Wed, Apr 22, 2026
[Archive]
Volume 24, Issue 2 (February 2026)
IJRM 2026, 24(2): 157-162 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Bahrami S, Bozorgian L, Ahankoub Nejad A, Rahmani A H. Gestational trophoblastic neoplasia following assisted reproductive technology in a gestational carrier: A case report. IJRM 2026; 24 (2) :157-162
URL: http://ijrm.ir/article-1-3678-en.html
URL: http://ijrm.ir/article-1-3678-en.html
1- Department of Obstetrics and Gynecology, Division of Perinatology, Imam Khomeini Hospital, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
2- Social Determinants of Health Research Center, Yasuj University of Medical Sciences, Yasuj, Iran.
3- Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran.
4- Faculty of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran. ,Rahmani-ah@ajums.ac.ir
2- Social Determinants of Health Research Center, Yasuj University of Medical Sciences, Yasuj, Iran.
3- Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran.
4- Faculty of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran. ,
Full-Text [PDF 1070 kb]
(86 Downloads)
| Abstract (HTML) (190 Views)
Full-Text: (9 Views)
1. Introduction
A hydatidiform mole is a rare pregnancy complication caused by the abnormal proliferation of trophoblast cells (1). Molar pregnancy may take the form of a complete or partial hydatidiform mole. Overall, hydatidiform moles occur in about 1 in 1000 pregnancies (2). Gestational trophoblastic neoplasia (GTN) refers to a group of malignant neoplasms that consist of abnormal proliferation of trophoblastic tissue and may follow a hydatidiform mole or a nonmolar pregnancy (3). However, the incidence of moles in pregnancies resulting from in vitro fertilization (IVF) is rare, although studies show that the risk of their occurrence is not eliminated (4, 5). The tendency for local invasion and metastasis varies in different types of molar pregnancy (6). This study aims to report an Iranian case diagnosed with a GTN following IVF.
2. Case Presentation
A 30-yr-old female, gravid 2, live 1, and abortion 0, with a surgical history of an elective cesarean section reported to Imam Khomeini hospital, Ahvaz, Iran. Her first pregnancy was spontaneous and without intervention. She had no family history of any disease or cancer, or even surgery other than laparoscopy and cesarean section. The case stated that the previous laparoscopy was performed due to suspected endometriosis.
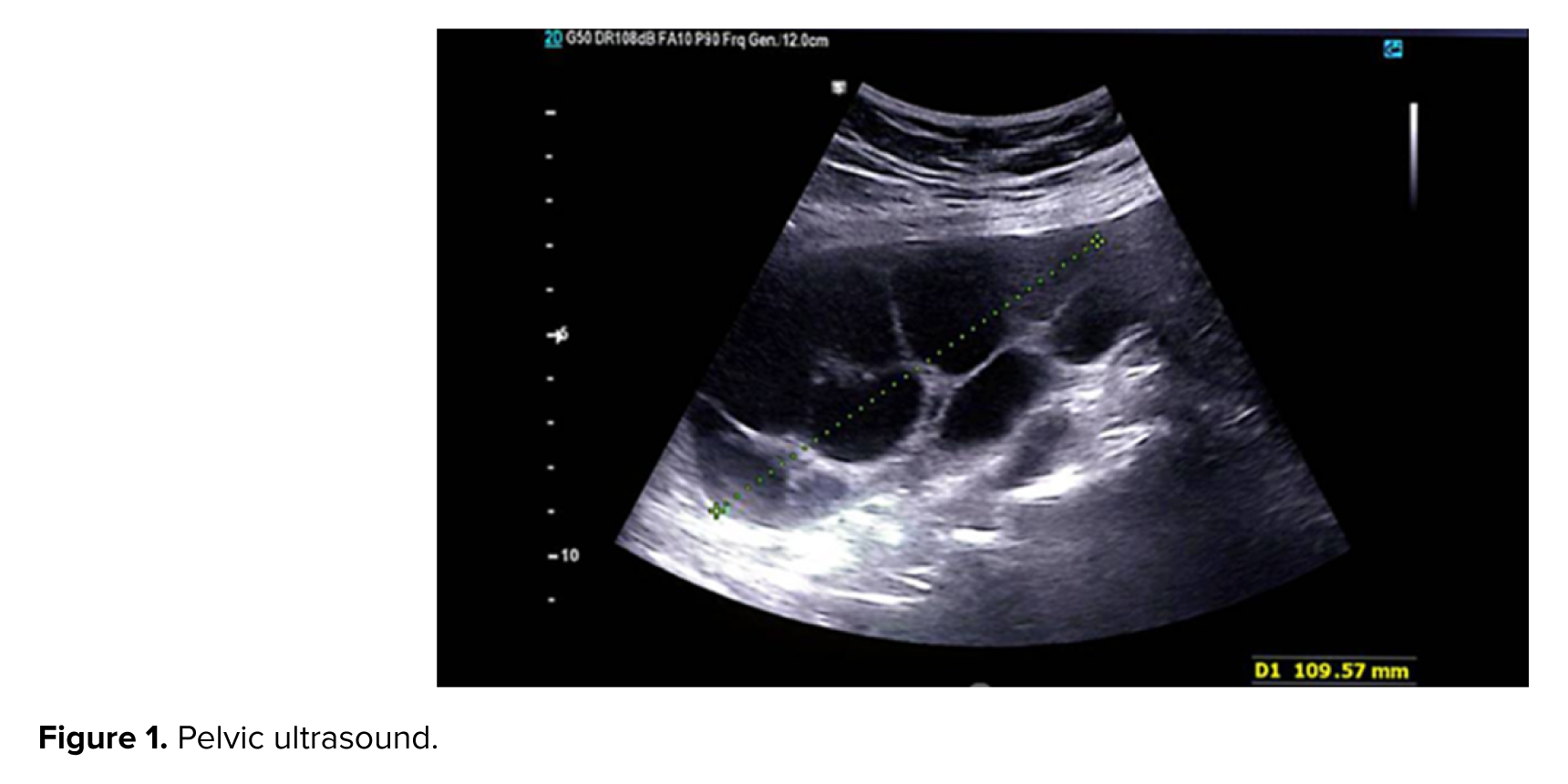
The patient conceived through IVF treatment after 10 yr of secondary infertility. According to history, on December 18, 2024, IVF was performed using 2 3-day frozen embryos. 4 wk later, the beta human chorionic gonadotropin (β-hCG) titer was 726. On January 20, 2025, at a gestational age of 10 wk, the case presented with an ultrasound showing an echogenic mass measuring 107 mm and 63 mm in the posterior part of the gestational sac, with abundant fibrocystic areas, indicating an incomplete mole or placental mesenchymal dysplasia, and a human chorionic gonadotropin hormone titer of 40,000. After initial investigations, tests (complete blood count, blood group Rh, liver function test, thyroid function tests, blood urea nitrogen, creatinine) and chest X-ray were requested and performed. The results of which were normal. Ultrasound-guided suction curettage was performed, and 48 hr later the initial suction curettage, a β-hCG titer was sent (the result of the β-hCG titer was 19120). A pelvic ultrasound was also performed. The ultrasound report included the following: observation of a heterogeneous structure measuring 20 and 14 mm in the endometrial fundus in favor of retained products of conception, some free fluid around the ovaries, and several ovarian cysts, the largest of which were 50 and 40 mm (Figure 1).
A sample of uterine tissue was sent to the laboratory for pathological examination. The endometrial tissue pathology results confirmed a partial hydatidiform mole (Figure 2).
The patient was discharged in good general condition and was advised to return to the doctor's office for follow-up with a β-hCG titer and a subsequent pelvic ultrasound.
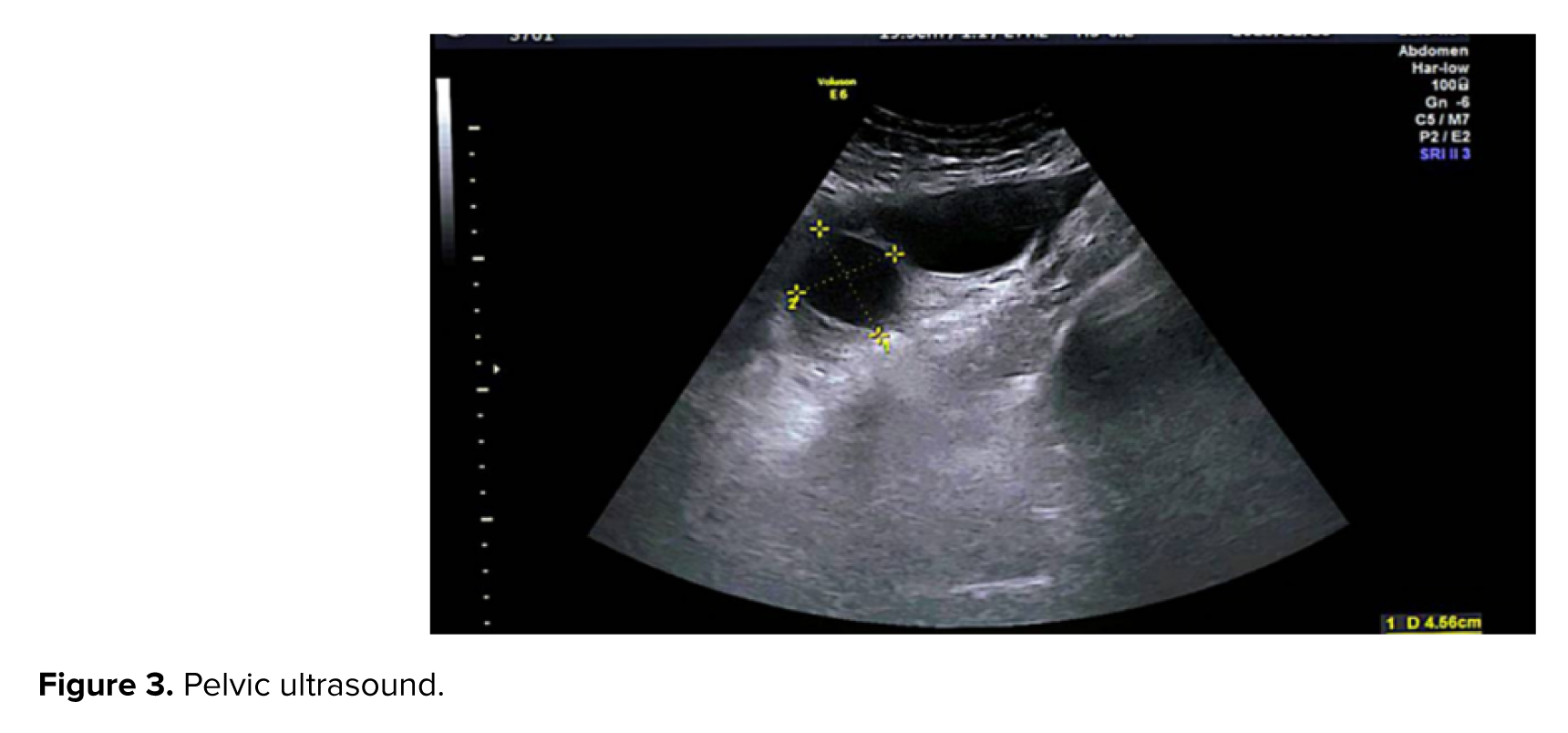
4 wk later, the case presented with complaints of severe vaginal bleeding and increased β-hCG titer (31037) after suction. Upon admission to the emergency department, vital signs were as follows: blood pressure, 110/75 mmHg; pulse, 84 bpm; temperature, 36.5°C; and respiratory rate, 18 breaths/min. Abdominal and pelvic examinations were normal. A pelvic ultrasound was performed. The ultrasound results showed heterogeneous remnants (63 and 50 mm) in the uterine cavity and 3 cysts (51 and 37, 48 and 51, and 27 and 45 mm) in the left ovary and 3 cysts (59 and 56, 60 & 53, 48 & 40) in the right ovary and some free fluid in the pelvic cavity (Figure 3).
Complete blood count, liver function test, thyroid function tests, blood urea nitrogen, and creatinine tests were requested again, along with a chest X-ray. The test results and chest X-rays were normal. Due to heavy bleeding, the case underwent ultrasound-guided suction curettage again. Because the bleeding did not stop after suction curettage, the uterine catheter was fixed in the uterus with 40 cc of normal saline. According to the computed tomography scan of the pelvis and abdomen, the ovaries were larger than normal and contained multiple cysts, suggesting a theca lutein cyst. An ultrasound was performed one day after suction curettage. According to the ultrasound report, the myometrium was observed to be heterogeneous in the fundus, with increased vascularity, which could be indicative of invasion.
The patient’s vital signs and vaginal bleeding were normal. Due to the diagnosis of GTN stage 1 and an increase in the β-hCG titer of more than 10%, during 3 weekly measurements and the normal results of chest X-ray, abdominal and pelvic CT, a single-drug chemotherapy regimen with weekly methotrexate 50 mg/m2 was started. The patient was discharged in good general condition, with the necessary advice and follow-up instructions, and was advised to visit the doctor's office.
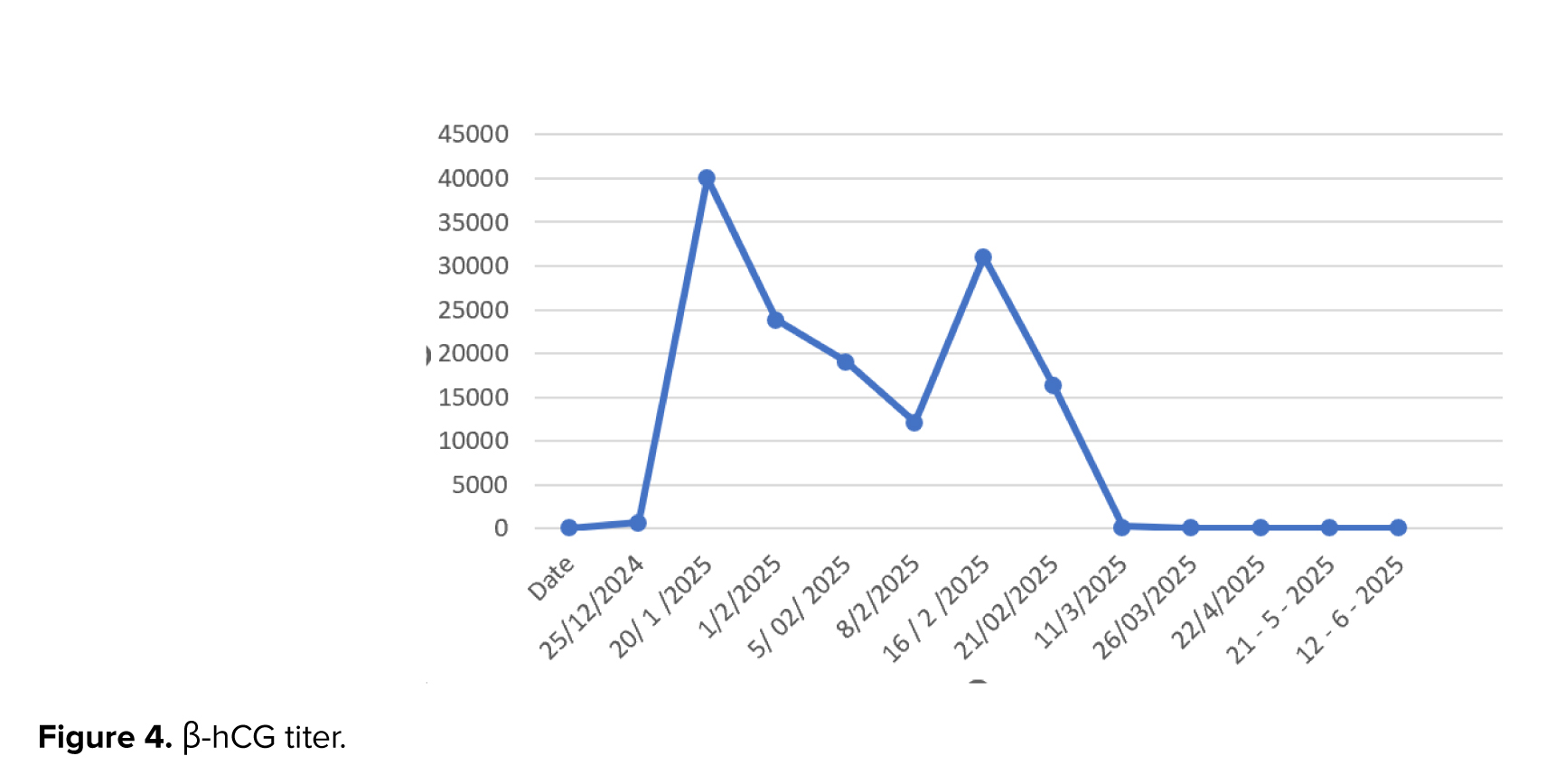
A week later, the patient returned complaining of relatively heavy vaginal bleeding. β-hCG titer was 16330. The patient was hospitalized, and the necessary tests were requested again. A uterine catheter was inserted into the uterus under ultrasound guidance. Oncology and interventional consultation were requested. According to the interventional report, color Doppler ultrasound showed no evidence of abnormal vascularity in the endometrium. Hence, she was started on a multi-day regimen every 2 wk by intramuscular methotrexate 90 mg. It was also injected on 28/02/2025, 02/03/2025, and 04/03/2025. Leucovorin 9 mg was injected intramuscularly on 27/02, 01/03, 03/03, and 05/03-2025. The patient was followed up with β-hCG titer checks until the level reached zero in 3 consecutive tests. Despite 3 negative β-hCG titers, the case continued to receive chemotherapy. It was recommended that the β-hCG titer follow-up be continued for the next 6 months. 3 months after completing chemotherapy, the patient’s beta-hash titer was negative, no vaginal bleeding was observed, and the patient was taking contraceptive low dose pills. β-hCG titer tests are reported in figure 4.

2.1. Ethical Considerations
Written informed consent was obtained from the participant to publish this case report.
3. Discussion
In recent years, the use of assisted reproductive techniques (ART) to treat infertility has increased (7). The risk of developing a molar pregnancy following ART is relatively rare. Studies show that the incidence of molar pregnancy is 1/4333 with a fresh IVF cycle and between 1/2317 and 1/28967 with a frozen cycle (8). Hydatidiform moles are divided into 2 categories: complete moles and partial moles. Partial moles result from fertilizing a haploid ovum by 2 separate sperm, resulting in a triploid genome found in this type of focal villi and embryonic parts. However, the complete mole is composed of chromosomes of paternal origin, which have a diploid androgenetic genome (9). In the case study, the molar pregnancy was partial.
Genetic abnormalities can be among the causes of molar pregnancy (10). Other factors that cause molar pregnancy include advanced age, failure to prevent multiple fertilization, immature eggs, and sperm abnormalities. Therefore, it appears that preimplantation genetic diagnosis may be necessary during IVF (9). Unintended fertilization of a diploid or triploid sperm following IVF can result in a molar pregnancy (8).
One study found that induction therapy and forced ovulation can cause over-release, which increases the incidence of nulliparous eggs (11). The results of a review study showed that in patients who had hydatidiform mole with a twin live fetus after ART, the incidence of gestational trophoblastic disease was 42.85%. The live birth rate was 39.28% despite the absence of gestational trophoblastic disease risk (12). Therefore, in pregnancies that occur after ART, the likelihood of having a normal child should be weighed against the risk of complications.
Preimplantation genetic diagnosis seems a reasonable proposition for an IVF candidate case (5). Therefore, decisions regarding the fate of a precious pregnancy due to complications such as a mole require a highly skilled individual. Also, timely intervention will be invaluable in preventing maternal mortality and complications.
4. Conclusion
Despite scientific advances and help in solving infertility using IVF techniques, it seems that there is a possibility of a molar pregnancy following IVF. Therefore, considering the possible harm to the mother, managing a molar pregnancy after IVF is essential.
Data Availability
All relevant data is contained in the article file and is publicly available.
Author Contributions
S. Bahrami: Collaboration in writing the article. L. Bozorgian: Writing the article and supervision. A. Ahankoub Nejad: Patient follow-up. AH. Rahmani: Collaboration in writing the article and language editing.
Acknowledgments
The woman whose story is described in this case report is thanked. No artificial intelligence was used.
Conflict of Interest
The authors declare that there is no conflict of interest.
A hydatidiform mole is a rare pregnancy complication caused by the abnormal proliferation of trophoblast cells (1). Molar pregnancy may take the form of a complete or partial hydatidiform mole. Overall, hydatidiform moles occur in about 1 in 1000 pregnancies (2). Gestational trophoblastic neoplasia (GTN) refers to a group of malignant neoplasms that consist of abnormal proliferation of trophoblastic tissue and may follow a hydatidiform mole or a nonmolar pregnancy (3). However, the incidence of moles in pregnancies resulting from in vitro fertilization (IVF) is rare, although studies show that the risk of their occurrence is not eliminated (4, 5). The tendency for local invasion and metastasis varies in different types of molar pregnancy (6). This study aims to report an Iranian case diagnosed with a GTN following IVF.
2. Case Presentation
A 30-yr-old female, gravid 2, live 1, and abortion 0, with a surgical history of an elective cesarean section reported to Imam Khomeini hospital, Ahvaz, Iran. Her first pregnancy was spontaneous and without intervention. She had no family history of any disease or cancer, or even surgery other than laparoscopy and cesarean section. The case stated that the previous laparoscopy was performed due to suspected endometriosis.
The patient conceived through IVF treatment after 10 yr of secondary infertility. According to history, on December 18, 2024, IVF was performed using 2 3-day frozen embryos. 4 wk later, the beta human chorionic gonadotropin (β-hCG) titer was 726. On January 20, 2025, at a gestational age of 10 wk, the case presented with an ultrasound showing an echogenic mass measuring 107 mm and 63 mm in the posterior part of the gestational sac, with abundant fibrocystic areas, indicating an incomplete mole or placental mesenchymal dysplasia, and a human chorionic gonadotropin hormone titer of 40,000. After initial investigations, tests (complete blood count, blood group Rh, liver function test, thyroid function tests, blood urea nitrogen, creatinine) and chest X-ray were requested and performed. The results of which were normal. Ultrasound-guided suction curettage was performed, and 48 hr later the initial suction curettage, a β-hCG titer was sent (the result of the β-hCG titer was 19120). A pelvic ultrasound was also performed. The ultrasound report included the following: observation of a heterogeneous structure measuring 20 and 14 mm in the endometrial fundus in favor of retained products of conception, some free fluid around the ovaries, and several ovarian cysts, the largest of which were 50 and 40 mm (Figure 1).
A sample of uterine tissue was sent to the laboratory for pathological examination. The endometrial tissue pathology results confirmed a partial hydatidiform mole (Figure 2).
The patient was discharged in good general condition and was advised to return to the doctor's office for follow-up with a β-hCG titer and a subsequent pelvic ultrasound.
4 wk later, the case presented with complaints of severe vaginal bleeding and increased β-hCG titer (31037) after suction. Upon admission to the emergency department, vital signs were as follows: blood pressure, 110/75 mmHg; pulse, 84 bpm; temperature, 36.5°C; and respiratory rate, 18 breaths/min. Abdominal and pelvic examinations were normal. A pelvic ultrasound was performed. The ultrasound results showed heterogeneous remnants (63 and 50 mm) in the uterine cavity and 3 cysts (51 and 37, 48 and 51, and 27 and 45 mm) in the left ovary and 3 cysts (59 and 56, 60 & 53, 48 & 40) in the right ovary and some free fluid in the pelvic cavity (Figure 3).
Complete blood count, liver function test, thyroid function tests, blood urea nitrogen, and creatinine tests were requested again, along with a chest X-ray. The test results and chest X-rays were normal. Due to heavy bleeding, the case underwent ultrasound-guided suction curettage again. Because the bleeding did not stop after suction curettage, the uterine catheter was fixed in the uterus with 40 cc of normal saline. According to the computed tomography scan of the pelvis and abdomen, the ovaries were larger than normal and contained multiple cysts, suggesting a theca lutein cyst. An ultrasound was performed one day after suction curettage. According to the ultrasound report, the myometrium was observed to be heterogeneous in the fundus, with increased vascularity, which could be indicative of invasion.
The patient’s vital signs and vaginal bleeding were normal. Due to the diagnosis of GTN stage 1 and an increase in the β-hCG titer of more than 10%, during 3 weekly measurements and the normal results of chest X-ray, abdominal and pelvic CT, a single-drug chemotherapy regimen with weekly methotrexate 50 mg/m2 was started. The patient was discharged in good general condition, with the necessary advice and follow-up instructions, and was advised to visit the doctor's office.
A week later, the patient returned complaining of relatively heavy vaginal bleeding. β-hCG titer was 16330. The patient was hospitalized, and the necessary tests were requested again. A uterine catheter was inserted into the uterus under ultrasound guidance. Oncology and interventional consultation were requested. According to the interventional report, color Doppler ultrasound showed no evidence of abnormal vascularity in the endometrium. Hence, she was started on a multi-day regimen every 2 wk by intramuscular methotrexate 90 mg. It was also injected on 28/02/2025, 02/03/2025, and 04/03/2025. Leucovorin 9 mg was injected intramuscularly on 27/02, 01/03, 03/03, and 05/03-2025. The patient was followed up with β-hCG titer checks until the level reached zero in 3 consecutive tests. Despite 3 negative β-hCG titers, the case continued to receive chemotherapy. It was recommended that the β-hCG titer follow-up be continued for the next 6 months. 3 months after completing chemotherapy, the patient’s beta-hash titer was negative, no vaginal bleeding was observed, and the patient was taking contraceptive low dose pills. β-hCG titer tests are reported in figure 4.




2.1. Ethical Considerations
Written informed consent was obtained from the participant to publish this case report.
3. Discussion
In recent years, the use of assisted reproductive techniques (ART) to treat infertility has increased (7). The risk of developing a molar pregnancy following ART is relatively rare. Studies show that the incidence of molar pregnancy is 1/4333 with a fresh IVF cycle and between 1/2317 and 1/28967 with a frozen cycle (8). Hydatidiform moles are divided into 2 categories: complete moles and partial moles. Partial moles result from fertilizing a haploid ovum by 2 separate sperm, resulting in a triploid genome found in this type of focal villi and embryonic parts. However, the complete mole is composed of chromosomes of paternal origin, which have a diploid androgenetic genome (9). In the case study, the molar pregnancy was partial.
Genetic abnormalities can be among the causes of molar pregnancy (10). Other factors that cause molar pregnancy include advanced age, failure to prevent multiple fertilization, immature eggs, and sperm abnormalities. Therefore, it appears that preimplantation genetic diagnosis may be necessary during IVF (9). Unintended fertilization of a diploid or triploid sperm following IVF can result in a molar pregnancy (8).
One study found that induction therapy and forced ovulation can cause over-release, which increases the incidence of nulliparous eggs (11). The results of a review study showed that in patients who had hydatidiform mole with a twin live fetus after ART, the incidence of gestational trophoblastic disease was 42.85%. The live birth rate was 39.28% despite the absence of gestational trophoblastic disease risk (12). Therefore, in pregnancies that occur after ART, the likelihood of having a normal child should be weighed against the risk of complications.
Preimplantation genetic diagnosis seems a reasonable proposition for an IVF candidate case (5). Therefore, decisions regarding the fate of a precious pregnancy due to complications such as a mole require a highly skilled individual. Also, timely intervention will be invaluable in preventing maternal mortality and complications.
4. Conclusion
Despite scientific advances and help in solving infertility using IVF techniques, it seems that there is a possibility of a molar pregnancy following IVF. Therefore, considering the possible harm to the mother, managing a molar pregnancy after IVF is essential.
Data Availability
All relevant data is contained in the article file and is publicly available.
Author Contributions
S. Bahrami: Collaboration in writing the article. L. Bozorgian: Writing the article and supervision. A. Ahankoub Nejad: Patient follow-up. AH. Rahmani: Collaboration in writing the article and language editing.
Acknowledgments
The woman whose story is described in this case report is thanked. No artificial intelligence was used.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Case Report |
Subject:
Reproductive Oncology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





