Thu, May 21, 2026
[Archive]
Volume 24, Issue 1 (January 2026)
IJRM 2026, 24(1): 37-44 |
Back to browse issues page
Ethics code: IR.ACECR.ROYAN.REC.1403.044
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Babaei M, Sadighi Gilani M A, Ramezani-Binabaj M, Arasteh H, Yahyazadeh S R. Outcomes of microdissection testicular sperm extraction in azoospermic men following chemotherapy: A cross-sectional study. IJRM 2026; 24 (1) :37-44
URL: http://ijrm.ir/article-1-3635-en.html
URL: http://ijrm.ir/article-1-3635-en.html
Mehdi Babaei1 

 , Mohammad Ali Sadighi Gilani1
, Mohammad Ali Sadighi Gilani1 

 , Mahdi Ramezani-Binabaj *2
, Mahdi Ramezani-Binabaj *2 

 , Hamid Arasteh1
, Hamid Arasteh1 

 , Seyed Reza Yahyazadeh3
, Seyed Reza Yahyazadeh3 




 , Mohammad Ali Sadighi Gilani1
, Mohammad Ali Sadighi Gilani1 

 , Mahdi Ramezani-Binabaj *2
, Mahdi Ramezani-Binabaj *2 

 , Hamid Arasteh1
, Hamid Arasteh1 

 , Seyed Reza Yahyazadeh3
, Seyed Reza Yahyazadeh3 


1- Department of Andrology, Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine, Tehran, Iran. & Department of Urology, Shariati Hospital, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran.
2- Department of Urology, Shariati Hospital, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran. ,m-ramezanib@razi.tums.ac.ir
3- Department of Urology, Shariati Hospital, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran.
2- Department of Urology, Shariati Hospital, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran. ,
3- Department of Urology, Shariati Hospital, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran.
Full-Text [PDF 385 kb]
(204 Downloads)
| Abstract (HTML) (218 Views)
Full-Text: (51 Views)
1. Introduction
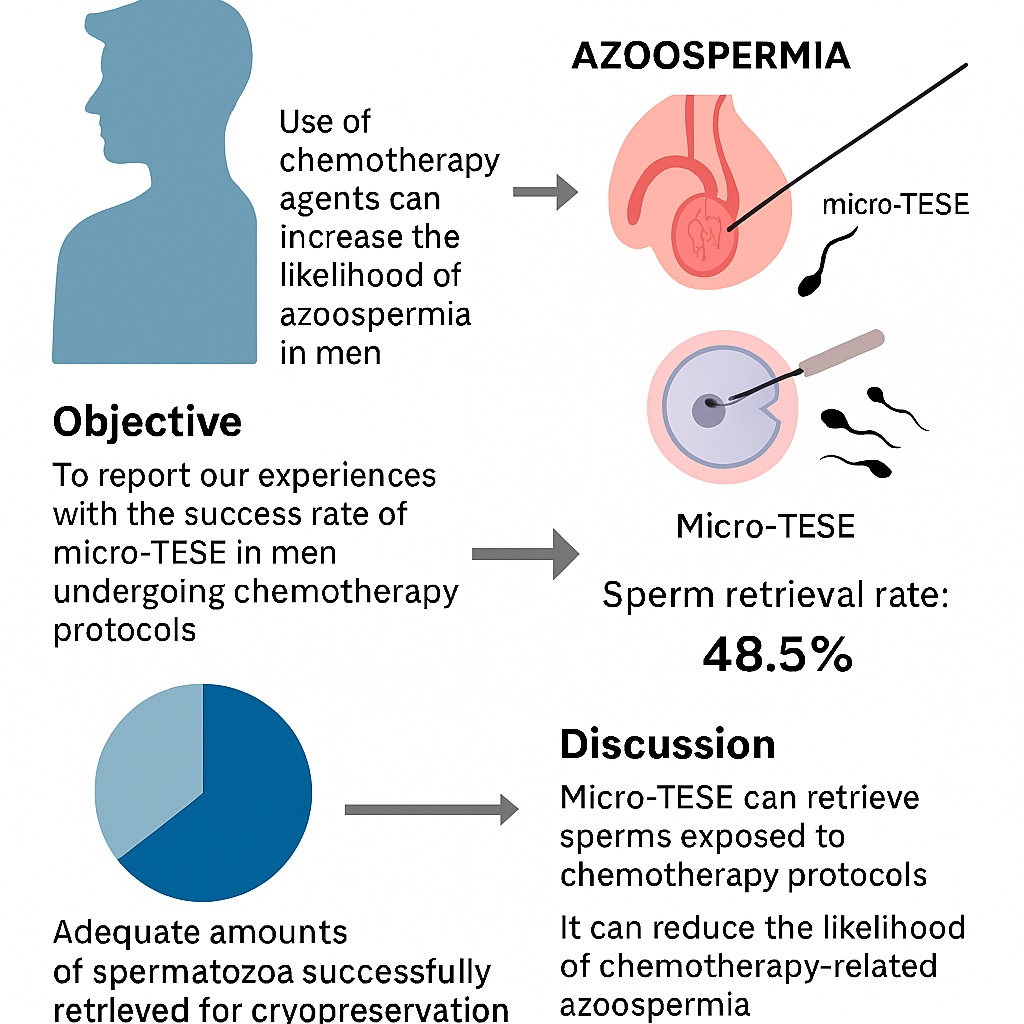
Developing innovative chemotherapeutic protocols holds significant promise for enhancing the survival rates of individuals afflicted with various malignancies. However, a crucial concern arises when considering appropriate treatment strategies for male cancer patients, namely the restoration of their fertilization ability and the achievement of parenthood (1). Unfortunately, a well-documented adverse effect of chemotherapy agents is non-obstructive azoospermia (NOA), which affects approximately 2-3rd of men undergoing chemotherapy treatments, with only a minority experiencing subsequent recovery of spermatogenesis post-treatment (2, 3). This undesirable consequence primarily arises from the utilization of alkylating chemotherapy agents such as cyclophosphamide, busulfan, procarbazine or chlorambucil (4, 5). The extent of gonadotoxic effects and subsequent germ cell damage is influenced by various factors, including the pre-treatment quality of spermatogenesis, the specific type of malignancy, the cancer treatment protocol employed, and individual susceptibility (6). Consequently, all men receiving such chemotherapy are strongly advised to consider sperm cryopreservation; however, this approach may be limited due to factors such as inadequate population awareness regarding chemotherapy-induced infertility, initial poor sperm quality, unavailability of sperm banking facilities, or prepubertal age of the male (7).
Fortunately, the development of assisted reproductive technologies has successfully addressed and mitigated this predicament. Among the available treatment options for affected men grappling with chemotherapy-induced infertility, the employment of micro-dissection testicular sperm extraction (micro-TESE) followed by intracytoplasmic sperm injection (ICSI) has emerged as the preferred and optimal approach (8, 9). This treatment modality proves invaluable for individuals lacking access to cryopreservation, as well as those who are exposed to gonadotoxic chemotherapy agents. A growing body of evidence supports the utilization of this protocol, with recent reports highlighting its remarkable success rates in retrieving sperm from men suffering from post-chemotherapy azoospermia (10). Notably, studies have demonstrated a significant increase in sperm retrieval rates (SRR), with success rates reaching up to 44% (11).
In the present study, we aim to provide a comprehensive analysis of our experiences with the micro-TESE protocol, focusing on the success rate of sperm restoration in men undergoing chemotherapy protocols.
2. Materials and Methods
In this cross-sectional study, we reviewed the medical records of 33 men with NOA secondary to chemotherapy for different types of malignancies, who underwent micro-TESE followed by ICSI, and were referred to Royan Institute for Reproductive Biomedicine (Tehran, Iran) between 2010 and 2023. Extracted data included baseline characteristics, such as age, type of malignancies, duration of chemotherapy treatment, serum levels of testosterone, follicle-stimulating hormone (FSH), and luteinizing hormone (LH), history of cigarette, alcohol, or hookah use, as well as diametric features of the testes (measured using an orchidometer and ultrasound examination). This study was retrospective in nature. Accordingly, the sample size was based on the total number of eligible patients referred to our institution and met the inclusion criteria. No sample size calculation was performed prior to data collection, and no interim analysis or stopping rules were applied.
Inclusion criteria were: 1) NOA resulting from the use of chemotherapy agents for different types of malignancies, 2) scheduled for micro-TESE followed by ICSI, 3) demonstrated normal sexual hormonal conditions, as indicated by normal serum levels of FSH and LH.
Exclusion criteria encompassed individuals with hypogonadotropic hypogonadism, any genetic or chromosomal defects affecting fertility (e.g., Klinefelter syndrome, microdeletion of azoospermia factors, or chromosomal translocations), or those with possible obstructive azoospermia. Andrological and hormonal evaluations were performed prior to the operation to determine the primary causes of azoospermia.
Azoospermia was defined as the absence of spermatozoa in 2 consecutive semen analyses and confirmed by at least 3 centrifuged semen pellet analyses.
Following initial assessment, micro-TESE was performed under general anesthesia according to the procedure previously described by Schlegel and Li (12). In this procedure, the testis is carefully opened, allowing for extensive dissection to explore all possible areas of sperm production within the divided halves of the testis. Successful sperm retrieval during micro-TESE was defined as the detection of at least one sperm cell by an experienced andrologist in the operating room. This was achieved through phase contrast examination of dispersed and minced testis tissue, which was carefully placed on a plain glass slide and covered with a slip. If viable sperm were successfully retrieved through microdissection, they were utilized for ICSI either on the same day or the following day after retrieval. Testicular biopsies were obtained and sent to a single laboratory for histopathological assessment, where a genitourinary pathologist examined them to determine the predominant histopathology. The study endpoint was to assess the outcomes of sperm extraction and related determinants.
2.1. Ethical Considerations
This study was approved by the Ethics Committee of Royan Institute for Reproductive Biomedicine, Tehran, Iran (Code: IR.ACECR.ROYAN.REC.1403.044). Given the retrospective nature of the research and use of de-identified participant data, the requirement for informed consent was waived by the committee. Data confidentiality was maintained, and participants’ personal information was protected from unauthorized access or disclosure.
2.2. Statistical Analysis
Quantitative variables were presented as mean ± standard deviation, while categorical variables were summarized by frequency (percentage). Continuous variables were compared using a t test or Mann-Whitney U test when the data did not appear to follow a normal distribution or when the assumption of equal variances was violated across the study groups. Categorical variables were compared using the Chi-Square test. A p ≤ 0.05 was considered statistically significant. All analyses were performed using Statistical Package for the Social Sciences, version 23.0 for Windows (IBM Corp., Armonk, NY, USA).
3. Results
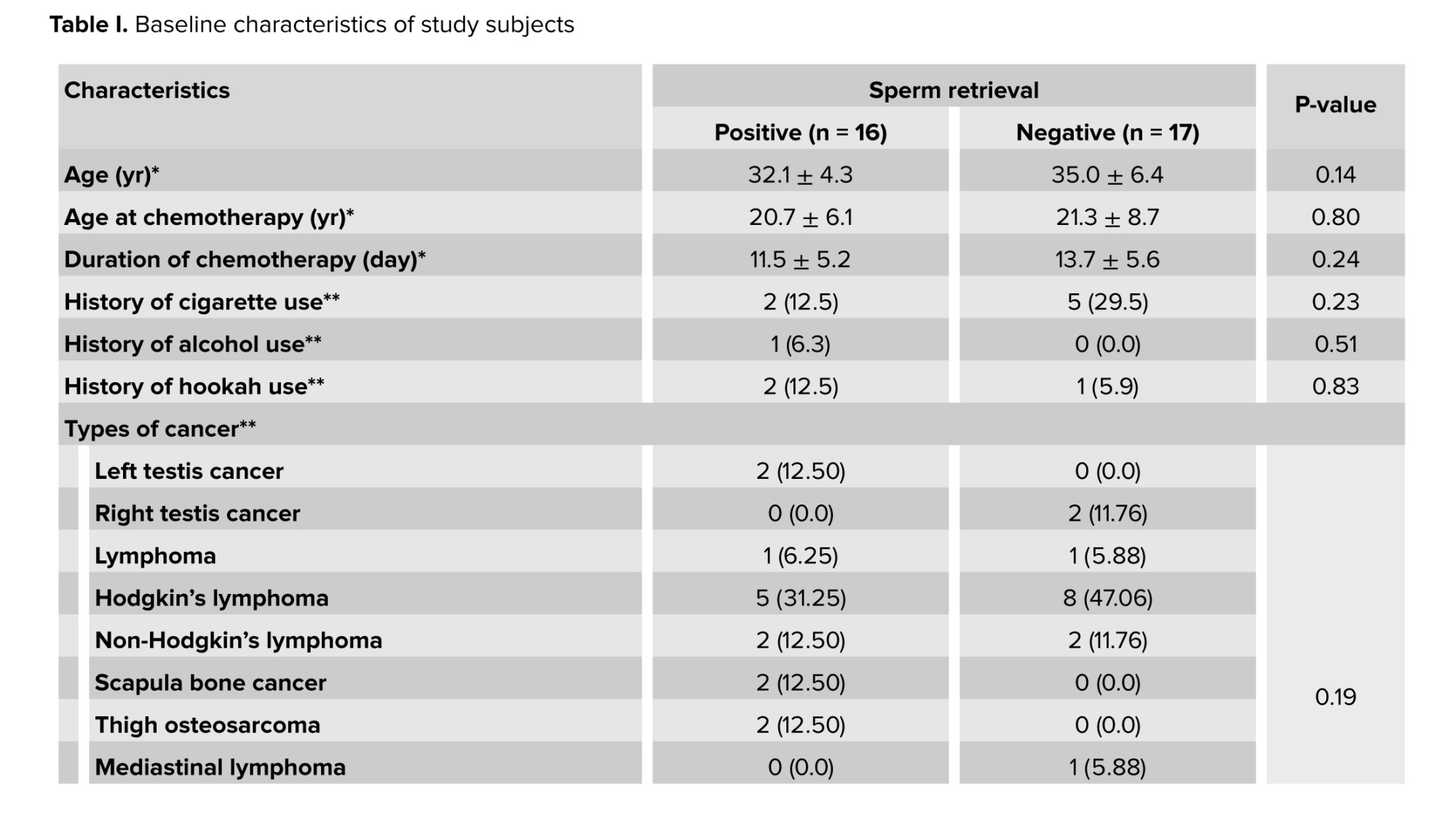
Data of 33 men suffering from post-chemotherapy NOA were included in this study. Table I represents baseline characteristics of study objects. Mean age at the start of chemotherapy and at the time of surgery were 21 ± 7.4 and 33.6 ± 5.6, respectively. Mean FSH, LH, and testosterone levels were 19.9 ± 13.5, 9.6 ± 5.3, and 3.4 ± 1.5, respectively, which indicates primary spermatogenic failure. The most dominant cancer among cases was Hodgkin's lymphoma (39.39%).
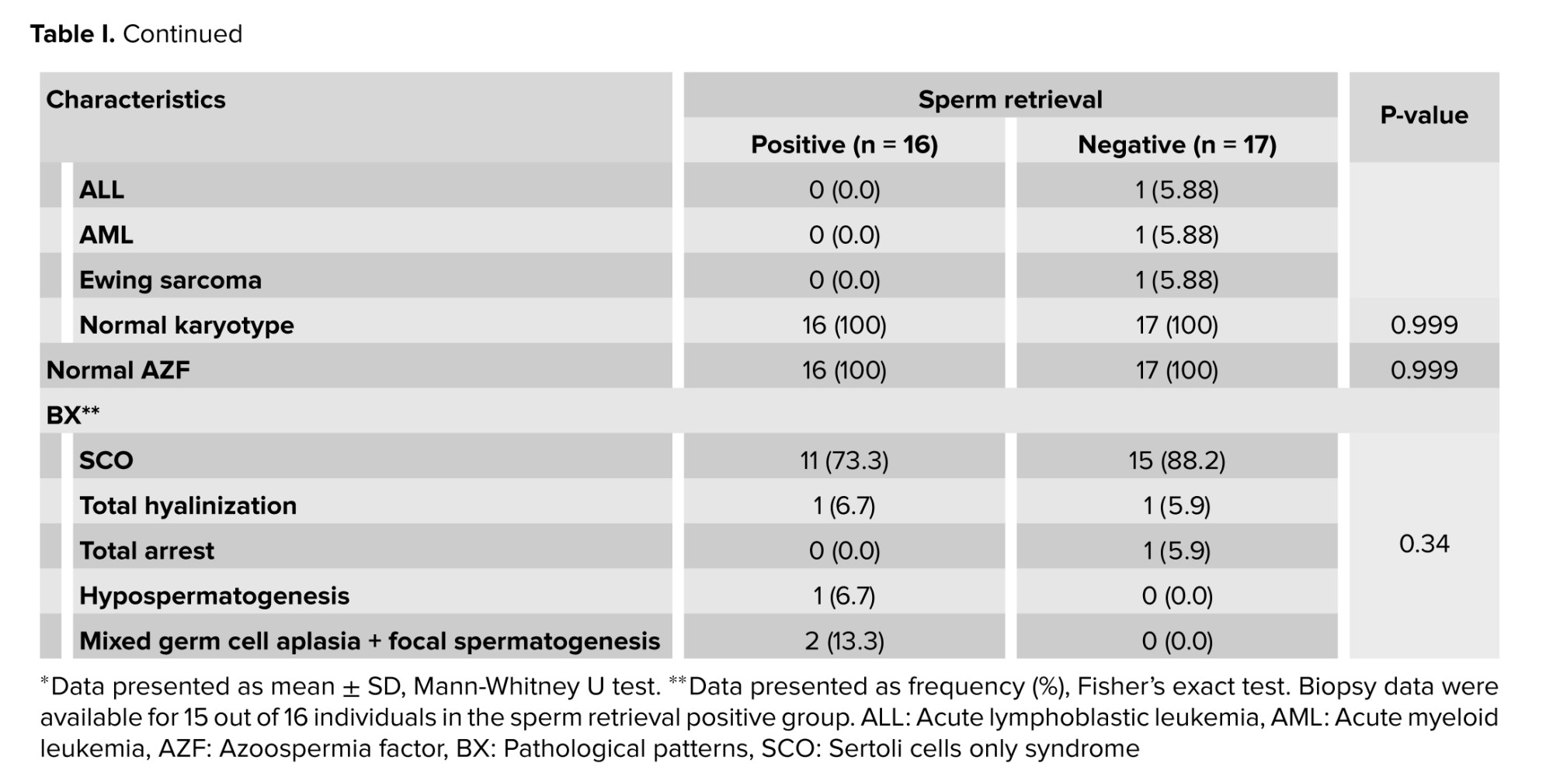
Adequate amounts of spermatozoa were successfully retrieved for cryopreservation and subsequent ICSI in 16 out of 33 men, leading SRR of 48.5%. According to this sperm extraction outcome, the cases were assigned into 2 positive and negative groups. No difference was observed between the positive and negative subgroups in baseline characteristics, including mean age, mean age at chemotherapy, type of malignancies, and the results of baseline karyotype analysis (Table I). The maximum number of samples taken from the right and left testicles were 64 and 85, respectively. Testicular biopsies were evaluated pathologically. The most pathological pattern was Sertoli cells only syndrome. The other pathological patterns included total hyalinization, total arrest, hypospermatogenesis, and mixed germ cell aplasia plus focal spermatogenesis. In terms of micro-TESE results, no significant association was observed between the positive and negative subgroups in testicular pathologies.
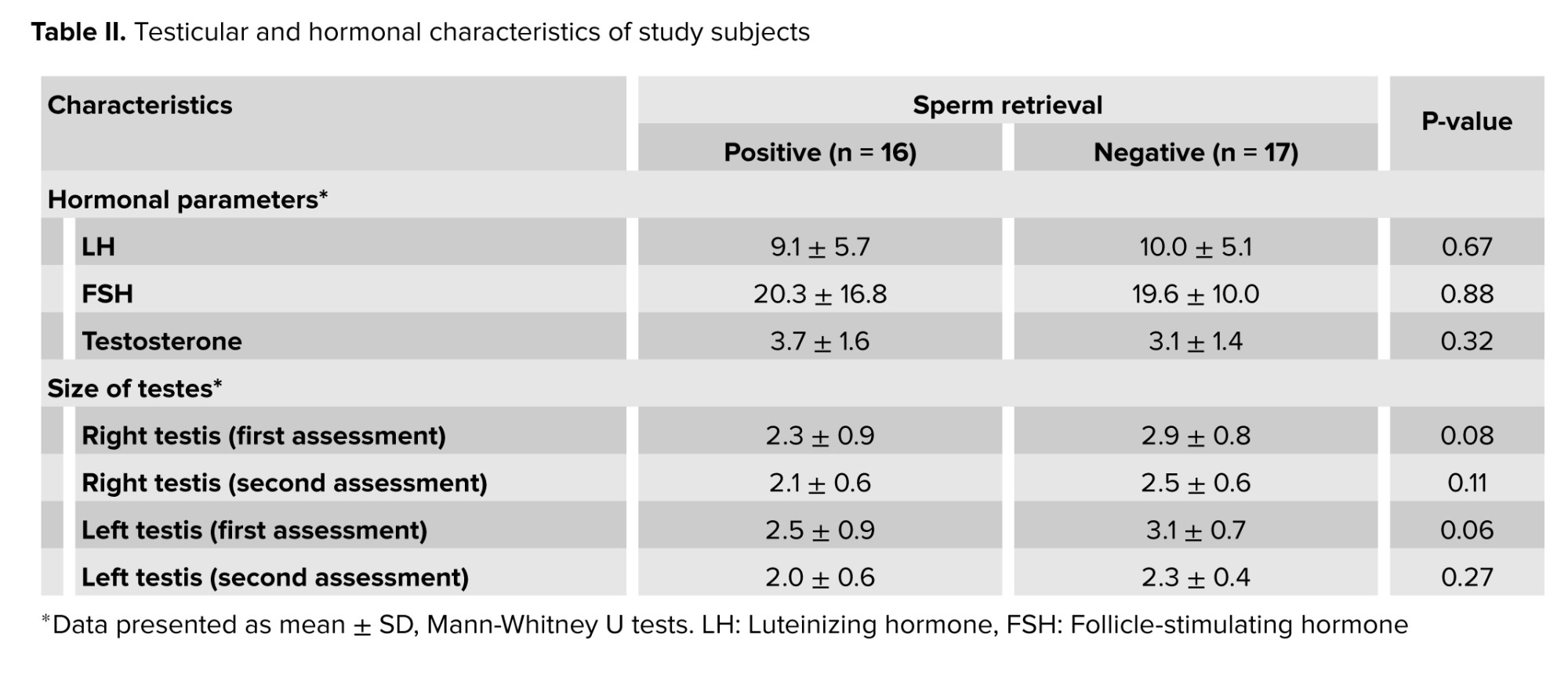
As presented in table II, orchidometer measurement revealed no statistically significant difference between the 2 groups with respect to left and right testicular size.
The serum levels of sexual hormones, including testosterone, FSH, and LH, were also not different between the 2 groups. Overall, none of the factors studied were significant between the 2 groups with or without sperm extraction.
4. Discussion
In this study, the most common malignancy was Hodgkin’s lymphoma. SRR occurred in 16 of 33 cases (48.5%), with no statistically significant differences in baseline characteristics, cancer type, karyotype, or azoospermia factor results between successful and unsuccessful groups. Histopathological examination revealed Sertoli cells only syndrome as the predominant finding, with additional patterns including complete hyalinization, spermatogenic arrest, hypospermatogenesis, and mixed germ cell aplasia with focal spermatogenesis. Statistical analysis revealed no significant differences between the 2 groups with respect to orchidometric measurements and serum sex hormone levels (LH, FSH, and testosterone).
The development of novel therapeutic protocols, including chemotherapy regimens for the treatment of malignancies, have significantly increased patients' long-term survival. However, the use of some of these agents might adversely affect reproductive abilities, especially in men. According to reports, certain chemotherapy regimens may lead to spermatogenic dysfunction by inducing defects in germ cells and spermatogonia differentiation. In other words, the rapid differentiation of these cells is highly sensitive to these cytotoxic medications. Therefore, within 2-3 months after beginning chemotherapy, the likelihood of azoospermia may be increased (13).
Based on the type, dosage, and duration of the chemotherapy regimen, azoospermia may be transient or irreversible. However, it is impossible to predict which chemotherapy regimen may lead to transient or permanent defects in spermatogenesis. Thus, considering the cryopreservation of sperm even before the initiation of chemotherapy can warrant post-treatment fertility potential in men. Although the protocol of micro-TESE followed by ICSI in azoospermic men after chemotherapy has resulted in successful preservation and recovery of spermatogenesis, the rate of this recovery has been reported to vary. As demonstrated in the present study, the SRR was found to be 48.5%. In a comparable study (14), sperm was successfully retrieved in 47%, which was close to our retrieval rate. In another study (15), sperm retrieval was successful in 42% of the individuals. However, they also demonstrated that men with post-chemotherapy azoospermia after testicular cancer treatment were able to achieve a high retrieval rate (75%), and exposure to alkylating agents resulted in lower SRR.
In a recent study, the authors evaluated the effectiveness of cyclophosphamide equivalent dose in sperm retrieval in men with persistent azoospermia following chemotherapy. SRR was lower in patients who received a chemotherapy regimen with alkylating agents. Patients with cyclophosphamide equivalent dose > 4000 mg/m2 did not have viable sperm in micro-TESE (16).
In another study, spermatozoa were successfully retrieved in 37% of men and in 42.9% of overall procedures (17). A fertilization rate of 57.1% (per injected oocyte) was achieved with ICSI, leading to a clinical pregnancy rate of 50% with an overall live birth rate of 42%.
Therefore, the introduction of these techniques is an important step in maintaining fertility for men undergoing chemotherapy, we also have a long way to go to prevent azoospermia after this protocol. Another important issue related to the success of micro-TESE followed by ICSI protocols in sperm retrieval is identifying the potential determinants that influence the success rate of these protocols. As indicated in the present study, none of the baseline parameters, including individuals age, time, and duration of chemotherapy, type of cancer, history of smoking or alcohol use, or baseline hormonal states, appeared to affect the rate of sperm retrieval. However, different studies have shown that some baseline parameters might affect the rate of sperm recovery. In line with previous findings, age, time since chemotherapy, FSH, and the initial diagnostic indication for chemotherapy did not affect the success of sperm retrieval. They also indicated that none of the abovementioned factors could predict TESE outcomes (14).
However, in some studies, bone marrow transplantation was an unfavorable factor affecting sperm retrieval following such procedures (14, 18). It has been reported that the rate of sperm recovery can be potentially affected by the type of alkylating agents (15). Therefore, regardless of what factors may affect the recovery rate of sperm, the use of these techniques is currently considered the only way to increase the chances of maintaining fertility after exposure to chemotherapy and therefore should improve the quality of these techniques.
5. Conclusion
Micro-TESE yielded a 48.5% SRR, closely mirroring previously published series. No demographic, oncologic, hormonal, or histopathological parameter significantly influenced success, highlighting the unpredictability of spermatogenic recovery after cytotoxic exposure. These results support micro-TESE followed by ICSI as an essential fertility preservation option for men undergoing chemotherapy. The absence of documented fertility status prior to chemotherapy is a limitation of this study.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Author Contributions
M. Babaei: Design of the work, acquisition, analysis, or interpretation of data, drafting the work, critical revising of the draft, final approval of the project. MA. Sadighi Gilani: Design of the work, acquisition, analysis, or interpretation of data, drafting the work, critical revising of the draft, final approval of the project. M. Ramezani-Binabaj: Design of the work, drafting the work, acquisition, analysis, or interpretation of data, critical revising of the draft, final approval of the project. H. Arasteh: Acquisition, analysis, or interpretation of data, drafting the work, critical revising of the draft, final approval of the project. SR. Yahyazadeh: Acquisition, analysis, or interpretation of data, drafting the work, critical revising of the draft, final approval of the project.
Acknowledgments
We sincerely appreciate the support of the Royan Institute for Reproductive Biomedicine, Tehran, Iran staff, Tehran, Iran. This project has not received any funding support. Artificial intelligence-based (Microsoft Copilot) grammar checking tools were used for some sentences.
Conflict of Interest
The author declares that there is no conflict of interest.
Developing innovative chemotherapeutic protocols holds significant promise for enhancing the survival rates of individuals afflicted with various malignancies. However, a crucial concern arises when considering appropriate treatment strategies for male cancer patients, namely the restoration of their fertilization ability and the achievement of parenthood (1). Unfortunately, a well-documented adverse effect of chemotherapy agents is non-obstructive azoospermia (NOA), which affects approximately 2-3rd of men undergoing chemotherapy treatments, with only a minority experiencing subsequent recovery of spermatogenesis post-treatment (2, 3). This undesirable consequence primarily arises from the utilization of alkylating chemotherapy agents such as cyclophosphamide, busulfan, procarbazine or chlorambucil (4, 5). The extent of gonadotoxic effects and subsequent germ cell damage is influenced by various factors, including the pre-treatment quality of spermatogenesis, the specific type of malignancy, the cancer treatment protocol employed, and individual susceptibility (6). Consequently, all men receiving such chemotherapy are strongly advised to consider sperm cryopreservation; however, this approach may be limited due to factors such as inadequate population awareness regarding chemotherapy-induced infertility, initial poor sperm quality, unavailability of sperm banking facilities, or prepubertal age of the male (7).
Fortunately, the development of assisted reproductive technologies has successfully addressed and mitigated this predicament. Among the available treatment options for affected men grappling with chemotherapy-induced infertility, the employment of micro-dissection testicular sperm extraction (micro-TESE) followed by intracytoplasmic sperm injection (ICSI) has emerged as the preferred and optimal approach (8, 9). This treatment modality proves invaluable for individuals lacking access to cryopreservation, as well as those who are exposed to gonadotoxic chemotherapy agents. A growing body of evidence supports the utilization of this protocol, with recent reports highlighting its remarkable success rates in retrieving sperm from men suffering from post-chemotherapy azoospermia (10). Notably, studies have demonstrated a significant increase in sperm retrieval rates (SRR), with success rates reaching up to 44% (11).
In the present study, we aim to provide a comprehensive analysis of our experiences with the micro-TESE protocol, focusing on the success rate of sperm restoration in men undergoing chemotherapy protocols.
2. Materials and Methods
In this cross-sectional study, we reviewed the medical records of 33 men with NOA secondary to chemotherapy for different types of malignancies, who underwent micro-TESE followed by ICSI, and were referred to Royan Institute for Reproductive Biomedicine (Tehran, Iran) between 2010 and 2023. Extracted data included baseline characteristics, such as age, type of malignancies, duration of chemotherapy treatment, serum levels of testosterone, follicle-stimulating hormone (FSH), and luteinizing hormone (LH), history of cigarette, alcohol, or hookah use, as well as diametric features of the testes (measured using an orchidometer and ultrasound examination). This study was retrospective in nature. Accordingly, the sample size was based on the total number of eligible patients referred to our institution and met the inclusion criteria. No sample size calculation was performed prior to data collection, and no interim analysis or stopping rules were applied.
Inclusion criteria were: 1) NOA resulting from the use of chemotherapy agents for different types of malignancies, 2) scheduled for micro-TESE followed by ICSI, 3) demonstrated normal sexual hormonal conditions, as indicated by normal serum levels of FSH and LH.
Exclusion criteria encompassed individuals with hypogonadotropic hypogonadism, any genetic or chromosomal defects affecting fertility (e.g., Klinefelter syndrome, microdeletion of azoospermia factors, or chromosomal translocations), or those with possible obstructive azoospermia. Andrological and hormonal evaluations were performed prior to the operation to determine the primary causes of azoospermia.
Azoospermia was defined as the absence of spermatozoa in 2 consecutive semen analyses and confirmed by at least 3 centrifuged semen pellet analyses.
Following initial assessment, micro-TESE was performed under general anesthesia according to the procedure previously described by Schlegel and Li (12). In this procedure, the testis is carefully opened, allowing for extensive dissection to explore all possible areas of sperm production within the divided halves of the testis. Successful sperm retrieval during micro-TESE was defined as the detection of at least one sperm cell by an experienced andrologist in the operating room. This was achieved through phase contrast examination of dispersed and minced testis tissue, which was carefully placed on a plain glass slide and covered with a slip. If viable sperm were successfully retrieved through microdissection, they were utilized for ICSI either on the same day or the following day after retrieval. Testicular biopsies were obtained and sent to a single laboratory for histopathological assessment, where a genitourinary pathologist examined them to determine the predominant histopathology. The study endpoint was to assess the outcomes of sperm extraction and related determinants.
2.1. Ethical Considerations
This study was approved by the Ethics Committee of Royan Institute for Reproductive Biomedicine, Tehran, Iran (Code: IR.ACECR.ROYAN.REC.1403.044). Given the retrospective nature of the research and use of de-identified participant data, the requirement for informed consent was waived by the committee. Data confidentiality was maintained, and participants’ personal information was protected from unauthorized access or disclosure.
2.2. Statistical Analysis
Quantitative variables were presented as mean ± standard deviation, while categorical variables were summarized by frequency (percentage). Continuous variables were compared using a t test or Mann-Whitney U test when the data did not appear to follow a normal distribution or when the assumption of equal variances was violated across the study groups. Categorical variables were compared using the Chi-Square test. A p ≤ 0.05 was considered statistically significant. All analyses were performed using Statistical Package for the Social Sciences, version 23.0 for Windows (IBM Corp., Armonk, NY, USA).
3. Results
Data of 33 men suffering from post-chemotherapy NOA were included in this study. Table I represents baseline characteristics of study objects. Mean age at the start of chemotherapy and at the time of surgery were 21 ± 7.4 and 33.6 ± 5.6, respectively. Mean FSH, LH, and testosterone levels were 19.9 ± 13.5, 9.6 ± 5.3, and 3.4 ± 1.5, respectively, which indicates primary spermatogenic failure. The most dominant cancer among cases was Hodgkin's lymphoma (39.39%).
Adequate amounts of spermatozoa were successfully retrieved for cryopreservation and subsequent ICSI in 16 out of 33 men, leading SRR of 48.5%. According to this sperm extraction outcome, the cases were assigned into 2 positive and negative groups. No difference was observed between the positive and negative subgroups in baseline characteristics, including mean age, mean age at chemotherapy, type of malignancies, and the results of baseline karyotype analysis (Table I). The maximum number of samples taken from the right and left testicles were 64 and 85, respectively. Testicular biopsies were evaluated pathologically. The most pathological pattern was Sertoli cells only syndrome. The other pathological patterns included total hyalinization, total arrest, hypospermatogenesis, and mixed germ cell aplasia plus focal spermatogenesis. In terms of micro-TESE results, no significant association was observed between the positive and negative subgroups in testicular pathologies.
As presented in table II, orchidometer measurement revealed no statistically significant difference between the 2 groups with respect to left and right testicular size.
The serum levels of sexual hormones, including testosterone, FSH, and LH, were also not different between the 2 groups. Overall, none of the factors studied were significant between the 2 groups with or without sperm extraction.



4. Discussion
In this study, the most common malignancy was Hodgkin’s lymphoma. SRR occurred in 16 of 33 cases (48.5%), with no statistically significant differences in baseline characteristics, cancer type, karyotype, or azoospermia factor results between successful and unsuccessful groups. Histopathological examination revealed Sertoli cells only syndrome as the predominant finding, with additional patterns including complete hyalinization, spermatogenic arrest, hypospermatogenesis, and mixed germ cell aplasia with focal spermatogenesis. Statistical analysis revealed no significant differences between the 2 groups with respect to orchidometric measurements and serum sex hormone levels (LH, FSH, and testosterone).
The development of novel therapeutic protocols, including chemotherapy regimens for the treatment of malignancies, have significantly increased patients' long-term survival. However, the use of some of these agents might adversely affect reproductive abilities, especially in men. According to reports, certain chemotherapy regimens may lead to spermatogenic dysfunction by inducing defects in germ cells and spermatogonia differentiation. In other words, the rapid differentiation of these cells is highly sensitive to these cytotoxic medications. Therefore, within 2-3 months after beginning chemotherapy, the likelihood of azoospermia may be increased (13).
Based on the type, dosage, and duration of the chemotherapy regimen, azoospermia may be transient or irreversible. However, it is impossible to predict which chemotherapy regimen may lead to transient or permanent defects in spermatogenesis. Thus, considering the cryopreservation of sperm even before the initiation of chemotherapy can warrant post-treatment fertility potential in men. Although the protocol of micro-TESE followed by ICSI in azoospermic men after chemotherapy has resulted in successful preservation and recovery of spermatogenesis, the rate of this recovery has been reported to vary. As demonstrated in the present study, the SRR was found to be 48.5%. In a comparable study (14), sperm was successfully retrieved in 47%, which was close to our retrieval rate. In another study (15), sperm retrieval was successful in 42% of the individuals. However, they also demonstrated that men with post-chemotherapy azoospermia after testicular cancer treatment were able to achieve a high retrieval rate (75%), and exposure to alkylating agents resulted in lower SRR.
In a recent study, the authors evaluated the effectiveness of cyclophosphamide equivalent dose in sperm retrieval in men with persistent azoospermia following chemotherapy. SRR was lower in patients who received a chemotherapy regimen with alkylating agents. Patients with cyclophosphamide equivalent dose > 4000 mg/m2 did not have viable sperm in micro-TESE (16).
In another study, spermatozoa were successfully retrieved in 37% of men and in 42.9% of overall procedures (17). A fertilization rate of 57.1% (per injected oocyte) was achieved with ICSI, leading to a clinical pregnancy rate of 50% with an overall live birth rate of 42%.
Therefore, the introduction of these techniques is an important step in maintaining fertility for men undergoing chemotherapy, we also have a long way to go to prevent azoospermia after this protocol. Another important issue related to the success of micro-TESE followed by ICSI protocols in sperm retrieval is identifying the potential determinants that influence the success rate of these protocols. As indicated in the present study, none of the baseline parameters, including individuals age, time, and duration of chemotherapy, type of cancer, history of smoking or alcohol use, or baseline hormonal states, appeared to affect the rate of sperm retrieval. However, different studies have shown that some baseline parameters might affect the rate of sperm recovery. In line with previous findings, age, time since chemotherapy, FSH, and the initial diagnostic indication for chemotherapy did not affect the success of sperm retrieval. They also indicated that none of the abovementioned factors could predict TESE outcomes (14).
However, in some studies, bone marrow transplantation was an unfavorable factor affecting sperm retrieval following such procedures (14, 18). It has been reported that the rate of sperm recovery can be potentially affected by the type of alkylating agents (15). Therefore, regardless of what factors may affect the recovery rate of sperm, the use of these techniques is currently considered the only way to increase the chances of maintaining fertility after exposure to chemotherapy and therefore should improve the quality of these techniques.
5. Conclusion
Micro-TESE yielded a 48.5% SRR, closely mirroring previously published series. No demographic, oncologic, hormonal, or histopathological parameter significantly influenced success, highlighting the unpredictability of spermatogenic recovery after cytotoxic exposure. These results support micro-TESE followed by ICSI as an essential fertility preservation option for men undergoing chemotherapy. The absence of documented fertility status prior to chemotherapy is a limitation of this study.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Author Contributions
M. Babaei: Design of the work, acquisition, analysis, or interpretation of data, drafting the work, critical revising of the draft, final approval of the project. MA. Sadighi Gilani: Design of the work, acquisition, analysis, or interpretation of data, drafting the work, critical revising of the draft, final approval of the project. M. Ramezani-Binabaj: Design of the work, drafting the work, acquisition, analysis, or interpretation of data, critical revising of the draft, final approval of the project. H. Arasteh: Acquisition, analysis, or interpretation of data, drafting the work, critical revising of the draft, final approval of the project. SR. Yahyazadeh: Acquisition, analysis, or interpretation of data, drafting the work, critical revising of the draft, final approval of the project.
Acknowledgments
We sincerely appreciate the support of the Royan Institute for Reproductive Biomedicine, Tehran, Iran staff, Tehran, Iran. This project has not received any funding support. Artificial intelligence-based (Microsoft Copilot) grammar checking tools were used for some sentences.
Conflict of Interest
The author declares that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Oncology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |