Sun, Apr 12, 2026
[Archive]
Volume 23, Issue 9 (September 2025)
IJRM 2025, 23(9): 689-700 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Ebrahimian A, Iravani M, Faal Siahkal S. Effects of World Health Organization labor care guide on maternal and neonatal outcomes: A systematic review and meta-analysis. IJRM 2025; 23 (9) :689-700
URL: http://ijrm.ir/article-1-3459-en.html
URL: http://ijrm.ir/article-1-3459-en.html
1- Reproductive Sciences and Sexual Health Research Center, Faculty of Nursing and Midwifery, Isfahan University of Medical Sciences, Isfahan, Iran.
2- Reproductive Health Promotion Research Center, Department of Midwifery and Reproductive Health, Nursing and Midwifery School, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran. ,iravani-m@ajums.ac.ir
3- Department of Midwifery, Mara.C., Islamic Azad University, Marand, Iran.
2- Reproductive Health Promotion Research Center, Department of Midwifery and Reproductive Health, Nursing and Midwifery School, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran. ,
3- Department of Midwifery, Mara.C., Islamic Azad University, Marand, Iran.
Keywords: World Health Organization, Labor care guide, Maternal outcomes, Neonatal outcomes, Systematic review.
Full-Text [PDF 641 kb]
(2226 Downloads)
| Abstract (HTML) (688 Views)
Full-Text: (328 Views)
1. Introduction
Ensuring safe delivery remains one of the most fundamental missions of healthcare systems worldwide (1). Maternal and neonatal outcomes are directly influenced by the quality of care provided during labor, which includes continuous monitoring of labor progress, timely recognition of complications, and appropriate management strategies (2). Optimal intrapartum care not only reduces preventable maternal and neonatal morbidity and mortality but also improves the overall childbirth experience for women and families. The World Health Organization (WHO) emphasizes that cesarean section should be performed only when medically indicated. While the expected population-based cesarean section rate is estimated at 10-15%, a steady and often alarming increase has been documented globally over the past few decades (3). Rising cesarean rates pose significant public health concerns, particularly because repeated cesarean deliveries are associated with long-term complications, including abnormal placentation, higher risks of hemorrhage, and adverse maternal and neonatal outcomes (4). WHO emphasizes that cesarean rates above 15% are unjustified and has issued guidelines for the safe and rational use of cesarean section (5).
Among the leading contributors to primary cesarean section, especially in nulliparous women, is labor dystocia or arrest of labor progression (6). Accurate diagnosis of labor protraction and decisions to intervene remain highly complex and sometimes controversial. According to the 1994 WHO partograph, cervical dilation progress of < 1 cm per hour over a minimum of 4 hr was defined as a dilatation disorder (7). However, the clinical applicability of this threshold has been debated for decades. Misclassification of slow but physiologically normal labor as dystocia may contribute to unnecessary cesarean deliveries. Recognizing these limitations, WHO revised its recommendations in 2020 and introduced the labor care guide (LCG), designed to replace the traditional partograph and provide clinicians with a more nuanced, evidence-based approach to labor monitoring and decision-making (8).
The LCG differs from the traditional partograph in key aspects. Whereas the partograph considered active labor to begin at 4 cm of cervical dilation, the LCG shifts this threshold to 5 cm, based on recent evidence regarding labor patterns. Unlike the partograph, which ends at full dilation, the LCG incorporates systematic monitoring of the second stage of labor and provides time-based “alert values” for both stages. In the first stage, remaining at 5 cm for 6 hr or longer is considered abnormal, with similar thresholds for 6-9 cm: ≥ 5 hr at 6 cm, ≥ 3 hr at 7 cm, ≥ 2.5 hr at 8 cm, and ≥ 2 hr at 9 cm (9). In the second stage, prolonged labor is over 3 hr for primiparous and 2 hr for multiparous women. Cervical dilation varies, and slower progress can still result in uncomplicated vaginal delivery. Studies comparing WHO partograph and Zhang’s guidelines show differences in dystocia diagnosis and management, though clinical impact remains uncertain (10, 11).
Given these debates and the need to limit unnecessary interventions, this study aims to evaluate the impact of the WHO LCG on maternal and neonatal outcomes.
2. Materials and Methods
2.1. Formulation of the research question
The research question for this meta-analysis was structured using the PICO framework to ensure clarity and precision in identifying relevant studies. The population (P) consisted of women in labor; the intervention (I) involved the implementation of the WHO LCG (WHO LCG); the comparison (C) was conventional intrapartum care not based on WHO LCG; and the outcomes (O) included maternal and neonatal indicators such as duration of labor, mode of delivery, maternal complications, neonatal Apgar scores, and admission to the neonatal intensive care unit (NICU).
2.2. Study design
This study is a meta-analysis of clinical trials conducted to evaluate the impact of implementing (WHO LCG) on maternal and neonatal outcomes. The study design and reporting adhered to the PRISMA guidelines (12).
2.3. Search strategy
A comprehensive and systematic search was conducted to identify relevant studies examining the impact of the WHO LCG on maternal and neonatal outcomes. The literature search was performed in the electronic databases PubMed, Scopus, Google Scholar, and Embase for articles published up to September 2024, without any language restrictions.
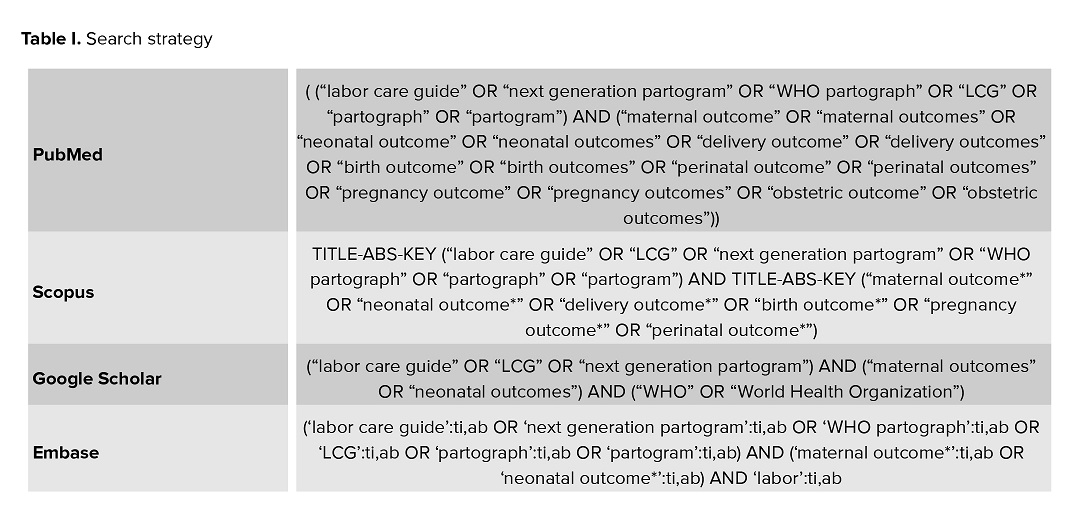
The search strategy combined both controlled vocabulary (Medical Subject Headings-MeSH) and relevant free-text keywords. Key terms included: “WHO Labor Care Guide”, “Labor Care Guide”, “next-generation partograph”, “labor outcomes”, “maternal outcomes”, “neonatal outcomes”, and related synonyms. Keywords were selected based on MeSH terms and were refined through consultation with domain experts in maternal health and obstetrics to enhance the sensitivity and specificity of the search. Boolean operators such as AND and OR were used to logically structure and expand the search queries. An example of the full search strategy is presented in table I. In addition to database searching, the reference lists of all included studies were manually reviewed to identify any potentially relevant articles that may have been missed in the electronic search.
2.4. Inclusion and exclusion criteria
2.4.1. Inclusion criteria
Randomized controlled trials or quasi-experimental studies, studies comparing the LCG intervention with standard intrapartum care, studies reporting at least one maternal or neonatal outcome, and full-text articles published in peer-reviewed journals.
2.4.2. Exclusion criteria
Protocols, editorials, qualitative studies, short communications, conference abstracts, studies lacking a control group or quantitative outcome data, and studies involving women with current high-risk pregnancies or a history of high-risk obstetric complications.
2.5. Study selection
2 independent reviewers conducted screening of titles and abstracts following the removal of duplicates. Full-text articles meeting potential eligibility were further assessed in line with the inclusion and exclusion criteria. Any disagreements were settled through discussion or, when necessary, by involving a third reviewer.
2.6. Data extraction
Data were independently extracted by 2 authors using a standardized form, including study characteristics (author, year, country, study design), sample size in intervention and control groups, maternal outcomes, neonatal outcomes, and effect measures (odds ratios, mean differences with 95% confidence intervals).
2.7. Data items
2.7.1. Types of participants
Women in labor, including those undergoing spontaneous or induced labor, were considered eligible for inclusion. Studies involving women with current high-risk pregnancies or a history of high-risk obstetric complications were excluded.
2.7.2. Types of interventions
Implementation of the WHO LCG during intrapartum care. This included studies explicitly mentioning the use of WHO LCG or next-generation partograph aligned with the WHO LCG guidelines.
2.7.3. Outcome measures
Studies were required to report at least one maternal or neonatal outcome. Maternal outcomes included mode of delivery, duration of the second stage of labor, severe perineal trauma, augmentation of labor, and postpartum hemorrhage. Neonatal outcomes included stillbirth, Apgar score, and admission to the NICU.
2.8. Quality assessments
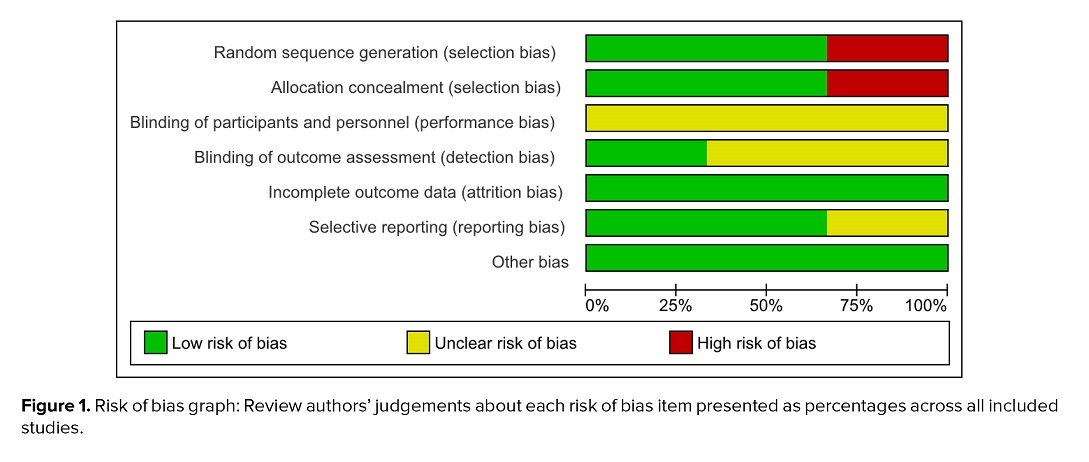
The methodological quality of the studies included in this systematic review and meta-analysis was independently assessed by 2 reviewers using the Cochrane risk of bias tool within review manager. 7 distinct domains are assessed using this tool, including random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and additional potential sources of bias (13).
Each study was categorized as having a low, moderate, or high risk of bias in each domain. In general, the included studies exhibited acceptable methodological rigor. While certain domains, such as blinding, demonstrated moderate to high risk of bias in some studies, other domains, particularly incomplete outcome data and other sources of bias, were predominantly rated as low risk. A summary of the risk of bias assessment is presented in figure 1, depicting the distribution of bias levels across all included studies and assessed domains.
2.9. Data analysis
Data analysis for this meta-analysis was conducted using RevMan version 5.3 software. For dichotomous outcomes, odds ratios were used, while for continuous outcomes, mean differences were applied. Statistical heterogeneity was assessed using the Chi-square test and the I² statistic, and a random-effects model was employed for all analyses. Forest plots were generated to visually display effect sizes and confidence intervals, and sensitivity analyses were conducted by removing studies with a high risk of bias. Due to the small number of studies, publication bias assessment was not performed, and a p < 0.05 was considered statistically significant. All data were entered and verified independently by 2 reviewers to ensure accuracy and consistency.
3. Results
3.1. Study selection and characteristics
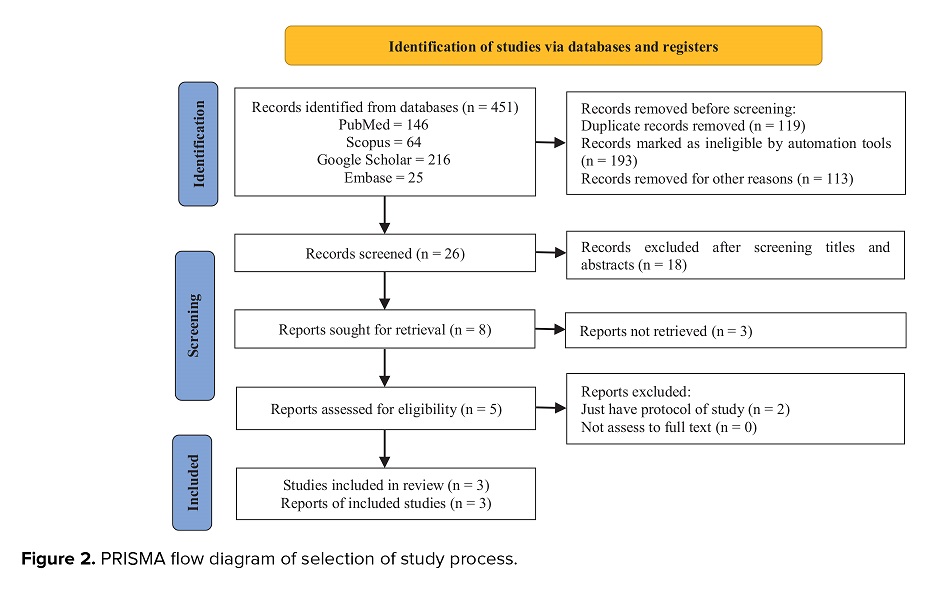
In the initial search, 451 articles were retrieved. Following the removal of duplicates and obviously irrelevant studies, 26 articles were screened using their titles and abstracts. Of these, 18 articles were excluded for specific reasons, including lack of an intervention group, nonclinical trial design, insufficient data, or irrelevance to the study objectives. 8 articles were selected for full-text assessment. After applying the inclusion criteria, 5 studies were further excluded. Ultimately, 3 clinical trial studies met all eligibility criteria and were included in the final analysis. The PRISMA flowchart illustrating the study selection process is shown in figure 2.
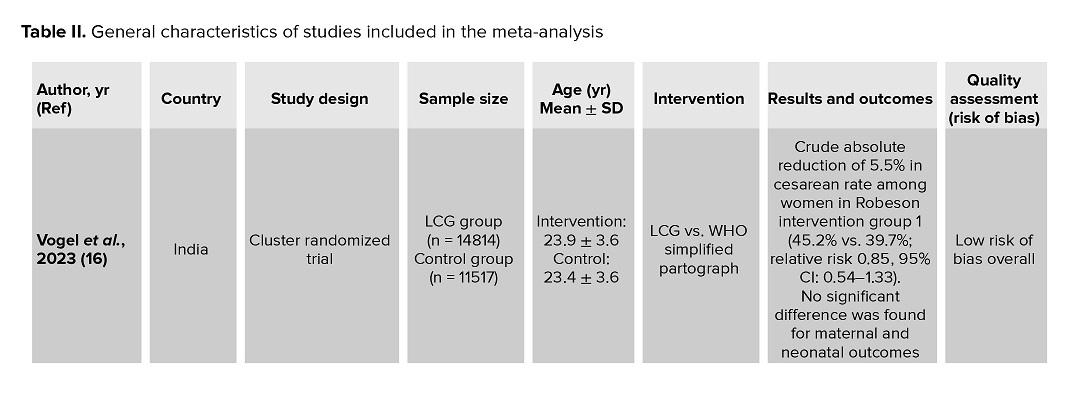
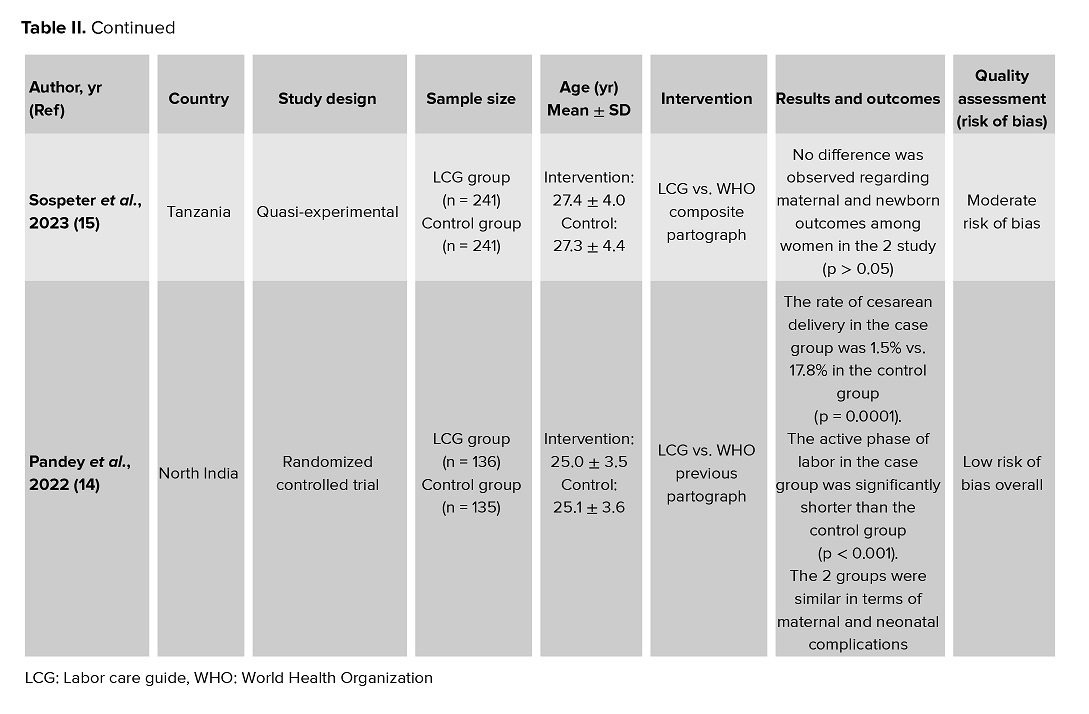
All 3 studies were conducted in public hospitals and involved staff training on the use of the LCG. 2 studies, included low-risk women with term pregnancies (37-40 wk gestation), cephalic presentation, and spontaneous labor onset, while women with high-risk pregnancies or prior high-risk obstetric histories were excluded (14, 15). Another study included pregnancies beyond 20 wk and applied the Robson classification to both intervention and control groups (16).
For the Vogel study, data from Robson groups 1 and 3 were included, representing nulliparous and multiparous women with single cephalic pregnancies, spontaneous labor, and term gestation (≥ 37 wk). These groups were selected to evaluate the intervention’s impact within a homogeneous, low-risk population for a more accurate assessment.
In all trials, the active phase of labor was defined as starting at 5 cm cervical dilation in the LCG group and at 3 cm in the control group (using the previous partograph). Maternal and fetal parameters were monitored according to LCG criteria and recorded on the LCG partograph. Cervical dilation alert thresholds and second-stage labor durations were followed based on WHO recommendations (Table II).
3.2. Maternal outcomes
3.2.1. Caesarean section rate
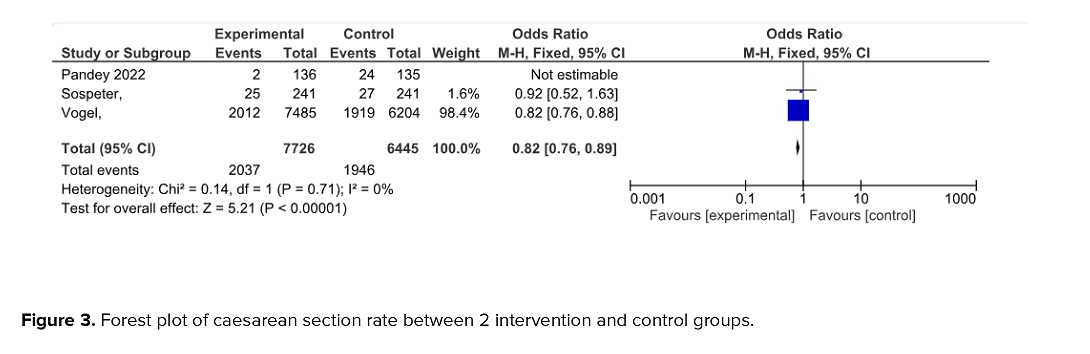
The meta-analysis showed a statistically significant reduction in the caesarean section rate in the intervention group compared to the control group (OR = 0.82; 95% CI: 0.76-0.89; Figure 3).
3.2.2. Duration of the second stage of labor
The meta-analysis revealed that the intervention group experienced a notable reduction in the second stage of labor, showing a mean difference of -5.28 min (95% CI: -7.25 to -3.32), as presented in figure 4.
3.2.3. Severe perineal trauma and postpartum hemorrhage
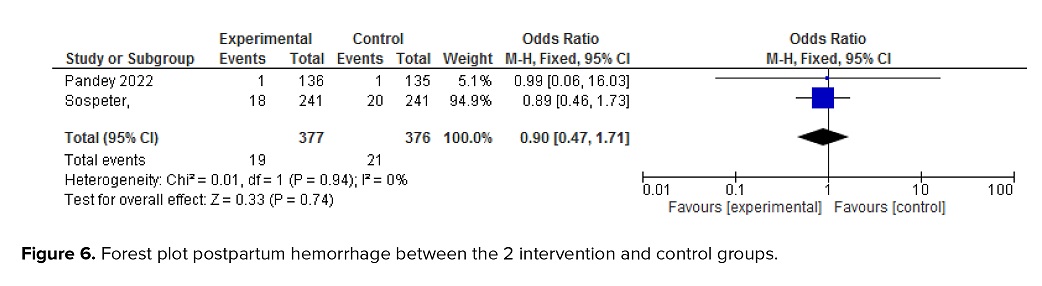
The meta-analysis indicated that there were no significant differences between the intervention and control groups in terms of severe perineal trauma (OR = 0.61; 95% CI: 0.35-1.04; Figure 5) or postpartum hemorrhage (OR = 0.90; 95% CI: 0.47-1.71; Figure 6).
3.2.4. Augmentation of labor
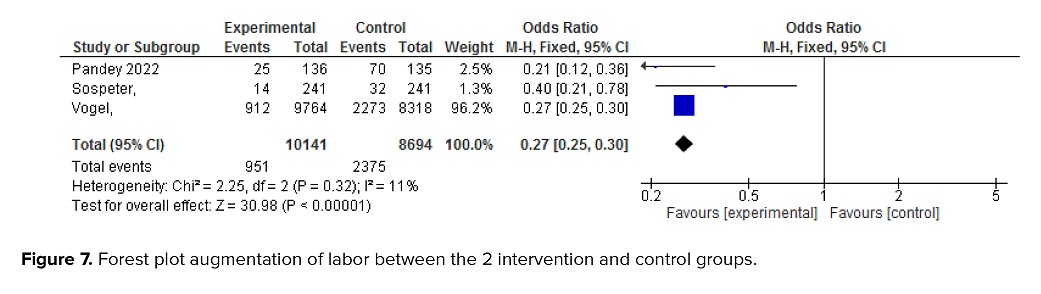
The meta-analysis demonstrated a significant reduction in the rate of labor augmentation in the intervention group, with an odds ratio of 0.27 (95% CI: 0.25-0.30) as shown in figure 7.
3.3. Neonatal outcomes
3.3.1. Stillbirth
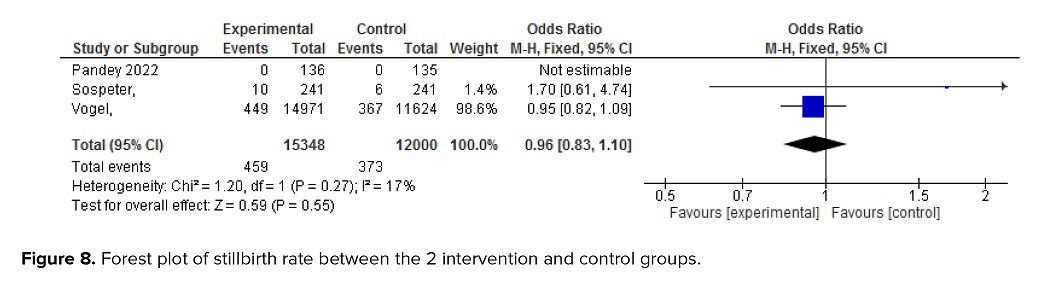
The meta-analysis indicated no significant difference in stillbirth rates between the intervention and control groups (OR = 0.96; 95% CI: 0.83-1.10; Figure 8).
3.3.2. Apgar score < 7 at 5 min
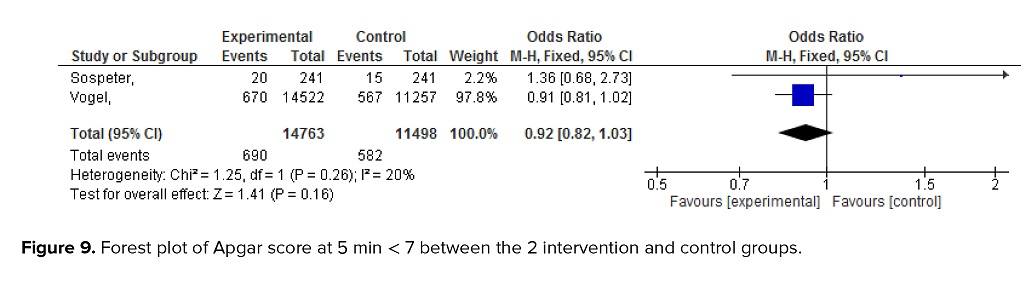
The meta-analysis revealed no significant difference in the rate of Apgar score < 7 at 5 min between the intervention and control groups (OR = 0.92; 95% CI: 0.82-1.03; Figure 9).
3.3.3. NICU admission
The meta-analysis found no statistically significant difference in NICU admissions after birth between the intervention and control groups (OR = 0.58; 95% CI: 0.30-1.12; Figure 10).



4. Discussion
This meta-analysis aimed to assess the impact of implementing the LCG on maternal and neonatal outcomes. The findings demonstrate that LCG use is associated with a significant reduction in cesarean section rates, the duration of the second stage of labor, and the need for labor augmentation, without increasing adverse maternal or neonatal events.
The observed reduction in cesarean rates aligns with the WHO objective of minimizing unnecessary obstetric interventions, underscoring the clinical value of the LCG in optimizing intrapartum care. Evidence suggests that the LCG promotes a more patient-centered approach by extending active labor thresholds and reducing premature decisions regarding cesarean delivery (17). This meta-analysis observed a shorter second stage of labor, which may be explained by more consistent and timely clinical decisions guided by the structured LCG criteria. This aligns with the WHO revision of labor definitions, which increased the threshold for the onset of active labor from 3-4 cm to 5 cm of cervical dilation to prevent premature diagnoses of labor arrest (18).
Similarly, the decreased need for labor augmentation suggests that the LCG can minimize unnecessary pharmacological interventions by providing clearer parameters for assessing labor progression. In addition, reviews indicate that many cesarean deliveries result from mistimed interventions (19). By offering structured guidance, the LCG may help clinicians make more accurate decisions, thereby reducing avoidable cesarean sections.
A key feature of the LCG is the raised cervical dilation threshold for identifying active labor, previously set at 3 cm in the modified partograph (20) and now at 5 cm in the LCG (21). The LCG allows for a maximum of approximately 18.5 hr from 5 cm dilation to full cervical dilation, effectively distinguishing abnormal from normal labor. This extended timeframe also helps reduce excessive interventions during labor.
While the thresholds in the LCG are physiologically appropriate, they are more complex to interpret than the standard 1 cm/hr rule during active labor (22), and the guidelines are primarily intended for low-risk pregnancies (23). Consequently, it remains crucial to identify abnormal labor patterns, such as slow progress, and to develop appropriate management plans -whether intervention or continued monitoring- for both clinical and medico-legal purposes (24).
Although there have been concerns that extended labor monitoring might increase maternal complications, our findings showed no significant difference in severe perineal trauma or postpartum hemorrhage, supporting the safety of this approach. This aligns with a WHO-led multicenter feasibility study, which reported that the LCG is both acceptable to clinicians and does not elevate maternal risk when properly implemented (25).
Consistent with evidence on the effectiveness of the LCG in improving health outcomes, research indicates that this guideline positively impacts both objectively measured clinical outcomes and patient-reported metrics (26). By clearly defining labor stages and establishing precise standards for intervention timing, the LCG improves clinical decision-making and reduces unnecessary interventions. The more accurate timelines for each stage of labor help prevent premature or unnecessary procedures, ultimately enhancing the overall quality of care.
LCG use has been shown to increase maternal satisfaction with childbirth and contribute to improved maternal and neonatal outcomes (27). These results underscore the importance of implementing the LCG to enhance labor care quality and achieve positive outcomes for both mothers and newborns. Other maternal and neonatal outcomes showed no significant differences between groups, indicating that the LCG reduces early cesarean sections without adversely affecting other outcomes, thereby preventing complications associated with cesarean delivery. Nevertheless, further evaluation through larger pilot studies is needed to confirm these findings. While the current data are promising, larger randomized trials and long-term implementation studies are essential for robust validation. Some heterogeneity in the included studies -such as differences in provider experience, clinical settings, or baseline cesarean rates- may affect the generalizability of the results.
4.1. Strengths and limitations
One of the strengths of this study is that it is the first systematic review examining the effects of the WHO LCG on maternal and neonatal outcomes. By collecting data from various studies, this review provides a comprehensive overview of the impacts of LCG. Limitations include the small number of interventional studies in this field, which may reduce the robustness of the evidence. Many of the studies lack long-term follow-up, limiting the ability to assess the sustained impacts of LCG. Additionally, most studies have focused on low-risk pregnancies, and further research is needed to explore the application of LCG in high-risk pregnancies.
5. Conclusion
This meta-analysis demonstrated that the implementation of the WHO LCG significantly reduced cesarean section rates, the duration of the second stage of labor, and the rate of labor augmentation with oxytocin. However, no significant effects were observed on neonatal outcomes, including stillbirth, Apgar scores below 7 at 5 min, or NICU admissions. These findings suggest that while the intervention can improve certain aspects of the labor process, further research is necessary to comprehensively evaluate its impact on neonatal outcomes and its long-term effects.
Data Availability
The data that support the results of this research can be obtained from the corresponding author upon reasonable request.
Acknowledgments
We thank the authors for helping us in drafting this article. In this study, artificial intelligence was not used at any stage. This study was funded by the Jundishapur University of Medical Sciences, Ahvaz, Iran (grant number: RHPRC-0124).
Conflict of Interest
The authors declare that there is no conflict of interest.
Ensuring safe delivery remains one of the most fundamental missions of healthcare systems worldwide (1). Maternal and neonatal outcomes are directly influenced by the quality of care provided during labor, which includes continuous monitoring of labor progress, timely recognition of complications, and appropriate management strategies (2). Optimal intrapartum care not only reduces preventable maternal and neonatal morbidity and mortality but also improves the overall childbirth experience for women and families. The World Health Organization (WHO) emphasizes that cesarean section should be performed only when medically indicated. While the expected population-based cesarean section rate is estimated at 10-15%, a steady and often alarming increase has been documented globally over the past few decades (3). Rising cesarean rates pose significant public health concerns, particularly because repeated cesarean deliveries are associated with long-term complications, including abnormal placentation, higher risks of hemorrhage, and adverse maternal and neonatal outcomes (4). WHO emphasizes that cesarean rates above 15% are unjustified and has issued guidelines for the safe and rational use of cesarean section (5).
Among the leading contributors to primary cesarean section, especially in nulliparous women, is labor dystocia or arrest of labor progression (6). Accurate diagnosis of labor protraction and decisions to intervene remain highly complex and sometimes controversial. According to the 1994 WHO partograph, cervical dilation progress of < 1 cm per hour over a minimum of 4 hr was defined as a dilatation disorder (7). However, the clinical applicability of this threshold has been debated for decades. Misclassification of slow but physiologically normal labor as dystocia may contribute to unnecessary cesarean deliveries. Recognizing these limitations, WHO revised its recommendations in 2020 and introduced the labor care guide (LCG), designed to replace the traditional partograph and provide clinicians with a more nuanced, evidence-based approach to labor monitoring and decision-making (8).
The LCG differs from the traditional partograph in key aspects. Whereas the partograph considered active labor to begin at 4 cm of cervical dilation, the LCG shifts this threshold to 5 cm, based on recent evidence regarding labor patterns. Unlike the partograph, which ends at full dilation, the LCG incorporates systematic monitoring of the second stage of labor and provides time-based “alert values” for both stages. In the first stage, remaining at 5 cm for 6 hr or longer is considered abnormal, with similar thresholds for 6-9 cm: ≥ 5 hr at 6 cm, ≥ 3 hr at 7 cm, ≥ 2.5 hr at 8 cm, and ≥ 2 hr at 9 cm (9). In the second stage, prolonged labor is over 3 hr for primiparous and 2 hr for multiparous women. Cervical dilation varies, and slower progress can still result in uncomplicated vaginal delivery. Studies comparing WHO partograph and Zhang’s guidelines show differences in dystocia diagnosis and management, though clinical impact remains uncertain (10, 11).
Given these debates and the need to limit unnecessary interventions, this study aims to evaluate the impact of the WHO LCG on maternal and neonatal outcomes.
2. Materials and Methods
2.1. Formulation of the research question
The research question for this meta-analysis was structured using the PICO framework to ensure clarity and precision in identifying relevant studies. The population (P) consisted of women in labor; the intervention (I) involved the implementation of the WHO LCG (WHO LCG); the comparison (C) was conventional intrapartum care not based on WHO LCG; and the outcomes (O) included maternal and neonatal indicators such as duration of labor, mode of delivery, maternal complications, neonatal Apgar scores, and admission to the neonatal intensive care unit (NICU).
2.2. Study design
This study is a meta-analysis of clinical trials conducted to evaluate the impact of implementing (WHO LCG) on maternal and neonatal outcomes. The study design and reporting adhered to the PRISMA guidelines (12).
2.3. Search strategy
A comprehensive and systematic search was conducted to identify relevant studies examining the impact of the WHO LCG on maternal and neonatal outcomes. The literature search was performed in the electronic databases PubMed, Scopus, Google Scholar, and Embase for articles published up to September 2024, without any language restrictions.
The search strategy combined both controlled vocabulary (Medical Subject Headings-MeSH) and relevant free-text keywords. Key terms included: “WHO Labor Care Guide”, “Labor Care Guide”, “next-generation partograph”, “labor outcomes”, “maternal outcomes”, “neonatal outcomes”, and related synonyms. Keywords were selected based on MeSH terms and were refined through consultation with domain experts in maternal health and obstetrics to enhance the sensitivity and specificity of the search. Boolean operators such as AND and OR were used to logically structure and expand the search queries. An example of the full search strategy is presented in table I. In addition to database searching, the reference lists of all included studies were manually reviewed to identify any potentially relevant articles that may have been missed in the electronic search.

2.4. Inclusion and exclusion criteria
2.4.1. Inclusion criteria
Randomized controlled trials or quasi-experimental studies, studies comparing the LCG intervention with standard intrapartum care, studies reporting at least one maternal or neonatal outcome, and full-text articles published in peer-reviewed journals.
2.4.2. Exclusion criteria
Protocols, editorials, qualitative studies, short communications, conference abstracts, studies lacking a control group or quantitative outcome data, and studies involving women with current high-risk pregnancies or a history of high-risk obstetric complications.
2.5. Study selection
2 independent reviewers conducted screening of titles and abstracts following the removal of duplicates. Full-text articles meeting potential eligibility were further assessed in line with the inclusion and exclusion criteria. Any disagreements were settled through discussion or, when necessary, by involving a third reviewer.
2.6. Data extraction
Data were independently extracted by 2 authors using a standardized form, including study characteristics (author, year, country, study design), sample size in intervention and control groups, maternal outcomes, neonatal outcomes, and effect measures (odds ratios, mean differences with 95% confidence intervals).
2.7. Data items
2.7.1. Types of participants
Women in labor, including those undergoing spontaneous or induced labor, were considered eligible for inclusion. Studies involving women with current high-risk pregnancies or a history of high-risk obstetric complications were excluded.
2.7.2. Types of interventions
Implementation of the WHO LCG during intrapartum care. This included studies explicitly mentioning the use of WHO LCG or next-generation partograph aligned with the WHO LCG guidelines.
2.7.3. Outcome measures
Studies were required to report at least one maternal or neonatal outcome. Maternal outcomes included mode of delivery, duration of the second stage of labor, severe perineal trauma, augmentation of labor, and postpartum hemorrhage. Neonatal outcomes included stillbirth, Apgar score, and admission to the NICU.
2.8. Quality assessments
The methodological quality of the studies included in this systematic review and meta-analysis was independently assessed by 2 reviewers using the Cochrane risk of bias tool within review manager. 7 distinct domains are assessed using this tool, including random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and additional potential sources of bias (13).
Each study was categorized as having a low, moderate, or high risk of bias in each domain. In general, the included studies exhibited acceptable methodological rigor. While certain domains, such as blinding, demonstrated moderate to high risk of bias in some studies, other domains, particularly incomplete outcome data and other sources of bias, were predominantly rated as low risk. A summary of the risk of bias assessment is presented in figure 1, depicting the distribution of bias levels across all included studies and assessed domains.

2.9. Data analysis
Data analysis for this meta-analysis was conducted using RevMan version 5.3 software. For dichotomous outcomes, odds ratios were used, while for continuous outcomes, mean differences were applied. Statistical heterogeneity was assessed using the Chi-square test and the I² statistic, and a random-effects model was employed for all analyses. Forest plots were generated to visually display effect sizes and confidence intervals, and sensitivity analyses were conducted by removing studies with a high risk of bias. Due to the small number of studies, publication bias assessment was not performed, and a p < 0.05 was considered statistically significant. All data were entered and verified independently by 2 reviewers to ensure accuracy and consistency.
3. Results
3.1. Study selection and characteristics
In the initial search, 451 articles were retrieved. Following the removal of duplicates and obviously irrelevant studies, 26 articles were screened using their titles and abstracts. Of these, 18 articles were excluded for specific reasons, including lack of an intervention group, nonclinical trial design, insufficient data, or irrelevance to the study objectives. 8 articles were selected for full-text assessment. After applying the inclusion criteria, 5 studies were further excluded. Ultimately, 3 clinical trial studies met all eligibility criteria and were included in the final analysis. The PRISMA flowchart illustrating the study selection process is shown in figure 2.

All 3 studies were conducted in public hospitals and involved staff training on the use of the LCG. 2 studies, included low-risk women with term pregnancies (37-40 wk gestation), cephalic presentation, and spontaneous labor onset, while women with high-risk pregnancies or prior high-risk obstetric histories were excluded (14, 15). Another study included pregnancies beyond 20 wk and applied the Robson classification to both intervention and control groups (16).
For the Vogel study, data from Robson groups 1 and 3 were included, representing nulliparous and multiparous women with single cephalic pregnancies, spontaneous labor, and term gestation (≥ 37 wk). These groups were selected to evaluate the intervention’s impact within a homogeneous, low-risk population for a more accurate assessment.
In all trials, the active phase of labor was defined as starting at 5 cm cervical dilation in the LCG group and at 3 cm in the control group (using the previous partograph). Maternal and fetal parameters were monitored according to LCG criteria and recorded on the LCG partograph. Cervical dilation alert thresholds and second-stage labor durations were followed based on WHO recommendations (Table II).


3.2. Maternal outcomes
3.2.1. Caesarean section rate
The meta-analysis showed a statistically significant reduction in the caesarean section rate in the intervention group compared to the control group (OR = 0.82; 95% CI: 0.76-0.89; Figure 3).
3.2.2. Duration of the second stage of labor
The meta-analysis revealed that the intervention group experienced a notable reduction in the second stage of labor, showing a mean difference of -5.28 min (95% CI: -7.25 to -3.32), as presented in figure 4.
3.2.3. Severe perineal trauma and postpartum hemorrhage
The meta-analysis indicated that there were no significant differences between the intervention and control groups in terms of severe perineal trauma (OR = 0.61; 95% CI: 0.35-1.04; Figure 5) or postpartum hemorrhage (OR = 0.90; 95% CI: 0.47-1.71; Figure 6).
3.2.4. Augmentation of labor
The meta-analysis demonstrated a significant reduction in the rate of labor augmentation in the intervention group, with an odds ratio of 0.27 (95% CI: 0.25-0.30) as shown in figure 7.
3.3. Neonatal outcomes
3.3.1. Stillbirth
The meta-analysis indicated no significant difference in stillbirth rates between the intervention and control groups (OR = 0.96; 95% CI: 0.83-1.10; Figure 8).
3.3.2. Apgar score < 7 at 5 min
The meta-analysis revealed no significant difference in the rate of Apgar score < 7 at 5 min between the intervention and control groups (OR = 0.92; 95% CI: 0.82-1.03; Figure 9).
3.3.3. NICU admission
The meta-analysis found no statistically significant difference in NICU admissions after birth between the intervention and control groups (OR = 0.58; 95% CI: 0.30-1.12; Figure 10).








4. Discussion
This meta-analysis aimed to assess the impact of implementing the LCG on maternal and neonatal outcomes. The findings demonstrate that LCG use is associated with a significant reduction in cesarean section rates, the duration of the second stage of labor, and the need for labor augmentation, without increasing adverse maternal or neonatal events.
The observed reduction in cesarean rates aligns with the WHO objective of minimizing unnecessary obstetric interventions, underscoring the clinical value of the LCG in optimizing intrapartum care. Evidence suggests that the LCG promotes a more patient-centered approach by extending active labor thresholds and reducing premature decisions regarding cesarean delivery (17). This meta-analysis observed a shorter second stage of labor, which may be explained by more consistent and timely clinical decisions guided by the structured LCG criteria. This aligns with the WHO revision of labor definitions, which increased the threshold for the onset of active labor from 3-4 cm to 5 cm of cervical dilation to prevent premature diagnoses of labor arrest (18).
Similarly, the decreased need for labor augmentation suggests that the LCG can minimize unnecessary pharmacological interventions by providing clearer parameters for assessing labor progression. In addition, reviews indicate that many cesarean deliveries result from mistimed interventions (19). By offering structured guidance, the LCG may help clinicians make more accurate decisions, thereby reducing avoidable cesarean sections.
A key feature of the LCG is the raised cervical dilation threshold for identifying active labor, previously set at 3 cm in the modified partograph (20) and now at 5 cm in the LCG (21). The LCG allows for a maximum of approximately 18.5 hr from 5 cm dilation to full cervical dilation, effectively distinguishing abnormal from normal labor. This extended timeframe also helps reduce excessive interventions during labor.
While the thresholds in the LCG are physiologically appropriate, they are more complex to interpret than the standard 1 cm/hr rule during active labor (22), and the guidelines are primarily intended for low-risk pregnancies (23). Consequently, it remains crucial to identify abnormal labor patterns, such as slow progress, and to develop appropriate management plans -whether intervention or continued monitoring- for both clinical and medico-legal purposes (24).
Although there have been concerns that extended labor monitoring might increase maternal complications, our findings showed no significant difference in severe perineal trauma or postpartum hemorrhage, supporting the safety of this approach. This aligns with a WHO-led multicenter feasibility study, which reported that the LCG is both acceptable to clinicians and does not elevate maternal risk when properly implemented (25).
Consistent with evidence on the effectiveness of the LCG in improving health outcomes, research indicates that this guideline positively impacts both objectively measured clinical outcomes and patient-reported metrics (26). By clearly defining labor stages and establishing precise standards for intervention timing, the LCG improves clinical decision-making and reduces unnecessary interventions. The more accurate timelines for each stage of labor help prevent premature or unnecessary procedures, ultimately enhancing the overall quality of care.
LCG use has been shown to increase maternal satisfaction with childbirth and contribute to improved maternal and neonatal outcomes (27). These results underscore the importance of implementing the LCG to enhance labor care quality and achieve positive outcomes for both mothers and newborns. Other maternal and neonatal outcomes showed no significant differences between groups, indicating that the LCG reduces early cesarean sections without adversely affecting other outcomes, thereby preventing complications associated with cesarean delivery. Nevertheless, further evaluation through larger pilot studies is needed to confirm these findings. While the current data are promising, larger randomized trials and long-term implementation studies are essential for robust validation. Some heterogeneity in the included studies -such as differences in provider experience, clinical settings, or baseline cesarean rates- may affect the generalizability of the results.
4.1. Strengths and limitations
One of the strengths of this study is that it is the first systematic review examining the effects of the WHO LCG on maternal and neonatal outcomes. By collecting data from various studies, this review provides a comprehensive overview of the impacts of LCG. Limitations include the small number of interventional studies in this field, which may reduce the robustness of the evidence. Many of the studies lack long-term follow-up, limiting the ability to assess the sustained impacts of LCG. Additionally, most studies have focused on low-risk pregnancies, and further research is needed to explore the application of LCG in high-risk pregnancies.
5. Conclusion
This meta-analysis demonstrated that the implementation of the WHO LCG significantly reduced cesarean section rates, the duration of the second stage of labor, and the rate of labor augmentation with oxytocin. However, no significant effects were observed on neonatal outcomes, including stillbirth, Apgar scores below 7 at 5 min, or NICU admissions. These findings suggest that while the intervention can improve certain aspects of the labor process, further research is necessary to comprehensively evaluate its impact on neonatal outcomes and its long-term effects.
Data Availability
The data that support the results of this research can be obtained from the corresponding author upon reasonable request.
Acknowledgments
We thank the authors for helping us in drafting this article. In this study, artificial intelligence was not used at any stage. This study was funded by the Jundishapur University of Medical Sciences, Ahvaz, Iran (grant number: RHPRC-0124).
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Review Article |
Subject:
Pregnancy Health
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







