Fri, Apr 24, 2026
[Archive]
Volume 24, Issue 2 (February 2026)
IJRM 2026, 24(2): 127-144 |
Back to browse issues page
Ethics code: IR.SSU.AEC.1401.027
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Azizi Khareshki M, Hosseini-sharifabad M, Fesahat F, Talebi A, Hassanpour Dehnavi A. Therapeutic effects of Foeniculum vulgare and Achillea millefolium on ovarian function in a mouse model of polycystic ovary syndrome: An experimental study. IJRM 2026; 24 (2) :127-144
URL: http://ijrm.ir/article-1-3562-en.html
URL: http://ijrm.ir/article-1-3562-en.html
Mohadeseh Azizi Khareshki1 

 , Mohamad Hosseini-sharifabad1
, Mohamad Hosseini-sharifabad1 

 , Farzaneh Fesahat2
, Farzaneh Fesahat2 

 , Alireza Talebi3
, Alireza Talebi3 

 , Azam Hassanpour Dehnavi *4
, Azam Hassanpour Dehnavi *4 




 , Mohamad Hosseini-sharifabad1
, Mohamad Hosseini-sharifabad1 

 , Farzaneh Fesahat2
, Farzaneh Fesahat2 

 , Alireza Talebi3
, Alireza Talebi3 

 , Azam Hassanpour Dehnavi *4
, Azam Hassanpour Dehnavi *4 


1- Department of Anatomical Sciences, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
2- Reproductive Immunology Research Center, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Research and Clinical Center for Infertility, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
4- Department of Anatomical Sciences, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Reproductive Immunology Research Center, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,a.hassanpour@stu.ssu.ac.ir
2- Reproductive Immunology Research Center, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Research and Clinical Center for Infertility, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
4- Department of Anatomical Sciences, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Reproductive Immunology Research Center, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,
Full-Text [PDF 5449 kb]
(160 Downloads)
| Abstract (HTML) (246 Views)
1. Introduction
Polycystic ovary syndrome (PCOS) is a common endocrine disorder that affects 4-12% of women of reproductive age and is one of the leading causes of infertility. It is characterized by hormonal imbalances, insulin resistance (IR), ovarian dysfunction, and impaired folliculogenesis. PCOS has a major impact on the health and quality of life of women. It also causes psychological distress, leading to a lower quality of life compared to healthy individuals (1, 2). Given the high prevalence of PCOS, current treatments like clomiphene citrate and metformin have limitations, especially for overweight or obese women. These treatments also come with various side effects, making it essential to find alternative therapies (3).
Emerging evidence indicates that genes such as insulin receptor (INSR), KIT ligand (KITL), chemerin (CM), and chemokine-like receptor 1 (CMKLR1) are crucial in PCOS development. CM and CMKLR1 are involved in inflammation and metabolism, leading to IR and hormonal imbalance. INSR maintains insulin sensitivity and metabolic balance, while KITL is essential for folliculogenesis and oocyte maturation. Dysregulation of these genes in PCOS contributes to impaired follicular development, elevated androgen levels, and metabolic dysfunction (4-7).
Plants have long been used to address human fertility issues, and one such plants are Foeniculum vulgare (fennel) and Achillea millefolium (yarrow) (8, 9). Fennel is a medicinal plant of the Apiaceae family and belongs to the genus Foeniculum, native to the Mediterranean region and widely cultivated throughout the world. This aromatic plant has yellow flowers, thin leaves, and seeds rich in essential oils. The main constituents of its oil include anethole, fenchone, and estragole, which have many medicinal properties. In traditional medicine, fennel is used as an antispasmodic, milk-inducing, menstrual regulator, and for the treatment of gynecological disorders (10, 11). Fennel is rich in phytoestrogen and can imitate estrogen. It regulates hormones, stimulates follicle growth, and thickens the endometrium. Studies report more ovarian follicles, higher progesterone, and fewer PCOS symptoms, such as IR and anovulation, after fennel extract intake (12). Yarrow is a perennial plant of the Asteraceae family that has been used in traditional medicine for over 3000 yr due to its extensive medicinal properties. The plant grows widely in temperate regions of the world and is rich in bioactive compounds, including flavonoids, phenols, tannins, and essential oils. Yarrow is known as a plant with anti-inflammatory, antibacterial, antioxidant, and estrogenic effects and is used in the treatment of digestive disorders, gynecological diseases, wounds, and to reduce menstrual pain (13). A study has shown that yarrow extract, with its flavonoids and phenolic compounds, improves hormonal balance, reduces androgen levels, and increases estrogen. These effects help to enhance folliculogenesis and improve ovarian function in women with PCOS. Also, the antioxidant properties of yarrow have beneficial effects on infertility by reducing oxidative stress in ovarian tissue and regulating the menstrual cycle (9).
This study aims to investigate the combined effects of fennel and yarrow on ovarian histomorphology, folliculogenesis-related gene expression, and hormonal parameters in a PCOS model.
2. Materials and Methods
2.1. Study design
Full-Text: (9 Views)
1. Introduction
Polycystic ovary syndrome (PCOS) is a common endocrine disorder that affects 4-12% of women of reproductive age and is one of the leading causes of infertility. It is characterized by hormonal imbalances, insulin resistance (IR), ovarian dysfunction, and impaired folliculogenesis. PCOS has a major impact on the health and quality of life of women. It also causes psychological distress, leading to a lower quality of life compared to healthy individuals (1, 2). Given the high prevalence of PCOS, current treatments like clomiphene citrate and metformin have limitations, especially for overweight or obese women. These treatments also come with various side effects, making it essential to find alternative therapies (3).
Emerging evidence indicates that genes such as insulin receptor (INSR), KIT ligand (KITL), chemerin (CM), and chemokine-like receptor 1 (CMKLR1) are crucial in PCOS development. CM and CMKLR1 are involved in inflammation and metabolism, leading to IR and hormonal imbalance. INSR maintains insulin sensitivity and metabolic balance, while KITL is essential for folliculogenesis and oocyte maturation. Dysregulation of these genes in PCOS contributes to impaired follicular development, elevated androgen levels, and metabolic dysfunction (4-7).
Plants have long been used to address human fertility issues, and one such plants are Foeniculum vulgare (fennel) and Achillea millefolium (yarrow) (8, 9). Fennel is a medicinal plant of the Apiaceae family and belongs to the genus Foeniculum, native to the Mediterranean region and widely cultivated throughout the world. This aromatic plant has yellow flowers, thin leaves, and seeds rich in essential oils. The main constituents of its oil include anethole, fenchone, and estragole, which have many medicinal properties. In traditional medicine, fennel is used as an antispasmodic, milk-inducing, menstrual regulator, and for the treatment of gynecological disorders (10, 11). Fennel is rich in phytoestrogen and can imitate estrogen. It regulates hormones, stimulates follicle growth, and thickens the endometrium. Studies report more ovarian follicles, higher progesterone, and fewer PCOS symptoms, such as IR and anovulation, after fennel extract intake (12). Yarrow is a perennial plant of the Asteraceae family that has been used in traditional medicine for over 3000 yr due to its extensive medicinal properties. The plant grows widely in temperate regions of the world and is rich in bioactive compounds, including flavonoids, phenols, tannins, and essential oils. Yarrow is known as a plant with anti-inflammatory, antibacterial, antioxidant, and estrogenic effects and is used in the treatment of digestive disorders, gynecological diseases, wounds, and to reduce menstrual pain (13). A study has shown that yarrow extract, with its flavonoids and phenolic compounds, improves hormonal balance, reduces androgen levels, and increases estrogen. These effects help to enhance folliculogenesis and improve ovarian function in women with PCOS. Also, the antioxidant properties of yarrow have beneficial effects on infertility by reducing oxidative stress in ovarian tissue and regulating the menstrual cycle (9).
This study aims to investigate the combined effects of fennel and yarrow on ovarian histomorphology, folliculogenesis-related gene expression, and hormonal parameters in a PCOS model.
2. Materials and Methods
2.1. Study design
This animal experiment study was carried out in the Department of Anatomical Sciences and Cell Biology, Shahid Sadoughi University of Medical Sciences, Yazd, Iran, from January to June 2024.
2.2. Plant material and extraction
2.2. Plant material and extraction
Dried fennel (Herbarium code: aau115) and yarrow (Herbarium code: ssu116) were purchased from an herbal shop in Isfahan, Iran. Botanical identity was authenticated by the herbarium expert of the Shahid Sadoughi University of Medical Sciences, Yazd, Iran. For extraction, fennel seeds and the flowers and terminal stems of yarrow were used. The plants were finely powdered using an electric grinder. To prepare the hydroalcoholic extract, the plant powder was soaked in 70% ethanol in a 1:1 ratio (w/v). The mixture was kept at a maximum temperature of 30°C for 24 hr. During this time, the mixture was periodically stirred using a glass stirrer or by shaking the container, ensuring the lid was tightly sealed to prevent alcohol evaporation.
After 24 hr, the extract was filtered to separate the plant residue. The filtered extract was then poured into glass plates and placed in an oven at a maximum temperature of 35°C. During this process, the water and alcohol content evaporated, leaving behind a dry, pure extract. The dried extract was scraped off, weighed, and stored for further use.
2.3. Animals
After 24 hr, the extract was filtered to separate the plant residue. The filtered extract was then poured into glass plates and placed in an oven at a maximum temperature of 35°C. During this process, the water and alcohol content evaporated, leaving behind a dry, pure extract. The dried extract was scraped off, weighed, and stored for further use.
2.3. Animals
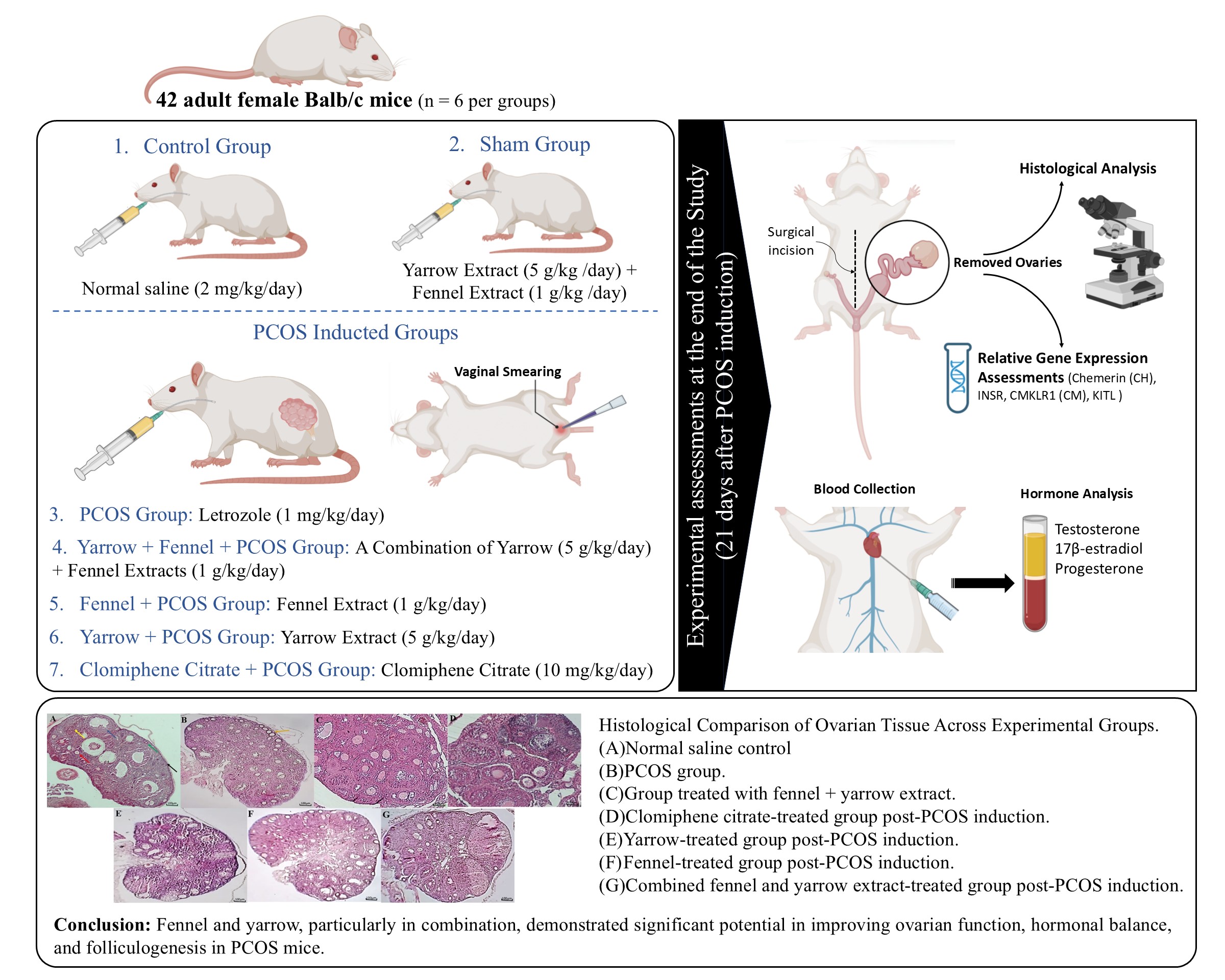
A total of 42 adult female Balb/c mice (25 ± 5 gr) were obtained from the animal nest of Royan Institute in Isfahan, Iran, and were included in the study. During the experiment, the animals were provided with standard laboratory chow and water.
After confirmation of the estrous cycle, animals were randomly assigned to 7 experimental groups (n = 6/each):
After confirmation of the estrous cycle, animals were randomly assigned to 7 experimental groups (n = 6/each):
- The control group, which received saline (2 mg/kg/day) for 21 days,
- The yarrow + fennel group, which received a combination of yarrow extract (5 gr/kg) and fennel extract (1 gr/kg) for 21 days,
- The PCOS group, which received letrozole (1 mg/kg/day) for 21 days to induce PCOS,
- The yarrow + fennel + PCOS group, which received a combination of yarrow (5 gr/kg/day) and fennel extracts (1 gr/kg/day) for 21 days after PCOS induction,
- The fennel + PCOS group, which received fennel extract (1 gr/kg/day) for 21 days after PCOS induction,
- The yarrow + PCOS group, which received yarrow extract (5 gr/kg/day) for 21 days after PCOS induction,
- The clomiphene citrate + PCOS group, which received clomiphene citrate (10 mg/kg/day) for 21 days after PCOS induction.
2.4. Vaginal smear and PCOS induction
2-3 mice from each group were randomly selected for vaginal smear tests over 4 consecutive days to confirm the estrous cycle. A drop of 0.9% saline was introduced into the vaginal canal, and the collected sample was smeared onto a slide for microscopic examination. PCOS was induced in 5 groups by administering letrozole (1 mg/kg/day) dissolved in saline (2 mg/kg/day) for 21 days. We performed vaginal smears for 10 consecutive days after letrozole treatment. The PCOS group exhibited persistent cornified (keratinized) epithelial cells, indicating a state of constant estrus and anovulation, a hallmark of this model. In addition, successful PCOS induction was confirmed by histological observation of a significant reduction in corpora lutea count and disrupted follicular architecture compared to control group.
2.5. Anesthesia and surgery
2.5. Anesthesia and surgery
After the treatment period (21 days for the first 3 groups and 42 days for others), the mice were anesthetized with a combination of ketamine (80 mg/kg) and xylazine (10 mg/kg) via intramuscular injection. The mice were disinfected, and a midline abdominal incision was made to expose the reproductive organs. The ovaries were carefully excised, with one ovary preserved in 10% formalin for histological analysis and the other in PBS for gene expression studies at -70°C.
2.6. Blood collection
2.6. Blood collection
Approximately 1 mL blood was collected from the left ventricle of the heart using a 2 mL syringe. The samples were centrifuged at 12,000 rpm for 2 min to separate the serum, which was stored at -70°C for hormonal analysis.
2.7. Histological analysis
2.7. Histological analysis
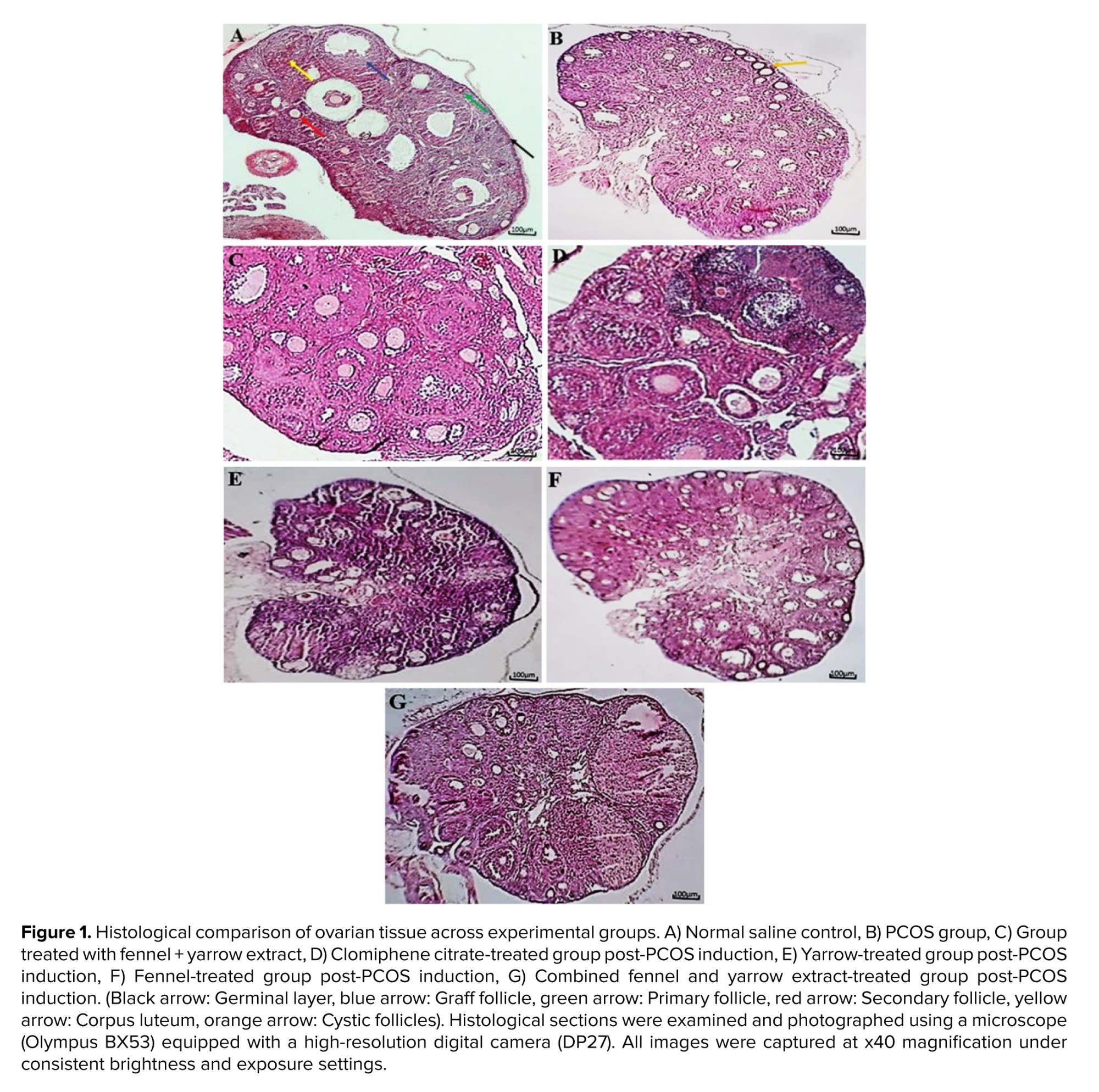
The ovaries designated for histological analysis were first dehydrated using a series of alcohol solutions, then cleared with xylene, and finally embedded in paraffin wax. Thin sections, each 5 μm in thickness, were cut and stained with hematoxylin and eosin. Follicle counting was performed on every 10th serial section (5 μm thick) to avoid double-counting of the same follicle. Only follicles with a visible oocyte nucleus were counted. For each ovary, 10 non-serial sections, spaced 50 μm apart, were analyzed from the central region of the ovary where the hilum was visible. 5 non-overlapping fields per section were examined at 400x magnification. The average count from these sections was calculated for each animal (Figure 1). All slides were coded by a researcher not involved in sample processing or group allocation, and histological evaluation was performed by an investigator blinded to the treatment groups to ensure unbiased assessment. Follicles in each section were counted, and the average count from the 10 sections of each ovary was calculated and compared across groups. Follicles were classified into 4 stages based on the number of granulosa cells (GC) layers surrounding the oocyte:
- Stage I: Primary follicles with a single GC layer.
- Stage II: Primary follicles with multiple GC layers.
- Stage III: Antral follicles containing one medium-sized or 2 small cavities.
- Stage IV: Graafian follicles with a large, distinct antral cavity (14, 15).
Cystic/arrested antral follicles were identified as antral-sized follicles with a thin, often disorganized GC layer, a thickened theca cell layer (theca cell hyperplasia), and the absence of a healthy cumulus-oocyte complex.
2.8. Relative gene expression assessments
2.8. Relative gene expression assessments
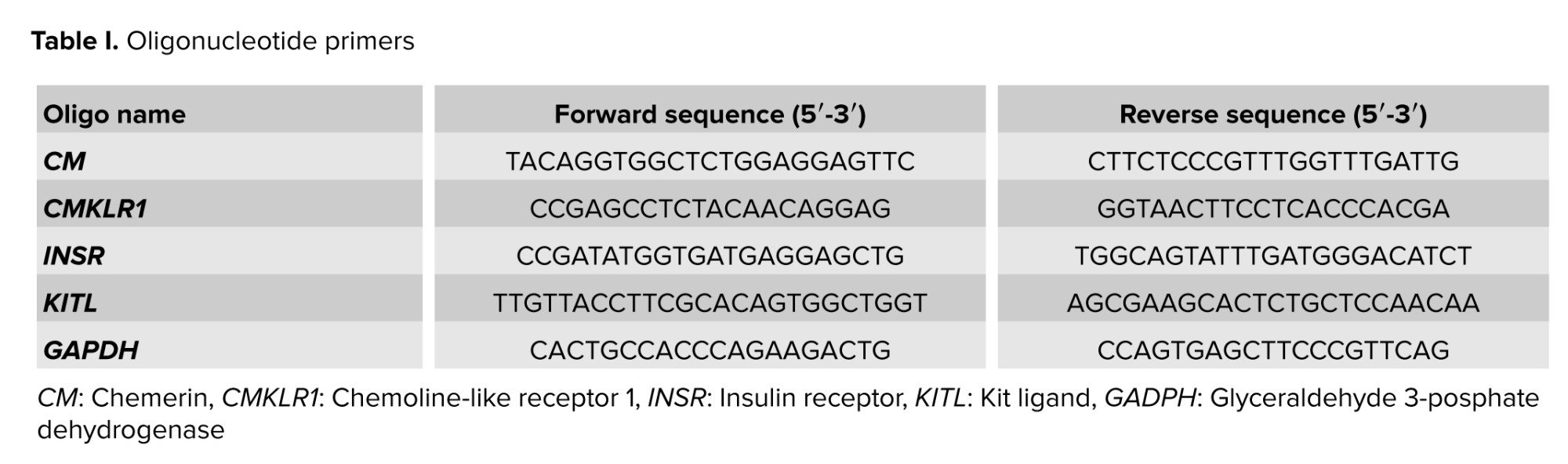
Gene expression assessments of certain genes (CM, CMKLR1, INSR, KITL) were analyzed using quantitative real-time polymerase chain reaction (RT-PCR). Total RNA was extracted from ovarian tissue using a total RNA extraction kit (Yekta Tajhiz Co., Iran) according to the manufacturer's instructions. The concentration of RNA was determined using spectrophotometry and adjusted to a concentration of 30 ng/μl. Subsequently, complementary DNA (cDNA) synthesis was performed as described below: the reaction was halted by incubating for 60 min at 37°C followed by 5 min at 70°C. Subsequently, the cDNA was utilized in quantitative RT-PCR employing SYBR green RT-PCR master mix and the one-step applied biosystem real-time thermocycler. The reference gene selected was glyceraldehyde 3-posphate dehydrogenase (GAPDH), as indicated in table I. Each PCR run was conducted 3 times. The RT-PCR process involved an initial denaturation at 95°C for 10 min, followed by 40 cycles consisting of 95°C for 15 sec, 60°C for CM, CMKLR1, INSR, KITL, and 64°C for CM for 20 sec, and 72°C for 30 sec. For each PCR, a 10 µl reaction mixture containing 1 µl of cDNA, along with 1 µl each of forward and reverse primers was used, mix 5 µl of master mix with 3 µl of diethyl pyrocarbonate treated water. The use of the GAPDH transcript helped ensure consistent results by minimizing differences in mRNA and cDNA levels. Gene expression levels between the treatment groups were assessed using the 2-∆∆CT method to quantify changes (16).
2.9. Hormonal assays
2.9. Hormonal assays
All serum samples were analyzed in duplicates, and the operator performing the enzyme-linked immunoassay (ELISA) assays was blinded to group identities. Sample codes were decoded only after data analysis was complete.
2.9.1. Testosterone measurement
2.9.1. Testosterone measurement
Testosterone, a 19-carbon steroid hormone, was measured using a competitive ELISA method (testosterone ELISA kit, Monobind Inc., USA). In this assay, serum samples, calibrators, or controls containing natural testosterone compete with horseradish peroxidase-conjugated testosterone for binding to biotinylated anti-testosterone antibodies. Streptavidin-coated wells were used to immobilize immune complexes. After washing, an enzyme substrate (horseradish peroxidase) was added, followed by a stop solution, and the absorbance was measured at 450 nm. The intensity of the color was inversely proportional to the concentration of testosterone in the sample, and concentrations were calculated using a standard curve.
2.9.2. Estrogen and progesterone measurement
2.9.2. Estrogen and progesterone measurement
Estrogen (17β-estradiol) and progesterone levels were determined using an ELISA kit (Monobind Inc., USA). The kit uses specific anti-estradiol and anti-progesterone antibodies, allowing direct measurement from serum or plasma without prior extraction. Biotinylated antibodies were mixed with the serum, allowing binding to antigens. After a short incubation, enzyme conjugates were added, creating a competitive reaction between the natural hormone in the sample and the enzyme-labeled hormone. The signal was measured using a substrate and stop solution, and the antigen concentration was determined via a dose-response curve.
2.10. Ethical Considerations


2.10. Ethical Considerations
All experimental procedures in this study were approved by the Ethical Committee of Shahid Sadoughi University of Medical Sciences, Yazd, Iran (Code: IR.SSU.AEC.1401.027). In order to observe ethical considerations and prevent animal suffering, all animals were anesthetized before performing surgical procedures. An attempt was made to use a minimum number of animals and perform various surgical procedures on anesthetized animals. Anesthesia was checked by pressing a part of the tail with forceps.
2.11. Statistical Analysis
2.11. Statistical Analysis
The normality of the data distribution for each variable was assessed using the Shapiro-Wilk test. Data that passed the normality test were analyzed using one-way analysis of variance (ANOVA) followed by Tukey's post-hoc test and were presented as mean ± SEM with overlaid individual data points. Data that were not normally distributed were analyzed using the Kruskal-Wallis test followed by Dunn's post-hoc test for multiple comparisons and are presented as box-and-whisker plots (showing the median, interquartile range, and min/max values). For all significant results, the exact p-value is reported. Effect sizes (η² for ANOVA/Cohen's d for post-hoc tests; ε² for Kruskal-Wallis) are provided in the figure legends.
3. Results
3.1. Mice weight and ovarian weights across study groups
3. Results
3.1. Mice weight and ovarian weights across study groups
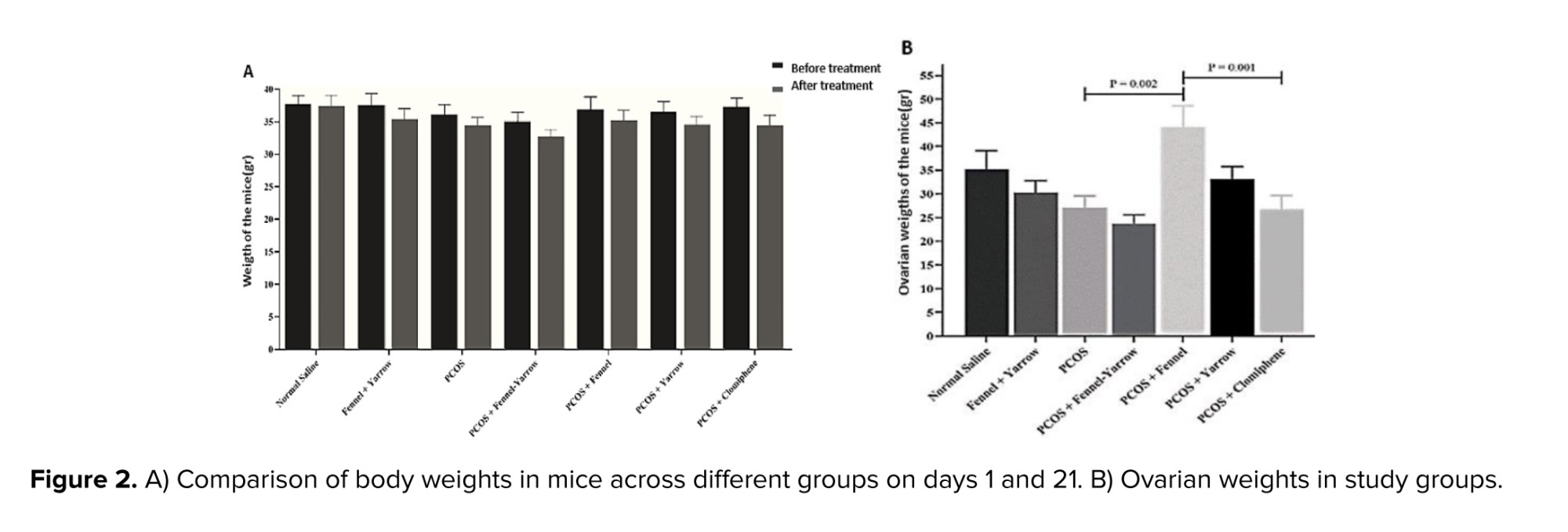
Based on the results of the present study, no significant difference was observed in the weight of mice in the normal saline group compared to the fennel-yarrow group and the PCOS group. Additionally, no significant changes were observed in the weight of mice in the treated PCOS groups (fennel, yarrow, fennel-yarrow combination, and clomiphene citrate) compared to the PCOS group and the normal saline group. Furthermore, an analysis of mouse weights on day 1 and day 21 across different groups revealed no significant differences (Figure 2A). The results of the present study showed that ovarian weight in the PCOS-fennel group significantly increased compared to the PCOS group (p = 0.002) and the PCOS-clomiphene citrate group (p = 0.001). Additionally, a significant difference was observed when comparing the PCOS-fennel group with the PCOS-fennel-yarrow group (p = 0.001). Despite a reduction in ovarian weight, the PCOS group did not show any significant difference compared to the normal saline group. Furthermore, no significant changes were observed in the remaining groups (Figure 2B).
3.2. Gene expression analysis
3.2. Gene expression analysis
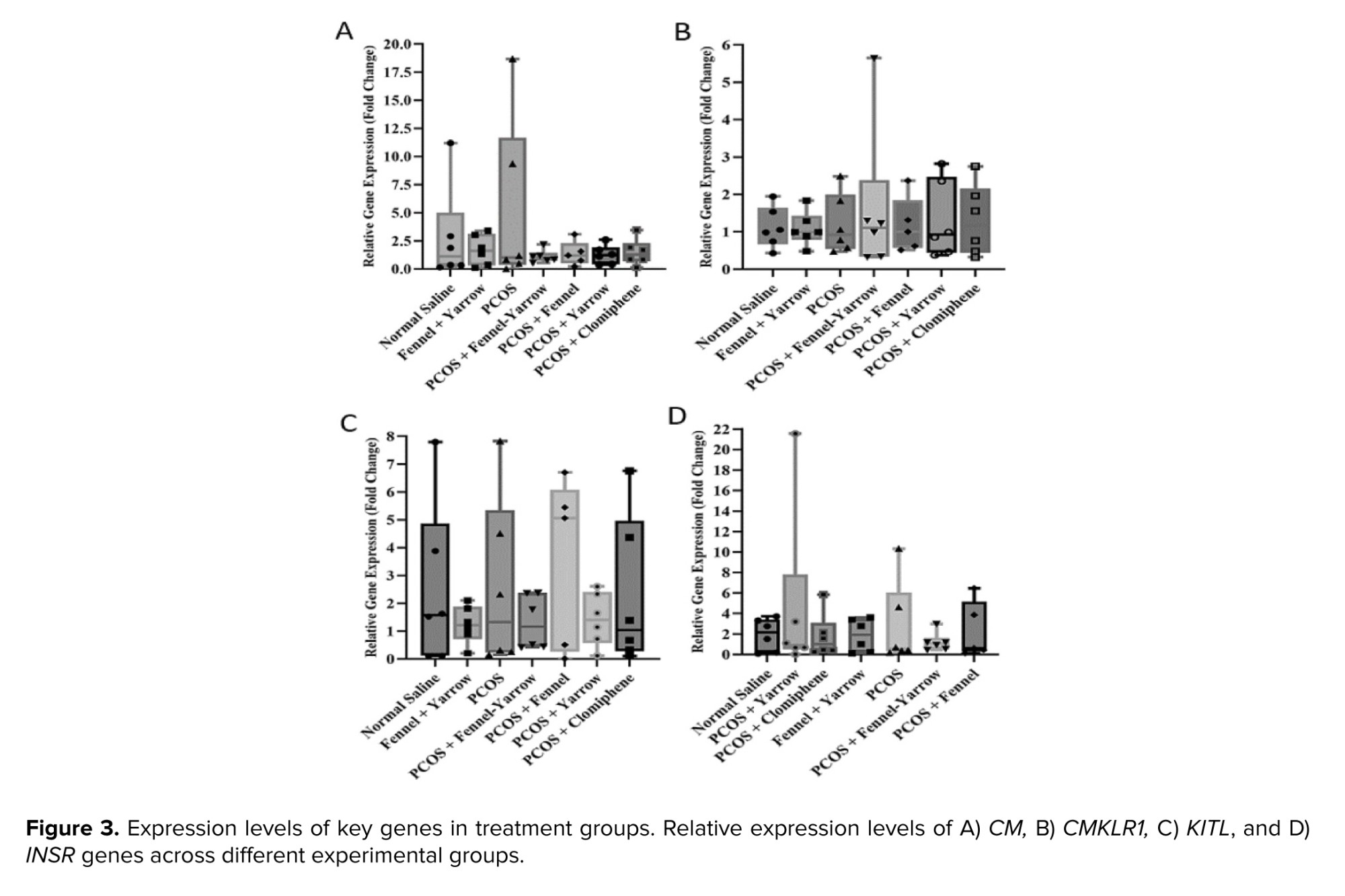
The results indicated no significant differences in the expression levels of the INSR, KITL, CM, and CMKLR1 genes between the normal saline group, the fennel-yarrow group, and the PCOS group. Additionally, the expression levels of these genes in the treated PCOS groups (fennel, yarrow, fennel-yarrow combination, and clomiphene citrate) showed no significant changes compared to the PCOS and normal saline groups (Figure 3).
3.3. Hormonal assays
3.3.1. Comparison of estrogen levels in study groups
3.3. Hormonal assays
3.3.1. Comparison of estrogen levels in study groups
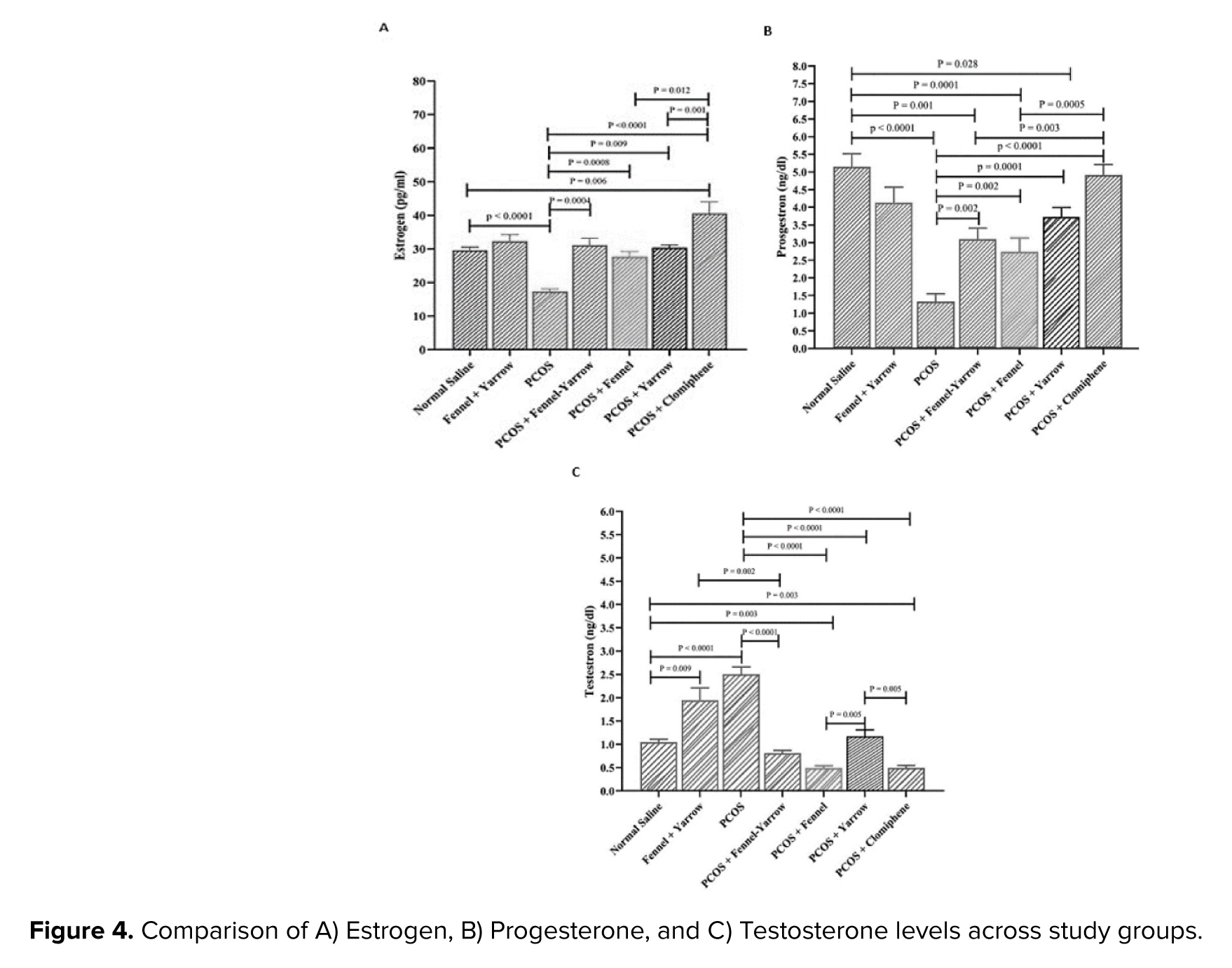
The results of the present study indicated a significant decrease in estrogen levels in the PCOS group compared to the normal saline group (p = 0.001). However, despite an increase in estrogen levels, the fennel-yarrow group did not show any significant change compared to the normal saline group. The PCOS-clomiphene citrate group showed a significant increase in estrogen levels compared to the normal saline group (p = 0.006), and the PCOS-yarrow group demonstrated a significant increase compared to the PCOS group (p = 0.009). Furthermore, significant increases in estrogen levels were observed in the PCOS-fennel group (p = 0.001), the PCOS-fennel-yarrow group (p = 0.001), and the PCOS-clomiphene citrate group (p = 0.001) compared to the PCOS group. Conversely, the PCOS-yarrow group exhibited significant decreases in estrogen levels when compared to the PCOS-clomiphene citrate group (p = 0.001), the PCOS-fennel group (p = 0.012), and the PCOS-fennel-yarrow group (p = 0.021) (Figure 4A).
3.3.2. Comparison of progesterone levels in study groups
3.3.2. Comparison of progesterone levels in study groups
The results demonstrated a significant decrease in progesterone levels in the PCOS group compared to the normal saline group (p = 0.001). Additionally, the fennel-yarrow group did not show significant changes compared to the normal saline group despite a slight increase in progesterone levels. The PCOS-yarrow group exhibited a significant decrease in progesterone levels compared to the normal saline group (p = 0.028), as did the PCOS-fennel group (p = 0.001) and the PCOS-fennel-yarrow group (p = 0.001). Furthermore, the PCOS-fennel group (p = 0.001) and the PCOS-fennel-yarrow group (p = 0.003) displayed significant decreases compared to the PCOS-clomiphene citrate group. In contrast, the PCOS-yarrow group (p = 0.001), the PCOS-fennel group (p = 0.02), the PCOS-fennel-yarrow group (p = 0.002), and the PCOS-clomiphene citrate group (p = 0.001) all showed significant increases in progesterone levels compared to the PCOS group (Figure 4B).
3.3.3. Comparison of testosterone levels in study groups
3.3.3. Comparison of testosterone levels in study groups
The results showed a significant increase in testosterone levels in the PCOS group compared to the normal saline group (p = 0.001). Similarly, the PCOS-fennel-yarrow group exhibited a significant increase in testosterone levels compared to the normal saline group (p = 0.009). However, a significant decrease in testosterone levels was observed in the PCOS-fennel-yarrow group compared to the fennel-yarrow group (p = 0.002). Significant reductions in testosterone levels were observed in the PCOS-fennel group (p = 0.003) and the PCOS-clomiphene citrate group (p = 0.003) compared to the normal saline group. Additionally, the PCOS-yarrow group (p = 0.001), the PCOS-fennel group (p = 0.001), the PCOS-fennel-yarrow group (p = 0.001), and the PCOS-clomiphene citrate group (p = 0.001) all exhibited significant decreases in testosterone levels compared to the PCOS group. Furthermore, the PCOS-yarrow group demonstrated significant increases in testosterone levels compared to the PCOS-clomiphene citrate group (p = 0.006) and notable changes compared to the PCOS-fennel group (p = 0.005) (Figure 4C).
3.4. Comparison of follicle counts
3.4.1. Comparison of primary follicle counts
3.4. Comparison of follicle counts
3.4.1. Comparison of primary follicle counts
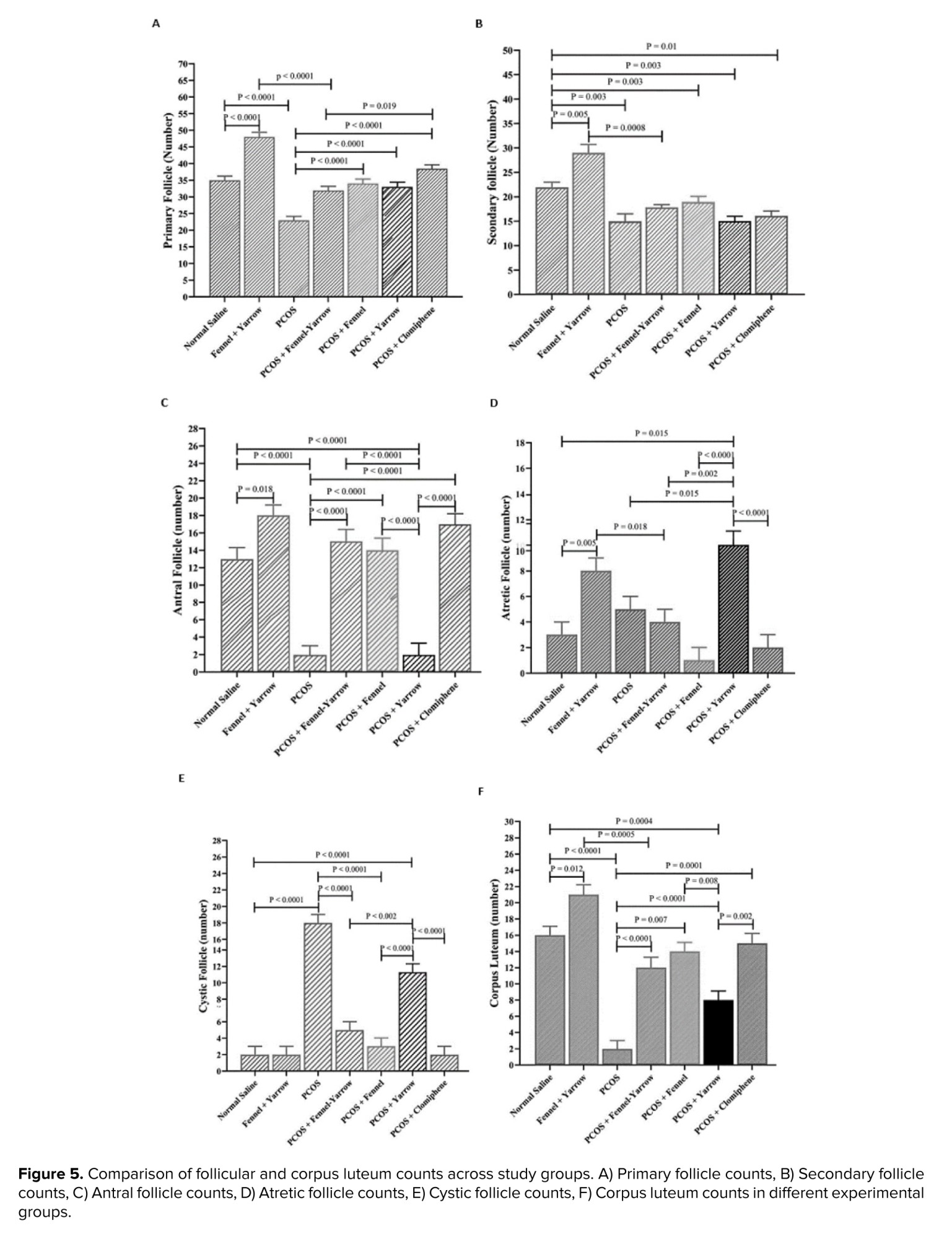
The results of the present study demonstrated that the number of primary follicles in the PCOS group significantly decreased compared to the normal saline group (p = 0.001). However, the PCOS-fennel group showed a notable increase in primary follicle count compared to the PCOS group (p = 0.001). Similarly, the PCOS-yarrow group also exhibited a significant increase compared to the PCOS group (p = 0.001). The PCOS-fennel-yarrow group showed a moderate increase in primary follicle count compared to the PCOS group (p = 0.0003). However, when compared to the PCOS-clomiphene citrate group, the PCOS-fennel-yarrow group displayed a significant decrease (p = 0.019). Additionally, the fennel-yarrow group showed a significant increase in primary follicle count compared to the normal saline group (p = 0.001) (Figure 5A).
3.4.2. Comparison of secondary follicle counts
3.4.2. Comparison of secondary follicle counts
The results of the present study demonstrated that the number of secondary follicles significantly decreased in the PCOS group compared to the normal saline group (p = 0.003). Similarly, the PCOS-fennel-yarrow group showed a notable decrease in secondary follicle count compared to the fennel-yarrow group (p = 0.001). However, the fennel-yarrow group exhibited a significant increase in secondary follicle count compared to the normal saline group (p = 0.005). The PCOS-yarrow group showed a significant decrease in secondary follicle count compared to the normal saline group (p = 0.003). Likewise, the PCOS-clomiphene citrate group displayed a significant reduction in secondary follicle count compared to the normal saline group (p = 0.01) (Figure 5B).
3.4.3. Comparison of antral follicle counts
3.4.3. Comparison of antral follicle counts
The results of the present study indicated that the number of antral follicles significantly decreased in the PCOS group compared to the normal saline group (p = 0.001). Conversely, the fennel-yarrow group showed a significant increase in antral follicle counts compared to the normal saline group (p = 0.018). The PCOS-yarrow group exhibited a marked decrease in antral follicle counts compared to the normal saline group (p = 0.001). In contrast, the PCOS-fennel-yarrow group (p = 0.001), the PCOS-fennel group (p = 0.001), and the PCOS-clomiphene citrate group (p = 0.001) all showed significant increases in antral follicle counts compared to the PCOS group. Furthermore, the PCOS-yarrow group demonstrated significant decreases in antral follicle counts compared to the PCOS-fennel group (p = 0.001), the PCOS-fennel-yarrow group (p = 0.001), and the PCOS-clomiphene citrate group (p = 0.001) (Figure 5C).
3.4.4. Comparison of atretic follicle counts in study groups
3.4.4. Comparison of atretic follicle counts in study groups
The results of the present study revealed no significant differences in the number of atretic follicles between the PCOS and normal saline groups. However, the fennel-yarrow group exhibited a significant increase in atretic follicle counts compared to the normal saline group (p = 0.005). Conversely, the PCOS-fennel-yarrow group displayed a notable decrease in atretic follicle counts compared to the fennel-yarrow group (p = 0.018). The PCOS-fennel group showed a significant increase in atretic follicle counts compared to the PCOS group (p = 0.001). Similarly, the PCOS-yarrow group demonstrated a substantial increase compared to the PCOS group (p = 0.001). Additionally, the PCOS-clomiphene citrate group exhibited significant reductions in atretic follicle counts compared to the PCOS group (p = 0.001) (Figure 5D).
3.4.5. Comparison of cystic follicle counts in study groups
3.4.5. Comparison of cystic follicle counts in study groups
The results of the present study showed that the number of cystic follicles significantly increased in the PCOS group compared to the normal saline group (p = 0.001). A similar significant increase was observed in the PCOS-yarrow group compared to the normal saline group (p = 0.001). Conversely, the PCOS-fennel group (p = 0.001), the PCOS-yarrow group (p = 0.003), the PCOS-fennel-yarrow group (p = 0.001), and the PCOS-clomiphene citrate group (p = 0.001) all showed significant decreases in cystic follicle counts compared to the PCOS group. Additionally, the PCOS-yarrow group demonstrated a notable increase in cystic follicle counts compared to the PCOS-fennel group (p = 0.001), the PCOS-fennel-yarrow group (p = 0.001), and the PCOS-clomiphene citrate group (p = 0.001) (Figure 5E).
3.4.6. Comparison of corpus luteum counts in study groups
3.4.6. Comparison of corpus luteum counts in study groups
The results of the present study indicated a significant decrease in the number of corpora lutea in the PCOS group compared to normal saline group (p = 0.001). Similarly, the PCOS-fennel-yarrow group exhibited a significant decrease in corpus luteum counts compared to the fennel-yarrow group (p = 0.001). However, the fennel-yarrow group showed a notable increase compared to the normal saline group (p = 0.012). The PCOS-yarrow group displayed a significant decrease in corpus luteum counts compared to the normal saline group (p = 0.001), the PCOS-fennel group (p = 0.008), and the PCOS-clomiphene citrate group (p = 0.002). Moreover, the PCOS-fennel group (p = 0.007), the PCOS-yarrow group (p = 0.001), the PCOS-fennel-yarrow group (p = 0.001), and the PCOS-clomiphene citrate group (p = 0.001) all demonstrated significant increases in corpus luteum counts compared to the PCOS group (Figure 5F).
4. Discussion




4. Discussion
The objective of this study was to evaluate the effects of 2 medicinal plants, fennel and yarrow, on histomorphological parameters of ovarian tissue, the expression levels of folliculogenesis-related genes (INSR, KITL, CM, CMKLR1), and serum levels of sex hormones in adult female mice with PCOS. This study aimed to assess these herbal treatments as alternatives or adjuncts to conventional drug therapies for PCOS, such as clomiphene citrate.
It should be noted that the present study employed a single dose of each plant extract administered for 21 days, selected based on prior studies demonstrating efficacy of F. vulgare and A. millefolium within a similar timeframe in PCOS models (8, 9, 17). This design enabled an initial evaluation of their therapeutic potential while maintaining adherence to ethical principles and minimizing animal distress associated with prolonged treatment. However, the use of a single dose and relatively short treatment duration represents a limitation of the study. Future investigations should therefore explore multiple dosing regimens and extended treatment durations (6-8 wk) to assess dose dependency, long-term efficacy, and safety profiles of these herbal therapies in PCOS management.
Another limitation of the present study is that metabolic parameters associated with PCOS, such as IR indices (fasting glucose, insulin, and HOMA-IR), were not assessed. Moreover, basic biochemical markers of hepatic and renal function were not evaluated to confirm systemic safety. The main objective of this work was to investigate reproductive and ovarian outcomes; therefore, metabolic and toxicity evaluations were beyond the initial study scope. Nevertheless, previous reports have shown no adverse hepatic or renal effects of F. vulgare and A. millefolium extracts at similar or higher doses (8, 9, 17). Future studies should therefore include detailed metabolic profiling and toxicity assessment to comprehensively elucidate the therapeutic potential and safety margins of these herbal preparations in PCOS management.
The results indicated no significant differences in body weight across groups. Although the PCOS group exhibited a slight increase in weight compared to the control, this change was not statistically significant. Similarly, no significant weight changes were observed in the groups treated with fennel, yarrow, or their combination compared to the PCOS and normal saline groups. These findings suggest that the treatments did not directly impact overall body weight. In contrast, ovarian weight increased significantly in the PCOS-fennel and PCOS-combined fennel-yarrow groups compared to the PCOS group. This increase may reflect the positive effects of these plants on follicular growth and ovarian function. Other groups showed no significant changes in ovarian weight. Previous studies have highlighted the estrogenic and antioxidant effects of fennel in increasing ovarian weight, while yarrow has been reported to decrease ovarian weight at higher doses (9, 17).
The results showed no significant differences in expression of the INSR gene across various treatment groups, which may indicate the lack of a direct effect of the treatments on this pathway or a low ovarian sensitivity to the regulation of this gene. The therapeutic effects of fennel and yarrow are likely mediated through pathways other than the modulation of INSR expression. The INSR gene, encoding the INSR, plays a crucial role in glucose metabolism and insulin sensitivity. In PCOS, alterations in INSR expression contribute to IR and exacerbate metabolic symptoms. Treatments such as metformin and minocycline have been shown to increase INSR gene expression and reduce IR. In one study, minocycline treatment in PCOS mouse models led to reduced CMKLR1 expression and increased INSR expression, which helped alleviate symptoms such as reduced estrogen levels and improved ovulation (18). Hyperinsulinemia and IR are critical pathological characteristics of PCOS. The mechanism of IR is significantly linked to the number and function of INSR receptors. IR compensatorily increases insulin secretion from beta cells, leading to hyperinsulinemia. When beta cells can no longer compensate for IR, hyperglycemia occurs, followed by glucose intolerance and type 2 diabetes. The causes of IR in PCOS remain unknown and may be attributed to defects in the INSR signaling pathway (19).
The findings of this study showed that the KITL gene expression levels did not significantly change across the treatment groups. This suggests that KITL, which plays a critical role in folliculogenesis, might not have been directly affected by the treatments or that any potential changes require a longer duration to manifest. It is also possible that the dosage or treatment duration was insufficient to induce noticeable effects, or that the therapeutic impact of fennel and yarrow occurs through pathways other than KITL. KITL protein is found in the ovarian cortex and human GCs (20).
Studies using Colocasia esculenta extract showed that it reduced cystic follicles and significantly increased the number of mature follicles and corpora lutea. These positive changes were associated with increased expression of folliculogenesis-related genes, including KITL. Similarly, Ecklonia cava and D-chiro-inositol improved ovarian function in PCOS models by upregulating KITL, facilitating follicular growth, and reducing cystic follicles (21, 22).
This study demonstrated no significant changes in the expression of CM and CH genes across the treatment groups (fennel, yarrow, combined fennel-yarrow, clomiphene citrate) compared to the normal saline or untreated PCOS groups. These findings suggest that the applied treatments had no substantial effect on these genes. CM, a key regulator of metabolic and immune processes, is elevated in PCOS and contributes to IR, chronic inflammation, and impaired folliculogenesis. Despite the treatments, CM expression remained unchanged, indicating potential resistance to modulation by herbal or pharmacological interventions. Research has shown that the CMKLR1 receptor, through its interaction with CM, plays a role in suppressing progesterone production in PCOS. Studies using CMKLR1 inhibitors have demonstrated the receptor's involvement in steroidogenesis dysregulation (23). CH, a less-studied gene in this context, also exhibited no significant changes across groups. Its specific regulatory mechanisms in PCOS remain unclear and warrant further investigation to elucidate its molecular pathways.
Although the expression levels of INSR, KITL, CM, and CH did not show significant differences across the experimental groups, the observed hormonal and histological improvements suggest that F. vulgare and A. millefolium may exert their beneficial effects through post-transcriptional mechanisms or via other molecular pathways not assessed in the present study. The selected genes primarily represent insulin signaling, follicular support, and inflammatory regulation, which were the main mechanistic targets of this experiment. However, it is possible that these extracts also influence steroidogenic (CYP19A1, STAR) and gonadotropin receptor (FSHR, LHR) pathways, as well as inflammatory mediators such as tumor necrosis factor alfa, which could further clarify their regulatory roles in ovarian function. Future investigations incorporating these genes and protein-level validation (western blotting or immunohistochemistry) are warranted to provide deeper mechanistic insight into the actions of these herbal agents in PCOS.
While the treatments yielded significant improvements in ovarian histomorphology and hormonal profiles, no corresponding significant changes were observed in the expression of INSR, KITL, CM, or CMKLR1. This suggests that the therapeutic effects of fennel and yarrow may not be mediated through the transcriptional regulation of these specific genes within the ovarian tissue at the measured time point. Instead, the benefits are likely conferred through alternative mechanisms; for example, reducing oxidative stress can improve ovarian function without necessarily altering the mRNA levels of these specific genes. Additionally, phytoestrogenic properties of fennel (anethole) might directly interact with estrogen receptors to restore hormonal feedback and follicular development, acting at a post-translational level.
The present study demonstrated a significant reduction in estrogen levels in the PCOS group compared to normal saline group. However, no significant changes in estrogen levels were observed in the groups treated with fennel or yarrow alone compared to the control group. This finding aligns with previous studies, such as one using Mahuang-Tang extract, which showed that herbal treatment reduced testosterone levels and restored estrogen levels depending on dose and duration (24). In contrast, the combination of fennel and yarrow significantly increased estrogen levels in the PCOS-fennel-yarrow group compared to the PCOS group. Similarly, clomiphene citrate treatment significantly increased estrogen levels compared to the PCOS group. The synergistic effects of fennel and yarrow appear to enhance estrogen levels, whereas these effects were not observed when the plants were used individually. Clomiphene citrate also directly stimulated the hypothalamic-pituitary-ovarian axis, significantly increasing estrogen levels (25, 26).
A marked reduction in progesterone levels was observed in the PCOS group. In contrast, significant improvements in progesterone levels were noted in groups treated with fennel, yarrow, and their combinations compared to PCOS group. These results are consistent with studies such as one investigating the effects of E. cava extract, which also improved progesterone levels in PCOS models (27). The reduced progesterone levels in the PCOS group are linked to disrupted ovulation and impaired ovarian function (28). Active compounds in fennel and yarrow were effective in reducing inflammation and restoring hormonal balance, aiding in progesterone production (29).
Testosterone levels were significantly elevated in the PCOS group compared to the normal saline group, reflecting hyperandrogenism and ovarian dysfunction commonly observed in PCOS (30). However, a significant reduction in testosterone levels was observed in the PCOS-fennel-yarrow group compared to the PCOS group, demonstrating the efficacy of the combined treatment in modulating androgen production. The notable decrease in testosterone levels in the fennel-yarrow group can be attributed to improved hormonal pathways and ovarian function (29). These results are consistent with previous studies demonstrating the regulatory effects of medicinal plants on sex hormone levels, although their efficacy depends on dosage and extract type.
The study findings revealed a significant decrease in the number of primary and secondary follicles in the PCOS group compared to the normal saline group. In contrast, the number of cystic follicles significantly increased. Treatment with fennel and yarrow resulted in significant improvements in primary and secondary follicle counts compared to the PCOS group. The combination of fennel and yarrow showed the most pronounced effect, significantly increasing primary and secondary follicles and reducing cystic follicles. The reduction in healthy follicles and increased cystic follicles in the PCOS group are attributed to hormonal imbalances and inflammation disrupting folliculogenesis (31).
The study findings revealed that in the PCOS group, the number of antral follicles and corpora lutea significantly decreased compared to the normal saline group. This reduction is likely due to the detrimental effects of PCOS, including hormonal imbalances, elevated androgen levels, and oxidative stress, which lead to cellular apoptosis and decreased ovarian activity. Additionally, the number of atretic follicles increased in some treatment groups, indicating either detrimental effects or insufficient efficacy of certain interventions in promoting follicular growth. In groups treated with a combination of fennel and yarrow, a significant increase was observed in the number of antral follicles and corpora lutea.
The positive effects can be attributed to the active compounds in these plants, such as the estrogenic properties of fennel and anti-inflammatory and antioxidant effects of yarrow. These compounds improve hormonal balance, reduce inflammation, and enhance ovarian tissue health, creating a favorable environment for folliculogenesis and ovulation.
Clomiphene citrate, as a standard treatment for ovulation induction, also showed a positive effect on the number of antral follicles and corpora lutea. This drug accelerates follicular growth and ovulation by stimulating the production of FSH and LH through the blockage of estrogen receptors in the hypothalamus. However, the combination of fennel and yarrow demonstrated comparable effects, suggesting its potential as a complementary therapy.
A related study investigated the effects of ethanol extract of yarrow on ovarian histology and serum hormone levels in a PCOS model induced by estradiol valerate. Follicles at various stages of development were observed in all ovarian groups. The results indicated no significant differences in the number of primordial, primary, mature, atretic follicles, and corpora lutea among the groups. However, the number of secondary follicles in the PCOS group (17.0 ± 1.5) was significantly lower than in the control group treated with 1.2 gr/kg of yarrow extract (25.0 ± 2.2, p = 0.05). Furthermore, the number of cysts increased in the PCOS group (9.0 ± 0.6), but significantly improved in mice receiving 5 gr/kg of yarrow extract (6.0 ± 0.7, p = 0.05) (9).
5. Conclusion
It should be noted that the present study employed a single dose of each plant extract administered for 21 days, selected based on prior studies demonstrating efficacy of F. vulgare and A. millefolium within a similar timeframe in PCOS models (8, 9, 17). This design enabled an initial evaluation of their therapeutic potential while maintaining adherence to ethical principles and minimizing animal distress associated with prolonged treatment. However, the use of a single dose and relatively short treatment duration represents a limitation of the study. Future investigations should therefore explore multiple dosing regimens and extended treatment durations (6-8 wk) to assess dose dependency, long-term efficacy, and safety profiles of these herbal therapies in PCOS management.
Another limitation of the present study is that metabolic parameters associated with PCOS, such as IR indices (fasting glucose, insulin, and HOMA-IR), were not assessed. Moreover, basic biochemical markers of hepatic and renal function were not evaluated to confirm systemic safety. The main objective of this work was to investigate reproductive and ovarian outcomes; therefore, metabolic and toxicity evaluations were beyond the initial study scope. Nevertheless, previous reports have shown no adverse hepatic or renal effects of F. vulgare and A. millefolium extracts at similar or higher doses (8, 9, 17). Future studies should therefore include detailed metabolic profiling and toxicity assessment to comprehensively elucidate the therapeutic potential and safety margins of these herbal preparations in PCOS management.
The results indicated no significant differences in body weight across groups. Although the PCOS group exhibited a slight increase in weight compared to the control, this change was not statistically significant. Similarly, no significant weight changes were observed in the groups treated with fennel, yarrow, or their combination compared to the PCOS and normal saline groups. These findings suggest that the treatments did not directly impact overall body weight. In contrast, ovarian weight increased significantly in the PCOS-fennel and PCOS-combined fennel-yarrow groups compared to the PCOS group. This increase may reflect the positive effects of these plants on follicular growth and ovarian function. Other groups showed no significant changes in ovarian weight. Previous studies have highlighted the estrogenic and antioxidant effects of fennel in increasing ovarian weight, while yarrow has been reported to decrease ovarian weight at higher doses (9, 17).
The results showed no significant differences in expression of the INSR gene across various treatment groups, which may indicate the lack of a direct effect of the treatments on this pathway or a low ovarian sensitivity to the regulation of this gene. The therapeutic effects of fennel and yarrow are likely mediated through pathways other than the modulation of INSR expression. The INSR gene, encoding the INSR, plays a crucial role in glucose metabolism and insulin sensitivity. In PCOS, alterations in INSR expression contribute to IR and exacerbate metabolic symptoms. Treatments such as metformin and minocycline have been shown to increase INSR gene expression and reduce IR. In one study, minocycline treatment in PCOS mouse models led to reduced CMKLR1 expression and increased INSR expression, which helped alleviate symptoms such as reduced estrogen levels and improved ovulation (18). Hyperinsulinemia and IR are critical pathological characteristics of PCOS. The mechanism of IR is significantly linked to the number and function of INSR receptors. IR compensatorily increases insulin secretion from beta cells, leading to hyperinsulinemia. When beta cells can no longer compensate for IR, hyperglycemia occurs, followed by glucose intolerance and type 2 diabetes. The causes of IR in PCOS remain unknown and may be attributed to defects in the INSR signaling pathway (19).
The findings of this study showed that the KITL gene expression levels did not significantly change across the treatment groups. This suggests that KITL, which plays a critical role in folliculogenesis, might not have been directly affected by the treatments or that any potential changes require a longer duration to manifest. It is also possible that the dosage or treatment duration was insufficient to induce noticeable effects, or that the therapeutic impact of fennel and yarrow occurs through pathways other than KITL. KITL protein is found in the ovarian cortex and human GCs (20).
Studies using Colocasia esculenta extract showed that it reduced cystic follicles and significantly increased the number of mature follicles and corpora lutea. These positive changes were associated with increased expression of folliculogenesis-related genes, including KITL. Similarly, Ecklonia cava and D-chiro-inositol improved ovarian function in PCOS models by upregulating KITL, facilitating follicular growth, and reducing cystic follicles (21, 22).
This study demonstrated no significant changes in the expression of CM and CH genes across the treatment groups (fennel, yarrow, combined fennel-yarrow, clomiphene citrate) compared to the normal saline or untreated PCOS groups. These findings suggest that the applied treatments had no substantial effect on these genes. CM, a key regulator of metabolic and immune processes, is elevated in PCOS and contributes to IR, chronic inflammation, and impaired folliculogenesis. Despite the treatments, CM expression remained unchanged, indicating potential resistance to modulation by herbal or pharmacological interventions. Research has shown that the CMKLR1 receptor, through its interaction with CM, plays a role in suppressing progesterone production in PCOS. Studies using CMKLR1 inhibitors have demonstrated the receptor's involvement in steroidogenesis dysregulation (23). CH, a less-studied gene in this context, also exhibited no significant changes across groups. Its specific regulatory mechanisms in PCOS remain unclear and warrant further investigation to elucidate its molecular pathways.
Although the expression levels of INSR, KITL, CM, and CH did not show significant differences across the experimental groups, the observed hormonal and histological improvements suggest that F. vulgare and A. millefolium may exert their beneficial effects through post-transcriptional mechanisms or via other molecular pathways not assessed in the present study. The selected genes primarily represent insulin signaling, follicular support, and inflammatory regulation, which were the main mechanistic targets of this experiment. However, it is possible that these extracts also influence steroidogenic (CYP19A1, STAR) and gonadotropin receptor (FSHR, LHR) pathways, as well as inflammatory mediators such as tumor necrosis factor alfa, which could further clarify their regulatory roles in ovarian function. Future investigations incorporating these genes and protein-level validation (western blotting or immunohistochemistry) are warranted to provide deeper mechanistic insight into the actions of these herbal agents in PCOS.
While the treatments yielded significant improvements in ovarian histomorphology and hormonal profiles, no corresponding significant changes were observed in the expression of INSR, KITL, CM, or CMKLR1. This suggests that the therapeutic effects of fennel and yarrow may not be mediated through the transcriptional regulation of these specific genes within the ovarian tissue at the measured time point. Instead, the benefits are likely conferred through alternative mechanisms; for example, reducing oxidative stress can improve ovarian function without necessarily altering the mRNA levels of these specific genes. Additionally, phytoestrogenic properties of fennel (anethole) might directly interact with estrogen receptors to restore hormonal feedback and follicular development, acting at a post-translational level.
The present study demonstrated a significant reduction in estrogen levels in the PCOS group compared to normal saline group. However, no significant changes in estrogen levels were observed in the groups treated with fennel or yarrow alone compared to the control group. This finding aligns with previous studies, such as one using Mahuang-Tang extract, which showed that herbal treatment reduced testosterone levels and restored estrogen levels depending on dose and duration (24). In contrast, the combination of fennel and yarrow significantly increased estrogen levels in the PCOS-fennel-yarrow group compared to the PCOS group. Similarly, clomiphene citrate treatment significantly increased estrogen levels compared to the PCOS group. The synergistic effects of fennel and yarrow appear to enhance estrogen levels, whereas these effects were not observed when the plants were used individually. Clomiphene citrate also directly stimulated the hypothalamic-pituitary-ovarian axis, significantly increasing estrogen levels (25, 26).
A marked reduction in progesterone levels was observed in the PCOS group. In contrast, significant improvements in progesterone levels were noted in groups treated with fennel, yarrow, and their combinations compared to PCOS group. These results are consistent with studies such as one investigating the effects of E. cava extract, which also improved progesterone levels in PCOS models (27). The reduced progesterone levels in the PCOS group are linked to disrupted ovulation and impaired ovarian function (28). Active compounds in fennel and yarrow were effective in reducing inflammation and restoring hormonal balance, aiding in progesterone production (29).
Testosterone levels were significantly elevated in the PCOS group compared to the normal saline group, reflecting hyperandrogenism and ovarian dysfunction commonly observed in PCOS (30). However, a significant reduction in testosterone levels was observed in the PCOS-fennel-yarrow group compared to the PCOS group, demonstrating the efficacy of the combined treatment in modulating androgen production. The notable decrease in testosterone levels in the fennel-yarrow group can be attributed to improved hormonal pathways and ovarian function (29). These results are consistent with previous studies demonstrating the regulatory effects of medicinal plants on sex hormone levels, although their efficacy depends on dosage and extract type.
The study findings revealed a significant decrease in the number of primary and secondary follicles in the PCOS group compared to the normal saline group. In contrast, the number of cystic follicles significantly increased. Treatment with fennel and yarrow resulted in significant improvements in primary and secondary follicle counts compared to the PCOS group. The combination of fennel and yarrow showed the most pronounced effect, significantly increasing primary and secondary follicles and reducing cystic follicles. The reduction in healthy follicles and increased cystic follicles in the PCOS group are attributed to hormonal imbalances and inflammation disrupting folliculogenesis (31).
The study findings revealed that in the PCOS group, the number of antral follicles and corpora lutea significantly decreased compared to the normal saline group. This reduction is likely due to the detrimental effects of PCOS, including hormonal imbalances, elevated androgen levels, and oxidative stress, which lead to cellular apoptosis and decreased ovarian activity. Additionally, the number of atretic follicles increased in some treatment groups, indicating either detrimental effects or insufficient efficacy of certain interventions in promoting follicular growth. In groups treated with a combination of fennel and yarrow, a significant increase was observed in the number of antral follicles and corpora lutea.
The positive effects can be attributed to the active compounds in these plants, such as the estrogenic properties of fennel and anti-inflammatory and antioxidant effects of yarrow. These compounds improve hormonal balance, reduce inflammation, and enhance ovarian tissue health, creating a favorable environment for folliculogenesis and ovulation.
Clomiphene citrate, as a standard treatment for ovulation induction, also showed a positive effect on the number of antral follicles and corpora lutea. This drug accelerates follicular growth and ovulation by stimulating the production of FSH and LH through the blockage of estrogen receptors in the hypothalamus. However, the combination of fennel and yarrow demonstrated comparable effects, suggesting its potential as a complementary therapy.
A related study investigated the effects of ethanol extract of yarrow on ovarian histology and serum hormone levels in a PCOS model induced by estradiol valerate. Follicles at various stages of development were observed in all ovarian groups. The results indicated no significant differences in the number of primordial, primary, mature, atretic follicles, and corpora lutea among the groups. However, the number of secondary follicles in the PCOS group (17.0 ± 1.5) was significantly lower than in the control group treated with 1.2 gr/kg of yarrow extract (25.0 ± 2.2, p = 0.05). Furthermore, the number of cysts increased in the PCOS group (9.0 ± 0.6), but significantly improved in mice receiving 5 gr/kg of yarrow extract (6.0 ± 0.7, p = 0.05) (9).
5. Conclusion
The findings of this study provide robust evidence for the therapeutic benefits of fennel and yarrow, particularly when combined, in treating PCOS. The combination treatment significantly improved ovarian histomorphology, increased primary and antral follicle counts, and enhanced the number of corpora lutea, suggesting improved ovulation potential. Notably, this group also demonstrated significant hormonal modulation, with increased estrogen and progesterone levels and reduced testosterone levels, comparable to the effects of clomiphene citrate. Despite the promising results, gene expression analysis revealed no significant changes in the expression of INSR, KITL, CM, and CMKLR1, implying that these treatments may exert their effects through non-genomic mechanisms such as anti-inflammatory and antioxidant pathways. The estrogenic properties of fennel and the anti-inflammatory and antioxidant effects of yarrow are likely responsible for these positive outcomes.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
M. Azizi Khareshki, A. Hassanpour Dehnavi, M. Hosseini-sharifabad and F. Fesahat designed the study and conducted the research. AR. Talebi and M. Hosseini-sharifabad monitored, evaluated, and analyzed the results of the study. Further, M. Azizi Khareshki, F. Fesahat, and A. Hassanpour Dehnavi reviewed the article. M. Azizi Khareshki, A. Hassanpour Dehnavi, and F. Fesahat carried out the drug preparation, specimen collections, gene expression assessments, and histopathological analysis. Drafting of the manuscript: M. Azizi Khareshki, A. Hassanpour Dehnavi, M. Hosseini-sharifabad, F. Fesahat and AR. Talebi. All authors approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgments
This article was extracted from a thesis written by Mohadeseh Azizi, M.Sc. candidate of Anatomy. We would like to express our deepest gratitude to the Reproductive Immunology Research Center and Research and Clinical Center for Infertility, Shahid Sadoughi University of Medical Sciences, Yazd, Iran, for their contribution. Regarding the use of artificial intelligence (AI), I would like to clarify that ChatGPT 4o was used for assisting with grammar checks and revisions.
Conflict of Interest
The authors declare that there is no conflict of interest.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
M. Azizi Khareshki, A. Hassanpour Dehnavi, M. Hosseini-sharifabad and F. Fesahat designed the study and conducted the research. AR. Talebi and M. Hosseini-sharifabad monitored, evaluated, and analyzed the results of the study. Further, M. Azizi Khareshki, F. Fesahat, and A. Hassanpour Dehnavi reviewed the article. M. Azizi Khareshki, A. Hassanpour Dehnavi, and F. Fesahat carried out the drug preparation, specimen collections, gene expression assessments, and histopathological analysis. Drafting of the manuscript: M. Azizi Khareshki, A. Hassanpour Dehnavi, M. Hosseini-sharifabad, F. Fesahat and AR. Talebi. All authors approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgments
This article was extracted from a thesis written by Mohadeseh Azizi, M.Sc. candidate of Anatomy. We would like to express our deepest gratitude to the Reproductive Immunology Research Center and Research and Clinical Center for Infertility, Shahid Sadoughi University of Medical Sciences, Yazd, Iran, for their contribution. Regarding the use of artificial intelligence (AI), I would like to clarify that ChatGPT 4o was used for assisting with grammar checks and revisions.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Pattology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |