Wed, Jul 2, 2025
[Archive]
Volume 22, Issue 2 (Febraory 2024)
IJRM 2024, 22(2): 101-110 |
Back to browse issues page
Ethics code: IR.UK.VETMED. REC. 1400.016
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Nikkhouy P, Abbasnejad M, Esmaeili-Mahani S, Kooshki R. Mode of delivery alters sensitivity to thermal and chemical stimuli in adult rats: An experimental study. IJRM 2024; 22 (2) :101-110
URL: http://ijrm.ir/article-1-3109-en.html
URL: http://ijrm.ir/article-1-3109-en.html
1- Department of Biology, Faculty of Sciences, Shahid Bahonar University of Kerman, Kerman, Iran.
2- Department of Biology, Faculty of Sciences, Shahid Bahonar University of Kerman, Kerman, Iran. Laboratory of Molecular Neuroscience, Kerman Neuroscience Research Center, Institute of Neuropharmacology, Kerman University of Medical Sciences, Kerman, Iran.
3- Department of Biology, Faculty of Sciences, Lorestan University, Khorramabad, Iran. ,kooshki.r@lu.ac.ir
2- Department of Biology, Faculty of Sciences, Shahid Bahonar University of Kerman, Kerman, Iran. Laboratory of Molecular Neuroscience, Kerman Neuroscience Research Center, Institute of Neuropharmacology, Kerman University of Medical Sciences, Kerman, Iran.
3- Department of Biology, Faculty of Sciences, Lorestan University, Khorramabad, Iran. ,
Full-Text [PDF 1598 kb]
(764 Downloads)
| Abstract (HTML) (642 Views)
Full-Text: (155 Views)
Cesarean section (C-section) primarily took place due to obstetrical complications or medical illness. However, in addition to inevitable cases, the tendency to elective primary C-sections with no apparent medical or obstetrical indication has intensely increased in recent years (1, 2).
The neonatal period is essential for the development of brain structures. Much-related evidence shows that the brain’s physiological function is affected by changes in brain development after childbirth (3, 4). Especially, the pain threshold in newborns is lower than that in adults due to the incomplete development of the sensory pathways (5).
It has been indicated that physiological function, including pain sensitivity, is somewhat affected by the type of delivery. The risk of postpartum infection and pulmonary embolism has been higher in planned C-sections than with vaginal delivery (6).
In addition, infants born by cesarean delivery have been shown to have a deficiency of immune system function (7). Instead, it has been found that the incidence of learning disabilities in cesarean children under neuraxial anesthesia is lower than vaginal delivery (8). C-section-delivered rats have been shown to have lower sensitivity to capsaicin-evoked pulpal nociception. Moreover, learning and memory performances are improved in C-sections than vaginally delivered rats (9). The mode of delivery changes the plasma concentration of opioid peptides in both mothers and infants (10).
Peripheral noxious stimulation has been shown to increase c-Fos protein expression in rats' lumbar neurons of spinal cord. The c-Fos and c-Jun proteins complex is involved in the regulation of transcription from promoters containing nuclear factor activator protein 1 (11, 12).
This study primarily evaluated whether a type of delivery, vaginal or C-section, could alter sensitivity to thermal or chemical stimuli in female rats in later life. In addition, the delivery-related changes in neuronal activity and nociception were evaluated by assessing c-Fos and c-Jun protein levels within the lumbar spinal cord of rats. Together, the aim of the current study was to evaluate the probable differences between offspring of this 2 type of delivery methods in rats.
2. Materials and Methods
The neonatal period is essential for the development of brain structures. Much-related evidence shows that the brain’s physiological function is affected by changes in brain development after childbirth (3, 4). Especially, the pain threshold in newborns is lower than that in adults due to the incomplete development of the sensory pathways (5).
It has been indicated that physiological function, including pain sensitivity, is somewhat affected by the type of delivery. The risk of postpartum infection and pulmonary embolism has been higher in planned C-sections than with vaginal delivery (6).
In addition, infants born by cesarean delivery have been shown to have a deficiency of immune system function (7). Instead, it has been found that the incidence of learning disabilities in cesarean children under neuraxial anesthesia is lower than vaginal delivery (8). C-section-delivered rats have been shown to have lower sensitivity to capsaicin-evoked pulpal nociception. Moreover, learning and memory performances are improved in C-sections than vaginally delivered rats (9). The mode of delivery changes the plasma concentration of opioid peptides in both mothers and infants (10).
Peripheral noxious stimulation has been shown to increase c-Fos protein expression in rats' lumbar neurons of spinal cord. The c-Fos and c-Jun proteins complex is involved in the regulation of transcription from promoters containing nuclear factor activator protein 1 (11, 12).
This study primarily evaluated whether a type of delivery, vaginal or C-section, could alter sensitivity to thermal or chemical stimuli in female rats in later life. In addition, the delivery-related changes in neuronal activity and nociception were evaluated by assessing c-Fos and c-Jun protein levels within the lumbar spinal cord of rats. Together, the aim of the current study was to evaluate the probable differences between offspring of this 2 type of delivery methods in rats.
2. Materials and Methods
-
- Animals
In this experimental study, 20 pregnant Wistar rats were used. The study was processed. The animals were preserved in the animal room of the Department of Biology, Shahid Bahonar University, Kerman, Iran. 9 rats gave birth vaginally, and 11 underwent C-sections. After giving birth, each mother with her offspring was kept in an individual cage until the 4th wk. Then, the female pups were moved in 4 per cage. A total of 76 female pups were involved. The pups were controlled for exclusion criteria, including abnormal behavioral performance and motor deficiency. None of the rats were excluded. The animals were housed in the same place under a 12-hr light/dark cycle with temperature-controlled at 22 ± 2oC. Food and water were available ad libitum. 56 adult female offspring (2 months, 200-220 gr) born by vaginal or cesarean procedure were used in experiments (n = 28/each). The rats were randomly divided into 8 groups (n = 7 in each group).
2.2. Cesarean procedure
The pregnant rats were transferred to the operation lab 21 days after the beginning of pregnancy. The rats were anesthetized with an intra-parietal injection of a mixture of ketamine (Alphason-Netherlands) (65 mg/kg) and xylazine (Alphason-Netherlands) (10 mg/kg); hysterectomies were performed to remove the uterine. Then, through a lateral incision of the uterine, the pups and all of their amniotic membrane were externalized. The mother's abdomen incision was closed with a sterile needle and thread stitches. Lastly, the mothers and pups were placed in a cage to feed and remove the placenta.
2.3. Formalin test
Formalin (2.5%) (Merck, USA) produced biphasic pain in the rat’s paw. Following formalin injection, the rats were placed in a 30×30×30 cm plexiglas box with a mirror on the floor at a 45o angle. The test consisted of phase 1 (0-10 min) and phase 2 (15-60 min). In each stage, cumulative time of biting and licking behaviors were recorded as nociceptive time (13). Moreover, 2 groups of rats were injected intraperitoneally with ibuprofen (10 mg/kg) 20 min before formalin injection to evaluate anti-inflammatory response.
2.4. Thermal nociception tests
The tail flick and hot plate tests were used to evaluate basal nociceptive threshold and morphine-induced thermal analgesia in rats. The morphine injection (10 mg/kg) was performed 20 min before nociceptive assessment.
2.4.1. Tail-flick test
The rats were gently controlled, and radiant heat was applied to the base of the tail. Tail-flick withdrawal latency time was verified for each animal 3 times with 2 min intervals between experiments, and the meantime was described. A cutoff time was set at 12 sec to avoid tissue injury (14).
2.4.2. Hot plate test
The hot plate test was used to measure thermal hyperalgesia. The rats were placed individually on a metal surface maintained at a constant temperature (52 ± 2oC). The latencies for licking, lifting, or jumping responses were recorded. And the mean of 3 times of the 1st animal reaction was reported as a test response. To avoid tissue damage, the cut-off time was considered 30 sec (15).
2.5. Western blot test
The lumbar portion of the rat’s spinal cord was homogenized at radioimmunoprecipitation assay buffer. Then, the sample was centrifuged at 12,000×g for 20 min at 4oC, and heated for 5 min at 100oC in Laemmli sample buffer. An equal amount (40 µg) of the sample was electrophoresed in sodium dodecyl sulfate-tris-glycine polyacrylamide gels for 90 min at 100 constant volts. After that, the proteins were transferred in an electrical field from the gel onto a polyvinylidene fluoride or polyvinylidene difluoride membrane. The transfer was conducted for 90 min at 220 constant mA. The membrane was blocked (overnight at 4oC) using a blocking buffer. Then, it was washed 3 times in tris-buffered saline with 0.1% tween® 20 detergent for 5 min each. The membrane was incubated with appropriate dilutions of primary antibodies against p-c-Fos (1/1000, Santa Crus Biotechnology, USA) and p-c-Jun (1/1000, Santa Crus Biotechnology, USA) in a blocking buffer at room temperature for 3 hr. It was then washed and incubated with conjugated secondary antibodies in blocking buffer at room temperature for 1 hr. An enhanced chemiluminescence reagent (GE Healthcare, USA) was used to expose the blots to x-ray film (Fuji, Japan). ImageJ software (version 2020) was used to evaluate the blot density (16).
2.6. Ethical considerations
All experiments followed the guidelines on ethical standards for the investigation of experimental pain in animals and were approved by the relevant Ethical Committee of Shahid Bahonar University of Kerman, Kerman, Iran (Code: IR.UK.VETMED.REC.1400.016).
2.7. Statistical analysis
The results were analyzed by Statistical Package for the Social Sciences, SPSS Inc., Chicago, Illinois, USA (version 19). The data were examined using one or two-way ANOVA followed by Tukey’s test. All values were reported as the mean ± standard error. A p < 0.05 was considered statistically significant.
3. Results
3.1. Formalin test
The animals in each group were subjected to different experiments as provided in the flow diagram (Figure 1). The time courses of formalin-induced pain in the vaginal delivery and C-section rats during a 60 min test period are shown in figure 2A. In the first phase, formalin-induced pain was significantly decreased in the C-section (2.17 ± 0.013 (group compared to vaginal delivery rats (2.26 ± 0.009) )p < 0.001(. Also, pre-treatment with intraperitoneal infusion of ibuprofen (10 mg/kg) decreased formalin-induced pain scores in C-section but not vaginal delivered rats (Figure 2B). In the second phase, the C-section group showed lower pain scores (1.45 ± 0.014) than the vaginal delivery group (1.71 ± 0.03) (p < 0.01(. Pre-treatment with ibuprofen decreased pain scores in both C-section (0.495 ± 0.011) and vaginal delivery (1.451 ± 0.014) groups (p < 0.001(. However, the ibuprofen induced more analgesic effects in the C-section than vaginal delivered rats (Figure 2B).
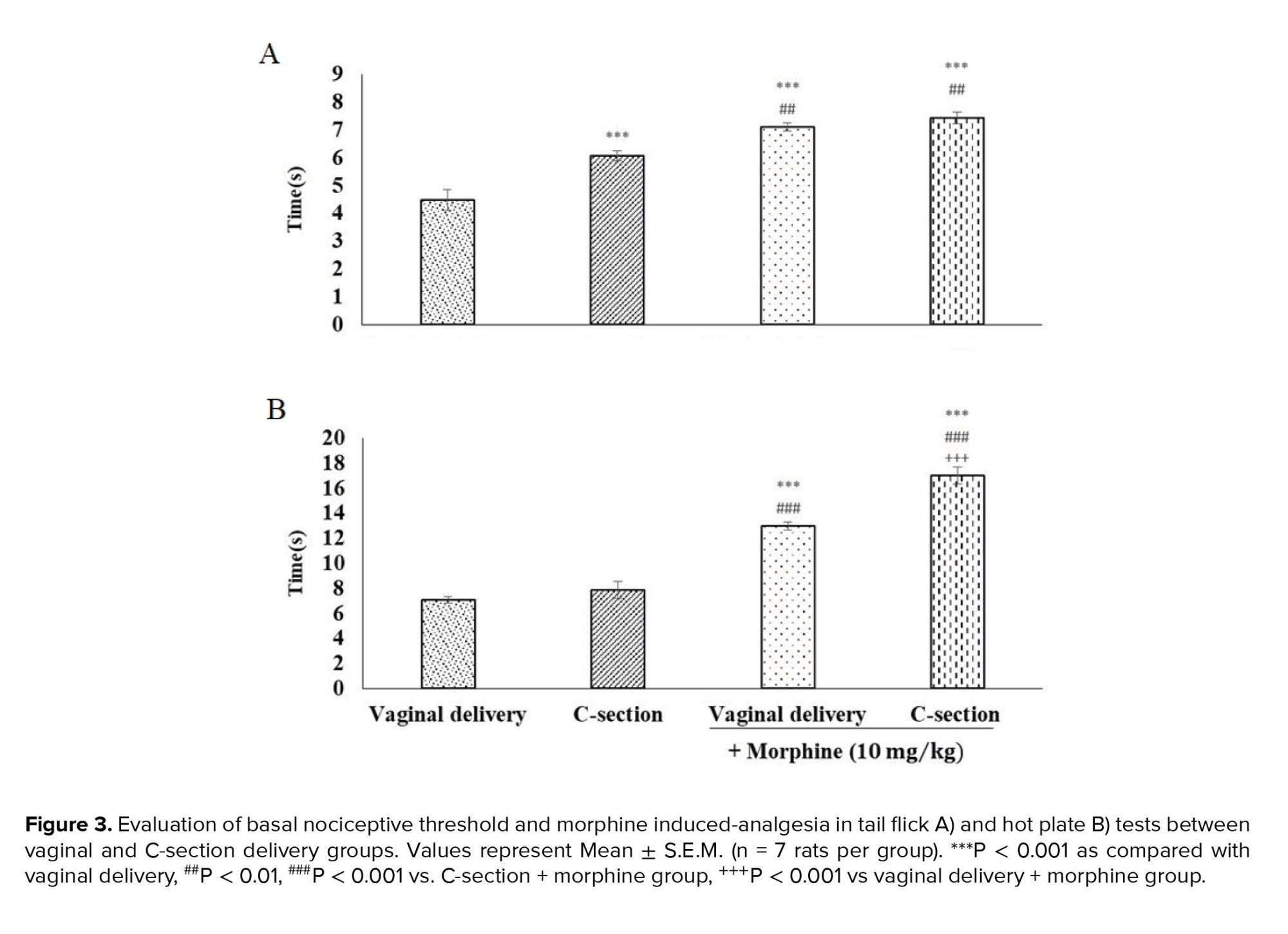
3.2. Thermal nociceptive tests
A significant difference was observed in the tail-flick nociceptive withdrawal threshold between the standard and cesarean delivery groups. As shown in figure 3A, the basal nociceptive threshold increased in the C-section group (5.85 ± 0.63) compared to the vaginal delivery group (4.6 ± 0.42) (p < 0.001). Morphine treatment (10 mg/kg, i.p.) elicited a significant increase, but at the same maximum level, in pain threshold in both experimental groups (Figure 3).
In the hot plate test, the nociceptive threshold did not alter in the C-section group compared to vaginal delivery rats. Morphine pre-treatment caused a significant increase in the baseline nociceptive thresholds in both vaginal (13.6 ± 0.307) (p < 0.05) and C-section (16.66 ± 0.95) (p < 0.001) rats. However, morphine-induced analgesia was more potent in the C-section delivered rats (p < 0.001) (Figure 3B).
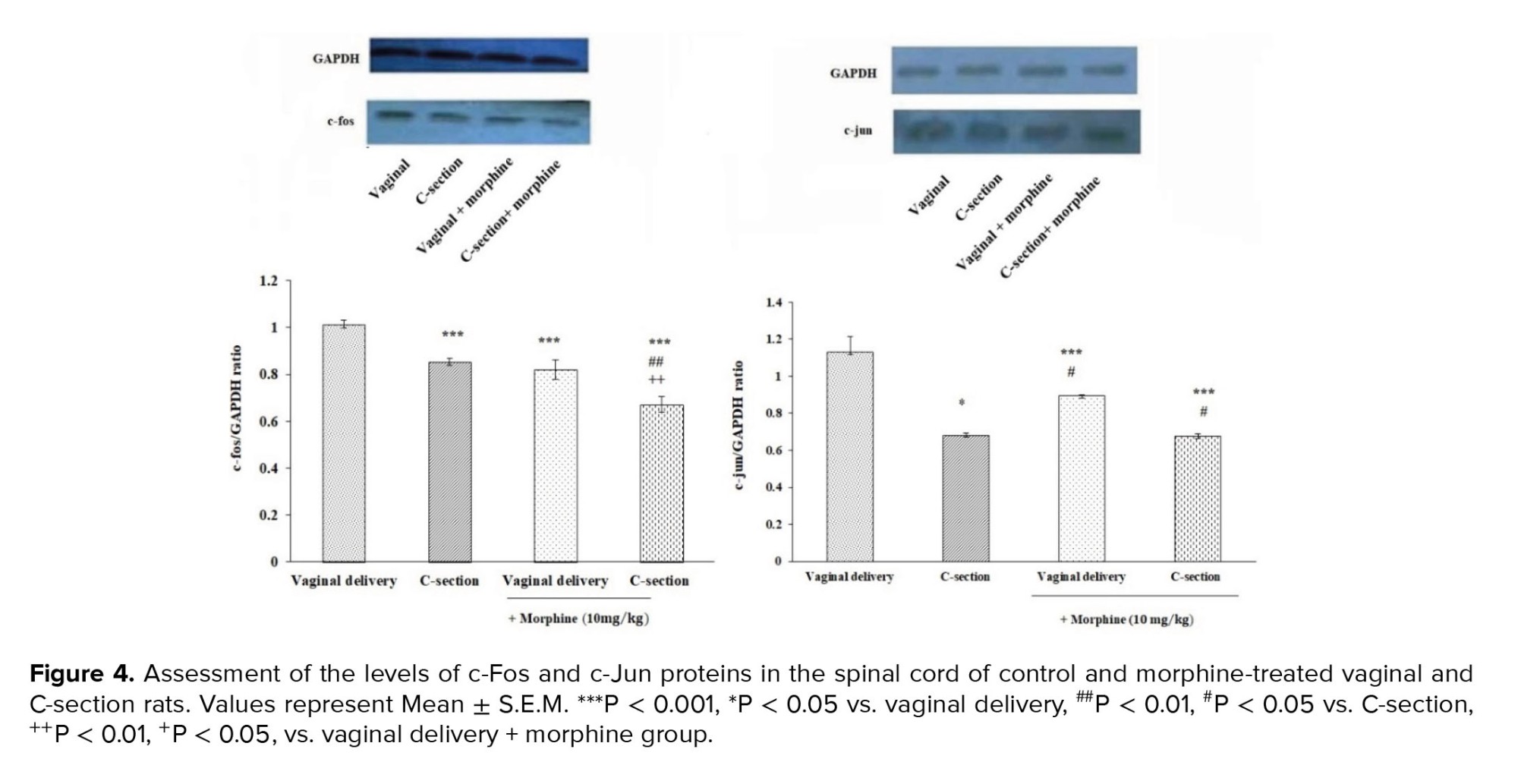
3.3. Immunoblot test
Immunoblot analysis showed a significant decrease in c-Fos and c-Jun protein values in the lumbar spinal cord of C-section delivered rats compared to vaginal delivery animals (p < 0.001). Besides, c-Fos protein levels were significantly decreased in morphine-treated C-section (0.672 ± 0.032) and vaginal delivery groups (0.821 ± 0.041) (p < 0.001). Morphine-reduced c-Fos protein value was more potent in C-section rats than vaginally delivered group. However, C-section (0.677 ± 0.012) and vaginal delivered (0.895 ± 0.04) rats pretreated with morphine did not show significant differences in the c-Jun protein level (Figure 4).


4. Discussion
The present study results indicated that basal sensitivity to thermal and inflammatory stimuli is decreased in the female adult rats born by C-section compared to vaginally delivered rats. Also, morphine-induced thermal analgesia was more noticeable in the C-section group. Moreover, the spinal cord level of c-Fos protein increased in normal and morphine-treated C-section rats compared to the vaginal delivery group. The level of c-Jun decreased in the control group but not in the morphine-treated C-section group compared with vaginal birth rats.
This study showed an association between mode of delivery and changes in pain perception in adult female offspring rats. Some previous studies are available for comparison with the present results. In contrast, capsaicin-evoked pulpal nociception has been shown to decrease in C-section male rats compared to vaginally delivered animals (9). However, a human study demonstrated that spontaneous pain behaviors and vocalization are reduced in infants born vaginally compared to C-section (17). Besides, the recent prospective cohort study revealed no significant differences between vaginal and C-section newborns in acute pain sensation (18).
In this study, the baseline nociceptive threshed and morphine-induced analgesia increased in female C-section rats. The exact mechanism(s) of lower pain sensitivity in C-section rats is not clear. However, differences in stress hormones during childbirth might be considered as one of many possible involving mechanisms. Vaginal delivery as major stress is associated with extensive changes in the number of stress hormones. It has been reported that the stress and pain response of neonates after vaginal birth by vacuum extraction is increased compared to C-section delivery (19). Exposure to endocrine stressors during vaginal childbirth alters the activity of the hypothalamus-pituitary axis in the later life (20). It may increase the risk of pathophysiologic situations such as high blood pressure and metabolic syndromes in the postnatal period.
The level of stress hormones, including adrenocorticotropin and cortisol, in mothers and neonates is significantly higher following spontaneous vaginal delivery compared to C-section birth (21). It reinforces the hypothesis that exposure to different levels of endogenous stressors during vaginal or C-section delivery may be reflected by a long-term effect on hypothalamus-pituitary axis function. It may alter emotional behaviors and stress responsiveness. Cesarean deliveries have been associated with many changes in the physiology and behavior of the offspring (22). Interestingly recent data findings show sex-dependent differences in C-section offspring mice (23).
c-Fos, is a specific marker of neuronal activity that is expressed in nociceptive pathways rapidly after noxious stimuli. It forms a complex with Jun, another oncogene protein, making connections with the activator protein 1 DNA site to induce gene transcription. c-Fos expression after noxious stimulation and tissue injury has been associated with activation of intracellular pathways involved in pain sensitization including PERK/MAPK (24). In this study, to endorse the results of behavioral tests, alteration in the c-Fos and c-Jun proteins were evaluated in the spinal cord of rats. In line with behavioral data, the levels of c-Fos and c-Jun proteins were decreased in control and C-section morphine-treated rats in comparison to vaginally delivered rats. In a related study, c-Fos expression has been increased in the piriform cortex, caudate nucleus, putamen and cerebellar cortex in vaginally delivered compared to C-section born mice (25). These data show mode of delivery interferences on neuronal exhibition as well as nociceptive development.
The circulating concentrations of cytokines like tumor necrosis factors alpha and interleukin 6 in mothers before delivery and infants’ in the first 4 days of life have increased in vaginal delivery compared to elective C-sections (26). The human study on maternal and fetal plasma prostaglandin levels showed that the plasma concentration of prostaglandin F is significantly elevated at vaginal delivery compared to C-section (27). The positive correlation between nociceptive and pro-inflammatory responses raised the possibility that C-section rats’ low sensitivity to nociceptive impulses could attenuate the inflammatory pathways.
Optimum oxygen supply is essential during parturition. It has been shown that oxygen deprivation during prolonged vaginal delivery increases hypoxia-ischemia-induced brain deficit that could be associated with long-lasting neurological inadequately (28, 29).
It has been indicated that hypoxia, even for a short time, may induce oxidative stress damage (30). Studies have shown that oxidative stress plays a vital role in the induction and preserves various pain conditions (31-33). The levels of antioxidant enzymes have been increased in umbilical cord blood of neonates born by elective C-sections compared to those of vaginally born neonates (21). So, mode of delivery-induced changes in pain sensitivity may be somewhat related to alteration in antioxidant enzyme activities. However, further studies are still required to approve the association between delivery mode and the variation in antioxidant enzymes.
5. Conclusion
Totally, the present study provides some evidences to support intervention of delivery mode on development of nociceptive behavior and alteration of basal neuronal activities in adults’ female rats. However, further research is still required to clarify the intricate underling mechanism(s).
Data availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author contributions
All the authors had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Mehdi Abbasnejad, acquisition, analysis, or interpretation of data: Razieh Kooshki, Parastoo Nikkhouy, drafting of the manuscript: Parastoo Nikkhouy, critical revision of the manuscript for important intellectual content: all authors, statistical analysis: Saeed Esmaeili-Mahani, supervision: Razieh Kooshki.
Acknowledgments
The authors would like to thank Shahid Bahonar University, Kerman, Iran for financial support.
Conflict of Interest
The authors declare that there is no conflict of interest.
2.2. Cesarean procedure
The pregnant rats were transferred to the operation lab 21 days after the beginning of pregnancy. The rats were anesthetized with an intra-parietal injection of a mixture of ketamine (Alphason-Netherlands) (65 mg/kg) and xylazine (Alphason-Netherlands) (10 mg/kg); hysterectomies were performed to remove the uterine. Then, through a lateral incision of the uterine, the pups and all of their amniotic membrane were externalized. The mother's abdomen incision was closed with a sterile needle and thread stitches. Lastly, the mothers and pups were placed in a cage to feed and remove the placenta.
2.3. Formalin test
Formalin (2.5%) (Merck, USA) produced biphasic pain in the rat’s paw. Following formalin injection, the rats were placed in a 30×30×30 cm plexiglas box with a mirror on the floor at a 45o angle. The test consisted of phase 1 (0-10 min) and phase 2 (15-60 min). In each stage, cumulative time of biting and licking behaviors were recorded as nociceptive time (13). Moreover, 2 groups of rats were injected intraperitoneally with ibuprofen (10 mg/kg) 20 min before formalin injection to evaluate anti-inflammatory response.
2.4. Thermal nociception tests
The tail flick and hot plate tests were used to evaluate basal nociceptive threshold and morphine-induced thermal analgesia in rats. The morphine injection (10 mg/kg) was performed 20 min before nociceptive assessment.
2.4.1. Tail-flick test
The rats were gently controlled, and radiant heat was applied to the base of the tail. Tail-flick withdrawal latency time was verified for each animal 3 times with 2 min intervals between experiments, and the meantime was described. A cutoff time was set at 12 sec to avoid tissue injury (14).
2.4.2. Hot plate test
The hot plate test was used to measure thermal hyperalgesia. The rats were placed individually on a metal surface maintained at a constant temperature (52 ± 2oC). The latencies for licking, lifting, or jumping responses were recorded. And the mean of 3 times of the 1st animal reaction was reported as a test response. To avoid tissue damage, the cut-off time was considered 30 sec (15).
2.5. Western blot test
The lumbar portion of the rat’s spinal cord was homogenized at radioimmunoprecipitation assay buffer. Then, the sample was centrifuged at 12,000×g for 20 min at 4oC, and heated for 5 min at 100oC in Laemmli sample buffer. An equal amount (40 µg) of the sample was electrophoresed in sodium dodecyl sulfate-tris-glycine polyacrylamide gels for 90 min at 100 constant volts. After that, the proteins were transferred in an electrical field from the gel onto a polyvinylidene fluoride or polyvinylidene difluoride membrane. The transfer was conducted for 90 min at 220 constant mA. The membrane was blocked (overnight at 4oC) using a blocking buffer. Then, it was washed 3 times in tris-buffered saline with 0.1% tween® 20 detergent for 5 min each. The membrane was incubated with appropriate dilutions of primary antibodies against p-c-Fos (1/1000, Santa Crus Biotechnology, USA) and p-c-Jun (1/1000, Santa Crus Biotechnology, USA) in a blocking buffer at room temperature for 3 hr. It was then washed and incubated with conjugated secondary antibodies in blocking buffer at room temperature for 1 hr. An enhanced chemiluminescence reagent (GE Healthcare, USA) was used to expose the blots to x-ray film (Fuji, Japan). ImageJ software (version 2020) was used to evaluate the blot density (16).
2.6. Ethical considerations
All experiments followed the guidelines on ethical standards for the investigation of experimental pain in animals and were approved by the relevant Ethical Committee of Shahid Bahonar University of Kerman, Kerman, Iran (Code: IR.UK.VETMED.REC.1400.016).
2.7. Statistical analysis
The results were analyzed by Statistical Package for the Social Sciences, SPSS Inc., Chicago, Illinois, USA (version 19). The data were examined using one or two-way ANOVA followed by Tukey’s test. All values were reported as the mean ± standard error. A p < 0.05 was considered statistically significant.
3. Results
3.1. Formalin test
The animals in each group were subjected to different experiments as provided in the flow diagram (Figure 1). The time courses of formalin-induced pain in the vaginal delivery and C-section rats during a 60 min test period are shown in figure 2A. In the first phase, formalin-induced pain was significantly decreased in the C-section (2.17 ± 0.013 (group compared to vaginal delivery rats (2.26 ± 0.009) )p < 0.001(. Also, pre-treatment with intraperitoneal infusion of ibuprofen (10 mg/kg) decreased formalin-induced pain scores in C-section but not vaginal delivered rats (Figure 2B). In the second phase, the C-section group showed lower pain scores (1.45 ± 0.014) than the vaginal delivery group (1.71 ± 0.03) (p < 0.01(. Pre-treatment with ibuprofen decreased pain scores in both C-section (0.495 ± 0.011) and vaginal delivery (1.451 ± 0.014) groups (p < 0.001(. However, the ibuprofen induced more analgesic effects in the C-section than vaginal delivered rats (Figure 2B).
3.2. Thermal nociceptive tests
A significant difference was observed in the tail-flick nociceptive withdrawal threshold between the standard and cesarean delivery groups. As shown in figure 3A, the basal nociceptive threshold increased in the C-section group (5.85 ± 0.63) compared to the vaginal delivery group (4.6 ± 0.42) (p < 0.001). Morphine treatment (10 mg/kg, i.p.) elicited a significant increase, but at the same maximum level, in pain threshold in both experimental groups (Figure 3).
In the hot plate test, the nociceptive threshold did not alter in the C-section group compared to vaginal delivery rats. Morphine pre-treatment caused a significant increase in the baseline nociceptive thresholds in both vaginal (13.6 ± 0.307) (p < 0.05) and C-section (16.66 ± 0.95) (p < 0.001) rats. However, morphine-induced analgesia was more potent in the C-section delivered rats (p < 0.001) (Figure 3B).
3.3. Immunoblot test
Immunoblot analysis showed a significant decrease in c-Fos and c-Jun protein values in the lumbar spinal cord of C-section delivered rats compared to vaginal delivery animals (p < 0.001). Besides, c-Fos protein levels were significantly decreased in morphine-treated C-section (0.672 ± 0.032) and vaginal delivery groups (0.821 ± 0.041) (p < 0.001). Morphine-reduced c-Fos protein value was more potent in C-section rats than vaginally delivered group. However, C-section (0.677 ± 0.012) and vaginal delivered (0.895 ± 0.04) rats pretreated with morphine did not show significant differences in the c-Jun protein level (Figure 4).




4. Discussion
The present study results indicated that basal sensitivity to thermal and inflammatory stimuli is decreased in the female adult rats born by C-section compared to vaginally delivered rats. Also, morphine-induced thermal analgesia was more noticeable in the C-section group. Moreover, the spinal cord level of c-Fos protein increased in normal and morphine-treated C-section rats compared to the vaginal delivery group. The level of c-Jun decreased in the control group but not in the morphine-treated C-section group compared with vaginal birth rats.
This study showed an association between mode of delivery and changes in pain perception in adult female offspring rats. Some previous studies are available for comparison with the present results. In contrast, capsaicin-evoked pulpal nociception has been shown to decrease in C-section male rats compared to vaginally delivered animals (9). However, a human study demonstrated that spontaneous pain behaviors and vocalization are reduced in infants born vaginally compared to C-section (17). Besides, the recent prospective cohort study revealed no significant differences between vaginal and C-section newborns in acute pain sensation (18).
In this study, the baseline nociceptive threshed and morphine-induced analgesia increased in female C-section rats. The exact mechanism(s) of lower pain sensitivity in C-section rats is not clear. However, differences in stress hormones during childbirth might be considered as one of many possible involving mechanisms. Vaginal delivery as major stress is associated with extensive changes in the number of stress hormones. It has been reported that the stress and pain response of neonates after vaginal birth by vacuum extraction is increased compared to C-section delivery (19). Exposure to endocrine stressors during vaginal childbirth alters the activity of the hypothalamus-pituitary axis in the later life (20). It may increase the risk of pathophysiologic situations such as high blood pressure and metabolic syndromes in the postnatal period.
The level of stress hormones, including adrenocorticotropin and cortisol, in mothers and neonates is significantly higher following spontaneous vaginal delivery compared to C-section birth (21). It reinforces the hypothesis that exposure to different levels of endogenous stressors during vaginal or C-section delivery may be reflected by a long-term effect on hypothalamus-pituitary axis function. It may alter emotional behaviors and stress responsiveness. Cesarean deliveries have been associated with many changes in the physiology and behavior of the offspring (22). Interestingly recent data findings show sex-dependent differences in C-section offspring mice (23).
c-Fos, is a specific marker of neuronal activity that is expressed in nociceptive pathways rapidly after noxious stimuli. It forms a complex with Jun, another oncogene protein, making connections with the activator protein 1 DNA site to induce gene transcription. c-Fos expression after noxious stimulation and tissue injury has been associated with activation of intracellular pathways involved in pain sensitization including PERK/MAPK (24). In this study, to endorse the results of behavioral tests, alteration in the c-Fos and c-Jun proteins were evaluated in the spinal cord of rats. In line with behavioral data, the levels of c-Fos and c-Jun proteins were decreased in control and C-section morphine-treated rats in comparison to vaginally delivered rats. In a related study, c-Fos expression has been increased in the piriform cortex, caudate nucleus, putamen and cerebellar cortex in vaginally delivered compared to C-section born mice (25). These data show mode of delivery interferences on neuronal exhibition as well as nociceptive development.
The circulating concentrations of cytokines like tumor necrosis factors alpha and interleukin 6 in mothers before delivery and infants’ in the first 4 days of life have increased in vaginal delivery compared to elective C-sections (26). The human study on maternal and fetal plasma prostaglandin levels showed that the plasma concentration of prostaglandin F is significantly elevated at vaginal delivery compared to C-section (27). The positive correlation between nociceptive and pro-inflammatory responses raised the possibility that C-section rats’ low sensitivity to nociceptive impulses could attenuate the inflammatory pathways.
Optimum oxygen supply is essential during parturition. It has been shown that oxygen deprivation during prolonged vaginal delivery increases hypoxia-ischemia-induced brain deficit that could be associated with long-lasting neurological inadequately (28, 29).
It has been indicated that hypoxia, even for a short time, may induce oxidative stress damage (30). Studies have shown that oxidative stress plays a vital role in the induction and preserves various pain conditions (31-33). The levels of antioxidant enzymes have been increased in umbilical cord blood of neonates born by elective C-sections compared to those of vaginally born neonates (21). So, mode of delivery-induced changes in pain sensitivity may be somewhat related to alteration in antioxidant enzyme activities. However, further studies are still required to approve the association between delivery mode and the variation in antioxidant enzymes.
5. Conclusion
Totally, the present study provides some evidences to support intervention of delivery mode on development of nociceptive behavior and alteration of basal neuronal activities in adults’ female rats. However, further research is still required to clarify the intricate underling mechanism(s).
Data availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author contributions
All the authors had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Mehdi Abbasnejad, acquisition, analysis, or interpretation of data: Razieh Kooshki, Parastoo Nikkhouy, drafting of the manuscript: Parastoo Nikkhouy, critical revision of the manuscript for important intellectual content: all authors, statistical analysis: Saeed Esmaeili-Mahani, supervision: Razieh Kooshki.
Acknowledgments
The authors would like to thank Shahid Bahonar University, Kerman, Iran for financial support.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Biology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







