Thu, May 21, 2026
[Archive]
Volume 24, Issue 1 (January 2026)
IJRM 2026, 24(1): 13-22 |
Back to browse issues page
Ethics code: IR.IAU.BABOL.REC.1404.046
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Jebeli Eshrat Abadi A, Gharib F Z, Hosseini S M. Ivermectin mitigates 7,12-dimethylbenz(a)anthracene-induced reproductive toxicity in female wistar rats: An experimental study. IJRM 2026; 24 (1) :13-22
URL: http://ijrm.ir/article-1-3737-en.html
URL: http://ijrm.ir/article-1-3737-en.html
1- Department of Pathology, Bab.C., Islamic Azad University, Babol, Iran.
2- Department of Clinical Sciences, Bab.C., Islamic Azad University, Babol, Iran. ,Fzgharib@iau.ac.ir
2- Department of Clinical Sciences, Bab.C., Islamic Azad University, Babol, Iran. ,
Full-Text [PDF 4041 kb]
(265 Downloads)
| Abstract (HTML) (256 Views)
Full-Text: (44 Views)
1. Introduction
Reproductive disorders are a major global health burden, with infertility affecting about 1 in 6 adults worldwide (approximately 17.5% of the population) according to the World Health Organization. Recent analyses estimate that over 165 million people lived with infertility in 2021, with prevalence projected to rise by 2040 (1). These conditions stem from genetic, infectious, and lifestyle factors, but environmental exposures such as polycyclic aromatic hydrocarbons (PAHs) are increasingly recognized as key contributors, disrupting ovarian function, sperm quality, and pregnancy outcomes (2, 3). PAHs threaten female reproductive health, with DMBA (a common pollutant from combustion and industrial sources) metabolized into reactive epoxides that form DNA adducts and free radicals (4). This process produces reactive oxygen species (ROS) and lipid peroxidation, overwhelms antioxidants, and leads to oxidative stress that emerges as the key mechanism behind DMBA’s toxicity and infertility effects (5).
Previous studies confirm that DMBA exposure causes extensive ovarian damage. For example, in cultured rat ovaries, DMBA induced DNA double-strand breaks, γH2AX and caspase-3 activation, and loss of all follicle stages (6). In vivo, single DMBA administration destroys preantral and antral follicles via apoptosis. DMBA’s toxicity extends to hormone disruption: chronic PAH exposure has been linked to reduced progesterone and altered gonadotropins in animal models and humans (7). Although Wistar rats may exhibit strain-specific resistance to some endocrine effects (8), a significant DMBA-induced hormonal imbalance can likely cause ovarian steroidogenesis impairment. Importantly, DMBA’s ability to induce oxidative stress and apoptosis makes it a useful model to test protective agents. Antioxidant and anti-apoptotic interventions have attenuated DMBA toxicity (9). For instance, dietary supplements protected mammalian tissues from DMBA by boosting antioxidant defenses and reducing apoptosis (10, 11). These studies suggest that counteracting oxidative stress can preserve ovarian integrity under DMBA insult.
Ivermectin is a macrocyclic lactone originally developed for parasitic infections (12). Beyond its anthelmintic role, Ivermectin is increasingly recognized to have broad pharmacologic effects. It inhibits inflammatory signaling (NF-κB pathway) and reduces pro-inflammatory cytokine production. Preclinical models show Ivermectin can mitigate inflammation and oxidative damage: e.g., in bleomycin-induced pulmonary fibrosis, Ivermectin reduced markers of oxidative stress and inflammatory cytokines, significantly reducing fibrosis (13). Ivermectin also exhibits anticancer effects in cell and animal studies, often through modulation of apoptotic pathways (14-16).
Given the urgent need for strategies to counteract environmental reproductive toxins, we hypothesized that Ivermectin could attenuate DMBA-induced ovarian injury. To test this, we treated female Wistar rats with DMBA alone or with concurrent Ivermectin administration. We then assessed oxidative stress, sex hormones (estradiol, progesterone, testosterone), apoptosis-related gene expression, and histopathology of ovaries and uterus. We predicted that Ivermectin’s anti-oxidative and anti-apoptotic effects would reduce DMBA toxicity, normalizing biochemical markers and preserving tissue morphology. This study explores Ivermectin repurposing as a potential protective agent against PAH-induced reproductive toxicity, with implications for environmental health interventions.
2. Materials and Methods
2.1. Study design
Ivermectin (Nasr Freeman Pharmaceutical Co.) and DMBA (Cas number: 57-97-6, Cat number: D3254, Sigma-Aldrich) were purchased. This experimental study was conducted in July 2025 at Amol city, Amol, Iran. 20 female Wistar albino rats (8 wk, 210 gr) were purchased from Shomal Pasteur Institute, Northern Iran and maintained at 20-23°C and 60-70% relative humidity under a 12-hr light/dark cycle under standard conditions with ad libitum food. After acclimation, rats were randomly assigned to 4 groups (n = 5/each): 1) control (received normal saline); 2) Ivermectin (1 mg/kg, orally, weekly) (17); 3) DMBA (single dose 80 mg/kg DMBA, intraperitoneally) (18); and 4) DMBA + Ivermectin (the same doses).
2.2. Biochemical assays
Reproductive disorders are a major global health burden, with infertility affecting about 1 in 6 adults worldwide (approximately 17.5% of the population) according to the World Health Organization. Recent analyses estimate that over 165 million people lived with infertility in 2021, with prevalence projected to rise by 2040 (1). These conditions stem from genetic, infectious, and lifestyle factors, but environmental exposures such as polycyclic aromatic hydrocarbons (PAHs) are increasingly recognized as key contributors, disrupting ovarian function, sperm quality, and pregnancy outcomes (2, 3). PAHs threaten female reproductive health, with DMBA (a common pollutant from combustion and industrial sources) metabolized into reactive epoxides that form DNA adducts and free radicals (4). This process produces reactive oxygen species (ROS) and lipid peroxidation, overwhelms antioxidants, and leads to oxidative stress that emerges as the key mechanism behind DMBA’s toxicity and infertility effects (5).
Previous studies confirm that DMBA exposure causes extensive ovarian damage. For example, in cultured rat ovaries, DMBA induced DNA double-strand breaks, γH2AX and caspase-3 activation, and loss of all follicle stages (6). In vivo, single DMBA administration destroys preantral and antral follicles via apoptosis. DMBA’s toxicity extends to hormone disruption: chronic PAH exposure has been linked to reduced progesterone and altered gonadotropins in animal models and humans (7). Although Wistar rats may exhibit strain-specific resistance to some endocrine effects (8), a significant DMBA-induced hormonal imbalance can likely cause ovarian steroidogenesis impairment. Importantly, DMBA’s ability to induce oxidative stress and apoptosis makes it a useful model to test protective agents. Antioxidant and anti-apoptotic interventions have attenuated DMBA toxicity (9). For instance, dietary supplements protected mammalian tissues from DMBA by boosting antioxidant defenses and reducing apoptosis (10, 11). These studies suggest that counteracting oxidative stress can preserve ovarian integrity under DMBA insult.
Ivermectin is a macrocyclic lactone originally developed for parasitic infections (12). Beyond its anthelmintic role, Ivermectin is increasingly recognized to have broad pharmacologic effects. It inhibits inflammatory signaling (NF-κB pathway) and reduces pro-inflammatory cytokine production. Preclinical models show Ivermectin can mitigate inflammation and oxidative damage: e.g., in bleomycin-induced pulmonary fibrosis, Ivermectin reduced markers of oxidative stress and inflammatory cytokines, significantly reducing fibrosis (13). Ivermectin also exhibits anticancer effects in cell and animal studies, often through modulation of apoptotic pathways (14-16).
Given the urgent need for strategies to counteract environmental reproductive toxins, we hypothesized that Ivermectin could attenuate DMBA-induced ovarian injury. To test this, we treated female Wistar rats with DMBA alone or with concurrent Ivermectin administration. We then assessed oxidative stress, sex hormones (estradiol, progesterone, testosterone), apoptosis-related gene expression, and histopathology of ovaries and uterus. We predicted that Ivermectin’s anti-oxidative and anti-apoptotic effects would reduce DMBA toxicity, normalizing biochemical markers and preserving tissue morphology. This study explores Ivermectin repurposing as a potential protective agent against PAH-induced reproductive toxicity, with implications for environmental health interventions.
2. Materials and Methods
2.1. Study design
Ivermectin (Nasr Freeman Pharmaceutical Co.) and DMBA (Cas number: 57-97-6, Cat number: D3254, Sigma-Aldrich) were purchased. This experimental study was conducted in July 2025 at Amol city, Amol, Iran. 20 female Wistar albino rats (8 wk, 210 gr) were purchased from Shomal Pasteur Institute, Northern Iran and maintained at 20-23°C and 60-70% relative humidity under a 12-hr light/dark cycle under standard conditions with ad libitum food. After acclimation, rats were randomly assigned to 4 groups (n = 5/each): 1) control (received normal saline); 2) Ivermectin (1 mg/kg, orally, weekly) (17); 3) DMBA (single dose 80 mg/kg DMBA, intraperitoneally) (18); and 4) DMBA + Ivermectin (the same doses).
2.2. Biochemical assays
After 8 wk, rats were anesthetized and blood was collected by cardiac puncture. Serum was separated and used to assess estradiol (E2 AccuBind ELISA Kit, 4925-300A), progesterone (Progesterone AccuBind ELISA Kit, 4825-300A), and testosterone (Testosterone AccuBind ELISA Kit, 3725-300A) (19).
2.3. Oxidative stress markers
The activity and levels of malondialdehyde (MDA), superoxide dismutase (SOD), and total antioxidant capacity (TAC) were assayed by Navand Salamat kits (Nalondi [product number: NS-15022], Nasdox [product number: NS-15034], and Naxifer [product number: NS-15012] kits, respectively) (20). All assays were performed in duplicate according to manufacturer protocols.
2.4. Gene expression
Ovarian tissue was excised and homogenized for RNA extraction. cDNA was synthesized, and quantitative real-time polymerase chain reaction (qRT-PCR) was performed for apoptosis genes: Bax, Bcl-2, Caspase-3, and p53. B-actin was used as an internal control (21). Relative expression was calculated by the 2^-ΔΔCt method.
2.5. Histopathology
Uterine and ovarian tissues were fixed in 10% formalin, embedded in paraffin, sectioned (5 μm), and stained with H&E. Sections were examined under light microscopy (22).
2.6. Ethical Considerations
Additionally, the Research Ethics Committees of Islamic Azad University, Babol Branch, babol, Iran found this project to be in accordance with the ethical principles and the national norms and standards for conducting medical research in Iran(Code: IR.IAU.BABOL.REC.1404.046) In addition, the ARRIVE guideline was followed.
2.7. Statistical Analysis
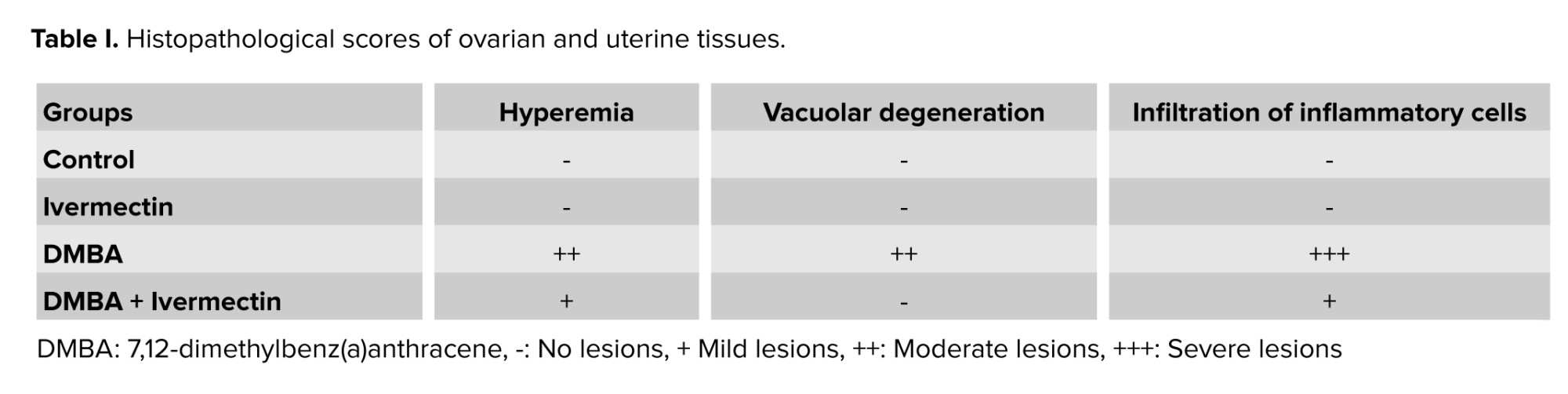
All the data related to hormones, oxidative stress markers, and gene expressions were analyzed using GraphPad software version 10 based on the one-way ANOVA and Tukey’s post hoc tests. P < 0.05 was considered as a significant difference. The results were expressed as the mean ± standard deviation (23). The histopathological evaluation was assessed based on earlier research (24). Briefly, the severity of lesions was defined as no lesions (-), mild lesions (+), moderate lesions (++), and severe lesion (+++) (25).
3. Results
3.1. Sex hormones
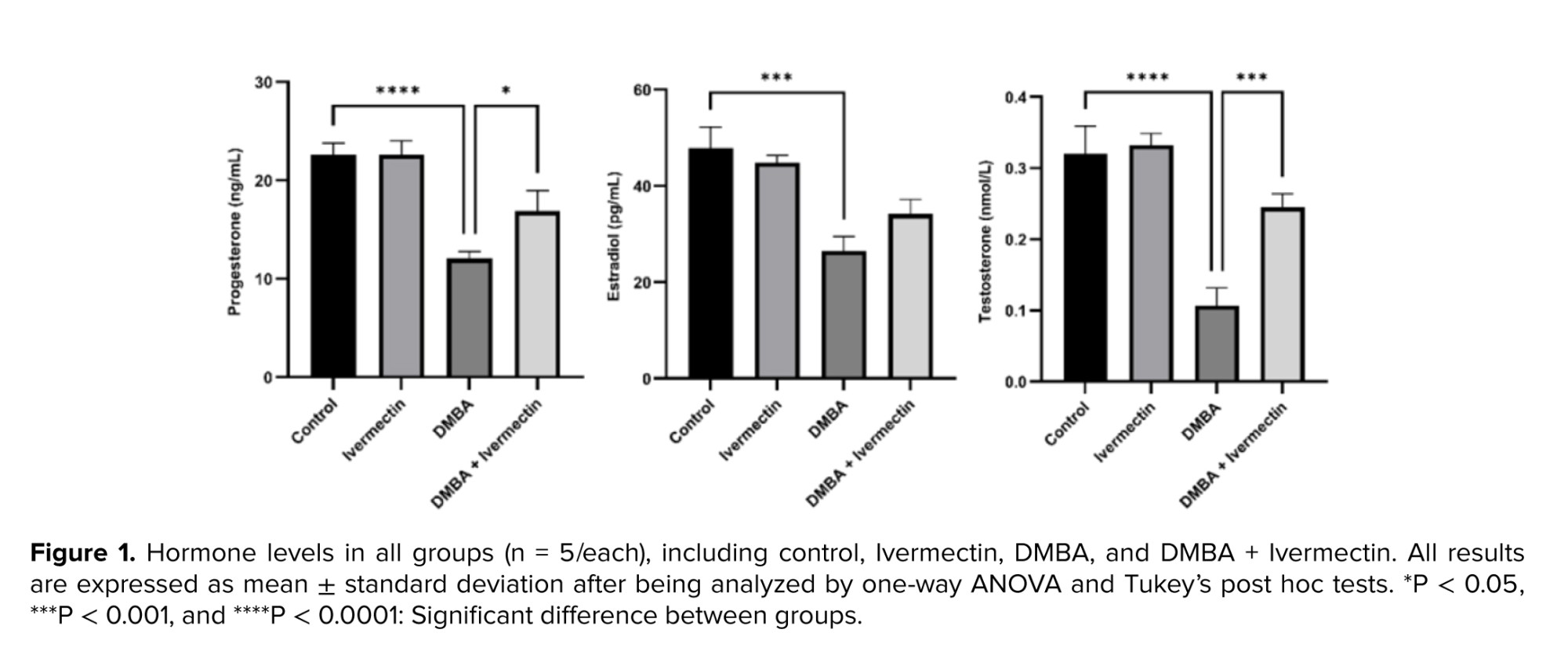
In comparison to the control group, all the hormone levels were found to be significantly decreased in the DMBA group (p < 0.001). Additionally, Ivermectin alone did not affect hormone levels. However, in the DMBA + Ivermectin group, progesterone level was significantly (p < 0.05) increased compared to the DMBA group. Similarly, testosterone levels also raised significantly (p < 0.001) compared to DMBA group (Figure 1).
3.2. Oxidative stress markers
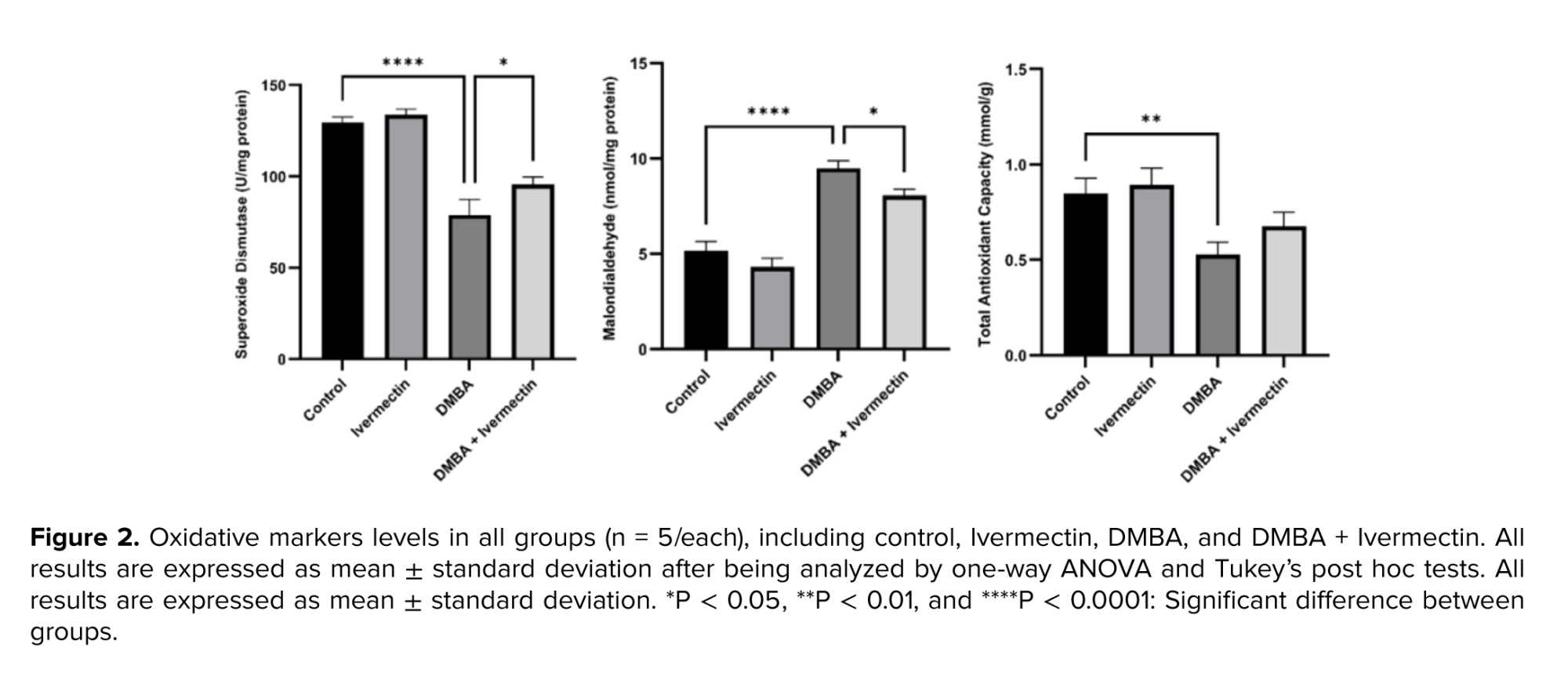
Serum MDA levels were significantly (p < 0.0001) elevated in the DMBA group vs. the control, indicating increased lipid peroxidation. Correspondingly, antioxidant defenses were depleted: SOD activity and TAC were significantly lower in DMBA rats (p < 0.0001 and p < 0.01, respectively). Moreover, Ivermectin treatment alone did not alter these markers vs. control. In contrast, in the DMBA + Ivermectin group, MDA was markedly reduced compared to DMBA alone (p < 0.05), while SOD and TAC levels were significantly higher in the treatment group compared to the DMBA group (Figure 2).
3.3. Apoptosis gene expression
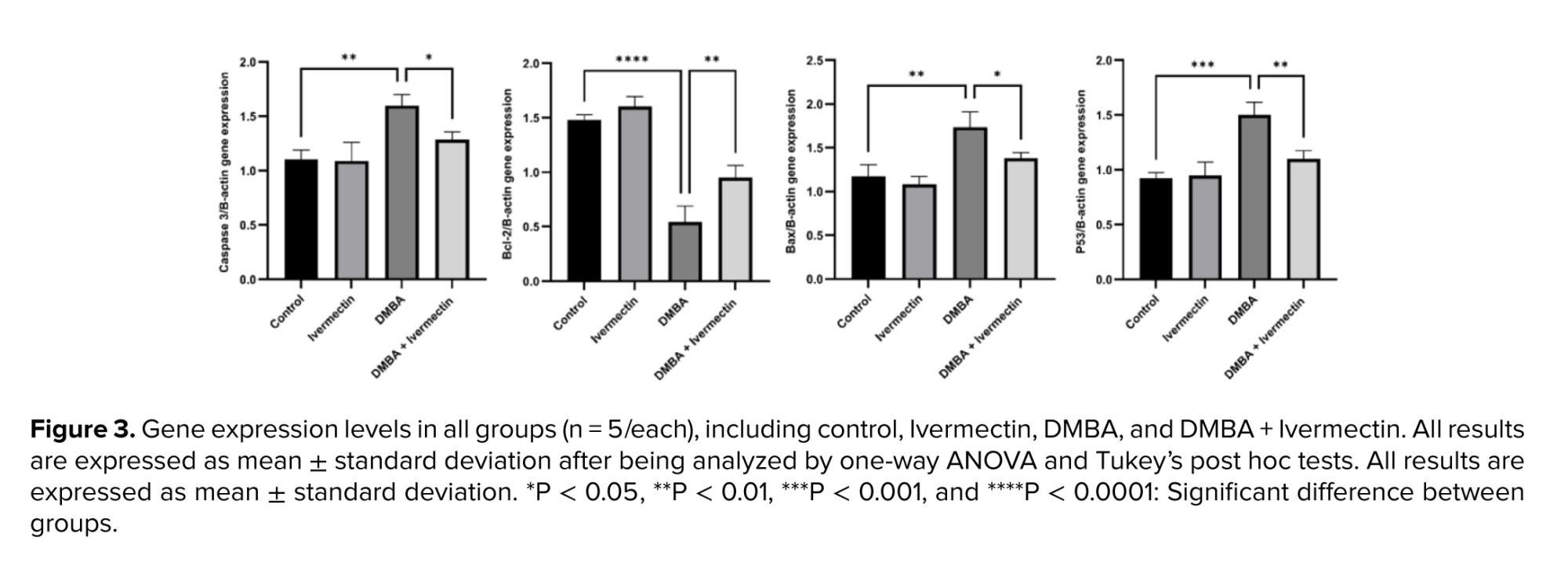
Ovarian tissue expressions of pro-apoptotic genes Bax, Caspase-3, and p53 were significantly upregulated by DMBA (p < 0.01). Anti-apoptotic Bcl-2 was downregulated (p < 0.0001). In contrast, Ivermectin co-treatment blunted these effects: Bax, Caspase-3, and p53 expressions were significantly lower in the DMBA + Ivermectin group compared to the DMBA-only (though still above control), and Bcl-2 was partly restored (Figure 3).
3.4. Histopathology
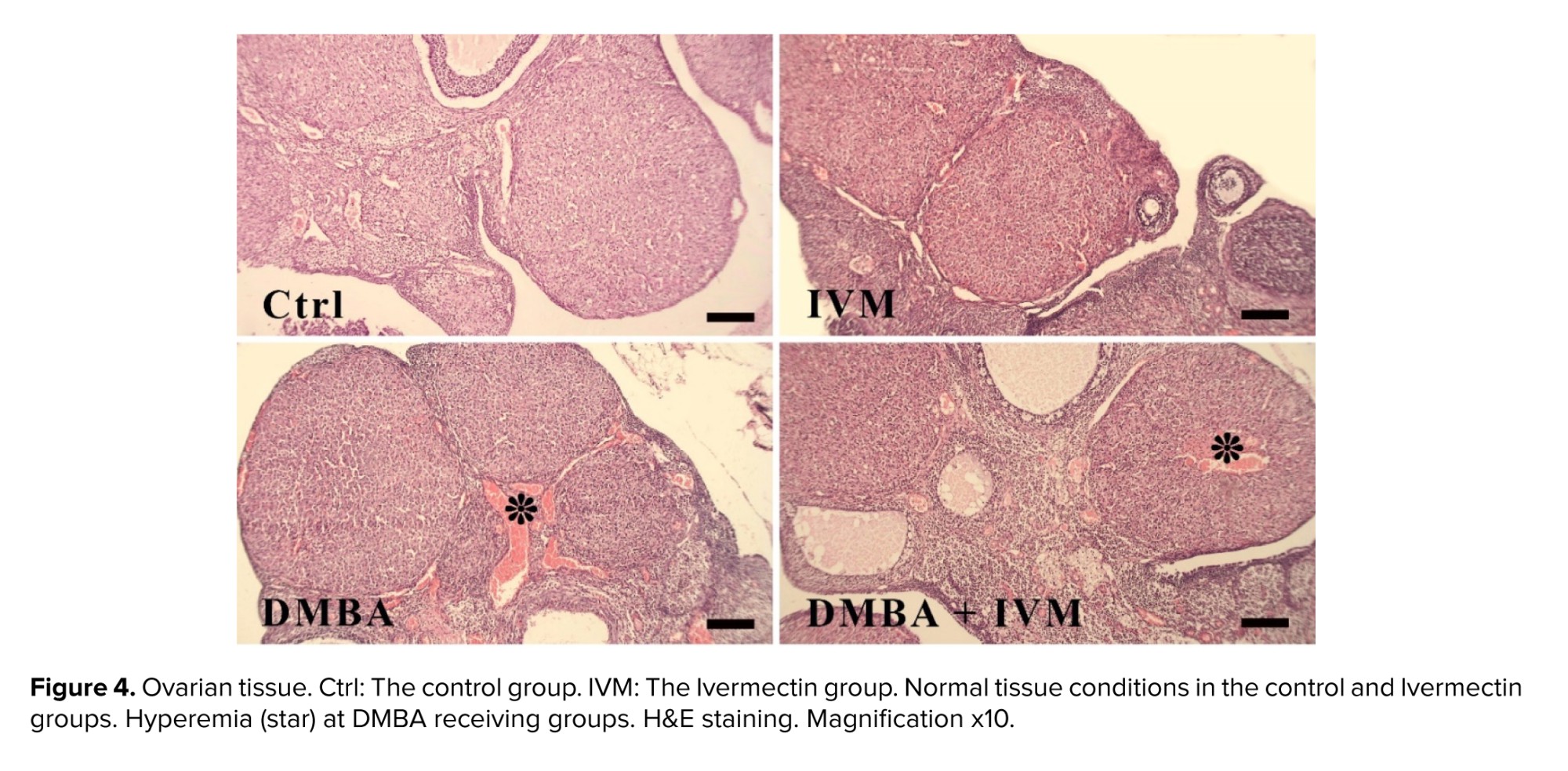
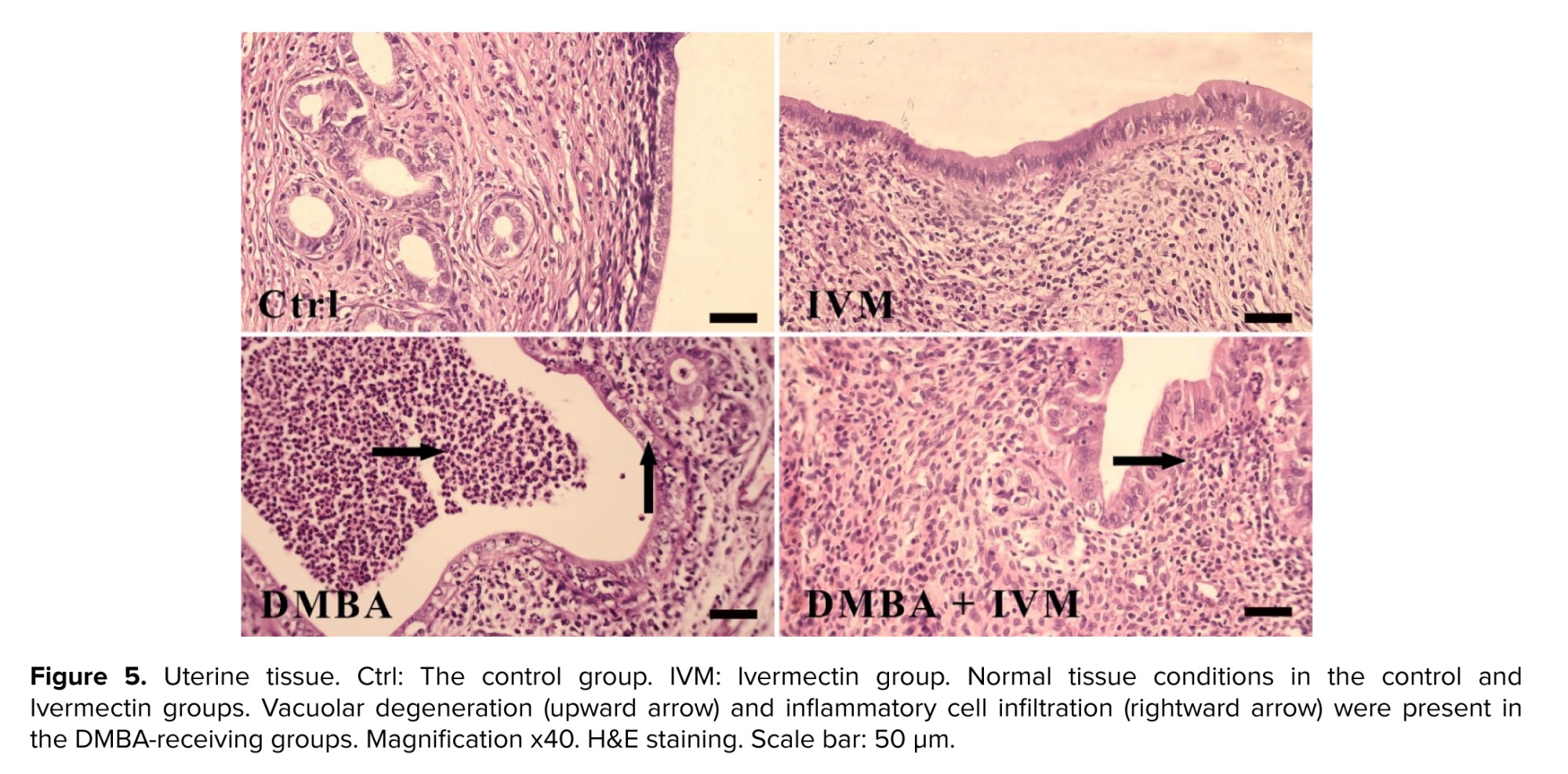
DMBA-treated ovary tissues showed moderate hemorrhage, while the epithelium of uterine was also disorganized, with severe infiltration of inflammatory cells and moderate vacuolar degeneration (Table I). In contrast, in the DMBA + Ivermectin group, the histology of both tissues was much closer to the figure for the normal group, with reduced amount of lesions. Moreover, Ivermectin alone caused no abnormalities (Figures 4, 5).
4. Discussion
This study demonstrates that Ivermectin can substantially attenuate DMBA-induced reproductive toxicity in female rats. Our findings reveal that Ivermectin co-administration significantly attenuates DMBA‑induced oxidative stress, hormonal disruption, apoptosis-related gene activation, and histopathological damage in female reproductive tissues.
DMBA-induced ovarian structural damage corresponded to decreased estradiol and progesterone with elevated testosterone, indicating impaired ovarian steroidogenesis. Ivermectin co-treatment normalized hormone levels, suggesting preserved granulosa cells and theca cell’s function. Comparable restorative endocrine effects have been reported with other antioxidants in DMBA models (18, 26, 27), which protected follicular architecture and steroidogenic enzyme expression, thereby stabilizing hormone profiles.
Consistent with prior reports, DMBA exposure triggered oxidative stress in our model, as evidenced by marked increases in serum MDA and depletion of antioxidant markers. These changes mirror the established mechanism of DMBA toxicity via ROS generation (28, 29). DMBA markedly elevated serum MDA while depleting SOD activity and TAC, indicating lipid peroxidation and impaired antioxidant enzymatic capacity. These findings confirm established mechanisms where PAHs induce ROS via cytochrome P450 bioactivation and subsequent oxidative injury in ovarian tissue (30). Ivermectin co-treatment reduced MDA and restored SOD/TAC. This suggests that ivermectin may upregulate endogenous antioxidant systems, reduce ROS generation, and decrease lipid peroxidation.
At the molecular level, DMBA drove apoptotic signaling. The increase in ovarian Bax, Caspase-3, and p53, along with Bcl-2 suppression, indicates activation of the intrinsic apoptosis pathway. Specifically, DMBA elevated pro-apoptotic genes (Bax, Caspase‑3, p53) and suppressed anti-apoptotic Bcl‑2, consistent with intrinsic mitochondrial apoptosis triggered by DNA damage and high ROS (14). Moreover, an earlier study reported DMBA-induced caspase-3 upregulation and DNA damage markers in ovarian tissue. The combined oxidative stress and DNA adducts produced by DMBA likely activate p53-mediated apoptosis (31). In contrast, Ivermectin co-treatment shifted the Bax/Bcl‑2 ratio and reduced Caspase‑3 and p53 expression. This protective anti-apoptotic effect contrasts with Ivermectin’s apoptotic activity in cancer cells, where it induces mitochondrial-derived apoptosis via ROS generation, collapse of mitochondrial membrane potential, cytochrome c release, and caspase activation (32-34). In our non-cancer tissue model, Ivermectin appears to reduce external ROS rather than generate it, preserving mitochondrial integrity and preventing activation of the intrinsic apoptosis pathway. Although not directly measured, Ivermectin’s known anti-inflammatory actions via NF‑κB suppression may reduce cytokine-driven ROS generation and subsequent apoptosis (35). By dampening inflammation, Ivermectin may limit secondary oxidative damage and preserve steroidogenic function. In addition, our results are parallel with studies in which antioxidants like resveratrol or phytoestrogens mitigated DMBA-induced ovarian injury by lowering ROS, inhibiting apoptosis, and preserving hormonal balance (36). Like these agents, Ivermectin appears to exert protection by mitigating oxidative insult and curbing apoptotic cascades.
Histopathology confirmed severe lesions and tissue degeneration in DMBA rats. Crucially, Ivermectin co-treatment abrogated many of these deleterious effects. The histological preservation of follicles in the Ivermectin group underscores its tissue-protective role. These findings highlight Ivermectin’s repurposing potential.
In summary, Ivermectin’s mitigation of DMBA-induced reproductive toxicity likely reflects a combination of antioxidant restoration, apoptotic pathway suppression, possible activation of protective autophagy, and anti-inflammatory actions. These mechanisms collectively preserve ovarian structure, endocrine function, and cellular viability in the face of environmental toxin insult.
Regarding the limitations and future directions, the precise signaling in ovarian tissue remains undefined. Future studies should assess markers of autophagy (LC3-II/I, Beclin-1, PINK1/Parkin), AKT/mTOR pathway activity, and NF‑κB/p65 expression to validate hypothesized mechanisms. Mitochondrial membrane potential assays or cytochrome c release assays in ovarian follicles could confirm mitochondrial protection. Dose-response studies and broader toxin panels would clarify Ivermectin’s generalizability.
Despite the valuable insights provided, this study has several limitations that warrant consideration. First, the relatively small sample size restricts the statistical power and generalizability of the findings. Second, the absence of a dose-response assessment limits the ability to determine threshold effects or establish causal relationships between exposure and outcome. Finally, mechanistic exploration was confined to gene expression analyses, without complementary assays at the protein or functional level, which constrains the depth of mechanistic interpretation. Future studies with a larger number of animals with both genders included, graded exposure designs and different timepoints, and integrated molecular and biochemical assays are needed to strengthen and extend these observations.
5. Conclusion
It can be concluded that Ivermectin significantly attenuated the adverse effects of DMBA in female Wistar rats. It blunted oxidative damage, normalized sex hormone levels, and reduced pro-apoptotic signaling in ovarian tissue. These results suggest that Ivermectin’s anti-inflammatory/antioxidant properties can be harnessed to protect against environmental reproductive toxins. This study supports further investigation of Ivermectin and similar agents as novel strategies for preserving female reproductive health in polluted environments.
Data Availability
The datasets are available from the corresponding author upon reasonable request.
Author Contributions
SM. Hosseini and FZ. Gharib designed the study and had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. A. Jebeli Eshrat Abadi conducted the research. All authors monitored, evaluated, and analyzed the result of the study. Further, all authors reviewed the article. All authors approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgements
We would like to express our sincere gratitude to Dr. Alireza Salehi for his invaluable guidance throughout revision of this study. His insightful feedback and expertise were instrumental in shaping this research. This study received no funding. In addition, artificial intelligence (Grammarly AI) was employed for grammar check.
Conflict of Interest
The authors declare that they have no competing interest.
2.3. Oxidative stress markers
The activity and levels of malondialdehyde (MDA), superoxide dismutase (SOD), and total antioxidant capacity (TAC) were assayed by Navand Salamat kits (Nalondi [product number: NS-15022], Nasdox [product number: NS-15034], and Naxifer [product number: NS-15012] kits, respectively) (20). All assays were performed in duplicate according to manufacturer protocols.
2.4. Gene expression
Ovarian tissue was excised and homogenized for RNA extraction. cDNA was synthesized, and quantitative real-time polymerase chain reaction (qRT-PCR) was performed for apoptosis genes: Bax, Bcl-2, Caspase-3, and p53. B-actin was used as an internal control (21). Relative expression was calculated by the 2^-ΔΔCt method.
2.5. Histopathology
Uterine and ovarian tissues were fixed in 10% formalin, embedded in paraffin, sectioned (5 μm), and stained with H&E. Sections were examined under light microscopy (22).
2.6. Ethical Considerations
Additionally, the Research Ethics Committees of Islamic Azad University, Babol Branch, babol, Iran found this project to be in accordance with the ethical principles and the national norms and standards for conducting medical research in Iran(Code: IR.IAU.BABOL.REC.1404.046) In addition, the ARRIVE guideline was followed.
2.7. Statistical Analysis
All the data related to hormones, oxidative stress markers, and gene expressions were analyzed using GraphPad software version 10 based on the one-way ANOVA and Tukey’s post hoc tests. P < 0.05 was considered as a significant difference. The results were expressed as the mean ± standard deviation (23). The histopathological evaluation was assessed based on earlier research (24). Briefly, the severity of lesions was defined as no lesions (-), mild lesions (+), moderate lesions (++), and severe lesion (+++) (25).
3. Results
3.1. Sex hormones
In comparison to the control group, all the hormone levels were found to be significantly decreased in the DMBA group (p < 0.001). Additionally, Ivermectin alone did not affect hormone levels. However, in the DMBA + Ivermectin group, progesterone level was significantly (p < 0.05) increased compared to the DMBA group. Similarly, testosterone levels also raised significantly (p < 0.001) compared to DMBA group (Figure 1).
3.2. Oxidative stress markers
Serum MDA levels were significantly (p < 0.0001) elevated in the DMBA group vs. the control, indicating increased lipid peroxidation. Correspondingly, antioxidant defenses were depleted: SOD activity and TAC were significantly lower in DMBA rats (p < 0.0001 and p < 0.01, respectively). Moreover, Ivermectin treatment alone did not alter these markers vs. control. In contrast, in the DMBA + Ivermectin group, MDA was markedly reduced compared to DMBA alone (p < 0.05), while SOD and TAC levels were significantly higher in the treatment group compared to the DMBA group (Figure 2).
3.3. Apoptosis gene expression
Ovarian tissue expressions of pro-apoptotic genes Bax, Caspase-3, and p53 were significantly upregulated by DMBA (p < 0.01). Anti-apoptotic Bcl-2 was downregulated (p < 0.0001). In contrast, Ivermectin co-treatment blunted these effects: Bax, Caspase-3, and p53 expressions were significantly lower in the DMBA + Ivermectin group compared to the DMBA-only (though still above control), and Bcl-2 was partly restored (Figure 3).
3.4. Histopathology
DMBA-treated ovary tissues showed moderate hemorrhage, while the epithelium of uterine was also disorganized, with severe infiltration of inflammatory cells and moderate vacuolar degeneration (Table I). In contrast, in the DMBA + Ivermectin group, the histology of both tissues was much closer to the figure for the normal group, with reduced amount of lesions. Moreover, Ivermectin alone caused no abnormalities (Figures 4, 5).






4. Discussion
This study demonstrates that Ivermectin can substantially attenuate DMBA-induced reproductive toxicity in female rats. Our findings reveal that Ivermectin co-administration significantly attenuates DMBA‑induced oxidative stress, hormonal disruption, apoptosis-related gene activation, and histopathological damage in female reproductive tissues.
DMBA-induced ovarian structural damage corresponded to decreased estradiol and progesterone with elevated testosterone, indicating impaired ovarian steroidogenesis. Ivermectin co-treatment normalized hormone levels, suggesting preserved granulosa cells and theca cell’s function. Comparable restorative endocrine effects have been reported with other antioxidants in DMBA models (18, 26, 27), which protected follicular architecture and steroidogenic enzyme expression, thereby stabilizing hormone profiles.
Consistent with prior reports, DMBA exposure triggered oxidative stress in our model, as evidenced by marked increases in serum MDA and depletion of antioxidant markers. These changes mirror the established mechanism of DMBA toxicity via ROS generation (28, 29). DMBA markedly elevated serum MDA while depleting SOD activity and TAC, indicating lipid peroxidation and impaired antioxidant enzymatic capacity. These findings confirm established mechanisms where PAHs induce ROS via cytochrome P450 bioactivation and subsequent oxidative injury in ovarian tissue (30). Ivermectin co-treatment reduced MDA and restored SOD/TAC. This suggests that ivermectin may upregulate endogenous antioxidant systems, reduce ROS generation, and decrease lipid peroxidation.
At the molecular level, DMBA drove apoptotic signaling. The increase in ovarian Bax, Caspase-3, and p53, along with Bcl-2 suppression, indicates activation of the intrinsic apoptosis pathway. Specifically, DMBA elevated pro-apoptotic genes (Bax, Caspase‑3, p53) and suppressed anti-apoptotic Bcl‑2, consistent with intrinsic mitochondrial apoptosis triggered by DNA damage and high ROS (14). Moreover, an earlier study reported DMBA-induced caspase-3 upregulation and DNA damage markers in ovarian tissue. The combined oxidative stress and DNA adducts produced by DMBA likely activate p53-mediated apoptosis (31). In contrast, Ivermectin co-treatment shifted the Bax/Bcl‑2 ratio and reduced Caspase‑3 and p53 expression. This protective anti-apoptotic effect contrasts with Ivermectin’s apoptotic activity in cancer cells, where it induces mitochondrial-derived apoptosis via ROS generation, collapse of mitochondrial membrane potential, cytochrome c release, and caspase activation (32-34). In our non-cancer tissue model, Ivermectin appears to reduce external ROS rather than generate it, preserving mitochondrial integrity and preventing activation of the intrinsic apoptosis pathway. Although not directly measured, Ivermectin’s known anti-inflammatory actions via NF‑κB suppression may reduce cytokine-driven ROS generation and subsequent apoptosis (35). By dampening inflammation, Ivermectin may limit secondary oxidative damage and preserve steroidogenic function. In addition, our results are parallel with studies in which antioxidants like resveratrol or phytoestrogens mitigated DMBA-induced ovarian injury by lowering ROS, inhibiting apoptosis, and preserving hormonal balance (36). Like these agents, Ivermectin appears to exert protection by mitigating oxidative insult and curbing apoptotic cascades.
Histopathology confirmed severe lesions and tissue degeneration in DMBA rats. Crucially, Ivermectin co-treatment abrogated many of these deleterious effects. The histological preservation of follicles in the Ivermectin group underscores its tissue-protective role. These findings highlight Ivermectin’s repurposing potential.
In summary, Ivermectin’s mitigation of DMBA-induced reproductive toxicity likely reflects a combination of antioxidant restoration, apoptotic pathway suppression, possible activation of protective autophagy, and anti-inflammatory actions. These mechanisms collectively preserve ovarian structure, endocrine function, and cellular viability in the face of environmental toxin insult.
Regarding the limitations and future directions, the precise signaling in ovarian tissue remains undefined. Future studies should assess markers of autophagy (LC3-II/I, Beclin-1, PINK1/Parkin), AKT/mTOR pathway activity, and NF‑κB/p65 expression to validate hypothesized mechanisms. Mitochondrial membrane potential assays or cytochrome c release assays in ovarian follicles could confirm mitochondrial protection. Dose-response studies and broader toxin panels would clarify Ivermectin’s generalizability.
Despite the valuable insights provided, this study has several limitations that warrant consideration. First, the relatively small sample size restricts the statistical power and generalizability of the findings. Second, the absence of a dose-response assessment limits the ability to determine threshold effects or establish causal relationships between exposure and outcome. Finally, mechanistic exploration was confined to gene expression analyses, without complementary assays at the protein or functional level, which constrains the depth of mechanistic interpretation. Future studies with a larger number of animals with both genders included, graded exposure designs and different timepoints, and integrated molecular and biochemical assays are needed to strengthen and extend these observations.
5. Conclusion
It can be concluded that Ivermectin significantly attenuated the adverse effects of DMBA in female Wistar rats. It blunted oxidative damage, normalized sex hormone levels, and reduced pro-apoptotic signaling in ovarian tissue. These results suggest that Ivermectin’s anti-inflammatory/antioxidant properties can be harnessed to protect against environmental reproductive toxins. This study supports further investigation of Ivermectin and similar agents as novel strategies for preserving female reproductive health in polluted environments.
Data Availability
The datasets are available from the corresponding author upon reasonable request.
Author Contributions
SM. Hosseini and FZ. Gharib designed the study and had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. A. Jebeli Eshrat Abadi conducted the research. All authors monitored, evaluated, and analyzed the result of the study. Further, all authors reviewed the article. All authors approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgements
We would like to express our sincere gratitude to Dr. Alireza Salehi for his invaluable guidance throughout revision of this study. His insightful feedback and expertise were instrumental in shaping this research. This study received no funding. In addition, artificial intelligence (Grammarly AI) was employed for grammar check.
Conflict of Interest
The authors declare that they have no competing interest.
Type of Study: Original Article |
Subject:
Reproductive Pattology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







