Thu, Jun 11, 2026
[Archive]
Volume 23, Issue 9 (September 2025)
IJRM 2025, 23(9): 713-722 |
Back to browse issues page
Ethics code: IR.ARAKMU.REC.1400.099
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Matin Manesh Z, Zeinali H, Ghanbari F, Mafi A, Goudarzi M, Ghoddoosi M, et al . Protective effect of chrysin on di (2-ethylhexyl) phthalate-induced toxicity in mice testis: An experimental study. IJRM 2025; 23 (9) :713-722
URL: http://ijrm.ir/article-1-3594-en.html
URL: http://ijrm.ir/article-1-3594-en.html
Zahra Matin Manesh1 

 , Hossein Zeinali2
, Hossein Zeinali2 

 , Fatemeh Ghanbari3
, Fatemeh Ghanbari3 

 , Afsaneh Mafi4
, Afsaneh Mafi4 

 , Mehdi Goudarzi5
, Mehdi Goudarzi5 

 , Mahdiieh Ghoddoosi6
, Mahdiieh Ghoddoosi6 

 , Saeed Hajihashemi7
, Saeed Hajihashemi7 

 , Hamid Heidari *8
, Hamid Heidari *8 




 , Hossein Zeinali2
, Hossein Zeinali2 

 , Fatemeh Ghanbari3
, Fatemeh Ghanbari3 

 , Afsaneh Mafi4
, Afsaneh Mafi4 

 , Mehdi Goudarzi5
, Mehdi Goudarzi5 

 , Mahdiieh Ghoddoosi6
, Mahdiieh Ghoddoosi6 

 , Saeed Hajihashemi7
, Saeed Hajihashemi7 

 , Hamid Heidari *8
, Hamid Heidari *8 


1- Student Research Committee, Faculty of Medicine, Arak University of Medical Sciences, Arak, Iran.
2- Cellular and Molecular Research Center, Qom University of Medical Sciences, Qom, Iran. & Department of Physiology, Faculty of Medicine, Qom University of Medical Sciences, Qom, Iran.
3- Department of Pharmacology, Islamic Azad University, Arak Branch, Arak, Iran.
4- Cellular and Molecular Research Center, Qom University of Medical Sciences, Qom, Iran.
5- Medicinal Plant Research Center, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
6- Department of Pathology, Shahid Beheshti Medical Center, Qom University of Medical Sciences, Qom, Iran.
7- Department of Physiology, Faculty of Medicine, Arak University of Medical Sciences, Arak, Iran.
8- Cellular and Molecular Research Center, Qom University of Medical Sciences, Qom, Iran. & Department of Physiology, Faculty of Medicine, Qom University of Medical Sciences, Qom, Iran. ,physiology_86@yahoo.com
2- Cellular and Molecular Research Center, Qom University of Medical Sciences, Qom, Iran. & Department of Physiology, Faculty of Medicine, Qom University of Medical Sciences, Qom, Iran.
3- Department of Pharmacology, Islamic Azad University, Arak Branch, Arak, Iran.
4- Cellular and Molecular Research Center, Qom University of Medical Sciences, Qom, Iran.
5- Medicinal Plant Research Center, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
6- Department of Pathology, Shahid Beheshti Medical Center, Qom University of Medical Sciences, Qom, Iran.
7- Department of Physiology, Faculty of Medicine, Arak University of Medical Sciences, Arak, Iran.
8- Cellular and Molecular Research Center, Qom University of Medical Sciences, Qom, Iran. & Department of Physiology, Faculty of Medicine, Qom University of Medical Sciences, Qom, Iran. ,
Full-Text [PDF 1483 kb]
(627 Downloads)
| Abstract (HTML) (738 Views)
3.3. The effects of CHr on DEHP-induced changes of sperm analysis
As shown in figures 2 and 3, the results of this study demonstrated a decline in sperm count upon the use of DEHP, but this change was not statistically significant. Moreover, treatment of CHr to normal and DEHP-received mice increased sperm count, but sperm count was not remarkably altered. Receiving DEHP could not cause any significant change in the percentage of sperm viability, but sperm viability elevated after the treatment of normal mice with CHr (p < 0.05).
3.4. The histopathological examination of testes
As shown in figure 4, the histological observations of tissue samples in the control group showed a healthy appearance of the seminiferous epithelium of testicular tubules. The seminiferous tubule was lined by a complex germinal epithelium, which is a highly modified stratified cuboidal epithelium. Also, there were a number of spermatozoa in the lumen of tubule. The loss of germinal epithelium cells, degenerated germ cells with vacuolated cytoplasm and deeply stained nuclei, wide intercellular spaces between germinal layers of seminiferous tubules, and loss of spermatozoa in the center of lumens were observed in the DEHP group. Also, both CHr-treated groups showed the restoration of germinal epithelium, increased height of epithelium, and spermatozoa in the lumens.

4. Discussion
The purpose of this study was to clarify the effects of CHr on DEHP-induced testicular damage in mice. We found that exposure to DEHP for 14 days led to testicular dysfunction and disrupts normal reproductive function in male mice, which was consistent with Goudarzi’s study (25). This was demonstrated by a decrease in serum testosterone, moderate changes in serum LH, and FSH levels. Furthermore, we also showed that the changes mentioned in biochemical parameters and serum hormones were in parallel with the sperm count and testicular histological results. Phthalates such as DEHP and dibutyl phthalate disrupt the endocrine aspect of the reproductive system (26). It is well established that the administration of DEHP could induce Leydig cell dysfunction and adversely affect steroidogenesis in these cells, which is associated with a reduction in testosterone levels, sperm analysis, and delays puberty (27, 28). DEHP reduces testicular total antioxidant capacity and glutathione and increases malondialdehyde level. DEHP is mostly metabolized into an active metabolite named mono-(2-ethylhexyl) phthalate, which induces oxidative stress in testes (29). Moreover, the overgeneration of reactive oxygen species (ROS) may have a serious role in DEHP-induced testicular damage (30). The overproduction of ROS, through the activation of transcription factors, could induce the expression of pro-inflammatory agents, such as tumor necrosis factor alpha, interleukin, and interferons, leading to inflammation in male reproductive tissues (31). In this way, DEHP increases production of these inflammatory cytokines in the testis (8). It is well established that these inflammatory mediators could induce apoptotic cell death and aggravate testicular tissue injury (32). It seems that DEHP disrupts the antioxidant defense mechanisms of Leydig cells, germ cells, and Sertoli cells, which in turn can increase the ROS content and activate apoptotic signaling pathways. Our results showed that DEHP exposure could affect gonadotropin levels to some extent, and also it might be acting on the hypothalamic-pituitary-testicular (HPT) axis. A study by Yu et al. showed that DEHP could increase the serum LH levels (33). Although the exact mechanisms for the effects of DEHP on the HPT axis remain unclear, studies have shown that DEHP causes the elevation of gonadotropin releasing hormone (GnRH) level in the hypothalamus and pituitary (34). It seems that GnRH, by increasing the synthesis and secretion of gonadotropins in the pituitary, led to increased serum LH and FSH levels. Considering the elevating effect of DEHP on hypothalamic GnRH levels and results of our study showing that DEHP cannot be similar to testosterone or reduce the levels of pituitary gonadotropins, rather the main toxic effects of DEHP are on the reproductive organ and does not have much negative destructive effects on brain tissues. Histopathological evaluation illustrated that seminiferous luminal, tubular diameters, and epithelial height were on a normal scale in the control group. Also, marked histological damages were seen in DEHP-received mice, which was consistent with previous reports that showed histological damages in testicular tissue following DEHP administration (35). In a recent study, damages included degenerated germ cells with vacuolated cytoplasm, wide intercellular spaces between germinal layers of seminiferous tubules, and loss of spermatozoa in the center of lumens. Also, it should be said that this is a proposed hypothesis and requires further investigation, and a complete conclusion cannot be reached with a small number of studies. Considering the damage to testicular tissue by DEHP, data histopathology, and the role of DEHP in causing oxidative stress and inflammation in testicular tissue, the slight increase in gonadotropins by DEHP (non-significant) failed to cause a significant change in sperm status. Our results demonstrated that CHr treatment at the dose of 50 mg/kg for 14 consecutive days, with its protective effect, could attenuate DEHP-induced testicular toxicity, by restoring the serum testosterone level. As the aromatase enzyme converts testosterone to estradiol, CHr has been reported to have boosting actions on testosterone through the inhibition of aromatase enzyme. Of course, CHr administration in our research elevated serum gonadotropin, which is in accordance with previous research (36). Although the exact mechanism of this effect of CHr on gonadotropins is unclear, it is likely that the relative increase in FSH and LH levels by CHr in the group receiving DEHP could be the result of 2 factors. First, a possible increase in GnRH by DEHP. Second, CHr itself, as an antioxidant, may enhance the function and secretory power of pituitary gland cells by reducing inflammatory mediators and oxidative stress in pituitary cells. Lastly, based on the antioxidant effects of CHr, it can improve the function of Leydig cells in production and secretion of testosterone by reducing oxidative stress. Of course, providing the correct cause requires further investigation into the intracellular and molecular mechanisms of Leydig and pituitary cells. In addition, we also showed protective effects of CHr, which correlated with testicular histological and, to some extent, sperm viability results. The protective effects of CHr were confirmed by histopathological studies of testes, indicating CHr repaired germinal epithelium, increased height of epithelium, and narrowed lumens containing number of spermatozoa. In the current study, sperm count did not show any significant change under the influence of CHr, but sperm vitality was strikingly increased by CHr which might be attributed to the strong antioxidant and anti-inflammatory effects of CHr mentioned in other studies. Considering the high sensitivity of sperms and Leydig cells to damaging factors, CHr may be able to increase sperm vitality by severely reducing oxidative damage. The antioxidant activities of CHr have been demonstrated in previous studies. CHr as a powerful antioxidant agent, increases the concentrations of antioxidant enzymes such as superoxide dismutase and glutathione peroxidase. Accordingly, the protective effect of CHr on DEHP-induced gonadotoxicity may happen by modulating oxidative stress in the testis (15, 37). Previous reports clearly demonstrated that CHr effectively decreased oxidative stress chain reactions and showed positive effects on reduction of inflammatory mediators such as tumor necrosis factor alpha, different types of interleukins, and interferon gamma, which can damage the testicles (16). There were some limitations, including further spermatogenic activity evaluation, which should be carried out in the future study. Also, the problems of maintaining the mice caused a shortening of the research duration.
5. Conclusion
According to our biochemical and histopathological analyses, DEHP led to severe testicular injury in mice. Our results suggest that CHr, as a free radical scavenger, by decreasing the tissue damage induced by DEHP, can be beneficial in alleviating the toxic effects of DEHP on the male reproductive system, and also can be useful in preventing DEHP effects. However, recommendations for therapeutic use of CHr, in DEHP-induced testis damage, need more detailed research, especially clinical trial studies.
Data Availability
The data are available from the corresponding author on reasonable request.
Author Contributions
M. Goudarzi, S. Hajihashemi, and H. Heidari: Concept and design of the study. Z. Matin Manesh, H. Zeinali, and H. Heidari: Acquisition statistical analysis or interpretation of data. S. Hajihashemi: Funding acquisition. F. Ghanbari and S. Hajihashemi: Supervision. Z. Matin Manesh and H. Heidari: Drafting of the manuscript. H. Zeinali: Laboratory workup in the methodology section, animal anesthesia, surgery, and tissue separation. A. Mafi and M. Ghoddoosi: Histopathological study or evaluation of mice testicles. All authors approved the final manuscript of this study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Since part of this project was conducted under the guidance of Dr. Saeed Hajihashemi at Arak University of Medical Sciences, Arak, Iran and the other part under the guidance of Dr. Hamid Heidari at Qom University of Medical Sciences, Qom, Iran this article has 2 corresponding authors.
Acknowledgments
We thank Research Deputy of Arak University of Medical Sciences, Arak, Iran for financial support (Code: IR.ARAKMU.REC.1400.099). The authors are also deeply thankful for the technical collaboration and staff of the Cellular and Molecular Research Center of Qom University of Medical Sciences, Qom, Iran. Artificial intelligence was not used in any part of this research.
Conflict of Interest
The authors declare that there is no conflict of interest.
Full-Text: (121 Views)
1. Introduction
More than 2 million tons of di (2-ethylhexyl) phthalate (DEHP) are produced worldwide annually (1). DEHP as a plasticizer is used in many polyvinyl chloride products such as footwear, food packaging, and toys (2, 3). DEHP is used in a number of medical devices such as gloves, blood bags, nasogastric tubes, and peritoneal dialysis bags (4). The results of a study by Hosseinzadeh shows that DEHP causes a decrease in body and testis weight and testosterone levels in mice (5).
DEHP, through injury to testis tissue, decreases in spermatogenesis, epididymal weights, and leads to the degeneration of germ cells such as spermatocytes and spermatids. In addition, it is significantly associated with a decrease in seminal fluid volume and sperm count, sperm concentration, percentage of morphologically normal sperm, and acrosin activity (2, 6). Patients undergoing frequent hemodialysis, catheterization, or extensive blood transfusions are at particular risk for DEHP toxicity (4). Since polyvinyl chloride is not chemically connected to the polymer, it can separate from the polymer during use, so it can be transmitted to humans through air, water, food, and even by using medical devices (7).
Previous research has indicated that the testicular injury and toxicity of DEHP can be attributed to inducing oxidative stress and reducing the antioxidant capacity of tissues (8). Hence the use of antioxidant agents like flavonoids with their free radical scavenging ability may affect DEHP-mediated toxicity (9). Chrysin ([CHr] 5,7-dihydroxyflavone), a natural flavonoid that can decrease the oxidative stress-induced injury, can be found in honey and propolis (a resinous substance produced by honeybees from various plants) (9, 10). Despite antioxidant effects (11), CHr has a wide range of unique biological properties like anti-inflammatory and anti-apoptotic effects (12). It has been reported in different studies that CHr could prevent cognitive impairments (13), improving focal brain ischemia/reperfusion damage (14) and cerebral hypoperfusion (15). The preventive role of CHr from diabetes mellitus, obesity, cardiovascular disorders, and inflammatory events, along with hepatoprotective functions, has been evident from different research (16). Some studies have indicated the effect of CHr on boosting reproductive health, including increasing motility and decreasing abnormalities, apoptosis of sperm (17), improving role in semen concentration, fertility (18), and glutathione levels (19). It is also associated with regulating the normal morphology of the testis (20).
In recent years, researchers have shown great interest in CHr because of its well-known biological properties. Apart from the widespread usage of medicinal plants containing CHr for its desirable effect, the synthetic CHr is also widely used. Therefore, the current study was carried out to assess the protective effects of CHr supplementation on DEHP-induced testicular injury of mice.
2. Materials and Methods
2.1. Chemicals
DEHP (CAS number: 117-81-7) was purchased from Sigma-Aldrich Chemical Co., Osaka, Japan. Also, CHr (CAS number: 502-65-8) was purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA).
2.2. Experimental animals
50 male mice (4-6 wk) were obtained from the animal house of the Pasteur Institute of Iran, Tehran, Iran. Animals were maintained in 2021 in the animal house of Qom University of Medical Sciences, Qom, Iran at 20-24°C with a 12-hr light/dark cycle and with access to food and water ad libitum during the study (21).
2.3. Sample size
The number of samples in each group was selected based on previous similar studies. Of course, in such animal studies, due to the high probability of some mice's mortality during the study, the number of mice in each group is considered larger, so that the desired result can be obtained from the remaining number.
2.4. Experimental design/groups
Animals were randomly divided into 5 groups (n = 10/each). All drugs and vehicles were administered through oral gavage for 2 wk. Group I received normal saline (5 ml/kg/day), group II received corn oil (5 ml/kg/day), group III received DEHP (2 gr/kg, dissolved in corn oil), group IV received DEHP (2 gr/kg/day, dissolved in corn oil) and CHr (50 mg/kg/day, dissolved in corn oil), and group V received CHr (50 mg/kg [15], dissolved in corn oil).
2.5. Biochemical parameters
Overnight after the end of the experiment, blood samples were directly collected by cardiac puncture under mild general anesthesia and centrifuged (3000 rpm, 15 min) to separate serum samples (Eppendorf centrifuge 5702, Germany). The resulting serum was then transferred to polypropylene tubes and stored at -80°C until the hormonal testing. Eliza kit (Pars Azmun, Iran) were used to evaluate the serum testosterone, luteinizing hormone (LH) and follicle-stimulating hormone (FSH) levels.
2.6. Mice sperm analysis
The testicles of animals were immediately separated, and also the testicular weight (mg) was carefully examined by a digital scale (Kern, Germany) in each group. In this stage, the epididymis cauda was separated from the testicles of all groups, cut into small pieces in a petri dish, followed by adding 1 ml of normal saline. For sperm count and vitality, one drop of the above solution containing the sperm was placed onto a neubauer hemocytometer chamber (HBG Co., Germany), and also sperm analysis was performed manually using a microscope (Optika, Italy) in white blood cell grids. At the end, the data were reported as sperm per milliliter (22). Eosin staining (Merk Chemicals, Germany) was used to measure mice sperms viability; that is, the assessment of dead (stained) and live (unstained) sperms. 20 μl of the eosin solution was added to sperm suspension on the blood cell chamber of the neubauer hemocytometer at room temperature for 5 min. Sperm viability (%) was determined by manually counting the number of live and dead spermatozoa within 2 min under optical magnification of 40 times (23).
2.7. Histopathological evaluation of the mice testicles
The testis tissues were then removed from the body of the mice for histopathological study. For this purpose, the samples were embedded in a solution containing 10% formalin for at least 24 hr for fixation, the samples were then dehydrated with a sequence of ethanol solutions, embedded in paraffin, cut into 5 μm sections, and stained with hematoxylin and eosin dye (H&E stain). Slides were observed under a light microscope (Olympus, Tokyo, Japan) and a 10x objective lens connected to a camera (Digital Microscope BMZ-04-DZ) in a blind manner (24).
2.8. Ethical Considerations
All animal-related procedures were approved by the Ethical Committee of Arak University of Medical Sciences, Arak, Iran (Code: IR.ARAKMU.REC.1400.099), and also ethical guidelines for working with laboratory animals were followed in accordance with the agenda of the Ethics Committee of the Medical Sciences University.
2.9. Statistical Analysis
The data were expressed as mean ± standard error of mean. All comparisons were made by analysis of variance (one-way ANOVA), followed by Tukey’s test. A p < 0.05 was considered to be statistically significant. The data were analyzed by SPSS, V. 17.01 (SPSS Inc, Chicago, USA).
3. Results
3.1. The effects of DEHP and CHr on plasma levels of LH, FSH, and testosterone
Due to DEHP-induced toxicity, the plasma level of testosterone was significantly reduced (p < 0.001), and the LH and FSH levels also increased in comparison with the control group (Table I). After treatment of normal mice with CHr, a significant increase in testosterone level (p < 0.01). In addition, receiving CHr by the DEHP-exposed group increased serum testosterone (p < 0.01). Furthermore, the administration of CHr to normal mice increased the serum levels of LH and FSH (p < 0.001 and p < 0.01, respectively). Also, moderate growth in LH and FSH levels was observed in DEHP-receiving mice after the administration of CHr.
3.2. The effects of DEHP and CHr on testicular weight
More than 2 million tons of di (2-ethylhexyl) phthalate (DEHP) are produced worldwide annually (1). DEHP as a plasticizer is used in many polyvinyl chloride products such as footwear, food packaging, and toys (2, 3). DEHP is used in a number of medical devices such as gloves, blood bags, nasogastric tubes, and peritoneal dialysis bags (4). The results of a study by Hosseinzadeh shows that DEHP causes a decrease in body and testis weight and testosterone levels in mice (5).
DEHP, through injury to testis tissue, decreases in spermatogenesis, epididymal weights, and leads to the degeneration of germ cells such as spermatocytes and spermatids. In addition, it is significantly associated with a decrease in seminal fluid volume and sperm count, sperm concentration, percentage of morphologically normal sperm, and acrosin activity (2, 6). Patients undergoing frequent hemodialysis, catheterization, or extensive blood transfusions are at particular risk for DEHP toxicity (4). Since polyvinyl chloride is not chemically connected to the polymer, it can separate from the polymer during use, so it can be transmitted to humans through air, water, food, and even by using medical devices (7).
Previous research has indicated that the testicular injury and toxicity of DEHP can be attributed to inducing oxidative stress and reducing the antioxidant capacity of tissues (8). Hence the use of antioxidant agents like flavonoids with their free radical scavenging ability may affect DEHP-mediated toxicity (9). Chrysin ([CHr] 5,7-dihydroxyflavone), a natural flavonoid that can decrease the oxidative stress-induced injury, can be found in honey and propolis (a resinous substance produced by honeybees from various plants) (9, 10). Despite antioxidant effects (11), CHr has a wide range of unique biological properties like anti-inflammatory and anti-apoptotic effects (12). It has been reported in different studies that CHr could prevent cognitive impairments (13), improving focal brain ischemia/reperfusion damage (14) and cerebral hypoperfusion (15). The preventive role of CHr from diabetes mellitus, obesity, cardiovascular disorders, and inflammatory events, along with hepatoprotective functions, has been evident from different research (16). Some studies have indicated the effect of CHr on boosting reproductive health, including increasing motility and decreasing abnormalities, apoptosis of sperm (17), improving role in semen concentration, fertility (18), and glutathione levels (19). It is also associated with regulating the normal morphology of the testis (20).
In recent years, researchers have shown great interest in CHr because of its well-known biological properties. Apart from the widespread usage of medicinal plants containing CHr for its desirable effect, the synthetic CHr is also widely used. Therefore, the current study was carried out to assess the protective effects of CHr supplementation on DEHP-induced testicular injury of mice.
2. Materials and Methods
2.1. Chemicals
DEHP (CAS number: 117-81-7) was purchased from Sigma-Aldrich Chemical Co., Osaka, Japan. Also, CHr (CAS number: 502-65-8) was purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA).
2.2. Experimental animals
50 male mice (4-6 wk) were obtained from the animal house of the Pasteur Institute of Iran, Tehran, Iran. Animals were maintained in 2021 in the animal house of Qom University of Medical Sciences, Qom, Iran at 20-24°C with a 12-hr light/dark cycle and with access to food and water ad libitum during the study (21).
2.3. Sample size
The number of samples in each group was selected based on previous similar studies. Of course, in such animal studies, due to the high probability of some mice's mortality during the study, the number of mice in each group is considered larger, so that the desired result can be obtained from the remaining number.
2.4. Experimental design/groups
Animals were randomly divided into 5 groups (n = 10/each). All drugs and vehicles were administered through oral gavage for 2 wk. Group I received normal saline (5 ml/kg/day), group II received corn oil (5 ml/kg/day), group III received DEHP (2 gr/kg, dissolved in corn oil), group IV received DEHP (2 gr/kg/day, dissolved in corn oil) and CHr (50 mg/kg/day, dissolved in corn oil), and group V received CHr (50 mg/kg [15], dissolved in corn oil).
2.5. Biochemical parameters
Overnight after the end of the experiment, blood samples were directly collected by cardiac puncture under mild general anesthesia and centrifuged (3000 rpm, 15 min) to separate serum samples (Eppendorf centrifuge 5702, Germany). The resulting serum was then transferred to polypropylene tubes and stored at -80°C until the hormonal testing. Eliza kit (Pars Azmun, Iran) were used to evaluate the serum testosterone, luteinizing hormone (LH) and follicle-stimulating hormone (FSH) levels.
2.6. Mice sperm analysis
The testicles of animals were immediately separated, and also the testicular weight (mg) was carefully examined by a digital scale (Kern, Germany) in each group. In this stage, the epididymis cauda was separated from the testicles of all groups, cut into small pieces in a petri dish, followed by adding 1 ml of normal saline. For sperm count and vitality, one drop of the above solution containing the sperm was placed onto a neubauer hemocytometer chamber (HBG Co., Germany), and also sperm analysis was performed manually using a microscope (Optika, Italy) in white blood cell grids. At the end, the data were reported as sperm per milliliter (22). Eosin staining (Merk Chemicals, Germany) was used to measure mice sperms viability; that is, the assessment of dead (stained) and live (unstained) sperms. 20 μl of the eosin solution was added to sperm suspension on the blood cell chamber of the neubauer hemocytometer at room temperature for 5 min. Sperm viability (%) was determined by manually counting the number of live and dead spermatozoa within 2 min under optical magnification of 40 times (23).
2.7. Histopathological evaluation of the mice testicles
The testis tissues were then removed from the body of the mice for histopathological study. For this purpose, the samples were embedded in a solution containing 10% formalin for at least 24 hr for fixation, the samples were then dehydrated with a sequence of ethanol solutions, embedded in paraffin, cut into 5 μm sections, and stained with hematoxylin and eosin dye (H&E stain). Slides were observed under a light microscope (Olympus, Tokyo, Japan) and a 10x objective lens connected to a camera (Digital Microscope BMZ-04-DZ) in a blind manner (24).
2.8. Ethical Considerations
All animal-related procedures were approved by the Ethical Committee of Arak University of Medical Sciences, Arak, Iran (Code: IR.ARAKMU.REC.1400.099), and also ethical guidelines for working with laboratory animals were followed in accordance with the agenda of the Ethics Committee of the Medical Sciences University.
2.9. Statistical Analysis
The data were expressed as mean ± standard error of mean. All comparisons were made by analysis of variance (one-way ANOVA), followed by Tukey’s test. A p < 0.05 was considered to be statistically significant. The data were analyzed by SPSS, V. 17.01 (SPSS Inc, Chicago, USA).
3. Results
3.1. The effects of DEHP and CHr on plasma levels of LH, FSH, and testosterone
Due to DEHP-induced toxicity, the plasma level of testosterone was significantly reduced (p < 0.001), and the LH and FSH levels also increased in comparison with the control group (Table I). After treatment of normal mice with CHr, a significant increase in testosterone level (p < 0.01). In addition, receiving CHr by the DEHP-exposed group increased serum testosterone (p < 0.01). Furthermore, the administration of CHr to normal mice increased the serum levels of LH and FSH (p < 0.001 and p < 0.01, respectively). Also, moderate growth in LH and FSH levels was observed in DEHP-receiving mice after the administration of CHr.
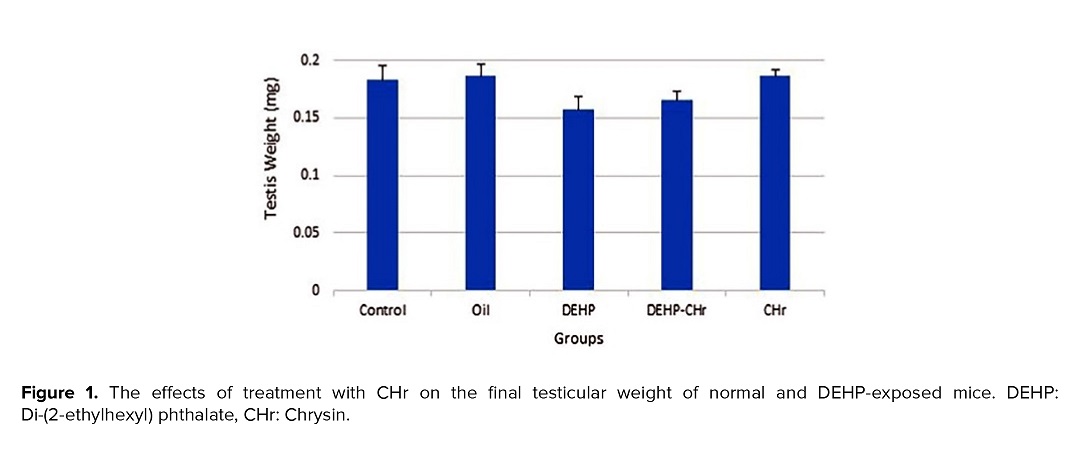
3.2. The effects of DEHP and CHr on testicular weight
The assessment of the testicles weight showed that the testis weight was diminished in the mice receiving DEHP compared to the intact control group, but this decrement was not statistically significant. The administration of CHr had no significant effect on the testicular weight of animals in the control and DEHP-received groups (Figure 1).
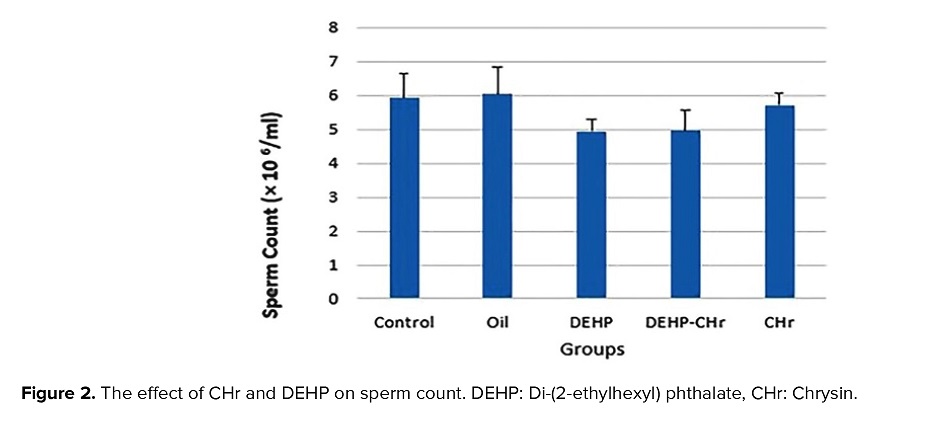
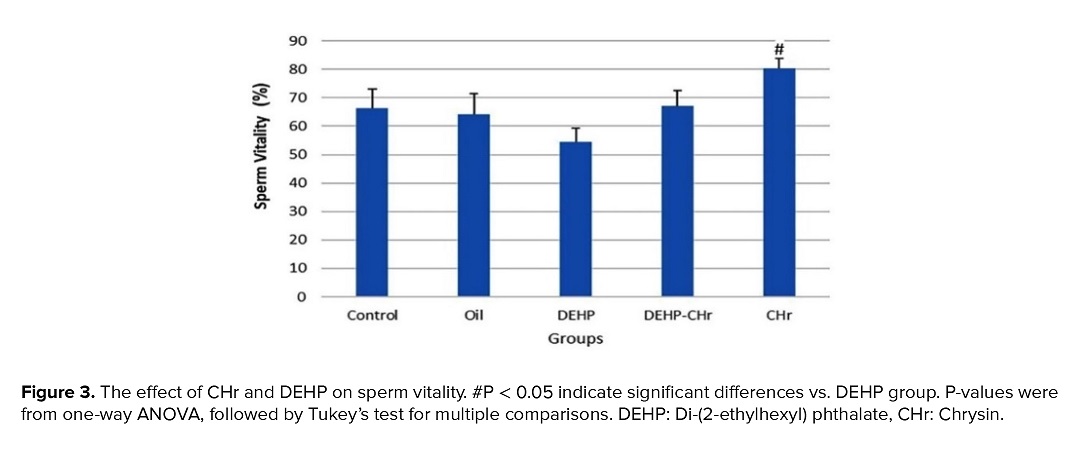
3.3. The effects of CHr on DEHP-induced changes of sperm analysis
As shown in figures 2 and 3, the results of this study demonstrated a decline in sperm count upon the use of DEHP, but this change was not statistically significant. Moreover, treatment of CHr to normal and DEHP-received mice increased sperm count, but sperm count was not remarkably altered. Receiving DEHP could not cause any significant change in the percentage of sperm viability, but sperm viability elevated after the treatment of normal mice with CHr (p < 0.05).
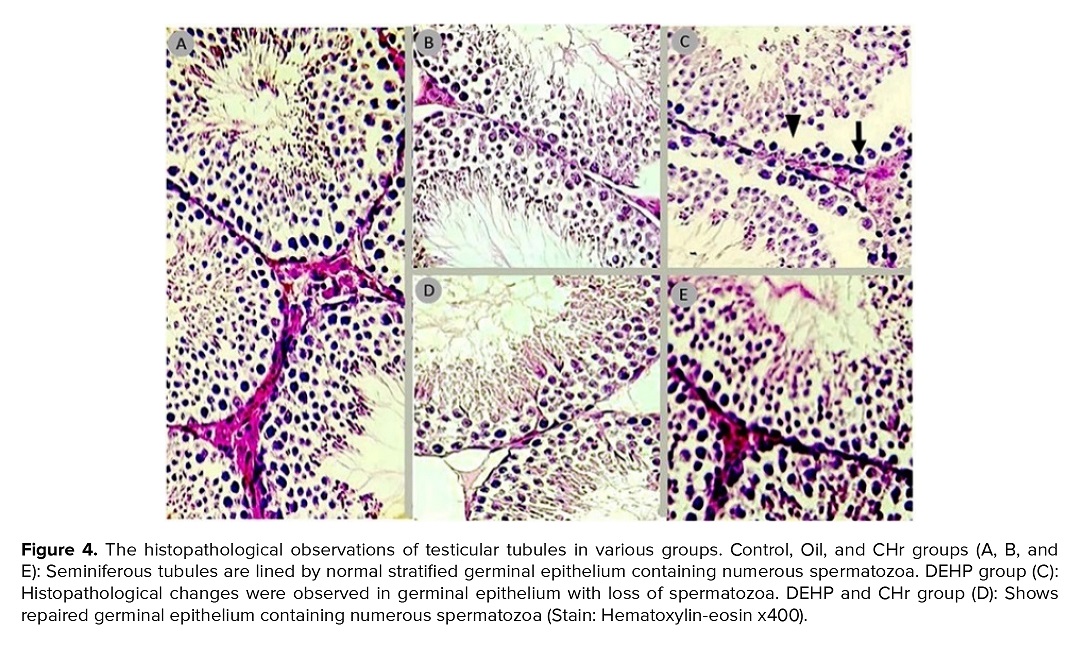
3.4. The histopathological examination of testes
As shown in figure 4, the histological observations of tissue samples in the control group showed a healthy appearance of the seminiferous epithelium of testicular tubules. The seminiferous tubule was lined by a complex germinal epithelium, which is a highly modified stratified cuboidal epithelium. Also, there were a number of spermatozoa in the lumen of tubule. The loss of germinal epithelium cells, degenerated germ cells with vacuolated cytoplasm and deeply stained nuclei, wide intercellular spaces between germinal layers of seminiferous tubules, and loss of spermatozoa in the center of lumens were observed in the DEHP group. Also, both CHr-treated groups showed the restoration of germinal epithelium, increased height of epithelium, and spermatozoa in the lumens.





4. Discussion
The purpose of this study was to clarify the effects of CHr on DEHP-induced testicular damage in mice. We found that exposure to DEHP for 14 days led to testicular dysfunction and disrupts normal reproductive function in male mice, which was consistent with Goudarzi’s study (25). This was demonstrated by a decrease in serum testosterone, moderate changes in serum LH, and FSH levels. Furthermore, we also showed that the changes mentioned in biochemical parameters and serum hormones were in parallel with the sperm count and testicular histological results. Phthalates such as DEHP and dibutyl phthalate disrupt the endocrine aspect of the reproductive system (26). It is well established that the administration of DEHP could induce Leydig cell dysfunction and adversely affect steroidogenesis in these cells, which is associated with a reduction in testosterone levels, sperm analysis, and delays puberty (27, 28). DEHP reduces testicular total antioxidant capacity and glutathione and increases malondialdehyde level. DEHP is mostly metabolized into an active metabolite named mono-(2-ethylhexyl) phthalate, which induces oxidative stress in testes (29). Moreover, the overgeneration of reactive oxygen species (ROS) may have a serious role in DEHP-induced testicular damage (30). The overproduction of ROS, through the activation of transcription factors, could induce the expression of pro-inflammatory agents, such as tumor necrosis factor alpha, interleukin, and interferons, leading to inflammation in male reproductive tissues (31). In this way, DEHP increases production of these inflammatory cytokines in the testis (8). It is well established that these inflammatory mediators could induce apoptotic cell death and aggravate testicular tissue injury (32). It seems that DEHP disrupts the antioxidant defense mechanisms of Leydig cells, germ cells, and Sertoli cells, which in turn can increase the ROS content and activate apoptotic signaling pathways. Our results showed that DEHP exposure could affect gonadotropin levels to some extent, and also it might be acting on the hypothalamic-pituitary-testicular (HPT) axis. A study by Yu et al. showed that DEHP could increase the serum LH levels (33). Although the exact mechanisms for the effects of DEHP on the HPT axis remain unclear, studies have shown that DEHP causes the elevation of gonadotropin releasing hormone (GnRH) level in the hypothalamus and pituitary (34). It seems that GnRH, by increasing the synthesis and secretion of gonadotropins in the pituitary, led to increased serum LH and FSH levels. Considering the elevating effect of DEHP on hypothalamic GnRH levels and results of our study showing that DEHP cannot be similar to testosterone or reduce the levels of pituitary gonadotropins, rather the main toxic effects of DEHP are on the reproductive organ and does not have much negative destructive effects on brain tissues. Histopathological evaluation illustrated that seminiferous luminal, tubular diameters, and epithelial height were on a normal scale in the control group. Also, marked histological damages were seen in DEHP-received mice, which was consistent with previous reports that showed histological damages in testicular tissue following DEHP administration (35). In a recent study, damages included degenerated germ cells with vacuolated cytoplasm, wide intercellular spaces between germinal layers of seminiferous tubules, and loss of spermatozoa in the center of lumens. Also, it should be said that this is a proposed hypothesis and requires further investigation, and a complete conclusion cannot be reached with a small number of studies. Considering the damage to testicular tissue by DEHP, data histopathology, and the role of DEHP in causing oxidative stress and inflammation in testicular tissue, the slight increase in gonadotropins by DEHP (non-significant) failed to cause a significant change in sperm status. Our results demonstrated that CHr treatment at the dose of 50 mg/kg for 14 consecutive days, with its protective effect, could attenuate DEHP-induced testicular toxicity, by restoring the serum testosterone level. As the aromatase enzyme converts testosterone to estradiol, CHr has been reported to have boosting actions on testosterone through the inhibition of aromatase enzyme. Of course, CHr administration in our research elevated serum gonadotropin, which is in accordance with previous research (36). Although the exact mechanism of this effect of CHr on gonadotropins is unclear, it is likely that the relative increase in FSH and LH levels by CHr in the group receiving DEHP could be the result of 2 factors. First, a possible increase in GnRH by DEHP. Second, CHr itself, as an antioxidant, may enhance the function and secretory power of pituitary gland cells by reducing inflammatory mediators and oxidative stress in pituitary cells. Lastly, based on the antioxidant effects of CHr, it can improve the function of Leydig cells in production and secretion of testosterone by reducing oxidative stress. Of course, providing the correct cause requires further investigation into the intracellular and molecular mechanisms of Leydig and pituitary cells. In addition, we also showed protective effects of CHr, which correlated with testicular histological and, to some extent, sperm viability results. The protective effects of CHr were confirmed by histopathological studies of testes, indicating CHr repaired germinal epithelium, increased height of epithelium, and narrowed lumens containing number of spermatozoa. In the current study, sperm count did not show any significant change under the influence of CHr, but sperm vitality was strikingly increased by CHr which might be attributed to the strong antioxidant and anti-inflammatory effects of CHr mentioned in other studies. Considering the high sensitivity of sperms and Leydig cells to damaging factors, CHr may be able to increase sperm vitality by severely reducing oxidative damage. The antioxidant activities of CHr have been demonstrated in previous studies. CHr as a powerful antioxidant agent, increases the concentrations of antioxidant enzymes such as superoxide dismutase and glutathione peroxidase. Accordingly, the protective effect of CHr on DEHP-induced gonadotoxicity may happen by modulating oxidative stress in the testis (15, 37). Previous reports clearly demonstrated that CHr effectively decreased oxidative stress chain reactions and showed positive effects on reduction of inflammatory mediators such as tumor necrosis factor alpha, different types of interleukins, and interferon gamma, which can damage the testicles (16). There were some limitations, including further spermatogenic activity evaluation, which should be carried out in the future study. Also, the problems of maintaining the mice caused a shortening of the research duration.
5. Conclusion
According to our biochemical and histopathological analyses, DEHP led to severe testicular injury in mice. Our results suggest that CHr, as a free radical scavenger, by decreasing the tissue damage induced by DEHP, can be beneficial in alleviating the toxic effects of DEHP on the male reproductive system, and also can be useful in preventing DEHP effects. However, recommendations for therapeutic use of CHr, in DEHP-induced testis damage, need more detailed research, especially clinical trial studies.
Data Availability
The data are available from the corresponding author on reasonable request.
Author Contributions
M. Goudarzi, S. Hajihashemi, and H. Heidari: Concept and design of the study. Z. Matin Manesh, H. Zeinali, and H. Heidari: Acquisition statistical analysis or interpretation of data. S. Hajihashemi: Funding acquisition. F. Ghanbari and S. Hajihashemi: Supervision. Z. Matin Manesh and H. Heidari: Drafting of the manuscript. H. Zeinali: Laboratory workup in the methodology section, animal anesthesia, surgery, and tissue separation. A. Mafi and M. Ghoddoosi: Histopathological study or evaluation of mice testicles. All authors approved the final manuscript of this study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Since part of this project was conducted under the guidance of Dr. Saeed Hajihashemi at Arak University of Medical Sciences, Arak, Iran and the other part under the guidance of Dr. Hamid Heidari at Qom University of Medical Sciences, Qom, Iran this article has 2 corresponding authors.
Acknowledgments
We thank Research Deputy of Arak University of Medical Sciences, Arak, Iran for financial support (Code: IR.ARAKMU.REC.1400.099). The authors are also deeply thankful for the technical collaboration and staff of the Cellular and Molecular Research Center of Qom University of Medical Sciences, Qom, Iran. Artificial intelligence was not used in any part of this research.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Fertility & Infertility
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |




