Thu, May 21, 2026
[Archive]
Volume 24, Issue 1 (January 2026)
IJRM 2026, 24(1): 1-12 |
Back to browse issues page
Ethics code: IR.SSU.SH.REC.1397.152
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Hooshmandi H, Ghadiri-Anari A, Fallahzadeh H, Ranjbar A M, Hosseinzadeh M, Nadjarzadeh A. Efficacy of licorice extract in combination with a low-calorie diet on sex hormones, sleep quality, depression, and appetite in overweight/obese women with polycystic ovary syndrome: An RCT. IJRM 2026; 24 (1) :1-12
URL: http://ijrm.ir/article-1-3474-en.html
URL: http://ijrm.ir/article-1-3474-en.html
Hadis Hooshmandi1 

 , Akram Ghadiri-Anari2
, Akram Ghadiri-Anari2 

 , Hossein Fallahzadeh3
, Hossein Fallahzadeh3 

 , Ali Mohammad Ranjbar4
, Ali Mohammad Ranjbar4 

 , Mahdieh Hosseinzadeh1
, Mahdieh Hosseinzadeh1 

 , Azadeh Nadjarzadeh *5
, Azadeh Nadjarzadeh *5 




 , Akram Ghadiri-Anari2
, Akram Ghadiri-Anari2 

 , Hossein Fallahzadeh3
, Hossein Fallahzadeh3 

 , Ali Mohammad Ranjbar4
, Ali Mohammad Ranjbar4 

 , Mahdieh Hosseinzadeh1
, Mahdieh Hosseinzadeh1 

 , Azadeh Nadjarzadeh *5
, Azadeh Nadjarzadeh *5 


1- Research Center for Food Hygiene and Safety, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Department of Nutrition, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
2- Research Center for Food Hygiene and Safety, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Diabetes Research Center, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Department of Biostatistics and Epidemiology, Research Center for Healthcare Data Modeling, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
4- Traditional Pharmacy and Pharmaceutical Sciences Research Center, Department of Pharmacognosy, Faculty of Pharmacy, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
5- Research Center for Food Hygiene and Safety, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Department of Nutrition, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,Azadehnajarzadeh@gmail.com; azadnajarzadeh@ssu.ac.ir
2- Research Center for Food Hygiene and Safety, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Diabetes Research Center, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Department of Biostatistics and Epidemiology, Research Center for Healthcare Data Modeling, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
4- Traditional Pharmacy and Pharmaceutical Sciences Research Center, Department of Pharmacognosy, Faculty of Pharmacy, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
5- Research Center for Food Hygiene and Safety, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Department of Nutrition, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,
Keywords: Polycystic ovary syndrome, Glycyrrhiza, Gonadal steroid hormones, Sleep quality, Depression, Appetite.
Full-Text [PDF 436 kb]
(327 Downloads)
| Abstract (HTML) (280 Views)
Full-Text: (52 Views)
1. Introduction
Women with polycystic ovary syndrome (PCOS) are more likely to suffer from anovulatory infertility due to dysfunction of follicular maturation, irregular menses, miscarriage, and hyperandrogenism, as well as dysfunction of hormones such as follicle-stimulating hormone (FSH) and luteinizing hormone (LH) (1). A substantial number of studies have shown that women with PCOS have a decreased quality of life due to abnormal hormonal changes (2-4). Accordingly, several recent studies have revealed that these women are more susceptible to depression and anxiety (5, 6). Furthermore, PCOS may also lead to obstructive sleep apnea, which is linked with depressive disorders and poor sleep quality (7). There is evidence that hyperandrogenism and polycystic ovaries could contribute to bulimic behavior through their influence on food cravings and impulsiveness (8). Also, a study has shown that women with PCOS typically have impaired cholecystokinin production and disordered appetite regulation (9).
Even though the exact cause is unknown, lifestyle, environment, and genetic factors are generally accepted to play a role in PCOS (10-12). Recently, the use of herbal remedies for PCOS has gained attention in traditional medicine as a form of lifestyle management that restores normal menstrual cycle and serum hormone levels (13). A reduction in testosterone and an increase in FSH and 17-estradiol were observed following their use (14, 15). Also, research has shown that they normalize reproductive cycles, enhance insulin sensitivity, and reduce polycystic ovaries and ovarian size (16).
As an herb, licorice, glycyrrhizae radix et rhizome, is considered to be an effective remedy for inflammation, liver failure, immune response, spasms, and women’s metabolic disorders (17, 18). There is limited human evidence that licorice or its ingredients are effective in the management of PCOS-related complications (19). However, licorice supplementation reduced serum testosterone in healthy women (20) and healthy men (21). In animal studies, it could decrease the size of ovarian cysts and enhance the fertilization rate of oocytes and embryos in PCOS mice (22). Also, the ethanolic extract of licorice inhibited the symptoms of PCOS by regulating hormone imbalance and ovarian follicle irregularity in female rats (23).
Despite these, as far as we know, no randomized clinical trial has investigated the effects of licorice supplementation on sex hormones and other health-related complications in women with PCOS. So, this research aimed to examine the effects of licorice extract in combination with a low-calorie diet on sex hormones, sleep quality, depression, and appetite compared to the placebo plus a low-calorie diet in women with PCOS.
2. Materials and Methods
2.1. Study design
Women with polycystic ovary syndrome (PCOS) are more likely to suffer from anovulatory infertility due to dysfunction of follicular maturation, irregular menses, miscarriage, and hyperandrogenism, as well as dysfunction of hormones such as follicle-stimulating hormone (FSH) and luteinizing hormone (LH) (1). A substantial number of studies have shown that women with PCOS have a decreased quality of life due to abnormal hormonal changes (2-4). Accordingly, several recent studies have revealed that these women are more susceptible to depression and anxiety (5, 6). Furthermore, PCOS may also lead to obstructive sleep apnea, which is linked with depressive disorders and poor sleep quality (7). There is evidence that hyperandrogenism and polycystic ovaries could contribute to bulimic behavior through their influence on food cravings and impulsiveness (8). Also, a study has shown that women with PCOS typically have impaired cholecystokinin production and disordered appetite regulation (9).
Even though the exact cause is unknown, lifestyle, environment, and genetic factors are generally accepted to play a role in PCOS (10-12). Recently, the use of herbal remedies for PCOS has gained attention in traditional medicine as a form of lifestyle management that restores normal menstrual cycle and serum hormone levels (13). A reduction in testosterone and an increase in FSH and 17-estradiol were observed following their use (14, 15). Also, research has shown that they normalize reproductive cycles, enhance insulin sensitivity, and reduce polycystic ovaries and ovarian size (16).
As an herb, licorice, glycyrrhizae radix et rhizome, is considered to be an effective remedy for inflammation, liver failure, immune response, spasms, and women’s metabolic disorders (17, 18). There is limited human evidence that licorice or its ingredients are effective in the management of PCOS-related complications (19). However, licorice supplementation reduced serum testosterone in healthy women (20) and healthy men (21). In animal studies, it could decrease the size of ovarian cysts and enhance the fertilization rate of oocytes and embryos in PCOS mice (22). Also, the ethanolic extract of licorice inhibited the symptoms of PCOS by regulating hormone imbalance and ovarian follicle irregularity in female rats (23).
Despite these, as far as we know, no randomized clinical trial has investigated the effects of licorice supplementation on sex hormones and other health-related complications in women with PCOS. So, this research aimed to examine the effects of licorice extract in combination with a low-calorie diet on sex hormones, sleep quality, depression, and appetite compared to the placebo plus a low-calorie diet in women with PCOS.
2. Materials and Methods
2.1. Study design
72 overweight/obese women with PCOS (18-45 yr) were enrolled in this 8 wk randomized, double-blind, placebo-controlled trial at Imam Ali outpatient clinic, Yazd, Iran, from September 2020-November 2020. More details about this research have been published previously (24). Briefly, we selected samples using a convenience sampling method and randomly divided eligible participants into 2 groups. Randomization was performed using permuted block randomization with a block size of 4, generated from a random numbers table. Supplements of licorice and placebo were added in the same capsules in terms of size, color, and shape. The intervention group received 3 capsules of licorice extract plus a low-calorie diet (n = 36), and the control group received 3 capsules of placebo plus a low-calorie diet (n = 36). The researcher prescribed supplements and placebos without knowing their types, and they were identical in shape and packaging. The primary endpoint in this investigation was serum testosterone concentration, while secondary endpoints included serum levels of LH, FSH, sex hormone binding globulin (SHBG), along with measures of depression, sleep quality, and appetite. All evaluated at baseline and 8-wk postintervention.
2.2. Population (inclusion and exclusion criteria)
The participants were included in the study if they were aged 18-45 yr, had a body mass index of 25-35 kg/m2, and had PCOS based on the Rotterdam criteria. Those with PCOS met at least 2 of the following criteria: A) no ovulation or oligo-ovulation (or fewer than 6 cycles in 12 months), B) clinical or biochemical signs of androgen elevation, including acne, hirsutism, alopecia, or elevated serum androgen levels, and C) polycystic ovary with at least 12 follicles per ovary with a diameter of 2-3 mm or an increase of < 11 cm3.
The study did not include participants with blood pressures ≥ 140/90 mmHg, diabetes, liver, thyroid, and cardiovascular and kidney diseases, smoking, regular drug use, taking vitamin-mineral, antioxidant, and herbal supplements during the past 3 months, eating > 300 gr of licorice per week, being pregnant or lactating, or adhering to a special diet. The study also did not include participants who had other causes of hyperandrogenism, including hereditary adrenal hyperplasia, Cushing's syndrome, hyperprolactinemia, or hypothyroidism, as well as an allergy to licorice or any of its components.
Regarding medication, the study did not include women taking estrogen or progesterone, but those who had previously been treated with metformin continued to receive it. Moreover, the people were not included if they had taken new drugs within 2 months before and during the study.
2.3. Intervention groups and implementation of the study
Participants were randomly assigned to either the intervention or control group using simple randomization at the individual level. Each participant was independently and randomly allocated, ensuring an equal probability of assignment across study groups. Randomization was performed using simple individual randomization, and allocation concealment was maintained by employing randomized tables and a balanced block method to ensure equal numbers in each group. Supplements of licorice and placebo were prepared in identical capsules in terms of size, color, and shape, and were encoded as A and B to maintain blinding.
In this study, the intervention group received 3 daily capsules of licorice extract supplement (500 mg per capsule, containing 36.5 mg glycyrrhizic acid) alongside a low-calorie diet (n = 36). The licorice extract was verified and prepared by a pharmacognosy specialist using spray-dried powder (Shirin Daroo Co., Shiraz, Iran) and encapsulated at the Faculty of Pharmacy, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. The control group received 3 identical-looking capsules containing 500 mg of corn starch daily, also combined with a low-calorie diet (n = 36). A total of 168 tablets were assigned to each participant in the study during 8 wk, half an hour before each primary meal. On day 1, each participant received 84 pills to consume during the following 4 wk. After this period, they were contacted for the second course. We counted and recorded unused capsules. During the second session, and if necessary, participants who failed to follow the treatment protocol were excluded. The third session was held after the study, and each person was asked to count the unused capsules they had. To prevent falls and reminders, people were contacted every week and asked about the consumption process; as well as their blood pressure was also checked. They were asked not to alter their medication, physical activity, or lifestyle during the study. They were excluded if they consumed < 70% of the pills. At the beginning and end of the study, a nutritionist completed a 24 hr food recall questionnaire to estimate energy intake, macronutrients, and micronutrients.
In this study, the 2 groups received the same low-calorie diet (restricted energy intake between 500 and 1000 kcal/day). There was a range of energy levels between 1200 and 1800 kcal per day for each participant. Each diet contained about 50-55% carbohydrates, 15-25% proteins, and 30% fats of the total calories. An increase in fiber intake was recommended for reducing dietary density and delaying gastric emptying in the individual; as well as alcohol and foods high in sugar were limited. During each visit, which was conducted every 4 wk, the rate of adherence to the diet was determined through a 24 hr recall and a 2 day food record.
Participants were randomly assigned to the intervention group (3 daily capsules of licorice extract, 500 mg per capsule containing 36.5 mg glycyrrhizic acid, Shirin Daroo Co., Shiraz, Iran) or the control group (3 identical capsules containing 500 mg corn starch). Capsules were identical in appearance, coded A and B, and administered in a double-blind manner. Each participant received 168 capsules (half before each meal for 8 wk) and adherence was monitored by capsule count and weekly follow-ups. Participants consuming < 70% of the capsules were excluded.
2.4. Sample size
2.2. Population (inclusion and exclusion criteria)
The participants were included in the study if they were aged 18-45 yr, had a body mass index of 25-35 kg/m2, and had PCOS based on the Rotterdam criteria. Those with PCOS met at least 2 of the following criteria: A) no ovulation or oligo-ovulation (or fewer than 6 cycles in 12 months), B) clinical or biochemical signs of androgen elevation, including acne, hirsutism, alopecia, or elevated serum androgen levels, and C) polycystic ovary with at least 12 follicles per ovary with a diameter of 2-3 mm or an increase of < 11 cm3.
The study did not include participants with blood pressures ≥ 140/90 mmHg, diabetes, liver, thyroid, and cardiovascular and kidney diseases, smoking, regular drug use, taking vitamin-mineral, antioxidant, and herbal supplements during the past 3 months, eating > 300 gr of licorice per week, being pregnant or lactating, or adhering to a special diet. The study also did not include participants who had other causes of hyperandrogenism, including hereditary adrenal hyperplasia, Cushing's syndrome, hyperprolactinemia, or hypothyroidism, as well as an allergy to licorice or any of its components.
Regarding medication, the study did not include women taking estrogen or progesterone, but those who had previously been treated with metformin continued to receive it. Moreover, the people were not included if they had taken new drugs within 2 months before and during the study.
2.3. Intervention groups and implementation of the study
Participants were randomly assigned to either the intervention or control group using simple randomization at the individual level. Each participant was independently and randomly allocated, ensuring an equal probability of assignment across study groups. Randomization was performed using simple individual randomization, and allocation concealment was maintained by employing randomized tables and a balanced block method to ensure equal numbers in each group. Supplements of licorice and placebo were prepared in identical capsules in terms of size, color, and shape, and were encoded as A and B to maintain blinding.
In this study, the intervention group received 3 daily capsules of licorice extract supplement (500 mg per capsule, containing 36.5 mg glycyrrhizic acid) alongside a low-calorie diet (n = 36). The licorice extract was verified and prepared by a pharmacognosy specialist using spray-dried powder (Shirin Daroo Co., Shiraz, Iran) and encapsulated at the Faculty of Pharmacy, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. The control group received 3 identical-looking capsules containing 500 mg of corn starch daily, also combined with a low-calorie diet (n = 36). A total of 168 tablets were assigned to each participant in the study during 8 wk, half an hour before each primary meal. On day 1, each participant received 84 pills to consume during the following 4 wk. After this period, they were contacted for the second course. We counted and recorded unused capsules. During the second session, and if necessary, participants who failed to follow the treatment protocol were excluded. The third session was held after the study, and each person was asked to count the unused capsules they had. To prevent falls and reminders, people were contacted every week and asked about the consumption process; as well as their blood pressure was also checked. They were asked not to alter their medication, physical activity, or lifestyle during the study. They were excluded if they consumed < 70% of the pills. At the beginning and end of the study, a nutritionist completed a 24 hr food recall questionnaire to estimate energy intake, macronutrients, and micronutrients.
In this study, the 2 groups received the same low-calorie diet (restricted energy intake between 500 and 1000 kcal/day). There was a range of energy levels between 1200 and 1800 kcal per day for each participant. Each diet contained about 50-55% carbohydrates, 15-25% proteins, and 30% fats of the total calories. An increase in fiber intake was recommended for reducing dietary density and delaying gastric emptying in the individual; as well as alcohol and foods high in sugar were limited. During each visit, which was conducted every 4 wk, the rate of adherence to the diet was determined through a 24 hr recall and a 2 day food record.
Participants were randomly assigned to the intervention group (3 daily capsules of licorice extract, 500 mg per capsule containing 36.5 mg glycyrrhizic acid, Shirin Daroo Co., Shiraz, Iran) or the control group (3 identical capsules containing 500 mg corn starch). Capsules were identical in appearance, coded A and B, and administered in a double-blind manner. Each participant received 168 capsules (half before each meal for 8 wk) and adherence was monitored by capsule count and weekly follow-ups. Participants consuming < 70% of the capsules were excluded.
2.4. Sample size
Based on data from Armanini et al. (20) assuming α = 0.05, power = 80%, σ = 6, and a mean difference of 4 units in testosterone levels between groups, the required sample size was estimated at 36 per group, calculated using the following formula:
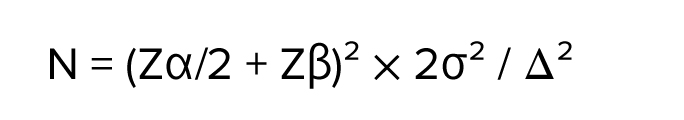
Samples were selected by convenience sampling method from women referred to the clinic. The eligible participants were then divided into 2 groups using a random number table (random allocation version 1). Supplementation and placebo were prescribed by the researcher without knowing their type, and their use by the participants in the study was without their knowledge. Supplements and placebo were same in shape and packaging.
2.5. Determination of basic data
As testosterone was designated and analyzed as the primary outcome in our previous publication, with the sample size calculation based on this parameter, the current manuscript focuses exclusively on the secondary outcomes.
In this study, after obtaining written consent, participants' general information was collected, including age, sex, and medical history. Anthropometric measurements, including weight and height, were taken at the beginning and end of the intervention. We measured the participants' body weight with minimal clothing and without shoes using a digital scale with 100 g sensitivity. Individuals' height was measured with 0.1 cm accuracy using a tape measure standing against the wall without shoes. The body mass index was calculated based on weight (kg)/height (m)2.
The basic data, including demographic variables, questionnaire responses, and physical measurements, were collected by a nutritionist during 2 months. Laboratory analyses were performed by a laboratory specialist at Sadra Laboratory, Yazd, Iran. Anthropometric indices (weight, height, body mass index) were measured at baseline and post-intervention by a trained nutritionist using standard methods.
2.6. Biochemical assessment
After fasting for 8-10 hr, 5 cc of venous blood was collected from each participant and stored at -73°C after serum separation. Blood samples were taken to examine the levels of LH, FSH, SHBG, and total testosterone, once at the beginning of the study and again at the end. Special kits were used for the measurements, and the results were recorded in laboratory forms. The ratio of LH to FSH (LH/FSH) was also calculated as a marker for PCOS diagnosis.
Following an 8-10 hr overnight fast, 5 mL venous blood samples were collected at baseline and after 8 wk. Serum LH, FSH, SHBG, and total testosterone concentrations were measured using standard ELISA kit, and LH/FSH ratio was calculated.
2.7. Assessment of sleep quality, appetite, and depression
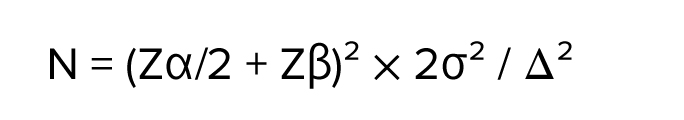
Samples were selected by convenience sampling method from women referred to the clinic. The eligible participants were then divided into 2 groups using a random number table (random allocation version 1). Supplementation and placebo were prescribed by the researcher without knowing their type, and their use by the participants in the study was without their knowledge. Supplements and placebo were same in shape and packaging.
2.5. Determination of basic data
As testosterone was designated and analyzed as the primary outcome in our previous publication, with the sample size calculation based on this parameter, the current manuscript focuses exclusively on the secondary outcomes.
In this study, after obtaining written consent, participants' general information was collected, including age, sex, and medical history. Anthropometric measurements, including weight and height, were taken at the beginning and end of the intervention. We measured the participants' body weight with minimal clothing and without shoes using a digital scale with 100 g sensitivity. Individuals' height was measured with 0.1 cm accuracy using a tape measure standing against the wall without shoes. The body mass index was calculated based on weight (kg)/height (m)2.
The basic data, including demographic variables, questionnaire responses, and physical measurements, were collected by a nutritionist during 2 months. Laboratory analyses were performed by a laboratory specialist at Sadra Laboratory, Yazd, Iran. Anthropometric indices (weight, height, body mass index) were measured at baseline and post-intervention by a trained nutritionist using standard methods.
2.6. Biochemical assessment
After fasting for 8-10 hr, 5 cc of venous blood was collected from each participant and stored at -73°C after serum separation. Blood samples were taken to examine the levels of LH, FSH, SHBG, and total testosterone, once at the beginning of the study and again at the end. Special kits were used for the measurements, and the results were recorded in laboratory forms. The ratio of LH to FSH (LH/FSH) was also calculated as a marker for PCOS diagnosis.
Following an 8-10 hr overnight fast, 5 mL venous blood samples were collected at baseline and after 8 wk. Serum LH, FSH, SHBG, and total testosterone concentrations were measured using standard ELISA kit, and LH/FSH ratio was calculated.
2.7. Assessment of sleep quality, appetite, and depression
Participants were asked to complete the Pittsburgh sleep quality index tool before and after the study period to determine their sleep quality. This standard, self-report, 19 item questionnaire assessed sleep quality over the past month. It was made up of several questions that asked about sleep disturbances and their scores, such as sleep disorders, sleep delay, mental quality of sleep, sleep duration, and use of sleep medications (25). The appetite of the participant were assessed using the council on nutrition appetite questionnaire. It contained 8 single-domain items that are graded using a 5-point Likert scale (the final score ranged between 8 and 40) (26). Moreover, we assessed depression with a Persian version of the 21 item Beck depression inventory questionnaire, which had been validated in previous studies. The questionnaire assesses various symptoms of depression, including crying, guilt, sadness, fear, hopelessness, sleep disturbances, and loss of appetite (27, 28).
Sleep quality was evaluated using the Pittsburgh sleep quality index, appetite by the council on nutrition appetite questionnaire, and depression by the 21 item Beck depression inventory. Validated Persian versions were used for all questionnaires.
2.8. Ethical Considerations
Sleep quality was evaluated using the Pittsburgh sleep quality index, appetite by the council on nutrition appetite questionnaire, and depression by the 21 item Beck depression inventory. Validated Persian versions were used for all questionnaires.
2.8. Ethical Considerations
This research was approved by the Medical Ethics Committee of Shahid Sadoughi University of Medical Sciences, Yazd, Iran (Code: IR.SSU.SH.REC.1397.152). The trial protocol was registered in the Iranian Registry of Clinical Trials (IRCT20200922048802N2 on 12/22/2020, updated on 12/03/2025). All study procedures were conducted in compliance with the Declaration of Helsinki and adhered to applicable guidelines and regulations. The trial followed CONSORT reporting standards, and all participants provided written informed consent prior to enrollment.
2.9. Statistical Analysis
2.9. Statistical Analysis
The normality of continuous variables was tested using the Kolmogorov-Smirnov test. Comparing continuous variables between the 2 groups was done using an independent sample t test or Mann-Whitney test. Comparing continuous variables between groups after adjusting for baseline values was done using analysis of covariance (ANCOVA). To compare baseline and post-intervention differences within groups, a paired t test was used. Analysis of the data was conducted with SPSS software (version 24, SPSS Inc., Chicago, IL, USA), and p < 0.05 was considered statistically significant.
Normality of data was assessed using the Kolmogorov-Smirnov test. Between-group comparisons were conducted using independent t test or Mann-Whitney U test, and within-group changes using paired t test. ANCOVA was used to adjust for baseline values. Analyses were performed using SPSS version 24 (SPSS Inc., Chicago, IL, USA), with significance set at p < 0.05.
3. Results
Normality of data was assessed using the Kolmogorov-Smirnov test. Between-group comparisons were conducted using independent t test or Mann-Whitney U test, and within-group changes using paired t test. ANCOVA was used to adjust for baseline values. Analyses were performed using SPSS version 24 (SPSS Inc., Chicago, IL, USA), with significance set at p < 0.05.
3. Results
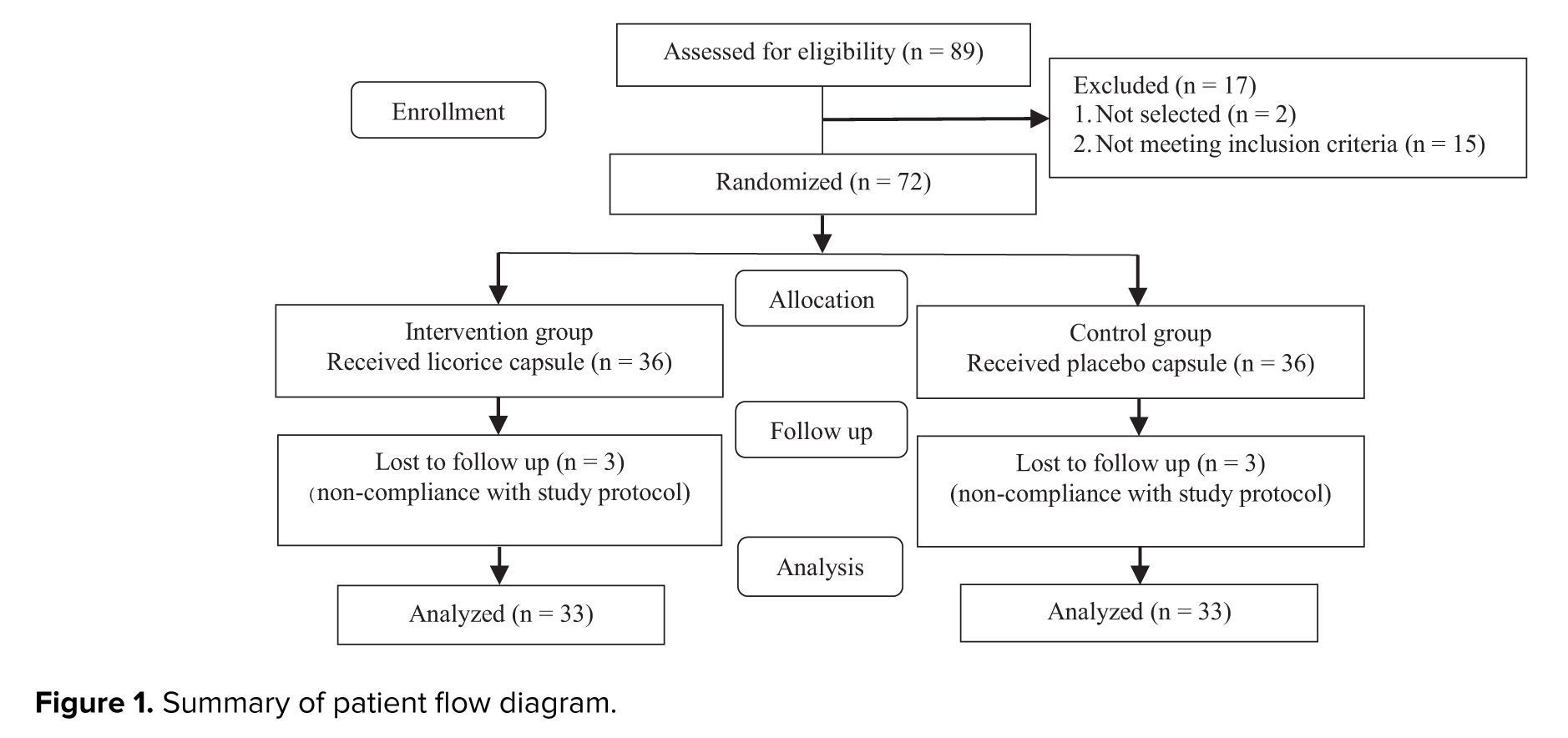
This study enrolled 72 overweight/obese women with PCOS, who were randomly allocated to either a licorice extract plus low-calorie diet group or a placebo plus low-calorie diet group (n = 36/group). 6 participants (3 from each group) were excluded from the final analysis due to non-compliance with study protocol, resulting in 33 participants per group for data analysis (Figure 1).
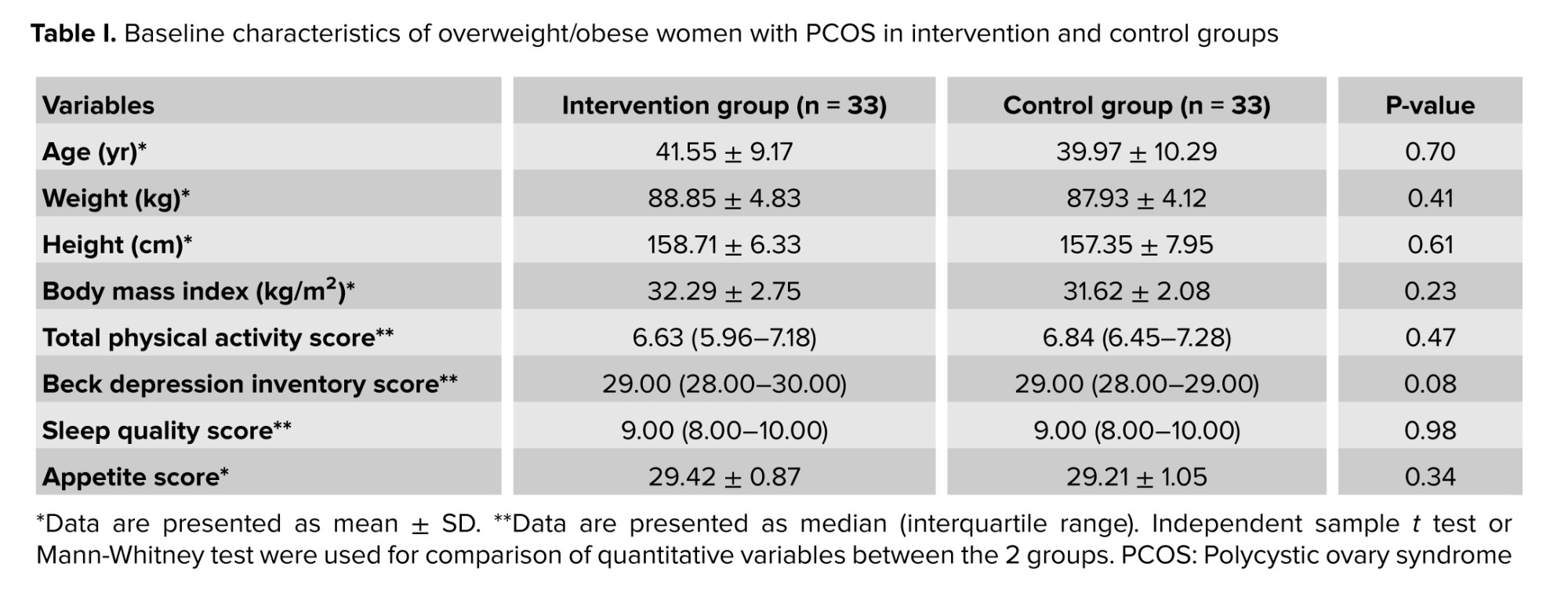
Baseline characteristics did not differ significantly between the 2 groups, as demonstrated in table I.
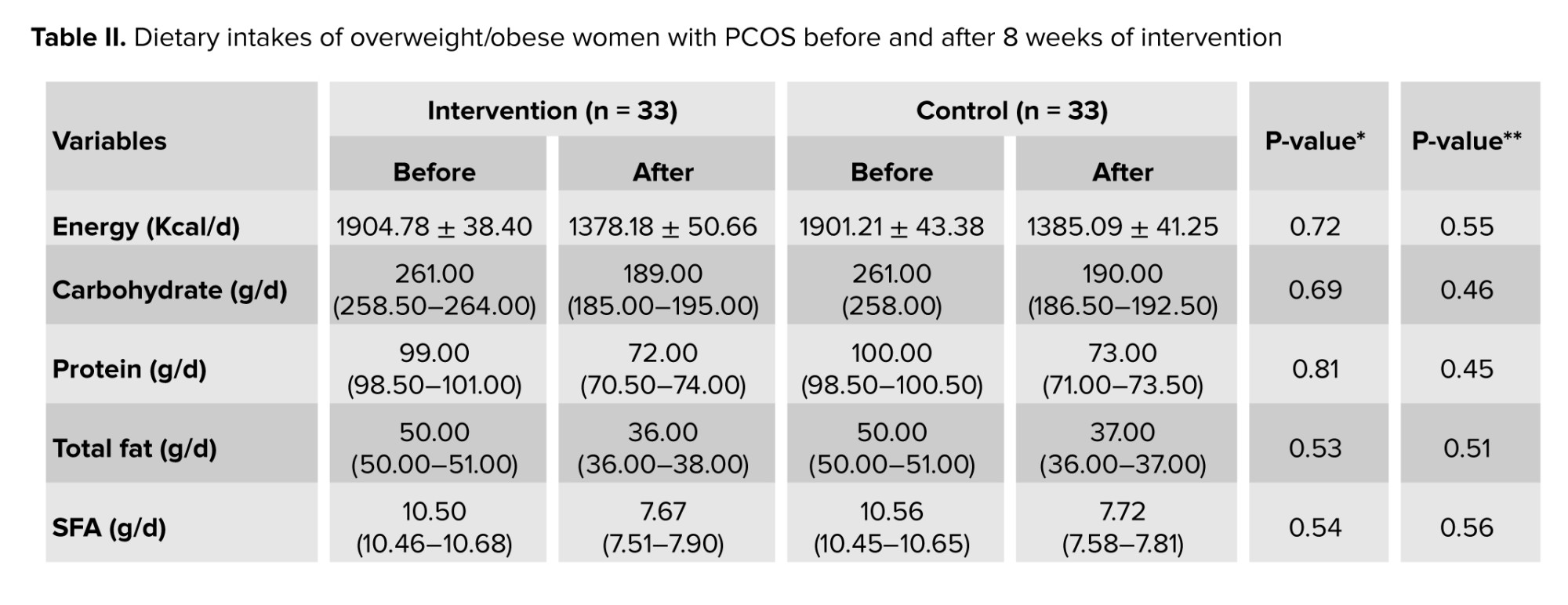
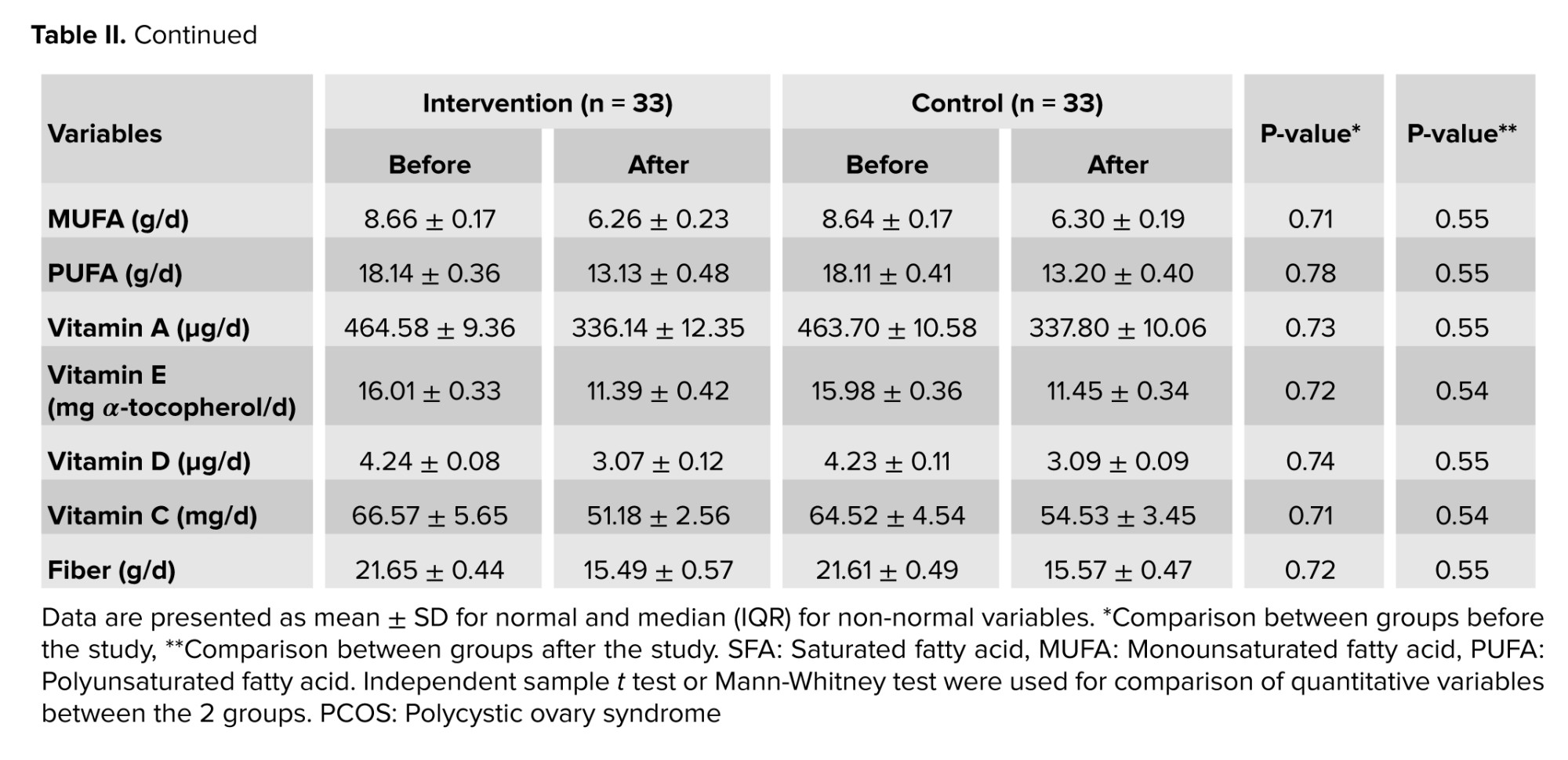
Table II presents changes in participants' dietary intake throughout the study. No statistically significant differences were observed between groups for any dietary parameters, either at baseline or following the 8 wk intervention period (p > 0.05).
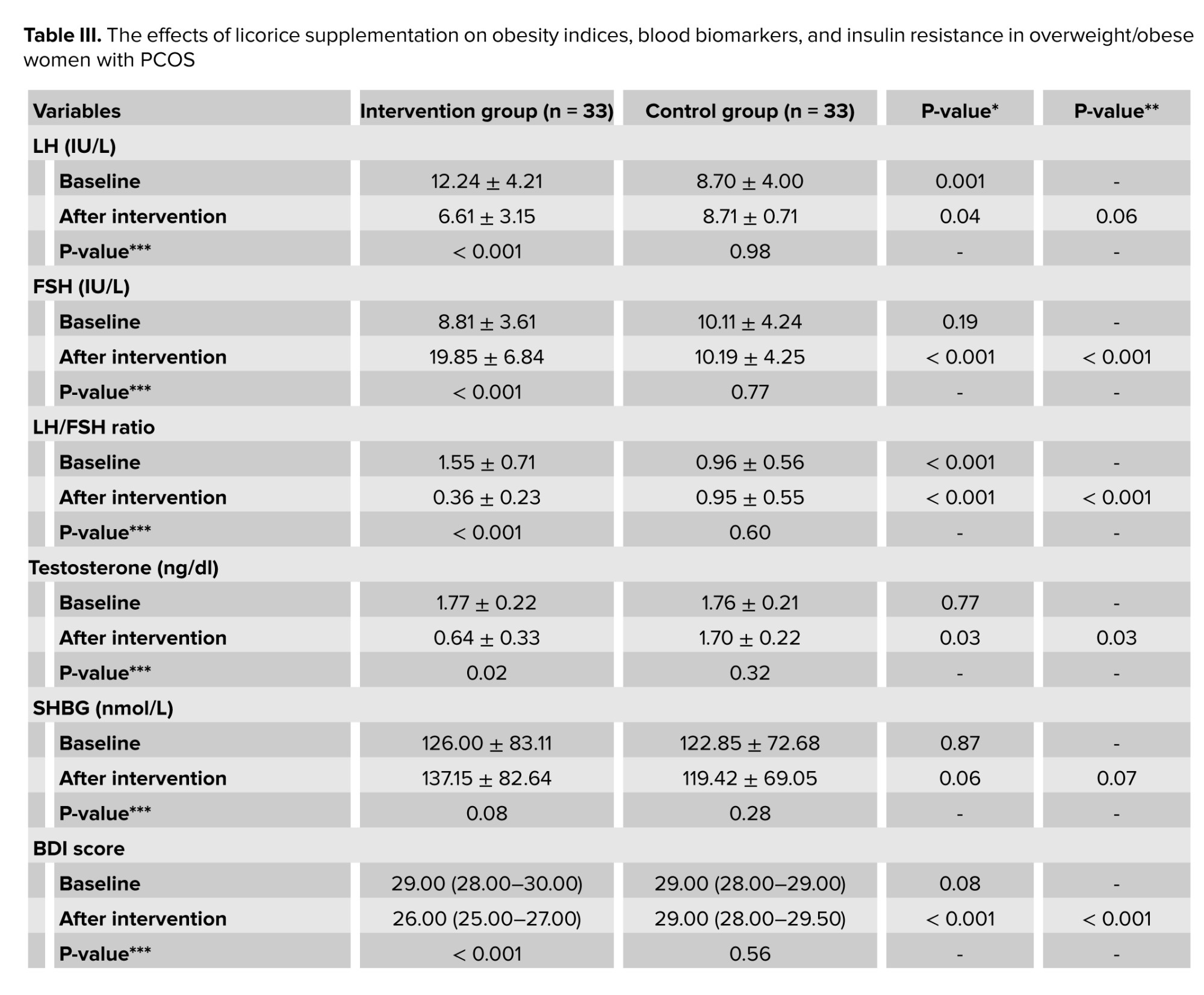
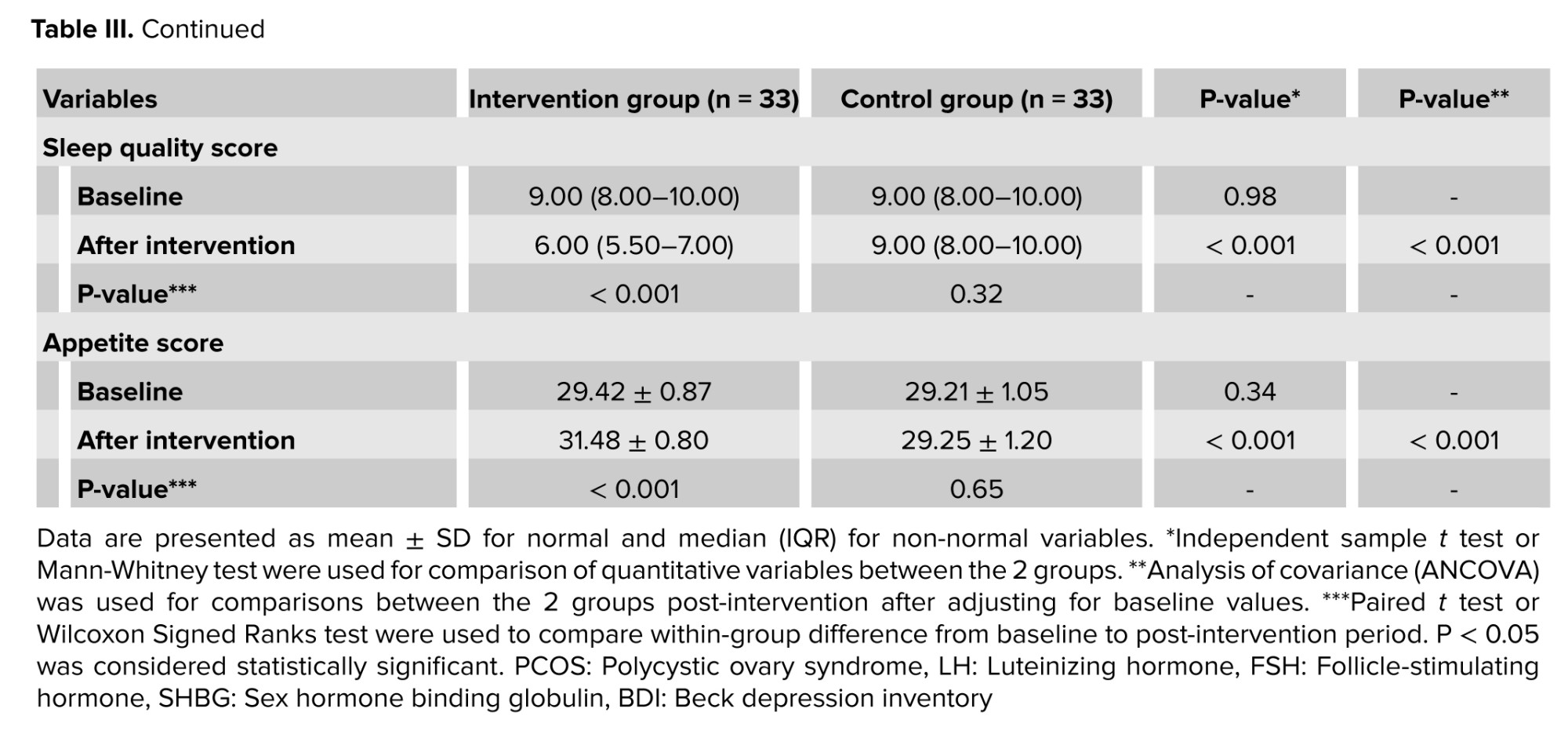
The effects of licorice supplementation on overweight/obese women with PCOS on sex hormones, depression, sleep quality, and appetite can be seen in table III. LH (p = 0.001) and LH/FSH ratios (p < 0.001) were significantly different between the groups at baseline. In the intervention group, LH (p < 0.001), FSH (p < 0.001), LH/FSH ratio (p < 0.001), testosterone (p = 0.02), depression score (p < 0.001), sleep quality (p < 0.001), and appetite (p < 0.001) significantly improved after 8 wk. The low-calorie diet did not have significant effects on all parameters in the control group (p > 0.05). After adjusting for baseline values, the licorice extract plus low-calorie diet group showed significantly greater improvements in terms of FSH (p < 0.001), LH/FSH ratio (p < 0.001), total testosterone (p = 0.03), depression score (p < 0.001), sleep quality (p < 0.001), and appetite (p < 0.001) compared to the control group.
4. Discussion
This randomized controlled trial evaluated the adjunctive effects of licorice extract (1.5 gr/day) to a low-calorie diet vs. diet alone in overweight/obese women with PCOS. The licorice supplementation group demonstrated significant improvements in endocrine parameters (FSH, LH/FSH ratio, and testosterone levels), sleep quality, depressive symptoms, and appetite control compared to the control group. Notably, the low-calorie diet alone failed to produce significant changes in these outcomes, suggesting that licorice extract may provide complementary therapeutic benefits for PCOS management beyond dietary modification. In this study, we found that licorice extract significantly improved sex hormones compared to a placebo. Limited human and animal research showed relatively positive effects of licorice and its components in the regulation of sex hormones (19-21). Accordingly, in a study, supplementation with a 3.5 gr/day licorice supplement for 2 cycles reduced total serum testosterone in healthy women. In that study, licorice supplementation could not change androstenedione, LH, and 17OH-progesterone levels (20). In another study, 8 days of intervention with licorice extract (7 gr/day) significantly reduced total serum testosterone in healthy men. The study also found that 17OH-progesterone and LH concentrations rose significantly, while free testosterone decreased slightly, but not significantly (21). Moreover, taking 10 mg/day of glabridin, an active ingredient in licorice, significantly decreased testosterone levels and increased SHBG levels, but did not significantly affect serum levels of LH or FSH (19). As seen, results from these studies may differ due to differences in intervention duration, study design, dosage, and type of licorice supplement.
Based on an animal study, a 4 wk intervention with licorice ethanolic extract (300 mg/kg/day) significantly boosted FSH levels and LH/FSH ratio but had no significant effect on LH concentrations in letrozole-induced PCOS in female rats (23). It was also found that 21 days of intervention with licorice (100 or 150 mg/kg/day) significantly increased the number of healthy follicles and corpus luteum, as well as significantly decreased the number of atretic follicles in a mice model of PCOS (22).
Several possible mechanisms can explain the effects of licorice extract on the regulation of sex hormones in women with PCOS. Firstly, evidence suggested that licorice could reduce serum testosterone probably through inhibition of 17β-hydroxysteroid dehydrogenase and 17,20 lyase (20). Secondly, licorice is a source of phytoestrogen which has been shown to decrease the serum level of androgens by increasing the level of sex SHBG (22). Lastly, there is evidence that the major components of licorice, glabridin and glabrene, may have estrogen-like properties (29).
In this study, we found that licorice extract significantly improved sleep quality and depression compared to the control group. A comprehensive literature review suggests there are no prior human studies assessing licorice's effects on health-related quality of life in women with PCOS. However, a substantial number of studies have shown that women with PCOS have a decreased bquality of life due to abnormal hormonal changes (2-4). Also, other studies have revealed that these women are more susceptible to depression and anxiety (5, 6). Furthermore, a recent systematic review and meta-analysis found that women with PCOS are more likely to have sleep disorders like hypersomnia and obstructive sleep apnea (30). Thus, it seems application of suitable dietary interventions like supplementation with natural foods and food ingredients, may help in the management of PCOS-related complications like sleep disorders and depression. A few experimental studies have shown the possible benefits of licorice ingredients on sleep quality, although no human studies have been conducted (31, 32). Accordingly, there has been evidence that licorice ethanolic extract imparts sedative-hypnotic effects in vivo and binds to γ-aminobutyric acid type A-benzodiazepine receptors in vitro (31). Also, licorice flavonoid isoliquiritigenin induces sleep by positively modulating the γ-aminobutyric acid type A-benzodiazepine receptors (32). However, due to limited available data, more research, especially on humans, is needed to make a conclusive decision.
We found that supplementation with licorice, compared to the placebo, significantly improved appetite in women with PCOS. A study has shown that women with PCOS typically have impaired cholecystokinin production and disordered appetite regulation (33). However, due to a lack of evidence, more studies are also warranted to highlight the possible mechanism of action of licorice in appetite regulation.
4.1. Strengths and Limitations
While this double-blind, placebo-controlled trial demonstrated high participant adherence, several limitations should be acknowledged. First, the 8 wk intervention period may have been insufficient to evaluate long-term effects, and the study design did not include a licorice-only arm without concurrent dietary modification. Second, mechanistic pathways remain unclear due to unmeasured plasma levels of bioactive licorice compounds (e.g., glabridin) and antioxidant enzymes that could elucidate hormonal regulation. Third, dietary assessments relied on 24 hr recall methodology, which is inherently limited by participant recall accuracy and reporting bias (34).
5. Conclusion
This study examined the effects of licorice supplementation combined with a low-calorie diet on quality of life in overweight/obese women with PCOS. Results demonstrated that licorice extract improved sex hormone levels, sleep quality, depressive symptoms, and appetite control compared to placebo. However, further research is required to confirm these findings due to study limitations.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Author Contributions
H. Hooshmandi conducted the experiments, handled the study materials, collected the data, and drafted the manuscript, A. Ghadiri-Anari contributed to study design, assisted in data collection, and critically revised the manuscript, AM. Ranjbar contributed to study design and provided guidance, H. Fallahzadeh analyzed and interpreted the data, M. Hosseinzadeh contributed to study design, provided guidance, and revised the manuscript, A. Nadjarzadeh designed the study, managed the project, supervised the manuscript, and provided guidance. All authors approved the final version and are accountable for the work.
Acknowledgments
The authors wish to thank all participants who kindly contributed to the study. This article was extracted from Master’s degree thesis (Hadis Hooshmandi). The costs of this study were supported by the Research Deputy of Shahid Sadoughi University of Medical Sciences, Yazd, Iran. No specific grant was received for this work. No funding for this project. During manuscript revision, AI was used only for grammar checking and enhancing English language clarity (Chat GPT). No scientific content, data analysis, or original text was generated by AI. The authors take full responsibility for the accuracy and integrity of the work.
Conflict of Interest
The authors declare that there is no conflict of interest.
Baseline characteristics did not differ significantly between the 2 groups, as demonstrated in table I.
Table II presents changes in participants' dietary intake throughout the study. No statistically significant differences were observed between groups for any dietary parameters, either at baseline or following the 8 wk intervention period (p > 0.05).
The effects of licorice supplementation on overweight/obese women with PCOS on sex hormones, depression, sleep quality, and appetite can be seen in table III. LH (p = 0.001) and LH/FSH ratios (p < 0.001) were significantly different between the groups at baseline. In the intervention group, LH (p < 0.001), FSH (p < 0.001), LH/FSH ratio (p < 0.001), testosterone (p = 0.02), depression score (p < 0.001), sleep quality (p < 0.001), and appetite (p < 0.001) significantly improved after 8 wk. The low-calorie diet did not have significant effects on all parameters in the control group (p > 0.05). After adjusting for baseline values, the licorice extract plus low-calorie diet group showed significantly greater improvements in terms of FSH (p < 0.001), LH/FSH ratio (p < 0.001), total testosterone (p = 0.03), depression score (p < 0.001), sleep quality (p < 0.001), and appetite (p < 0.001) compared to the control group.






4. Discussion
This randomized controlled trial evaluated the adjunctive effects of licorice extract (1.5 gr/day) to a low-calorie diet vs. diet alone in overweight/obese women with PCOS. The licorice supplementation group demonstrated significant improvements in endocrine parameters (FSH, LH/FSH ratio, and testosterone levels), sleep quality, depressive symptoms, and appetite control compared to the control group. Notably, the low-calorie diet alone failed to produce significant changes in these outcomes, suggesting that licorice extract may provide complementary therapeutic benefits for PCOS management beyond dietary modification. In this study, we found that licorice extract significantly improved sex hormones compared to a placebo. Limited human and animal research showed relatively positive effects of licorice and its components in the regulation of sex hormones (19-21). Accordingly, in a study, supplementation with a 3.5 gr/day licorice supplement for 2 cycles reduced total serum testosterone in healthy women. In that study, licorice supplementation could not change androstenedione, LH, and 17OH-progesterone levels (20). In another study, 8 days of intervention with licorice extract (7 gr/day) significantly reduced total serum testosterone in healthy men. The study also found that 17OH-progesterone and LH concentrations rose significantly, while free testosterone decreased slightly, but not significantly (21). Moreover, taking 10 mg/day of glabridin, an active ingredient in licorice, significantly decreased testosterone levels and increased SHBG levels, but did not significantly affect serum levels of LH or FSH (19). As seen, results from these studies may differ due to differences in intervention duration, study design, dosage, and type of licorice supplement.
Based on an animal study, a 4 wk intervention with licorice ethanolic extract (300 mg/kg/day) significantly boosted FSH levels and LH/FSH ratio but had no significant effect on LH concentrations in letrozole-induced PCOS in female rats (23). It was also found that 21 days of intervention with licorice (100 or 150 mg/kg/day) significantly increased the number of healthy follicles and corpus luteum, as well as significantly decreased the number of atretic follicles in a mice model of PCOS (22).
Several possible mechanisms can explain the effects of licorice extract on the regulation of sex hormones in women with PCOS. Firstly, evidence suggested that licorice could reduce serum testosterone probably through inhibition of 17β-hydroxysteroid dehydrogenase and 17,20 lyase (20). Secondly, licorice is a source of phytoestrogen which has been shown to decrease the serum level of androgens by increasing the level of sex SHBG (22). Lastly, there is evidence that the major components of licorice, glabridin and glabrene, may have estrogen-like properties (29).
In this study, we found that licorice extract significantly improved sleep quality and depression compared to the control group. A comprehensive literature review suggests there are no prior human studies assessing licorice's effects on health-related quality of life in women with PCOS. However, a substantial number of studies have shown that women with PCOS have a decreased bquality of life due to abnormal hormonal changes (2-4). Also, other studies have revealed that these women are more susceptible to depression and anxiety (5, 6). Furthermore, a recent systematic review and meta-analysis found that women with PCOS are more likely to have sleep disorders like hypersomnia and obstructive sleep apnea (30). Thus, it seems application of suitable dietary interventions like supplementation with natural foods and food ingredients, may help in the management of PCOS-related complications like sleep disorders and depression. A few experimental studies have shown the possible benefits of licorice ingredients on sleep quality, although no human studies have been conducted (31, 32). Accordingly, there has been evidence that licorice ethanolic extract imparts sedative-hypnotic effects in vivo and binds to γ-aminobutyric acid type A-benzodiazepine receptors in vitro (31). Also, licorice flavonoid isoliquiritigenin induces sleep by positively modulating the γ-aminobutyric acid type A-benzodiazepine receptors (32). However, due to limited available data, more research, especially on humans, is needed to make a conclusive decision.
We found that supplementation with licorice, compared to the placebo, significantly improved appetite in women with PCOS. A study has shown that women with PCOS typically have impaired cholecystokinin production and disordered appetite regulation (33). However, due to a lack of evidence, more studies are also warranted to highlight the possible mechanism of action of licorice in appetite regulation.
4.1. Strengths and Limitations
While this double-blind, placebo-controlled trial demonstrated high participant adherence, several limitations should be acknowledged. First, the 8 wk intervention period may have been insufficient to evaluate long-term effects, and the study design did not include a licorice-only arm without concurrent dietary modification. Second, mechanistic pathways remain unclear due to unmeasured plasma levels of bioactive licorice compounds (e.g., glabridin) and antioxidant enzymes that could elucidate hormonal regulation. Third, dietary assessments relied on 24 hr recall methodology, which is inherently limited by participant recall accuracy and reporting bias (34).
5. Conclusion
This study examined the effects of licorice supplementation combined with a low-calorie diet on quality of life in overweight/obese women with PCOS. Results demonstrated that licorice extract improved sex hormone levels, sleep quality, depressive symptoms, and appetite control compared to placebo. However, further research is required to confirm these findings due to study limitations.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Author Contributions
H. Hooshmandi conducted the experiments, handled the study materials, collected the data, and drafted the manuscript, A. Ghadiri-Anari contributed to study design, assisted in data collection, and critically revised the manuscript, AM. Ranjbar contributed to study design and provided guidance, H. Fallahzadeh analyzed and interpreted the data, M. Hosseinzadeh contributed to study design, provided guidance, and revised the manuscript, A. Nadjarzadeh designed the study, managed the project, supervised the manuscript, and provided guidance. All authors approved the final version and are accountable for the work.
Acknowledgments
The authors wish to thank all participants who kindly contributed to the study. This article was extracted from Master’s degree thesis (Hadis Hooshmandi). The costs of this study were supported by the Research Deputy of Shahid Sadoughi University of Medical Sciences, Yazd, Iran. No specific grant was received for this work. No funding for this project. During manuscript revision, AI was used only for grammar checking and enhancing English language clarity (Chat GPT). No scientific content, data analysis, or original text was generated by AI. The authors take full responsibility for the accuracy and integrity of the work.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Fertility & Infertility
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |




