the zygote to the blastocyst, development of the early cleavage stages was stimulated by the non-essential amino acids and glutamine, but was not effected by the essential amino acids (34). Steeves and Gardner (1994) have revealed not only that the bovine embryos has a requirement for amino acids, but also that amino acids have both a temporal and differential effect during development from the 1-to-2-cell zygote to the blastocyst. They suggest that the requirement for amino acids changes according to the developmental stage of embryos and the metabolic requirements are different in different developmental stage (30). Koo et al. (1997) reported that the addition of amino acids to NCSU 23 enhanced in vitro development of 1-to 2–cell stage porcine embryos to the hatching stage, because events related to protein synthesis in the hatching process that occur prior to morula formation may be dependent upon the availability of certain amino acids (1).
In our experiments EAA alone tended to increase blastocyst development, while when it combined with NEAA, blastocyst production and hatching were superior to the results using other treatments (table IV). Our results confirm Rosenkrans et al. findings (37). This is in contrast to previous reports for hamster embryo (38), which shows that EAA alone tended to depress blastocyst development, but similar with reports on addition of EAA and NEAA in culture medium for mouse (23). Gardner et al. (1994) have shown that all Eagle's amino acids significantly increase blastocyst formation, hatching and cell number in sheep (24). Eagle's essential amino acids were inhibitory when present before the 8-cell stage, but promoted blastocyst development and cell number when present after the 8-cell stage (36). During development from the 8-cell stage to the blastocyst, the non-essential amino acids and glutamine stimulated blastocyst formation and hatching, while the essential amino acids increased blastocyst cell number and differentiation of cells into the inner cell mass (30). Gardner and lane (1993) have demonstrated that the inclusion of EAA in the medium had no effect on mouse blastocyst hatching, in contrast, when all of Eagle's amino acids were present (23), hatching was significantly increased and further increases in the hatching rates were observed when only the non-essential amino acids, with or without glutamine, were present. This discrepancy might be due to a species-specific or stage-specific requirement for amino acids during oocyte maturation and embryo development.
Conclusion
In conclusion, bovine oocytes can be successfully developed in a chemically defined, protein free medium supplemented with EAA and NEAA, from maturation through fertilization and culture. These data indicate that the presence of amino acids, especially essential amino acids, in the maturation medium is beneficial to oocyte cytoplasmic maturation and subsequent early embryonic development in vitro. However, additional research is needed to compare protein synthesis in oocyte cytoplasm among each group and to compare the changes in poly (A) tail length of maternal transcripts in each group.
Acknowledgement
The authors would like to thank Jin-tea Chung and Ahmad-Kamal Abduljalil for their technical assistance.
Type of Study: Original Article |
References
1. Koo DB, kim NH, Lee HT, Chany KS. Effects of fetal calf serum, amino acids, vitamins and insulin on blastocoel formation and hatching of in vivo and lVM/IVF-derived porcine embryos developing in vitro. Theriogenology 1997; 48: 791-802. [DOI:10.1016/S0093-691X(97)00302-6]
2. Lane M, and Gardner DK. Amino acids and vitamins prevent culture-induced metabolic perturbations and associatrd loss of viability of mouse blastocysts. Human Reproduction 1998; 13(4): 991-997. [DOI:10.1093/humrep/13.4.991] [PMID]
3. Swain JE, Bormann CL, Clark SG,Walters EM, Wheeler MB, Krisher RL. Use of energy substrates by various stage preimplantation pig embryos produced in vivo and in vitro. Reproduction 2002; 123(2): 253-260. [DOI:10.1530/rep.0.1230253] [PMID]
4. Anbari K, Schultz RM. Effect of sodium and betaine in culture media on development and relative rates of protein synthesis in preimplantation mouse embryos in vitro. Mol Reprod Dev 1993; 35: 24-28. [DOI:10.1002/mrd.1080350105] [PMID]
5. Edwards LJ, Williams DA, Gardner DK. Intracellular PH of the mouse preimplantation embryo: amino acids act as buffers of intracellular PH. Hum Reprod 1998;13: 344-348. [DOI:10.1093/humrep/13.12.3441] [PMID]
6. Bavister BD. Culture of preimplantation embryos: facts and artifacts. Hum Reprod update 1995; 1:91-148. [DOI:10.1093/humupd/1.2.91] [PMID]
7. Bae IH, and Foote RH. Carbohydrate and amino acid requirements and ammonia production of rabbit follicular oocytes matured in vitro. Exp Cell Res1975; 91(1): 113-118. [DOI:10.1016/0014-4827(75)90148-2]
8. Gwatkin and Haidri. Requirements for the maturation of hamster oocytes in vitro. Exp Cell Res 1973; 76(1): 1-7. [DOI:10.1016/0014-4827(73)90411-4]
9. Ka HH, Sawai K, Wang WH, Im KS, Niwa K. Amino acids in maturation medium and presence of cumulus cells at fertilization promote male pronuclear formation in porcine oocytes matured and penetrated in vitro. Biol Reprod1997; 57(6): 1478-1483. [DOI:10.1095/biolreprod57.6.1478] [PMID]
10. Rose-Hellekant TA, Libersky-Williamson E.A. Bavister B.D: Energy substrates and amino acids provided during in vitro maturation of bovine oocytyes alter acquisition of developmental competence. Zygote 1998; 60: 285-294. [DOI:10.1017/S0967199498000239] [PMID]
11. Chian RC, Chung J, Downey BR, Tan SL. Maturational and developmental competence of immature oocytes retrieved from bovine ovaries at different phases of folliculogenesis. Reproductive Biomedicine Online 2002; 4(2): 129-134. [DOI:10.1016/S1472-6483(10)61929-3]
12. Downs SM, and Mastropolo AM. Culture conditions affect meiotic regulation in cumulus-cell enclosed mouse oocytes. Mol Reprod Dev 1997; 46: 551-566.
https://doi.org/10.1002/(SICI)1098-2795(199704)46:4<551::AID-MRD13>3.0.CO;2-Z [DOI:10.1002/(SICI)1098-2795(199704)46:43.0.CO;2-Z]
13. Kito S, and Bavister BD. Maturation of hamster oocytes under chemically defined conditions and sperm penetration through the zona pelucida. Zygote 1997; 4: 199-210. [DOI:10.1017/S0967199400003117] [PMID]
14. Hardy K, Wright CS, Franks S, Winston RML. In vitro maturation of oocytes. British Medical Bulletin 2000; 56(3): 588-602. [DOI:10.1258/0007142001903391] [PMID]
15. Lim Jm, Lee BC, Lee EC, Chung HM, Koj J, Paik SE, et al. In vitro maturation and in vitro Fertilization of bovine oocytes cultured in a chemichally defined, protein free medium: effects of carbohydrates and amino acids. Reprod Fertil Dev 1999; 11(2): 127-132. [DOI:10.1071/RD99001] [PMID]
16. Downs SM, and Hudson ED. Energy substrates and the completion of spontaneous meiotic maturation. Zygote 2000; 8: 339-351. [DOI:10.1017/S0967199400001131] [PMID]
17. Krisher RL, Bavister BD. Enhanced glycolysis after maturation of bovine oocytes in vitro is associated with increased developmental competence. Mol Reprod Dev 1999; 53(1): 19-26.
https://doi.org/10.1002/(SICI)1098-2795(199905)53:1<19::AID-MRD3>3.0.CO;2-U [DOI:10.1002/(SICI)1098-2795(199905)53:13.0.CO;2-U]
18. Zheng p, Bavister BD, Ji WZ. Amino acid requirements for muturation of rhesus monkey (Mac acca mulatta) oocytes in culture. Reproduction 2002; 124: 515-522. [DOI:10.1530/rep.0.1240515] [PMID]
19. Hong JY, Yong HY, Lee BC, Hwang WS, Lim JM, Lee ES. Effects of amino acids on maturation, fertilization and embryo development of pig follicular oocytes in two IVM media. Theriogenology 2004; 62(8): 1473-1482. [DOI:10.1016/j.theriogenology.2004.02.013] [PMID]
20. Li J, and Foote KH. Effect of inositol with increasing sodium chloride and constant osmolarity on cultured rabbit embryos. J Assisted Reprod Genetics 1995; 12: 141-146. [DOI:10.1007/BF02211384] [PMID]
21. Bavister BD, Leibfried ML, Leiberman G. Development of preimplantation embryos of the golden hamster in a defined culture medium. Biol Reprod 1983; 28(1): 235-247. [DOI:10.1095/biolreprod28.1.235] [PMID]
22. Corney EW, and Bavister BD. Stimulatory and inhibitory effects of amino acids on the development of hamster 8-cell embryo in vitro. J In Vitro Fertile and Embryo Transfer 1987; 4: 162-167. [DOI:10.1007/BF01555464] [PMID]
23. Gardner DK, and Lane M. Amino acids and ammonium regulate mouse embryo development in culture. Biol Reprod 1993; 48: 377-385. [DOI:10.1095/biolreprod48.2.377] [PMID]
24. Gardner DK, Lane M, Spitzer A, Batt P. Enhanced rates of cleavage and development for sheep zygotes cultured to the blastocyst stage in vitro in the absence of serum and somatic cells, amino acids, vitamins and culturiny embryos in groups stimulate development. Biol Reprod 1994; 50: 390-400. [DOI:10.1095/biolreprod50.2.390] [PMID]
25. Liu z, and Foote RH. Effects of amino acids on the development of in vitro maturation in vitro fertilization bovine embryos in a simple protein-free medium. Human Reproduction 1995; 10(ll): 2925-2991. [DOI:10.1093/oxfordjournals.humrep.a135834] [PMID]
26. Elhassan YM, Wu G, Leanez AC, Tasca RJ, Watson AJ, Westhusin ME. Amino acid concentrations in fluids from the bovine oviduct and uterus and in ksom-based culture media. Theriogenology 2001; 55:1907-1918. [DOI:10.1016/S0093-691X(01)00532-5]
27. Van Winkle LJ. Amino acid transport regulation and early embryo development. Biology of Reproduction 2001; 64: 1-12. [DOI:10.1095/biolreprod64.1.1] [PMID]
28. Liu Zishu, Foote Robert H, simkin Michael E. Effect of amino acids and α-Amanitin on the development of Rabbit Embryos in Modified Protein-Free KSOM with Hepes. Molecular Reproduction and development 1996; 45: 157-162.
https://doi.org/10.1002/(SICI)1098-2795(199610)45:2<157::AID-MRD8>3.0.CO;2-S [DOI:10.1002/(SICI)1098-2795(199610)45:23.0.CO;2-S]
29. Bolten VN, Oades PJ, Johnson MH. The relationship between cleavage, DNA replication, and gene expression in the mouse 2-cell embryo. J Emoryol EXP Morphol 1984; 75: 139-169.
30. Steeves TE, and Gardner DK. Temporal and differential effects of amino acids on bovine embryo development in culture. Biology of Reproduction GL 1999; 61(3): 731-740. [DOI:10.1095/biolreprod61.3.731] [PMID]
31. Watson AJ, De Sousa P, Eaveneyn A, Barcroft LC, Natale D, Urguhard J, et al. Impact of bovine oocyte maturation media on oocyte transcript levels, blastocyst development, cell number and apoptosis. Biol Reprod 2000; 62: 353-364. [DOI:10.1095/biolreprod62.2.355] [PMID]
32. Schultz GA, Kaye PL, Mckay DJ, Johnson MH. Endogenous amino acid pool size in mouse eggs and preimplantation embryos. J Reprod Fertil 1981; 61: 387-393. [DOI:10.1530/jrf.0.0610387] [PMID]
33. Pinyopummintr T, and Bavister BD. Effects of amino acids on development in vitro of cleavage stage bovine embryos into blastocysts. Reprod Fertil Dev 1996; 8: 835-841. [DOI:10.1071/RD9960835] [PMID]
34. Lane M, and Gardner DK. Nonessential amino acids and glutamine decrease time of the first three cleavage divisions and increase compaction of mouse zygotes in vitro. J Assist Reprod Genet 1997a; 14(7): 398- 403. [DOI:10.1007/BF02766148] [PMID] [PMCID]
35. Kim JH, Niwa k, Lim JM, Okuda k. Effects of Phosphute, Energy Substrates and amino acids on development of in vitro matured, in vitro fertilized bovine oocytes in a chemically defined, protein free culture medium. Biol Reprod 1993; 48: 1320-1325. [DOI:10.1095/biolreprod48.6.1320] [PMID]
36. Lane M, and Gardner DK. Differential regulation of mouse embryo development and viability by amino acids. J Reprod Fertil Dev 1997 b; 109: 153-164. [DOI:10.1530/jrf.0.1090153] [PMID]
37. Rosenkrans CF Jr, Dans DL, Milliken G. Pig blastocyst development in vitro is affected by amino acids. J Anim Sci 1993; 67: 1503-1508. [DOI:10.2527/jas1989.6761503x] [PMID]
38. Bavister BD, McKiernan SH. Regulation of hamster embryo development in vitro by amino acids. In: Bavister BD (ed.), Preimplantation Embryo Development. New York: Springer-Verlag; 1993: 57-72. [DOI:10.1007/978-1-4613-9317-7_5]
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |




