Thu, Jun 11, 2026
[Archive]
Volume 23, Issue 9 (September 2025)
IJRM 2025, 23(9): 723-738 |
Back to browse issues page
Ethics code: TCMF1–2021074
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Tu X, Zhang H, Liu Z, Feng J, Zhang Y. Investigation of a novel absorbable protein stent for microsurgical vasovasostomy in rats: An experimental study. IJRM 2025; 23 (9) :723-738
URL: http://ijrm.ir/article-1-3494-en.html
URL: http://ijrm.ir/article-1-3494-en.html
1- Department of Urology, The First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou, China.
2- Chair of Endocrinology and Medical Sexology (ENDOSEX), Department of Experimental Medicine, University of Rome Tor Vergata, Rome, Italy. & Department of Infertility and Sexual Medicine, Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou, China.
3- Department of Infertility and Sexual Medicine, Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou, China.
4- Department of Infertility and Sexual Medicine, Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou, China. ,zhxml@sina.com
2- Chair of Endocrinology and Medical Sexology (ENDOSEX), Department of Experimental Medicine, University of Rome Tor Vergata, Rome, Italy. & Department of Infertility and Sexual Medicine, Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou, China.
3- Department of Infertility and Sexual Medicine, Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou, China.
4- Department of Infertility and Sexual Medicine, Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou, China. ,
Full-Text [PDF 4628 kb]
(612 Downloads)
| Abstract (HTML) (700 Views)
Full-Text: (159 Views)
1. Introduction
Microsurgical vasovasostomy is acknowledged as a highly effective and cost-efficient procedure, but requires extensive training (1, 2). Since its initial proposal by Silber in the 1970s, microsurgical vasovasostomy has emerged as the gold standard technique for vas deferens anastomosis (3, 4). Despite the effectiveness demonstrated by the traditional 2-layer anastomosis technique, it is limited by its extensive surgical duration and high technical demands.
To simplify the surgical procedure and reduce operation time, several researchers have proposed a single-layer anastomosis technique. Although this method reduces surgical complexity, it may lead to inadequate mucosal anastomosis of the vas deferens, potentially causing stenosis or obstruction at the anastomosis site, as well as granuloma formation, thereby affecting postoperative patency and pregnancy rates (5-7). Moreover, in recent years, robot-assisted vas deferens anastomosis has gained attention due to its potential to enhance surgical precision and reduce technical challenges. However, the application of this technology remains limited due to high costs and the absence of direct tactile feedback (8-10).
Stent-assisted anastomosis offers several advantages, including the capacity for customization according to the condition of both vasal ends, and the structural support provided by the stent that facilitates more accurate suturing. Vrijhof previously proposed a novel non-absorbable hollow vas deferens stent, which demonstrated excellent surgical efficiency and favorable patency (11). Nevertheless, the persistent presence of non-absorbable materials gives rise to concerns regarding long-term safety, thus necessitating continuous follow-up.
In recent years, increasing application of biodegradable materials has been observed in the field of andrology. For instance, the utilization of biodegradable stents in urological reconstruction has been demonstrated to exhibit excellent biocompatibility and absorbability (12). These developments provide a solid theoretical foundation and promising prospects for exploring new materials for vas deferens stents.
In this study, rats were selected as the experimental model based on their anatomical and functional similarity of the vas deferens to humans, combined with low ethical burden and ease of manipulation (13). The objective of this study was to provide experimental evidence and theoretical support for further optimizing minimally invasive fertility restoration surgery by evaluating the application of a biodegradable protein stent in microsurgical vasovasostomy.
2. Materials and Methods
2.1. Preparation of the stent and animals
This study introduces an absorbable, natural protein stent derived from otter tail tendons (Medical Device Registration No: CFDA 20173654672) for use in stent-assisted microsurgical vasovasostomy for the first time. Developed and provided by Hunan Ranyuan Medical High-tech Protein Suture Co., Ltd., the stent represents an innovative strategy in the application of biomaterials. It is refined from structural proteins (mainly collagen, with trace amounts of elastin), which are found in abundance in natural tendon tissue. The preparation process involves ethanol dehydration, impurity removal using sodium hydroxide, multiple purification steps, and low-temperature sterilization to maximally preserve the natural conformation and biological activity of the proteins. The resulting material offers excellent tissue affinity and biodegradability. After implantation, the stent gradually degrades into nontoxic amino acid molecules within 4-8 wk, thereby avoiding chronic inflammatory responses caused by long-term foreign body retention. Its unique microfibrous mesh structure provides sufficient initial mechanical support to maintain the stability of the anastomosis and acts as a biological scaffold during degradation. This guides the regeneration of surrounding tissue, promoting cell adhesion, proliferation, and organized reconstruction. This realizes a true ‘stent-guidance-disappearance’ tri-functional transformation. Compared to nondegradable synthetic materials or traditional microsurgical 2-layer vasovasostomy, this protein stent has significant advantages in terms of supporting precise tissue reconstruction, reducing foreign body reactions, and lowering the risk of postoperative complications, particularly in microsurgical procedures where high levels of precision and biocompatibility are essential.
The experimental animals were obtained from the Guangdong Laboratory Animal Center, Guangdong, China and included 54 adult male Sprague-Dawley rats (8-9 wk, 300 ± 15 gr) and 12 adult female rats (7-8 wk, 250 ± 13 gr), all of which were in a specific pathogen-free state. The rats were housed in the SPF animal laboratory under controlled conditions of 24 ± 2°C with a 12-hr light/dark cycle.
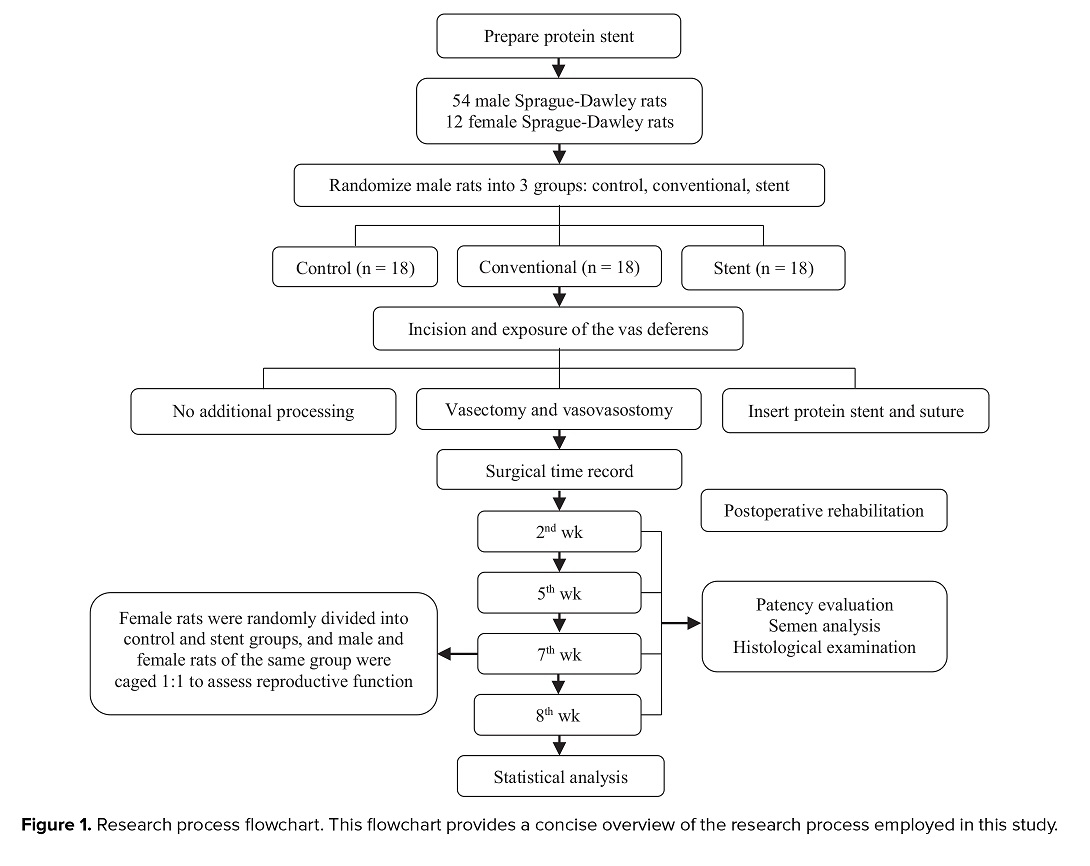
The male rats were randomly assigned to one of 3 groups: control, conventional, and stent. Each of these groups included 18 rats. The female rats were assigned to either the conventional or stent group, with 6 rats in each group. The randomization process ensured that the distribution across groups was equal (Figure 1). The sample size was determined based on multigroup comparisons, multiple time-point measurements, and variability of key outcome indicators. This process involved reference to prior animal studies and guidance from power analysis principles (14). This animal study was designed as a randomized controlled experiment and conducted in accordance with the ARRIVE guidelines (https://arriveguidelines.org). The experimental protocols received ethical approval from the Animal Ethics Committee of the First Affiliated hospital of Guangzhou University of Chinese Medicine (Approval No: TCMF1-2021074). Experiments were performed in the Animal Facility of the First Affiliated hospital of Guangzhou University of Chinese Medicine, Guangzhou, Guangdong Province, China, from January 2023-June 2024.
2.2. Surgical procedure
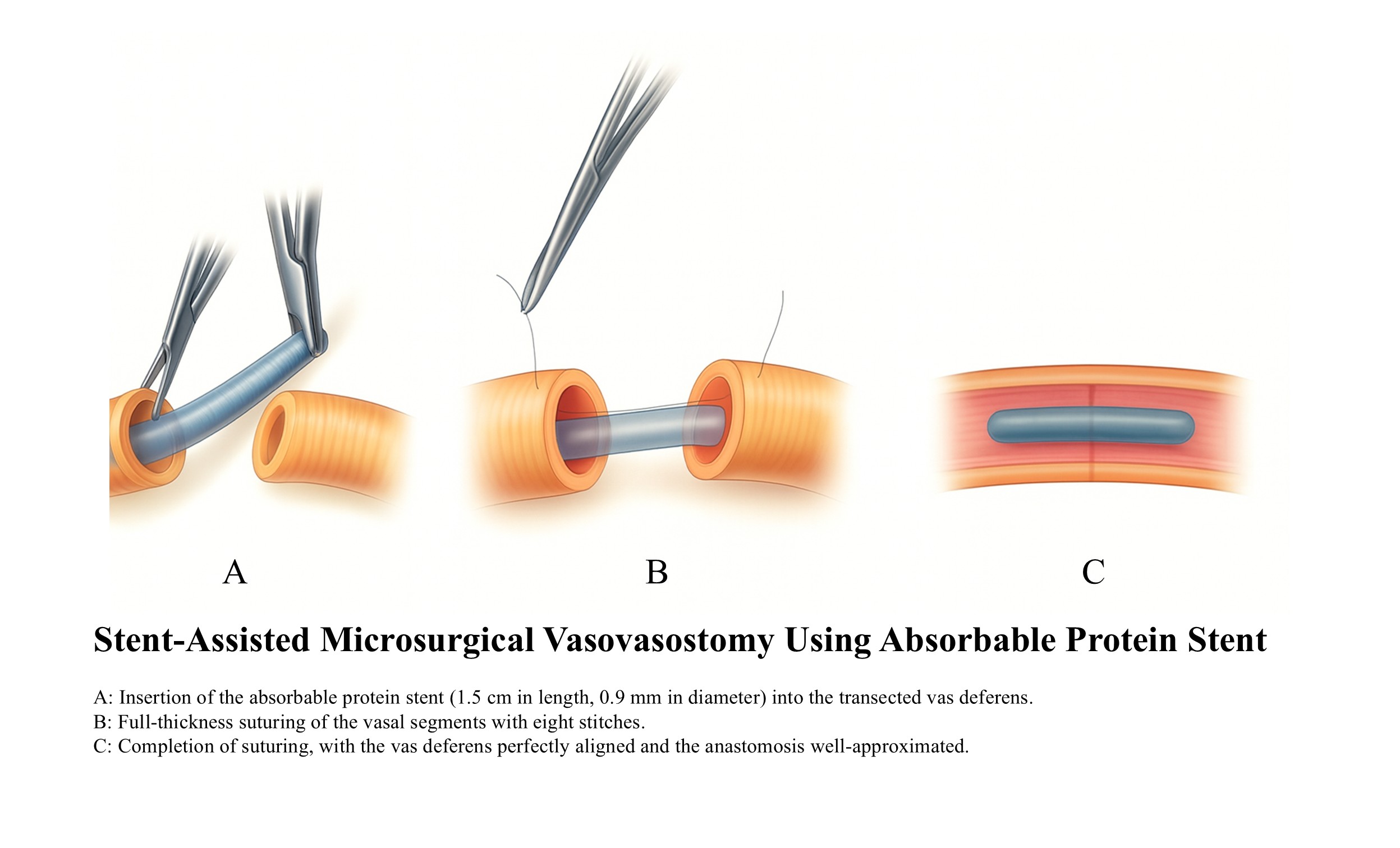
The surgical procedure was conducted in a sterile environment. The anesthetic was induced with 4% isoflurane (RWD LIFE SCIENCE, 100 mL, purity ≥ 98%) and maintained at 2% isoflurane with an oxygen flow rate of 1 L/min. A 1.5 cm abdominal incision was made to expose and ligate the left vas deferens, after which it was returned to its original position. In the control group, following the exposure of the right vas deferens, no further manipulation was conducted. In the conventional group, the right vas deferens was transected, and a 2-layer microsurgical vasovasostomy was performed, with the mucosal and muscular layers separately sutured using 6 and 8 stitches, respectively (15). In the stent group, the right vas deferens was transected, and a prepared protein stent (1.5 cm in length, 0.9 mm in diameter) was employed (Figure 2A). The stent was inserted into the vas deferens with precision, with half of it placed into one end while the mucosal anatomy was preserved. The remaining portion of the stent was then inserted into the opposite segment of the vas deferens. Subsequently, the full-thickness vasal segments were sutured uniformly with 8 stitches. The right vas deferens was repositioned to its original location, and the incision was closed. The duration of the surgical procedure was documented. To ensure procedural consistency, all surgeries were independently performed by the same surgeon (Zhang Hui). Following the surgical procedure, the rats were relocated to a warm, clean, and quiet environment to alleviate any discomfort. The postoperative recovery period was uneventful, with no complications observed (Figure 2B).
2.3. Sample size and timepoint allocation
A total of 54 male Sprague-Dawley rats were randomly divided into 3 groups: control groups, conventional groups, and stent groups, with 18 rats in each group. The groups were pre-allocated to specific timepoints: 6 rats per group were sacrificed and evaluated at postoperative week 2; 6 rats per group were sacrificed and evaluated at postoperative week 5; and the remaining 6 rats per group were sacrificed and evaluated at postoperative week 8, with a total of 18 rats per group. Additionally, 12 female rats were used solely for the cohabitation fertility assessment at postoperative week 7. These rats were co-housed with 6 male rats from the conventional and stent groups (one female rat per male rat) and evaluated at week 8. The control group did not undergo cohabitation. The male rats, after cohabitation at week 7, underwent final evaluation at week 8.
2.4. Postoperative evaluation at weeks 2 and 5: Physiological, immunological, reproductive, and histological assessments
2.4.1. Measurement of physiological parameters
At postoperative weeks 2 and 5, 6 male rats from each group were randomly selected, weighed, and blood samples were collected via tail vein into anticoagulant tubes. The measurements of hemoglobin (Hb) and white blood cell (WBC) counts were conducted using an automated hematology analyzer, in accordance with the instructions provided by the instrument manufacturer (16). Serum alanine aminotransferase (ALT) levels were determined using a biochemical analyser (based on the Reitman-Frankel method) (17), and serum potassium (K⁺) concentration was assessed with an electrolyte analyzer. Serum creatinine (Cr) levels were quantified using a commercially available enzyme-linked immunosorbent assay (ELISA) kit for rats (MEIMIAN, 48T, Guangzhou, China), following the manufacturer's instructions (18).
2.4.2. Measurement of reproductive hormone levels and immunological markers
Blood samples were centrifugated at 3000 rpm for 10 min to obtain the serum. The serum levels of follicle-stimulating hormone, luteinizing hormone, and testosterone were quantified using commercial rat ELISA kits (CUSABIO, Wuhan, China) in accordance with the instructions provided by the manufacturer. Estradiol (E2) concentrations were quantified using a rat ELISA kit (MyBioSource, San Diego, CA, United States) (19). Serum C-reactive protein (CRP) was quantified using a rat ELISA kit (MEIMIAN, Guangzhou, China), and serum immunoglobulin G (IgG) concentration was determined using a rat ELISA kit (LunChangShou Biotech, Guangzhou, China) (20). For anti-sperm antibody (AsAb) titration, serum samples were incubated with primary mouse antihuman AsAb monoclonal IgG and secondary alkaline phosphatase-labeled goat antimouse IgG (Chemicon, Temecula, CA, USA), and subsequently analyzed in accordance with the Quanta Biotech protocol (Surrey, UK) (21).
2.4.3. Patency of the vas deferens, histological evaluation, and epididymal semen analysis
The rats were anesthetized via intraperitoneal injection of 30 mg/kg sodium pentobarbital (Cayman Chemical, 5 mg, purity ≥ 98%). The right vas deferens, right epididymis, and bilateral testes were then exposed and isolated. The patency of the vas deferens was confirmed through a water injection test (Figure 3), with careful observation for any remaining stent remnants or potential obstructions. Subsequently, the vas deferens tissues were fixed in 4% formalin, embedded in paraffin after 48 hr, and sectioned at a thickness of 4 µ for hematoxylin-eosin staining (22). The sections were examined under 40× and 100× magnification with a light microscope (Olympus BX53, Olympus Corporation, Tokyo, Japan) to assess incision sites, vasal wall thickness, epithelial integrity, luminal patency, and the presence of sperm granulomas (13, 23). The right epididymal tissues were minced into small fragments and incubated in a semen-washing medium (Vitrolife, Sweden) at 37°C for 10 min to promote sperm release (24). The processed semen samples were evaluated using a computer-assisted sperm analysis system (HTM-TOX IVOS, Hamilton-Thorne, Beverly, MA, USA) to assess sperm count, motility, and morphology. Additionally, testicular weight was recorded. Upon completion of the aforementioned evaluations, the rats were euthanized with an overdose of sodium pentobarbital (150 mg/kg) administered intravenously.
2.5. Fertility assessment at postoperative week 7
At postoperative week 7, 6 male rats from both the conventional group and the stent group, which were reserved for evaluation at week 8, were co-housed with 12 female rats in a 1:1 ratio for 1 wk. Subsequently, the female rats were housed separately, and pregnancy status was monitored (pregnancy rate, number of offspring, and the number of live births) (25). The control group did not undergo cohabitation. The male rats were then evaluated at the end of week 8.
2.6. Postoperative evaluation at week 8: Physiological, immunological, reproductive, and histological assessments
In the 8 postoperative week, the remaining 6 male rats in each group underwent assessments of physiological, immunological, reproductive, and histological parameters in accordance with the procedures outlined in sections 3.1, 3.2, and 3.3. Once the evaluations had been completed, the animals were euthanized.


2.7. Ethical Considerations
Throughout the research process, we strictly adhered to the 3R principles (replacement, reduction, and refinement) to ensure the ethical integrity and scientific rigor of the experiments. The rats were provided with sterile food and water and housed in a controlled environment at the First Affiliated hospital of Guangzhou University of Chinese Medicine, Guangzhou, China. This study was conducted in accordance with the ARRIVE guidelines (https://arriveguidelines.org) and received ethical approval from the Animal Ethics Committee of the First Affiliated hospital of Guangzhou University of Chinese Medicine, Guangzhou, China (Code: TCMF1-2021074). All experimental procedures involving animals complied with ethical standards for the use of experimental animals. Since this study involved only animal experiments, participant consent is not applicable.
2.8. Statistical Analysis
The data were analyzed using the statistical software package Statistical Package for the Social Sciences version 20.0. Continuous variables were expressed as mean ± standard deviation. To evaluate differences between groups, an independent sample t test and Fisher's exact test were employed. In the case of multiple group comparisons, an ANOVA was employed, followed by Tukey's post hoc test. The level of statistical significance was set at p < 0.05.
3. Results
3.1. Operative time
The conventional group exhibited an average total operative time of 70.4 ± 4.0 min, with 44.1 ± 6.5 min dedicated to mucosal suturing and 26.3 ± 4.6 min to muscular layer suturing. In contrast, the average total operative time for the stent group was 28.8 ± 5.2 min. A significant difference in total operative time was observed between groups (p < 0.001) (Table I), indicating that the use of a stent in microsurgical vasovasostomy is associated with a reduction in the time required for the procedure and an increase in its simplicity compared to conventional 2-layer microsurgical vasovasostomy.
3.2. Patency rate and postoperative complications
The assessment of vas deferens patency was conducted at postoperative weeks 2, 5, and 8 (Figure 3). At week 2, the patency rate in the stent groups was found to be significantly lower than in the conventional groups (0% vs. 83.3%; p < 0.001). By week 5, the patency rate in the stent groups was 83.3%, compared to 100% in the conventional groups (p > 0.999), while at week 8, the stent groups reached a patency rate of 100%, with the conventional groups at 83.3% (p > 0.999) (Table I). A detailed analysis of stent absorption indicated that at week 2, the stent retention rate was 100% (6/6). However, by week 5, this had decreased to 33.3% (2/6), and by week 8, no stent remnants were detected in the vas deferens. This suggests a gradual reduction in stent retention over time. With regard to instances of patency failure, one rat in the stent group developed a sperm granuloma in the vicinity of the vasal suture at week 5. In contrast, another rat in the conventional groups exhibited a similar granuloma near the suture line by week 8 (Table I). The vas deferens of the 2 rats failed the patency test due to the presence of granulomas. Furthermore, both the stent and conventional groups exhibited typical gross anatomical features in the abdominal and inguinal regions, with no discernible masses or adhesions.
3.3. Measurement of blood physiological parameters
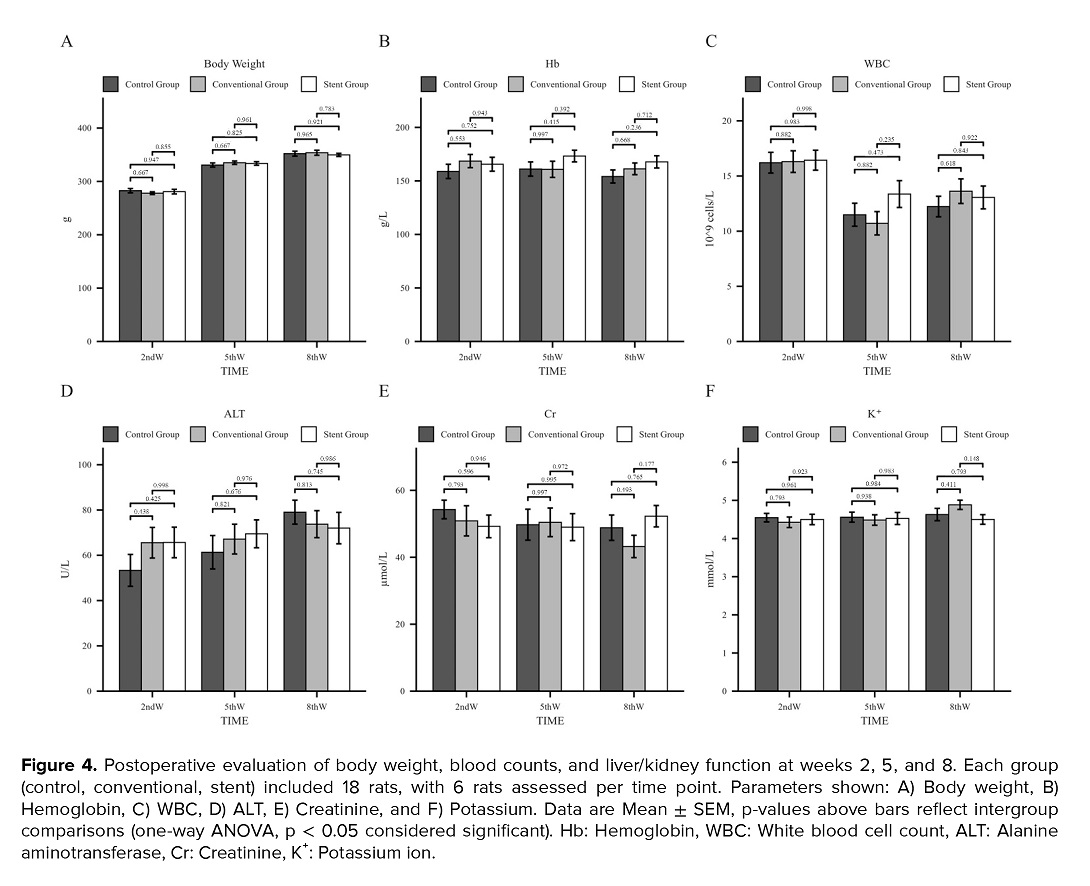
At postoperative weeks 2, 5, and 8, hematological and physiological parameters were monitored in each group of rats. The parameters monitored included body weight (Figure 4A), Hb levels (Figure 4B), WBC counts (Figure 4C), ALT activity (Figure 4D), Cr levels (Figure 4E), and serum K⁺ concentrations (Figure 4F). It is noteworthy that WBC counts exhibited a slight increase across all groups at week 2 postsurgery, which may indicate an early postoperative inflammatory response. However, the differences between the groups were not statistically significant (p > 0.05). Overall, the hematological physiological parameters demonstrated a tendency to stabilize during the postoperative recovery period, with no significant abnormal changes observed. Furthermore, no intergroup differences were identified (p > 0.05).
3.4. Evaluation of histopathology and immune response indicators
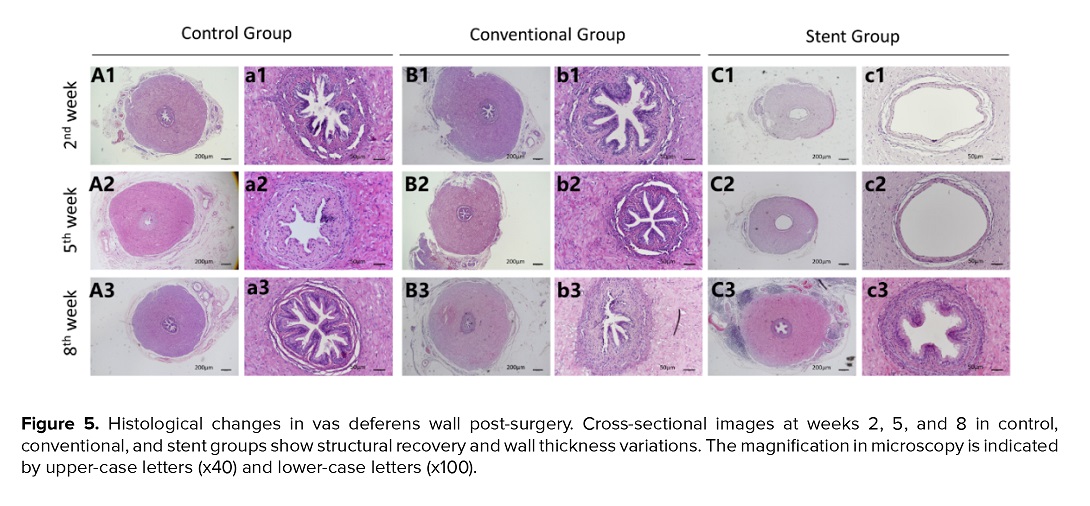
As illustrated in figure 5, by the second postoperative week, the stent had not yet been absorbed. Upon removal of the unabsorbed stent, the vasal mucosa in the stent groups remained intact, with a round and smooth lumen. A significant difference in wall thickness was observed between the 2 groups (205.0 ± 5.3 μm vs. 86.3 ± 5.8 μm, p < 0.001), suggesting that the stent exerted compressive pressure on the vas deferens, potentially preventing sperm leakage. By the fifth week, partial recovery of the mucosa was evident in the stent groups; however, wall thickness remained significantly lower than that of the conventional groups (103.0 ± 6.0 μm vs. 206.3 ± 7.6 μm, p < 0.001). By the 8 week, the vasal structure in the stent group had nearly returned to normal, with a stellate lumen morphology comparable to the conventional groups. No significant difference in wall thickness was noted between the groups (209.3 ± 6.1 μm vs. 205.3 ± 5.7 μm, p > 0.05) (Table I).
In a few rats with failed anastomosis, sperm granulomas were observed in both groups. Histological examination revealed the presence of epithelioid cells, lymphocytes, histiocytes, multinucleated giant cells, and centrally located degenerated spermatozoa (Figure 6). In contrast, no inflammatory swelling of the vas deferens was observed postoperatively (Figure 7A). Serum CRP levels were elevated in the early postoperative phase, indicating an acute inflammatory response, but gradually returned to baseline (Figure 7B). No significant differences were observed in antisperm antibody (AsAb) or IgG levels between the groups (Figure 7C, 7D).
3.5. Assessment of fertility
To evaluate the effects of different postoperative treatments on male reproductive function, epididymal sperm parameters and serum reproductive hormone levels were assessed at 2, 5, and 8 wk postsurgery. Results showed no significant differences among the control, conventional suture, and stent groups in sperm count, motility, or morphology at any time point (p > 0.05) (Figure 8A-C). At week 8, sperm concentrations were 24.6 ± 3.6, 25.6 ± 3.2, and 26.8 ± 3.8 ×10⁶/mL (p > 0.05); sperm motility was 71.2 ± 4.6%, 70.2 ± 3.3%, and 73.5 ± 2.6% (p > 0.05); and sperm malformation rates were 6.0 ± 1.1%, 5.3 ± 0.8%, and 5.3 ± 0.9%, respectively (p > 0.05).
Similarly, no significant differences were observed among groups in serum levels of testosterone, estradiol, luteinizing hormone, or follicle-stimulating hormone (p > 0.05) (Figure 8D-G). At week 8, testosterone levels were 13.2 ± 1.1, 13.3 ± 1.1, and 13.3 ± 1.1 ng/mL (p > 0.05), and estradiol concentrations were 53.9 ± 1.6, 49.9 ± 4.3, and 50.6 ± 5.1 pg/mL (p > 0.05). The weight of the left testis remained stable among groups, with all averaging 1.3 ± 0.2 gr (p > 0.05) (Figure 8H).
To further assess reproductive capacity, male rats were co-housed with females starting from week 7, and pregnancy outcomes were recorded at week 8. The pregnancy rate in both the conventional and stent groups was 83.3%, with average litter sizes of 5.0 ± 2.6 and 5.7 ± 3.2, live births of 4.3 ± 2.3 and 4.8 ± 2.6, and live birth rates of 71.9% and 73.1%, respectively, with no significant differences between groups (p > 0.05) (Figure 8I-L).




4. Discussion
This study innovatively proposed and validated a stent-assisted microsurgical vasovasostomy technique using a biodegradable protein stent, aiming to address the challenges of traditional microsurgical 2-layer vasovasostomy, such as complex procedures, high technical thresholds, and prolonged operative time. The results demonstrated that the protein stent significantly improved surgical efficiency, simplified the suturing process, enhanced postoperative vas patency, and reduced the risk of complications, while exhibiting good tissue compatibility and reproductive system biosafety.
Firstly, in terms of surgical efficiency, we found that the average duration of traditional microsurgical 2-layer vasovasostomy was 70.4 ± 4.0 min, whereas with the protein stent, the procedure took only 28.8 ± 5.2 min, a reduction of over 50%. This significant improvement in efficiency was mainly attributed to the stent’s guiding function during anastomosis, allowing the surgeon to quickly align the vas deferens and reduce the number of sutures required (from 14-8). These results suggest that the protein stent not only simplifies the surgical process but also lowers the technical barrier, making it potentially applicable in healthcare settings with lower surgical proficiency. Moreover, the shortened operative time helps reduce the physical burden on patients and minimizes surgery-related risks (15, 22).
With regard to postoperative patency, the stent group attained patency rates of 83.3% and 100% in postoperative weeks 5 and 8, respectively, which were significantly higher than the 16.7% recorded at week 2. The results obtained from this study suggest that the protein stent exhibits excellent biodegradability. In the early postoperative phase, the nonabsorbed stent exerted moderate pressure around the anastomotic site due to its appropriate diameter, thereby effectively preventing sperm leakage, as confirmed by histological examination. This moderate compression also helped reduce the incidence of postoperative vasal stricture. As the anastomosis healed, the stent was fully absorbed, thereby ensuring long-term patency. This time-dependent improvement trend provides a reference for postoperative clinical management and outcome expectations.
In contrast to preceding studies, certain researchers have endeavored to employ auxiliary materials with a view to reducing the complexity of microsurgical anastomosis. This has included the utilization of absorbable gut sutures as guiding structures (26) and nonabsorbable hollow stents to facilitate alignment (11). However, these materials exhibit significant drawbacks: gut sutures are soft and prone to deformation, making insertion difficult and alignment imprecise, which can lead to ductal distortion or injury during the procedure; nonabsorbable stents may cause chronic inflammation, granuloma formation, or even reduced long-term patency due to foreign body retention. In contrast, the protein stent under investigation in this study is composed of natural amino acids, which offer good plasticity, degradability, and low immunogenicity. The material exhibits a reduction in hardness upon contact with water, thus facilitating insertion into the lumen. The stent provides necessary mechanical support during anastomosis and is completely absorbed postoperatively, thereby avoiding the aforementioned issues.
In terms of biosafety, no significant immune response, abnormal serum hormone levels, or impaired fertility were observed in the stent group of the rat model. The cohabitation test provided further confirmation of normal reproductive capacity and healthy offspring development in rats from the stent group, thereby supporting the hypothesis that the stent exhibits excellent compatibility with the reproductive system. This finding is of crucial significance for the clinical translational potential of the stent in reproductive surgeries.
Although most animals in the stent group exhibited a positive postoperative recovery, localized sperm granulomas were still observed in a few rats at postoperative week 5. This phenomenon may be attributed to the reduced number of sutures or the uneven distribution of local stress. As demonstrated in previous studies, a correlation was observed between low anastomotic precision or significant foreign body stimulation and an increased likelihood of granulomatous tissue proliferation at the vasal anastomotic site (5, 23). Consequently, future research may involve moderately adjusting the suturing strategy, for instance by implementing a symmetrical reinforcement of suture point distribution, without significantly increasing surgical complexity. This would serve to further optimize anastomotic stability and reduce complication risks.
This study also contributes to the exploration of the potential mechanism of “anastomotic site pressure regulation” in postoperative recovery. It was observed that in the early postoperative period, prior to complete stent absorption, the stent applied moderate pressure to the tissues surrounding the anastomotic site. This pressure helped to prevent sperm leakage and promoted tissue adhesion and regeneration at the anastomosis. Histological analysis further corroborated the presence of mild inflammation and favorable tissue remodeling at the anastomotic site, thereby indicating that the mechanical support and degradation timeline of the stent synergistically promote anastomotic healing.
In conclusion, the biodegradable protein stent proposed in this study, as a novel anastomosis-assisting material, demonstrated superior performance over traditional microsurgical vasovasostomy and other auxiliary materials in terms of ease of operation, biosafety, tissue compatibility, and postoperative recovery. The device is particularly well-suited for utilization in minimally invasive microsurgery, in settings where resources are limited, and in the training of novices in surgery. It is recommended that future studies be conducted to explore the potential applications of the subject in other urological or reproductive tubular anastomosis procedures. Furthermore, the promotion of its clinical translation through long-term animal follow-up and preclinical research is advised.
5. Conclusion
Absorbable protein stents achieved patency rates comparable to traditional 2-layer vasovasostomy, while simplifying the procedure, reducing operative time, and maintaining reproductive safety in rats. These results support their potential as an effective alternative in microsurgical vasovasostomy.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon request.
Author Contributions
Y. Zhang and H. Zhang conceptualized and designed the study. H. Zhang, X. Tu, Z. Liu, and J. Feng performed the experiments. X. Tu conducted the statistical analysis of the data and drafted the manuscript. Y. Zhang and H. Zhang supervised the research, providing critical revisions for important intellectual content. Both X. Tu and H. Zhang made equal and substantial contributions to the design, implementation, and interpretation of the study. Therefore, they are both designated as first authors. This shared first authorship reflects their equal leadership roles in driving the research forward and producing the manuscript. All authors had full access to the data and take responsibility for the integrity of the data and the accuracy of the analysis. All authors read and approved the final manuscript.
Acknowledgments
We would like to express our sincere gratitude to the Traditional Chinese Medicine Bureau of Guangdong Province (No. 20221086) and the First Affiliated hospital of Guangzhou University of Chinese Medicine, Guangzhou, China (No. 2022QN23) for their financial support of this study. We also acknowledge that artificial intelligence tools were not used in this study for translation, proofreading, or grammar checking. All work was performed manually by the authors. The study was conducted in Guangzhou, China.
Conflict of Interest
The authors declare that there is no conflict of interest.
Microsurgical vasovasostomy is acknowledged as a highly effective and cost-efficient procedure, but requires extensive training (1, 2). Since its initial proposal by Silber in the 1970s, microsurgical vasovasostomy has emerged as the gold standard technique for vas deferens anastomosis (3, 4). Despite the effectiveness demonstrated by the traditional 2-layer anastomosis technique, it is limited by its extensive surgical duration and high technical demands.
To simplify the surgical procedure and reduce operation time, several researchers have proposed a single-layer anastomosis technique. Although this method reduces surgical complexity, it may lead to inadequate mucosal anastomosis of the vas deferens, potentially causing stenosis or obstruction at the anastomosis site, as well as granuloma formation, thereby affecting postoperative patency and pregnancy rates (5-7). Moreover, in recent years, robot-assisted vas deferens anastomosis has gained attention due to its potential to enhance surgical precision and reduce technical challenges. However, the application of this technology remains limited due to high costs and the absence of direct tactile feedback (8-10).
Stent-assisted anastomosis offers several advantages, including the capacity for customization according to the condition of both vasal ends, and the structural support provided by the stent that facilitates more accurate suturing. Vrijhof previously proposed a novel non-absorbable hollow vas deferens stent, which demonstrated excellent surgical efficiency and favorable patency (11). Nevertheless, the persistent presence of non-absorbable materials gives rise to concerns regarding long-term safety, thus necessitating continuous follow-up.
In recent years, increasing application of biodegradable materials has been observed in the field of andrology. For instance, the utilization of biodegradable stents in urological reconstruction has been demonstrated to exhibit excellent biocompatibility and absorbability (12). These developments provide a solid theoretical foundation and promising prospects for exploring new materials for vas deferens stents.
In this study, rats were selected as the experimental model based on their anatomical and functional similarity of the vas deferens to humans, combined with low ethical burden and ease of manipulation (13). The objective of this study was to provide experimental evidence and theoretical support for further optimizing minimally invasive fertility restoration surgery by evaluating the application of a biodegradable protein stent in microsurgical vasovasostomy.
2. Materials and Methods
2.1. Preparation of the stent and animals
This study introduces an absorbable, natural protein stent derived from otter tail tendons (Medical Device Registration No: CFDA 20173654672) for use in stent-assisted microsurgical vasovasostomy for the first time. Developed and provided by Hunan Ranyuan Medical High-tech Protein Suture Co., Ltd., the stent represents an innovative strategy in the application of biomaterials. It is refined from structural proteins (mainly collagen, with trace amounts of elastin), which are found in abundance in natural tendon tissue. The preparation process involves ethanol dehydration, impurity removal using sodium hydroxide, multiple purification steps, and low-temperature sterilization to maximally preserve the natural conformation and biological activity of the proteins. The resulting material offers excellent tissue affinity and biodegradability. After implantation, the stent gradually degrades into nontoxic amino acid molecules within 4-8 wk, thereby avoiding chronic inflammatory responses caused by long-term foreign body retention. Its unique microfibrous mesh structure provides sufficient initial mechanical support to maintain the stability of the anastomosis and acts as a biological scaffold during degradation. This guides the regeneration of surrounding tissue, promoting cell adhesion, proliferation, and organized reconstruction. This realizes a true ‘stent-guidance-disappearance’ tri-functional transformation. Compared to nondegradable synthetic materials or traditional microsurgical 2-layer vasovasostomy, this protein stent has significant advantages in terms of supporting precise tissue reconstruction, reducing foreign body reactions, and lowering the risk of postoperative complications, particularly in microsurgical procedures where high levels of precision and biocompatibility are essential.
The experimental animals were obtained from the Guangdong Laboratory Animal Center, Guangdong, China and included 54 adult male Sprague-Dawley rats (8-9 wk, 300 ± 15 gr) and 12 adult female rats (7-8 wk, 250 ± 13 gr), all of which were in a specific pathogen-free state. The rats were housed in the SPF animal laboratory under controlled conditions of 24 ± 2°C with a 12-hr light/dark cycle.
The male rats were randomly assigned to one of 3 groups: control, conventional, and stent. Each of these groups included 18 rats. The female rats were assigned to either the conventional or stent group, with 6 rats in each group. The randomization process ensured that the distribution across groups was equal (Figure 1). The sample size was determined based on multigroup comparisons, multiple time-point measurements, and variability of key outcome indicators. This process involved reference to prior animal studies and guidance from power analysis principles (14). This animal study was designed as a randomized controlled experiment and conducted in accordance with the ARRIVE guidelines (https://arriveguidelines.org). The experimental protocols received ethical approval from the Animal Ethics Committee of the First Affiliated hospital of Guangzhou University of Chinese Medicine (Approval No: TCMF1-2021074). Experiments were performed in the Animal Facility of the First Affiliated hospital of Guangzhou University of Chinese Medicine, Guangzhou, Guangdong Province, China, from January 2023-June 2024.
2.2. Surgical procedure
The surgical procedure was conducted in a sterile environment. The anesthetic was induced with 4% isoflurane (RWD LIFE SCIENCE, 100 mL, purity ≥ 98%) and maintained at 2% isoflurane with an oxygen flow rate of 1 L/min. A 1.5 cm abdominal incision was made to expose and ligate the left vas deferens, after which it was returned to its original position. In the control group, following the exposure of the right vas deferens, no further manipulation was conducted. In the conventional group, the right vas deferens was transected, and a 2-layer microsurgical vasovasostomy was performed, with the mucosal and muscular layers separately sutured using 6 and 8 stitches, respectively (15). In the stent group, the right vas deferens was transected, and a prepared protein stent (1.5 cm in length, 0.9 mm in diameter) was employed (Figure 2A). The stent was inserted into the vas deferens with precision, with half of it placed into one end while the mucosal anatomy was preserved. The remaining portion of the stent was then inserted into the opposite segment of the vas deferens. Subsequently, the full-thickness vasal segments were sutured uniformly with 8 stitches. The right vas deferens was repositioned to its original location, and the incision was closed. The duration of the surgical procedure was documented. To ensure procedural consistency, all surgeries were independently performed by the same surgeon (Zhang Hui). Following the surgical procedure, the rats were relocated to a warm, clean, and quiet environment to alleviate any discomfort. The postoperative recovery period was uneventful, with no complications observed (Figure 2B).
2.3. Sample size and timepoint allocation
A total of 54 male Sprague-Dawley rats were randomly divided into 3 groups: control groups, conventional groups, and stent groups, with 18 rats in each group. The groups were pre-allocated to specific timepoints: 6 rats per group were sacrificed and evaluated at postoperative week 2; 6 rats per group were sacrificed and evaluated at postoperative week 5; and the remaining 6 rats per group were sacrificed and evaluated at postoperative week 8, with a total of 18 rats per group. Additionally, 12 female rats were used solely for the cohabitation fertility assessment at postoperative week 7. These rats were co-housed with 6 male rats from the conventional and stent groups (one female rat per male rat) and evaluated at week 8. The control group did not undergo cohabitation. The male rats, after cohabitation at week 7, underwent final evaluation at week 8.
2.4. Postoperative evaluation at weeks 2 and 5: Physiological, immunological, reproductive, and histological assessments
2.4.1. Measurement of physiological parameters
At postoperative weeks 2 and 5, 6 male rats from each group were randomly selected, weighed, and blood samples were collected via tail vein into anticoagulant tubes. The measurements of hemoglobin (Hb) and white blood cell (WBC) counts were conducted using an automated hematology analyzer, in accordance with the instructions provided by the instrument manufacturer (16). Serum alanine aminotransferase (ALT) levels were determined using a biochemical analyser (based on the Reitman-Frankel method) (17), and serum potassium (K⁺) concentration was assessed with an electrolyte analyzer. Serum creatinine (Cr) levels were quantified using a commercially available enzyme-linked immunosorbent assay (ELISA) kit for rats (MEIMIAN, 48T, Guangzhou, China), following the manufacturer's instructions (18).
2.4.2. Measurement of reproductive hormone levels and immunological markers
Blood samples were centrifugated at 3000 rpm for 10 min to obtain the serum. The serum levels of follicle-stimulating hormone, luteinizing hormone, and testosterone were quantified using commercial rat ELISA kits (CUSABIO, Wuhan, China) in accordance with the instructions provided by the manufacturer. Estradiol (E2) concentrations were quantified using a rat ELISA kit (MyBioSource, San Diego, CA, United States) (19). Serum C-reactive protein (CRP) was quantified using a rat ELISA kit (MEIMIAN, Guangzhou, China), and serum immunoglobulin G (IgG) concentration was determined using a rat ELISA kit (LunChangShou Biotech, Guangzhou, China) (20). For anti-sperm antibody (AsAb) titration, serum samples were incubated with primary mouse antihuman AsAb monoclonal IgG and secondary alkaline phosphatase-labeled goat antimouse IgG (Chemicon, Temecula, CA, USA), and subsequently analyzed in accordance with the Quanta Biotech protocol (Surrey, UK) (21).
2.4.3. Patency of the vas deferens, histological evaluation, and epididymal semen analysis
The rats were anesthetized via intraperitoneal injection of 30 mg/kg sodium pentobarbital (Cayman Chemical, 5 mg, purity ≥ 98%). The right vas deferens, right epididymis, and bilateral testes were then exposed and isolated. The patency of the vas deferens was confirmed through a water injection test (Figure 3), with careful observation for any remaining stent remnants or potential obstructions. Subsequently, the vas deferens tissues were fixed in 4% formalin, embedded in paraffin after 48 hr, and sectioned at a thickness of 4 µ for hematoxylin-eosin staining (22). The sections were examined under 40× and 100× magnification with a light microscope (Olympus BX53, Olympus Corporation, Tokyo, Japan) to assess incision sites, vasal wall thickness, epithelial integrity, luminal patency, and the presence of sperm granulomas (13, 23). The right epididymal tissues were minced into small fragments and incubated in a semen-washing medium (Vitrolife, Sweden) at 37°C for 10 min to promote sperm release (24). The processed semen samples were evaluated using a computer-assisted sperm analysis system (HTM-TOX IVOS, Hamilton-Thorne, Beverly, MA, USA) to assess sperm count, motility, and morphology. Additionally, testicular weight was recorded. Upon completion of the aforementioned evaluations, the rats were euthanized with an overdose of sodium pentobarbital (150 mg/kg) administered intravenously.
2.5. Fertility assessment at postoperative week 7
At postoperative week 7, 6 male rats from both the conventional group and the stent group, which were reserved for evaluation at week 8, were co-housed with 12 female rats in a 1:1 ratio for 1 wk. Subsequently, the female rats were housed separately, and pregnancy status was monitored (pregnancy rate, number of offspring, and the number of live births) (25). The control group did not undergo cohabitation. The male rats were then evaluated at the end of week 8.
2.6. Postoperative evaluation at week 8: Physiological, immunological, reproductive, and histological assessments
In the 8 postoperative week, the remaining 6 male rats in each group underwent assessments of physiological, immunological, reproductive, and histological parameters in accordance with the procedures outlined in sections 3.1, 3.2, and 3.3. Once the evaluations had been completed, the animals were euthanized.



2.7. Ethical Considerations
Throughout the research process, we strictly adhered to the 3R principles (replacement, reduction, and refinement) to ensure the ethical integrity and scientific rigor of the experiments. The rats were provided with sterile food and water and housed in a controlled environment at the First Affiliated hospital of Guangzhou University of Chinese Medicine, Guangzhou, China. This study was conducted in accordance with the ARRIVE guidelines (https://arriveguidelines.org) and received ethical approval from the Animal Ethics Committee of the First Affiliated hospital of Guangzhou University of Chinese Medicine, Guangzhou, China (Code: TCMF1-2021074). All experimental procedures involving animals complied with ethical standards for the use of experimental animals. Since this study involved only animal experiments, participant consent is not applicable.
2.8. Statistical Analysis
The data were analyzed using the statistical software package Statistical Package for the Social Sciences version 20.0. Continuous variables were expressed as mean ± standard deviation. To evaluate differences between groups, an independent sample t test and Fisher's exact test were employed. In the case of multiple group comparisons, an ANOVA was employed, followed by Tukey's post hoc test. The level of statistical significance was set at p < 0.05.
3. Results
3.1. Operative time
The conventional group exhibited an average total operative time of 70.4 ± 4.0 min, with 44.1 ± 6.5 min dedicated to mucosal suturing and 26.3 ± 4.6 min to muscular layer suturing. In contrast, the average total operative time for the stent group was 28.8 ± 5.2 min. A significant difference in total operative time was observed between groups (p < 0.001) (Table I), indicating that the use of a stent in microsurgical vasovasostomy is associated with a reduction in the time required for the procedure and an increase in its simplicity compared to conventional 2-layer microsurgical vasovasostomy.
3.2. Patency rate and postoperative complications
The assessment of vas deferens patency was conducted at postoperative weeks 2, 5, and 8 (Figure 3). At week 2, the patency rate in the stent groups was found to be significantly lower than in the conventional groups (0% vs. 83.3%; p < 0.001). By week 5, the patency rate in the stent groups was 83.3%, compared to 100% in the conventional groups (p > 0.999), while at week 8, the stent groups reached a patency rate of 100%, with the conventional groups at 83.3% (p > 0.999) (Table I). A detailed analysis of stent absorption indicated that at week 2, the stent retention rate was 100% (6/6). However, by week 5, this had decreased to 33.3% (2/6), and by week 8, no stent remnants were detected in the vas deferens. This suggests a gradual reduction in stent retention over time. With regard to instances of patency failure, one rat in the stent group developed a sperm granuloma in the vicinity of the vasal suture at week 5. In contrast, another rat in the conventional groups exhibited a similar granuloma near the suture line by week 8 (Table I). The vas deferens of the 2 rats failed the patency test due to the presence of granulomas. Furthermore, both the stent and conventional groups exhibited typical gross anatomical features in the abdominal and inguinal regions, with no discernible masses or adhesions.
3.3. Measurement of blood physiological parameters
At postoperative weeks 2, 5, and 8, hematological and physiological parameters were monitored in each group of rats. The parameters monitored included body weight (Figure 4A), Hb levels (Figure 4B), WBC counts (Figure 4C), ALT activity (Figure 4D), Cr levels (Figure 4E), and serum K⁺ concentrations (Figure 4F). It is noteworthy that WBC counts exhibited a slight increase across all groups at week 2 postsurgery, which may indicate an early postoperative inflammatory response. However, the differences between the groups were not statistically significant (p > 0.05). Overall, the hematological physiological parameters demonstrated a tendency to stabilize during the postoperative recovery period, with no significant abnormal changes observed. Furthermore, no intergroup differences were identified (p > 0.05).
3.4. Evaluation of histopathology and immune response indicators
As illustrated in figure 5, by the second postoperative week, the stent had not yet been absorbed. Upon removal of the unabsorbed stent, the vasal mucosa in the stent groups remained intact, with a round and smooth lumen. A significant difference in wall thickness was observed between the 2 groups (205.0 ± 5.3 μm vs. 86.3 ± 5.8 μm, p < 0.001), suggesting that the stent exerted compressive pressure on the vas deferens, potentially preventing sperm leakage. By the fifth week, partial recovery of the mucosa was evident in the stent groups; however, wall thickness remained significantly lower than that of the conventional groups (103.0 ± 6.0 μm vs. 206.3 ± 7.6 μm, p < 0.001). By the 8 week, the vasal structure in the stent group had nearly returned to normal, with a stellate lumen morphology comparable to the conventional groups. No significant difference in wall thickness was noted between the groups (209.3 ± 6.1 μm vs. 205.3 ± 5.7 μm, p > 0.05) (Table I).
In a few rats with failed anastomosis, sperm granulomas were observed in both groups. Histological examination revealed the presence of epithelioid cells, lymphocytes, histiocytes, multinucleated giant cells, and centrally located degenerated spermatozoa (Figure 6). In contrast, no inflammatory swelling of the vas deferens was observed postoperatively (Figure 7A). Serum CRP levels were elevated in the early postoperative phase, indicating an acute inflammatory response, but gradually returned to baseline (Figure 7B). No significant differences were observed in antisperm antibody (AsAb) or IgG levels between the groups (Figure 7C, 7D).
3.5. Assessment of fertility
To evaluate the effects of different postoperative treatments on male reproductive function, epididymal sperm parameters and serum reproductive hormone levels were assessed at 2, 5, and 8 wk postsurgery. Results showed no significant differences among the control, conventional suture, and stent groups in sperm count, motility, or morphology at any time point (p > 0.05) (Figure 8A-C). At week 8, sperm concentrations were 24.6 ± 3.6, 25.6 ± 3.2, and 26.8 ± 3.8 ×10⁶/mL (p > 0.05); sperm motility was 71.2 ± 4.6%, 70.2 ± 3.3%, and 73.5 ± 2.6% (p > 0.05); and sperm malformation rates were 6.0 ± 1.1%, 5.3 ± 0.8%, and 5.3 ± 0.9%, respectively (p > 0.05).
Similarly, no significant differences were observed among groups in serum levels of testosterone, estradiol, luteinizing hormone, or follicle-stimulating hormone (p > 0.05) (Figure 8D-G). At week 8, testosterone levels were 13.2 ± 1.1, 13.3 ± 1.1, and 13.3 ± 1.1 ng/mL (p > 0.05), and estradiol concentrations were 53.9 ± 1.6, 49.9 ± 4.3, and 50.6 ± 5.1 pg/mL (p > 0.05). The weight of the left testis remained stable among groups, with all averaging 1.3 ± 0.2 gr (p > 0.05) (Figure 8H).
To further assess reproductive capacity, male rats were co-housed with females starting from week 7, and pregnancy outcomes were recorded at week 8. The pregnancy rate in both the conventional and stent groups was 83.3%, with average litter sizes of 5.0 ± 2.6 and 5.7 ± 3.2, live births of 4.3 ± 2.3 and 4.8 ± 2.6, and live birth rates of 71.9% and 73.1%, respectively, with no significant differences between groups (p > 0.05) (Figure 8I-L).






4. Discussion
This study innovatively proposed and validated a stent-assisted microsurgical vasovasostomy technique using a biodegradable protein stent, aiming to address the challenges of traditional microsurgical 2-layer vasovasostomy, such as complex procedures, high technical thresholds, and prolonged operative time. The results demonstrated that the protein stent significantly improved surgical efficiency, simplified the suturing process, enhanced postoperative vas patency, and reduced the risk of complications, while exhibiting good tissue compatibility and reproductive system biosafety.
Firstly, in terms of surgical efficiency, we found that the average duration of traditional microsurgical 2-layer vasovasostomy was 70.4 ± 4.0 min, whereas with the protein stent, the procedure took only 28.8 ± 5.2 min, a reduction of over 50%. This significant improvement in efficiency was mainly attributed to the stent’s guiding function during anastomosis, allowing the surgeon to quickly align the vas deferens and reduce the number of sutures required (from 14-8). These results suggest that the protein stent not only simplifies the surgical process but also lowers the technical barrier, making it potentially applicable in healthcare settings with lower surgical proficiency. Moreover, the shortened operative time helps reduce the physical burden on patients and minimizes surgery-related risks (15, 22).
With regard to postoperative patency, the stent group attained patency rates of 83.3% and 100% in postoperative weeks 5 and 8, respectively, which were significantly higher than the 16.7% recorded at week 2. The results obtained from this study suggest that the protein stent exhibits excellent biodegradability. In the early postoperative phase, the nonabsorbed stent exerted moderate pressure around the anastomotic site due to its appropriate diameter, thereby effectively preventing sperm leakage, as confirmed by histological examination. This moderate compression also helped reduce the incidence of postoperative vasal stricture. As the anastomosis healed, the stent was fully absorbed, thereby ensuring long-term patency. This time-dependent improvement trend provides a reference for postoperative clinical management and outcome expectations.
In contrast to preceding studies, certain researchers have endeavored to employ auxiliary materials with a view to reducing the complexity of microsurgical anastomosis. This has included the utilization of absorbable gut sutures as guiding structures (26) and nonabsorbable hollow stents to facilitate alignment (11). However, these materials exhibit significant drawbacks: gut sutures are soft and prone to deformation, making insertion difficult and alignment imprecise, which can lead to ductal distortion or injury during the procedure; nonabsorbable stents may cause chronic inflammation, granuloma formation, or even reduced long-term patency due to foreign body retention. In contrast, the protein stent under investigation in this study is composed of natural amino acids, which offer good plasticity, degradability, and low immunogenicity. The material exhibits a reduction in hardness upon contact with water, thus facilitating insertion into the lumen. The stent provides necessary mechanical support during anastomosis and is completely absorbed postoperatively, thereby avoiding the aforementioned issues.
In terms of biosafety, no significant immune response, abnormal serum hormone levels, or impaired fertility were observed in the stent group of the rat model. The cohabitation test provided further confirmation of normal reproductive capacity and healthy offspring development in rats from the stent group, thereby supporting the hypothesis that the stent exhibits excellent compatibility with the reproductive system. This finding is of crucial significance for the clinical translational potential of the stent in reproductive surgeries.
Although most animals in the stent group exhibited a positive postoperative recovery, localized sperm granulomas were still observed in a few rats at postoperative week 5. This phenomenon may be attributed to the reduced number of sutures or the uneven distribution of local stress. As demonstrated in previous studies, a correlation was observed between low anastomotic precision or significant foreign body stimulation and an increased likelihood of granulomatous tissue proliferation at the vasal anastomotic site (5, 23). Consequently, future research may involve moderately adjusting the suturing strategy, for instance by implementing a symmetrical reinforcement of suture point distribution, without significantly increasing surgical complexity. This would serve to further optimize anastomotic stability and reduce complication risks.
This study also contributes to the exploration of the potential mechanism of “anastomotic site pressure regulation” in postoperative recovery. It was observed that in the early postoperative period, prior to complete stent absorption, the stent applied moderate pressure to the tissues surrounding the anastomotic site. This pressure helped to prevent sperm leakage and promoted tissue adhesion and regeneration at the anastomosis. Histological analysis further corroborated the presence of mild inflammation and favorable tissue remodeling at the anastomotic site, thereby indicating that the mechanical support and degradation timeline of the stent synergistically promote anastomotic healing.
In conclusion, the biodegradable protein stent proposed in this study, as a novel anastomosis-assisting material, demonstrated superior performance over traditional microsurgical vasovasostomy and other auxiliary materials in terms of ease of operation, biosafety, tissue compatibility, and postoperative recovery. The device is particularly well-suited for utilization in minimally invasive microsurgery, in settings where resources are limited, and in the training of novices in surgery. It is recommended that future studies be conducted to explore the potential applications of the subject in other urological or reproductive tubular anastomosis procedures. Furthermore, the promotion of its clinical translation through long-term animal follow-up and preclinical research is advised.
5. Conclusion
Absorbable protein stents achieved patency rates comparable to traditional 2-layer vasovasostomy, while simplifying the procedure, reducing operative time, and maintaining reproductive safety in rats. These results support their potential as an effective alternative in microsurgical vasovasostomy.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon request.
Author Contributions
Y. Zhang and H. Zhang conceptualized and designed the study. H. Zhang, X. Tu, Z. Liu, and J. Feng performed the experiments. X. Tu conducted the statistical analysis of the data and drafted the manuscript. Y. Zhang and H. Zhang supervised the research, providing critical revisions for important intellectual content. Both X. Tu and H. Zhang made equal and substantial contributions to the design, implementation, and interpretation of the study. Therefore, they are both designated as first authors. This shared first authorship reflects their equal leadership roles in driving the research forward and producing the manuscript. All authors had full access to the data and take responsibility for the integrity of the data and the accuracy of the analysis. All authors read and approved the final manuscript.
Acknowledgments
We would like to express our sincere gratitude to the Traditional Chinese Medicine Bureau of Guangdong Province (No. 20221086) and the First Affiliated hospital of Guangzhou University of Chinese Medicine, Guangzhou, China (No. 2022QN23) for their financial support of this study. We also acknowledge that artificial intelligence tools were not used in this study for translation, proofreading, or grammar checking. All work was performed manually by the authors. The study was conducted in Guangzhou, China.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Andrology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |