Sat, May 23, 2026
[Archive]
Volume 17, Issue 3 (March 2019 2019)
IJRM 2019, 17(3): 20-209 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Rashidi B H, Kabodmehri R, Shariat M, Shahrokh Tehraninejad E, Abdollahi A, Bagheri M et al . Luteinizing hormone changes in gonadotropin-releasing hormone antagonist protocol in in vitro fertilization cycles: A cross-sectional study. IJRM 2019; 17 (3) :20-209
URL: http://ijrm.ir/article-1-1454-en.html
URL: http://ijrm.ir/article-1-1454-en.html
Batool Hosein Rashidi1 

 , Roya Kabodmehri *2
, Roya Kabodmehri *2 

 , Mamak Shariat1
, Mamak Shariat1 

 , Ensieh Shahrokh Tehraninejad1
, Ensieh Shahrokh Tehraninejad1 

 , Alireza Abdollahi1
, Alireza Abdollahi1 

 , Maryam Bagheri1
, Maryam Bagheri1 

 , Fedieh Hagholahi1
, Fedieh Hagholahi1 




 , Roya Kabodmehri *2
, Roya Kabodmehri *2 

 , Mamak Shariat1
, Mamak Shariat1 

 , Ensieh Shahrokh Tehraninejad1
, Ensieh Shahrokh Tehraninejad1 

 , Alireza Abdollahi1
, Alireza Abdollahi1 

 , Maryam Bagheri1
, Maryam Bagheri1 

 , Fedieh Hagholahi1
, Fedieh Hagholahi1 


1- Health Reproductive Research Center, Imam Khomeini Hospital, Tehran University of Medical Sciences, Tehran, Iran.
2- Health Reproductive Research Center, Imam Khomeini Hospital, Tehran University of Medical Sciences, Tehran, Iran. ,royakabodmehri@yahoo.com
2- Health Reproductive Research Center, Imam Khomeini Hospital, Tehran University of Medical Sciences, Tehran, Iran. ,
Full-Text [PDF 287 kb]
(1578 Downloads)
| Abstract (HTML) (4391 Views)
Full-Text: (673 Views)
1. Introduction
According to the two-cell-gonadotropin hypothesis, androgen plays a crucial role in making of estradiol (E2) by granulosa cell. Luteinizing hormone (LH) directly contributes to the control of granulosa cell function (1). The role of LH in folliculogenesis includes stimulation of theca cell, androgen production, triggering ovulation, supporting the corpus luteum, and development of late follicular stage (2). There is a basic and clinical evidence that show late development of follicle needs an LH, but there is a threshold for LH requirements during folliculogenesis (1). It is known that <1% of LH receptor needs to be occupied to provoke a best steroidogenic response (3), thus resting level of LH (1–10 IU/L) must be adequate to offer maximal stimulation to theca cell (3).
The role of in vitro fertilization was accepted as an effective way for the treatment of infertile patients (4). It has been well-known that suppressing the release of a pituitary follicle stimulatory hormone (FSH) and LH in controlled ovarian hyper stimulation cycles with Gonadotropinreleasing hormone (GnRH) analogs is an important factor relevant to the success of IVF treatment. In some IVF cycle, premature surge was a big problem. This problem was solved by Schally in 1971 through introduction of a GnRH agonist (5). Although GnRH-agonist managed premature LH surge, it was associated with creating new problems such as occurrence of cyst formation at the start of induction ovulation and ovarian hyper stimulation syndrome (OHSS) (1). The use of GnRH agonist associated with long duration of administration, low E2 level and side effect due to estrogen deficiency (1). Despite the availability of GnRH antagonist, it was not used for this purpose due to allergies in 1971 (6). In the antagonist protocol, several important advantages may be noted which include the following: in the polycystic ovarian syndrome (PCOS) patients, GnRH agonist can be used as a trigger in this protocol and reduce the risk of OHSS (7, 8). Several studies compared this two analog and the use of GnRH-antagonist was associated with shorter stimulation period and decreased the occurrence of OHSS by more than 50% (9, 10). One of the problems with taking GnRH antagonist seems to be a reduction in the blood level of LH.
There is a wide range of pituitary responses to GnRH antagonists. In some patients that received a single dose of GnRH-antagonist (3 mg), LH level may be decreased below a threshold level (9, 11). Some studies showed that the fast response to all doses of GnRH antagonists cause a drop in LH levels, which is similar in among all doses. However, a large change in LH levels is detected for the pituitary recovery 24 h later. While, 24 h after injection the low antagonist doses allow a quick recovery and high doses result are a partial recovery in pre-treatment LH levels (12). Maximum patients have a normal response; however, “hypo responders” might ovulate prematurely, while “over-suppressed” patients may have impaired follicular development (12).
Therefore, the aim of this study is the evaluation of LH change in antagonist protocol and identify patients who benefit from adding LH.
2. Materials and Methods
In this prospective cross-sectional study, 70 volunteers for antagonist protocol in IVF cycle (due to PCOS, a poor responder in the previous cycle and Egg donor) who were referred to the Reproductive Health Research Center, Tehran University of Medical Sciences between July 2016 and February 2016 were enrolled. PCOS defined by Rotterdam criteria: clinical and/or biochemical hyperandrogenism and oligo-amenorrhea and/or ultrasonographic criteria ≥12 follicles (2–9 mm diameter) and/or ovarian volume ≥10 mL (1). Based on ESHRE meeting the poor responder patients are defined according to two criteria of the following criteria:
I. Advanced maternal age (≥40 y) or any other risk factor.
II. A previous POR (cycles canceled or ≤3 oocytes with a conventional protocol).
III. An abnormal ovarian reserve test (antral follicle count <5–7 follicles or anti-Mullerian hormone (AMH) <0.5–1.1 ng/ml) (13).
Exclusion criteria was allergy to antagonist and patients with long agonist protocol. On the first day of menstrual cycle, suppression of ovary was confirmed by ultrasonography. The hormonal assessment was performed on the 2nd day of menstrual cycle including FSH, LH, and E2. For all patients gonadotropin (Follistim, Merck; GonalF, EMD-Serono) was started from the 2nd day of menstruation according to AMH and antral follicular countage, previous response to gonadotropin. Six days after the start of gonadotropin, ultrasonography was performed for estimate follicular size and repeated if necessary. When follicle reached ≥13 mm, we used GnRH-antagonist (Cetrotide, EMDSerono 0.25 mg/day SC). Serum LH level was estimated 24 hr before and after first Cetrotide injection. When two follicles were <18 mm, we used Human chorionic gonadotropin (10.000 IU; Pregnyl, Organon, Oss, Netherlands) intramuscular as a trigger; 36 hr after trigger, oocyte retrieval was performed under ultrasound guide. Primary outcome was the serum level of LH and its change in the three groups and the secondary outcome was Egg and Embryo quality.
2.1. Ethical consideration
The study was approved by the Ethics Committee Review Board of Tehran University (No: IRTUMS.VCR.REC.1395.1405). Written inform consent was obtained from all participants.
2.2. Statistical analysis
The data were analyzed using Statistical Package for the Social Sciences, version 20, SPSS Inc, Chicago, Illinois, USA (SPSS) software. Independent t-test, Man–Whitney test, and Kruskal– Wallis was used to compare the parametric and nonparametric parameters between groups. Comparison of qualitative variables between groups was performed using Chi-square test. The quantitative parameters were reported as mean±SEM. A p< 0.05 was taken as statistically significant. Spearman correlation test was used to assess the correlation of LH parameters with other factors.
3. Results
Table I shows the demographic characteristics and basal Hormonal assay. As expected, the average age of the poor responder group is more than two other groups (37.8±4.43) (p= 0.001). According to Kruskal–Wallis (nonparametric test), the serum level of basal LH that was measured on the second day of menstruation shows significant difference (p= 0.004). As expected serum level of LH is higher in PCOS (6.7±4.3) than poor responder (5.18±3) and normal responder groups (3.67±4) (Figure 1). As well as the level of AMH is high in PCOS groups. Other variables examined (basal FSH, E2, BMI, duration of infertility, number of previous IVF) in three groups showed no significant difference. Results of Table II show the significant differences between the three groups containing follicle-number, oocyte number, metaphase 2 (M2) oocyte, Germinal vesicle (GV) oocyte, number of transferred embryos and the number of consumed Gonadotropin ampule. To determine the difference between the three groups, we used Post Hoc and Bonferroni test. The number of follicles, GV oocyte and transferred embryo show significant difference between the PCO and poor responder group and the PCO and normal responder only, but there was no difference between Poor and normal responder. The number of the eggs and M2 oocyte has only significant difference between the PCO and Poor responder patients. This difference cannot be seen in each other. But it was significantly different between the three groups together in the number of gonadotropins ampule, and gonadotropin consumed in poor responder group was more than other groups (p= 0.0001). There was no significant difference (p= 0.45, p= 0.843) in LH levels 24 h before and after the injection of the antagonist between the three groups. As well as, the LH ratio and LH change not differ in the three groups (p= 0.077, p= 0.486). According to the Chi-square test, the LH difference percent in the three groups was not significant (p= 0.81). Spearman correlation test was used to assess the correlation of LH parameters with the number of follicle and oocyte and GV oocyte, metaphase 1 (M1) oocyte, M2 oocyte, endometrial thickness. The only significant relationship was seen between LH ratio and M1 (r= 0.31, p= 0.008) and LH change and M1(r= 0.26, p= 0.025). In other words, if the LH (24 hours after administration of the antagonist) is high, that is, the LH suppression by the antagonist is lower and the rate of M1 oocyte was more. According to Mann– Whitney and Fisher test, the correlation of the LH decrease were not significantly associated with GV, M1, M2 oocyte, the number of the follicle, the number of the oocyte, endometrial thickness (Table III). In the other words, the lower levels of LH after the injection had no effect on outcomes.
Table I: Characteristics of patients.
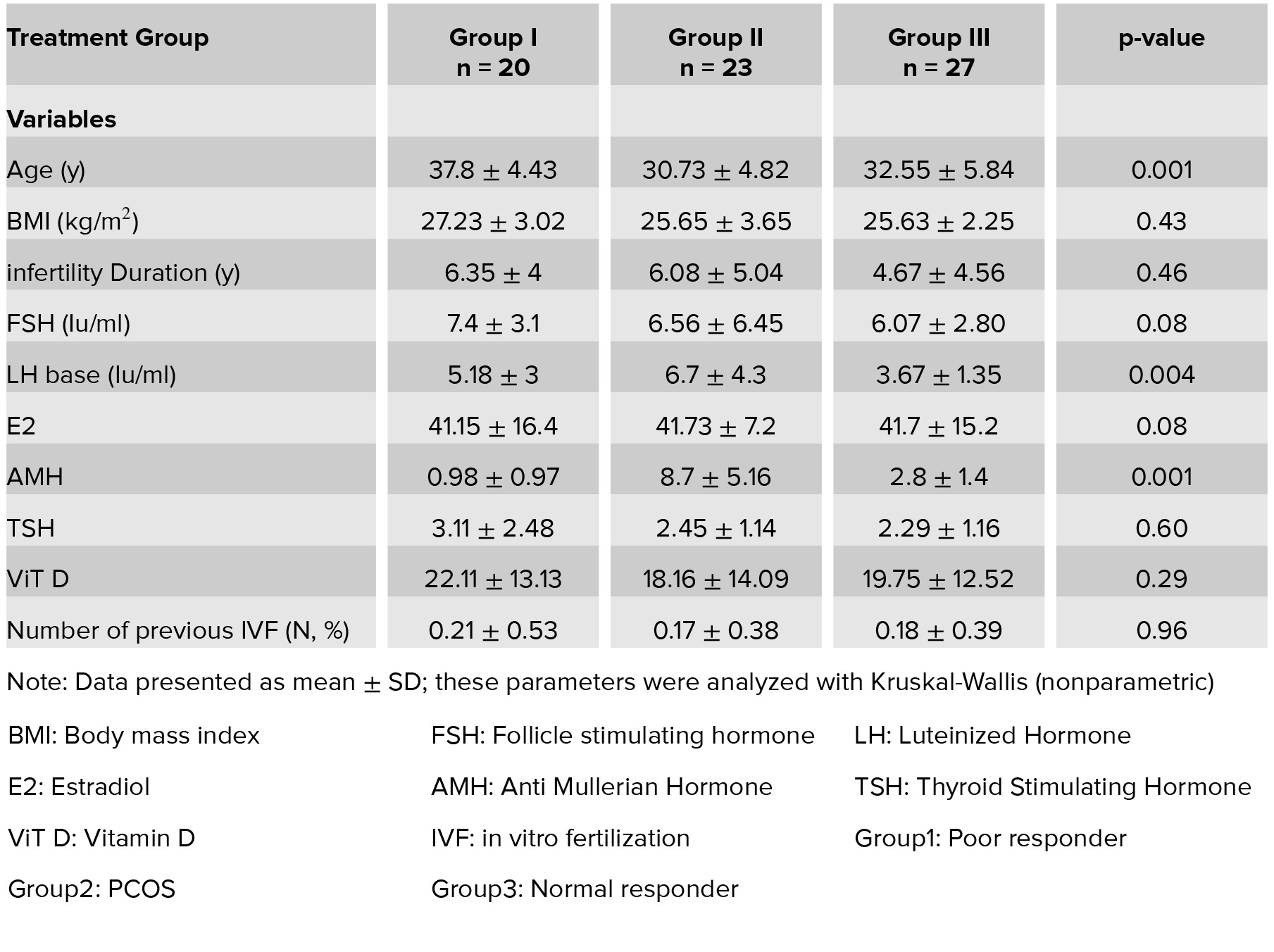
Table II: Treatment outcomes in a patient in three groups.
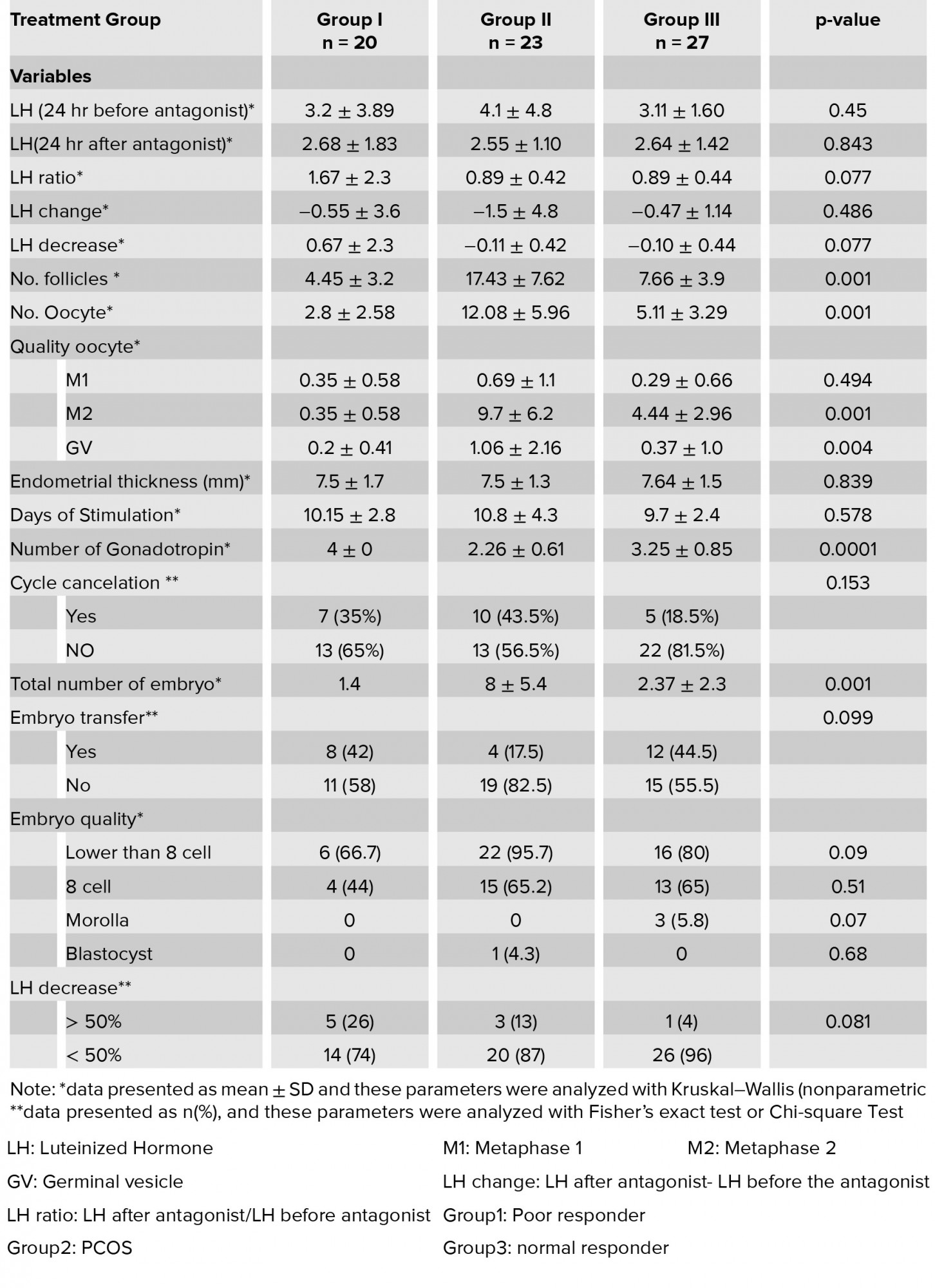
Table III: Correlation of LH decreases percent with treatment outcome in total group.
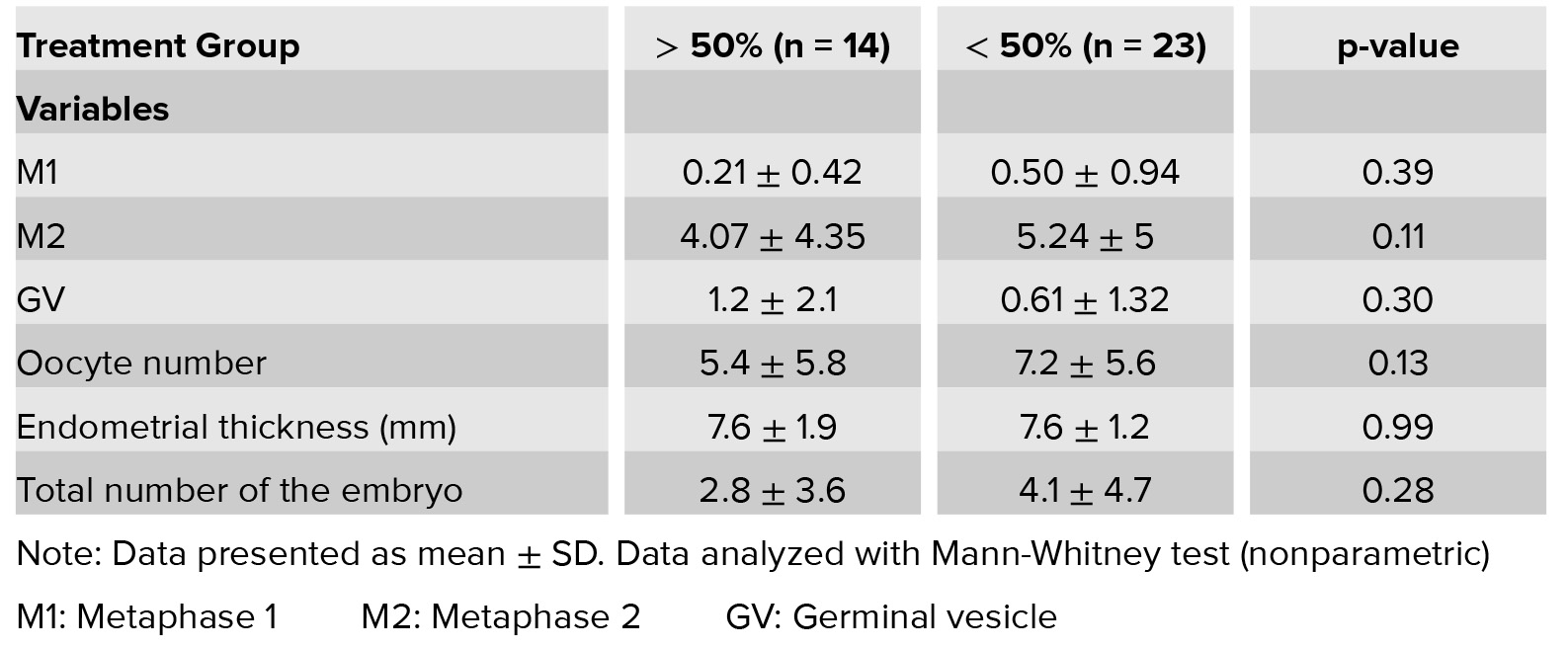
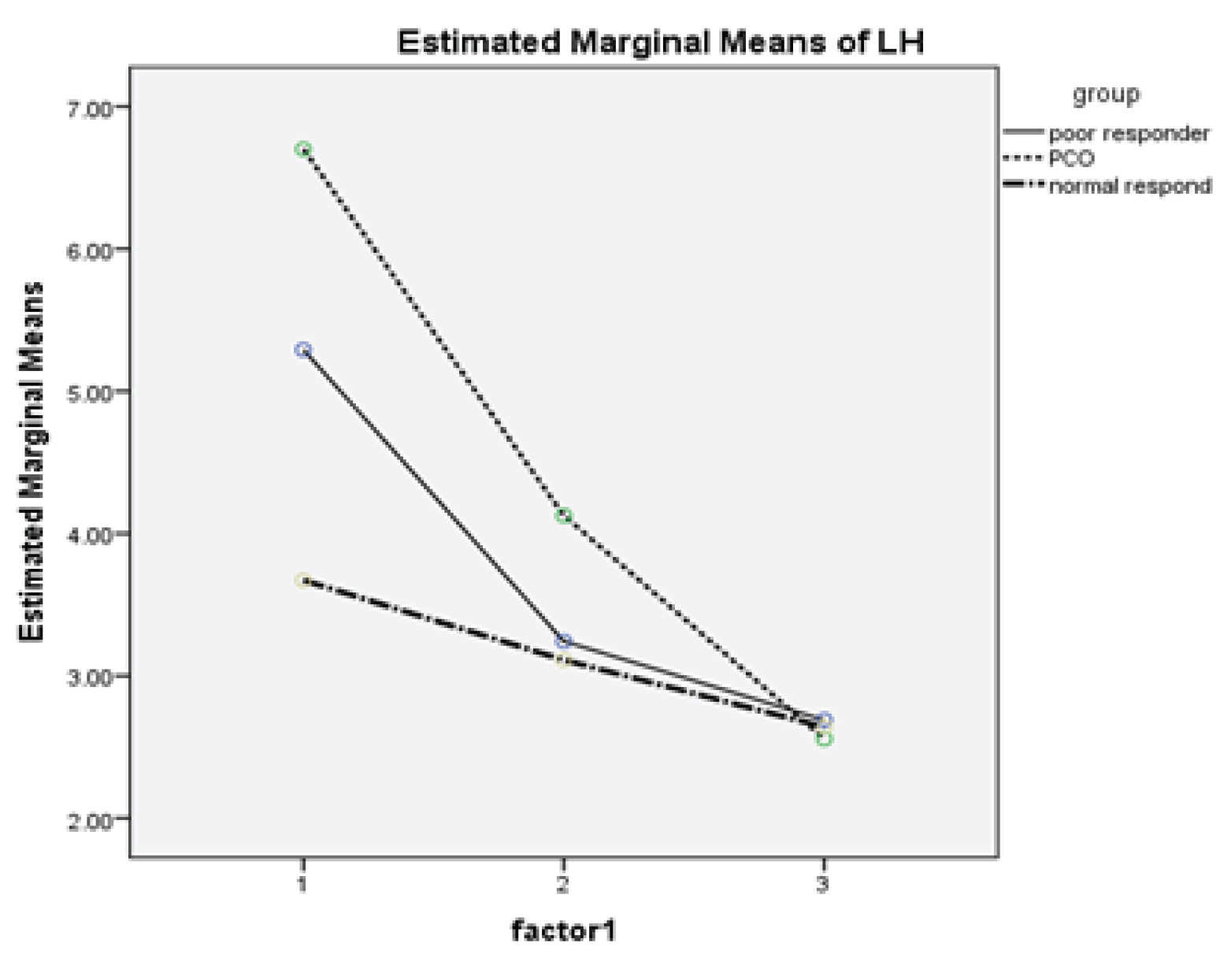
Figure 1: Slope or shot in the PCO is significantly higher than the other two groups, except in accordance with the above figure are more tangible between PCO and Normal.
4. Discussion
We evaluate the changes of serum LH in the three groups of patients who were entered in antagonist protocol. In none of the patients in the three groups, the blood level of the LH followed the GnRH antagonist reached below one. There was no reduction in more than 50% of the LH levels in any group following a GnRH antagonist. In our study, there was no significant difference in the LH levels 24 h before and after injection of the antagonist between three groups, as well as on the LH ratio we didn’t find a significant difference. Changes in LH more than 50% (drop more than 50% or even increase) was not significantly associated with GV, M1, M2 oocyte, the number of the follicle, the number of the oocyte, endometrial thickness. Several studies have evaluated the role of LH in antagonist protocol (14). Studies in hypogonadotropic patients using r-FSH established that FSH can induce follicular development in the preovulatory phase, but E2 and androstenedione remain very low, and this advocates that follicular development depends on the action of LH to induce androgen biosynthesis as a substrate for aromatase activity (15). Suitable folliculogenesis and steroid genesis are necessary for successful fertilization and implantation. Therefore, it depends upon a certain threshold level of LH. Although, the amount of LH necessary for standard follicle and oocyte maturation is not known (16), it is likely to be very low since a best steroidogenic response can be prompted when <1% of follicular LH receptors are occupied (16). There is also evidence that excessive level of LH can have an adverse effect on follicular development (17). So, there is a window for LH while the low and high level of LH is a harmful effect in follicular maturation (18). This is an update of a Cochrane review in 2001, and previously updated in 2006 and 2011 (19). In the study by Huirne and coworkers, they evaluated the change of plasma level in LH in five groups with different dosage of GnRH–antagonist, and effect of these changes on IVF outcome. In this study, no pregnancies were observed in relation to either very high or low LH which protects the window hypothesis (20). According to this study, excessive fluctuation in LH levels might disrupt this balance and associate with adverse effect (20). Overall it seems that LH may have a valuable effect through a mechanism that recovers oocyte maturation (20). In the studies by Gomez–Palomares and coworkers (1) and Lisi and coworker, significant improvement in IVF outcome in poor responder patients was found (21). Because of the unexpected and often dramatic inhibition of LH secretion related with the usage of GnRH-anta, there has been a probable need for exogenous LH supplementation (1). The meta analysis from six clinical randomized trials showed that normogomadotropic patients did not benefit from adding LH (22). In the cohort retrospective study in poor responder or AMA (advanced maternal age) did not protect add LH supplementation (22). In the study by Hill and coworkers, adding exogenous LH in AMA patients improved implantation and pregnancy rate (23). In the study by Kol, patients who were hyper sensitive to GnRH– antagonist benefitted from r-LH (12). In the study by Younis and coworkers, r-LH supplementation to rFSH following GnRH-antagonist administration did not seem to significantly increase serum E2 level on the day of Human chorionic gonadotropin administration in AMA patients. This recommends that endogenous serum LH levels following GnRHantagonist beginning are adequate for suitable late follicular ovarian steroidogenesis (14). The novelty of our study is evaluating the LH change in three subgroups including hyper responder (PCOS patients), hypo responds (poor ovarian reserve), and normal responder (donor patients). Limitations of the study:
1. Small sample size may affect results; however, further clinical trial studies with large sample size are needed.
2. Because in some patients we had frozen embryos, there was no possibility of assessing pregnancy.
5. Conclusion
The results showed no difference in the levels of LH and changes of LH in antagonist protocol between three groups, so it is not practical to assess the LH level in ART cycles.
Acknowledgments
In this study there was no financial support.
Conflict of Interest
In this study there was no conflict of interest.
According to the two-cell-gonadotropin hypothesis, androgen plays a crucial role in making of estradiol (E2) by granulosa cell. Luteinizing hormone (LH) directly contributes to the control of granulosa cell function (1). The role of LH in folliculogenesis includes stimulation of theca cell, androgen production, triggering ovulation, supporting the corpus luteum, and development of late follicular stage (2). There is a basic and clinical evidence that show late development of follicle needs an LH, but there is a threshold for LH requirements during folliculogenesis (1). It is known that <1% of LH receptor needs to be occupied to provoke a best steroidogenic response (3), thus resting level of LH (1–10 IU/L) must be adequate to offer maximal stimulation to theca cell (3).
The role of in vitro fertilization was accepted as an effective way for the treatment of infertile patients (4). It has been well-known that suppressing the release of a pituitary follicle stimulatory hormone (FSH) and LH in controlled ovarian hyper stimulation cycles with Gonadotropinreleasing hormone (GnRH) analogs is an important factor relevant to the success of IVF treatment. In some IVF cycle, premature surge was a big problem. This problem was solved by Schally in 1971 through introduction of a GnRH agonist (5). Although GnRH-agonist managed premature LH surge, it was associated with creating new problems such as occurrence of cyst formation at the start of induction ovulation and ovarian hyper stimulation syndrome (OHSS) (1). The use of GnRH agonist associated with long duration of administration, low E2 level and side effect due to estrogen deficiency (1). Despite the availability of GnRH antagonist, it was not used for this purpose due to allergies in 1971 (6). In the antagonist protocol, several important advantages may be noted which include the following: in the polycystic ovarian syndrome (PCOS) patients, GnRH agonist can be used as a trigger in this protocol and reduce the risk of OHSS (7, 8). Several studies compared this two analog and the use of GnRH-antagonist was associated with shorter stimulation period and decreased the occurrence of OHSS by more than 50% (9, 10). One of the problems with taking GnRH antagonist seems to be a reduction in the blood level of LH.
There is a wide range of pituitary responses to GnRH antagonists. In some patients that received a single dose of GnRH-antagonist (3 mg), LH level may be decreased below a threshold level (9, 11). Some studies showed that the fast response to all doses of GnRH antagonists cause a drop in LH levels, which is similar in among all doses. However, a large change in LH levels is detected for the pituitary recovery 24 h later. While, 24 h after injection the low antagonist doses allow a quick recovery and high doses result are a partial recovery in pre-treatment LH levels (12). Maximum patients have a normal response; however, “hypo responders” might ovulate prematurely, while “over-suppressed” patients may have impaired follicular development (12).
Therefore, the aim of this study is the evaluation of LH change in antagonist protocol and identify patients who benefit from adding LH.
2. Materials and Methods
In this prospective cross-sectional study, 70 volunteers for antagonist protocol in IVF cycle (due to PCOS, a poor responder in the previous cycle and Egg donor) who were referred to the Reproductive Health Research Center, Tehran University of Medical Sciences between July 2016 and February 2016 were enrolled. PCOS defined by Rotterdam criteria: clinical and/or biochemical hyperandrogenism and oligo-amenorrhea and/or ultrasonographic criteria ≥12 follicles (2–9 mm diameter) and/or ovarian volume ≥10 mL (1). Based on ESHRE meeting the poor responder patients are defined according to two criteria of the following criteria:
I. Advanced maternal age (≥40 y) or any other risk factor.
II. A previous POR (cycles canceled or ≤3 oocytes with a conventional protocol).
III. An abnormal ovarian reserve test (antral follicle count <5–7 follicles or anti-Mullerian hormone (AMH) <0.5–1.1 ng/ml) (13).
Exclusion criteria was allergy to antagonist and patients with long agonist protocol. On the first day of menstrual cycle, suppression of ovary was confirmed by ultrasonography. The hormonal assessment was performed on the 2nd day of menstrual cycle including FSH, LH, and E2. For all patients gonadotropin (Follistim, Merck; GonalF, EMD-Serono) was started from the 2nd day of menstruation according to AMH and antral follicular countage, previous response to gonadotropin. Six days after the start of gonadotropin, ultrasonography was performed for estimate follicular size and repeated if necessary. When follicle reached ≥13 mm, we used GnRH-antagonist (Cetrotide, EMDSerono 0.25 mg/day SC). Serum LH level was estimated 24 hr before and after first Cetrotide injection. When two follicles were <18 mm, we used Human chorionic gonadotropin (10.000 IU; Pregnyl, Organon, Oss, Netherlands) intramuscular as a trigger; 36 hr after trigger, oocyte retrieval was performed under ultrasound guide. Primary outcome was the serum level of LH and its change in the three groups and the secondary outcome was Egg and Embryo quality.
2.1. Ethical consideration
The study was approved by the Ethics Committee Review Board of Tehran University (No: IRTUMS.VCR.REC.1395.1405). Written inform consent was obtained from all participants.
2.2. Statistical analysis
The data were analyzed using Statistical Package for the Social Sciences, version 20, SPSS Inc, Chicago, Illinois, USA (SPSS) software. Independent t-test, Man–Whitney test, and Kruskal– Wallis was used to compare the parametric and nonparametric parameters between groups. Comparison of qualitative variables between groups was performed using Chi-square test. The quantitative parameters were reported as mean±SEM. A p< 0.05 was taken as statistically significant. Spearman correlation test was used to assess the correlation of LH parameters with other factors.
3. Results
Table I shows the demographic characteristics and basal Hormonal assay. As expected, the average age of the poor responder group is more than two other groups (37.8±4.43) (p= 0.001). According to Kruskal–Wallis (nonparametric test), the serum level of basal LH that was measured on the second day of menstruation shows significant difference (p= 0.004). As expected serum level of LH is higher in PCOS (6.7±4.3) than poor responder (5.18±3) and normal responder groups (3.67±4) (Figure 1). As well as the level of AMH is high in PCOS groups. Other variables examined (basal FSH, E2, BMI, duration of infertility, number of previous IVF) in three groups showed no significant difference. Results of Table II show the significant differences between the three groups containing follicle-number, oocyte number, metaphase 2 (M2) oocyte, Germinal vesicle (GV) oocyte, number of transferred embryos and the number of consumed Gonadotropin ampule. To determine the difference between the three groups, we used Post Hoc and Bonferroni test. The number of follicles, GV oocyte and transferred embryo show significant difference between the PCO and poor responder group and the PCO and normal responder only, but there was no difference between Poor and normal responder. The number of the eggs and M2 oocyte has only significant difference between the PCO and Poor responder patients. This difference cannot be seen in each other. But it was significantly different between the three groups together in the number of gonadotropins ampule, and gonadotropin consumed in poor responder group was more than other groups (p= 0.0001). There was no significant difference (p= 0.45, p= 0.843) in LH levels 24 h before and after the injection of the antagonist between the three groups. As well as, the LH ratio and LH change not differ in the three groups (p= 0.077, p= 0.486). According to the Chi-square test, the LH difference percent in the three groups was not significant (p= 0.81). Spearman correlation test was used to assess the correlation of LH parameters with the number of follicle and oocyte and GV oocyte, metaphase 1 (M1) oocyte, M2 oocyte, endometrial thickness. The only significant relationship was seen between LH ratio and M1 (r= 0.31, p= 0.008) and LH change and M1(r= 0.26, p= 0.025). In other words, if the LH (24 hours after administration of the antagonist) is high, that is, the LH suppression by the antagonist is lower and the rate of M1 oocyte was more. According to Mann– Whitney and Fisher test, the correlation of the LH decrease were not significantly associated with GV, M1, M2 oocyte, the number of the follicle, the number of the oocyte, endometrial thickness (Table III). In the other words, the lower levels of LH after the injection had no effect on outcomes.
Table I: Characteristics of patients.

Table II: Treatment outcomes in a patient in three groups.

Table III: Correlation of LH decreases percent with treatment outcome in total group.


Figure 1: Slope or shot in the PCO is significantly higher than the other two groups, except in accordance with the above figure are more tangible between PCO and Normal.
4. Discussion
We evaluate the changes of serum LH in the three groups of patients who were entered in antagonist protocol. In none of the patients in the three groups, the blood level of the LH followed the GnRH antagonist reached below one. There was no reduction in more than 50% of the LH levels in any group following a GnRH antagonist. In our study, there was no significant difference in the LH levels 24 h before and after injection of the antagonist between three groups, as well as on the LH ratio we didn’t find a significant difference. Changes in LH more than 50% (drop more than 50% or even increase) was not significantly associated with GV, M1, M2 oocyte, the number of the follicle, the number of the oocyte, endometrial thickness. Several studies have evaluated the role of LH in antagonist protocol (14). Studies in hypogonadotropic patients using r-FSH established that FSH can induce follicular development in the preovulatory phase, but E2 and androstenedione remain very low, and this advocates that follicular development depends on the action of LH to induce androgen biosynthesis as a substrate for aromatase activity (15). Suitable folliculogenesis and steroid genesis are necessary for successful fertilization and implantation. Therefore, it depends upon a certain threshold level of LH. Although, the amount of LH necessary for standard follicle and oocyte maturation is not known (16), it is likely to be very low since a best steroidogenic response can be prompted when <1% of follicular LH receptors are occupied (16). There is also evidence that excessive level of LH can have an adverse effect on follicular development (17). So, there is a window for LH while the low and high level of LH is a harmful effect in follicular maturation (18). This is an update of a Cochrane review in 2001, and previously updated in 2006 and 2011 (19). In the study by Huirne and coworkers, they evaluated the change of plasma level in LH in five groups with different dosage of GnRH–antagonist, and effect of these changes on IVF outcome. In this study, no pregnancies were observed in relation to either very high or low LH which protects the window hypothesis (20). According to this study, excessive fluctuation in LH levels might disrupt this balance and associate with adverse effect (20). Overall it seems that LH may have a valuable effect through a mechanism that recovers oocyte maturation (20). In the studies by Gomez–Palomares and coworkers (1) and Lisi and coworker, significant improvement in IVF outcome in poor responder patients was found (21). Because of the unexpected and often dramatic inhibition of LH secretion related with the usage of GnRH-anta, there has been a probable need for exogenous LH supplementation (1). The meta analysis from six clinical randomized trials showed that normogomadotropic patients did not benefit from adding LH (22). In the cohort retrospective study in poor responder or AMA (advanced maternal age) did not protect add LH supplementation (22). In the study by Hill and coworkers, adding exogenous LH in AMA patients improved implantation and pregnancy rate (23). In the study by Kol, patients who were hyper sensitive to GnRH– antagonist benefitted from r-LH (12). In the study by Younis and coworkers, r-LH supplementation to rFSH following GnRH-antagonist administration did not seem to significantly increase serum E2 level on the day of Human chorionic gonadotropin administration in AMA patients. This recommends that endogenous serum LH levels following GnRHantagonist beginning are adequate for suitable late follicular ovarian steroidogenesis (14). The novelty of our study is evaluating the LH change in three subgroups including hyper responder (PCOS patients), hypo responds (poor ovarian reserve), and normal responder (donor patients). Limitations of the study:
1. Small sample size may affect results; however, further clinical trial studies with large sample size are needed.
2. Because in some patients we had frozen embryos, there was no possibility of assessing pregnancy.
5. Conclusion
The results showed no difference in the levels of LH and changes of LH in antagonist protocol between three groups, so it is not practical to assess the LH level in ART cycles.
Acknowledgments
In this study there was no financial support.
Conflict of Interest
In this study there was no conflict of interest.
Type of Study: Original Article |
References
1. [1] Shoham Z. The clinical therapeutic window for luteinizing hormone in controlled ovarian stimulation. Fertil Steril 2002; 77: 1170-1177. [DOI:10.1016/S0015-0282(02)03157-6]
2. [2] Hillier SG. Gonadotropic control of ovarian follicular growth and development. Mol Cell Endocrinol 2001; 179: 39-46. [DOI:10.1016/S0303-7207(01)00469-5]
3. [3] Chappel SC, Howles C. Review Reevaluation of the roles of luteinizing hormone and follicle-stimulating hormone in the ovulatory process. Hum Reprod 1991; 6: 1206-1212.
4. [4] Trounson AO, Leeton JF, Wood C, Webb J, Wood J. Pregnancies in humans by fertilization in vitro and embryo transfer in the controlled ovulatory cycle. Science 1981; 212: 681-682. [DOI:10.1126/science.7221557]
5. [5] Schally AV, Arimura A, Baba Y, Nair RM, Matsuo H, Redding TW, et al. Isolation and properties of the FSH and LH-releasing hormone. Biochem Biophys Res Commun 1971; 43: 393-399. [DOI:10.1016/0006-291X(71)90766-2]
6. [6] Hahn DW, McGuire JL, Vale WW, Rivier J. Reproductive/ endocrine and anaphylactoid properties of an LHRHantagonist, ORF 18260 [Ac-DNAL1 (2), 4FDPhe2, D-Trp3, D-Arg6]-GnRH. Life Sci 1985; 37: 505-514. [DOI:10.1016/0024-3205(85)90462-X]
7. [7] Fischer R. Understanding the role of LH: myths and facts. Reprod Biomed Online 2007; 15: 468-477. [DOI:10.1016/S1472-6483(10)60375-6]
8. [8] Fluker MR, Hooper WM, Yuzpe AA. Withholding gonadotropins ("coasting") to minimize the risk of ovarian hyperstimulation during superovulation and in vitro fertilization-embryo transfer cycles. Fertil Steril 1999; 71: 294-301. [DOI:10.1016/S0015-0282(98)00438-5]
9. [9] Al-Inany HG, Youssef MA, Aboulghar M, Broekmans F, Sterrenburg M, Smit J, et al. Gonadotrophin-releasing hormone antagonists for assisted reproductive technology. Cochrane Database Syst Rev 2011; 11: CD001750. [DOI:10.1002/14651858.CD001750.pub3]
10. [10] Kolibianakis EM, Collins J, Tarlatzis BC, Devroey P, Diedrich K, Griesinger G. Among patients treated for IVF with gonadotrophins and GnRH analogues, is the probability of live birth dependent on the type of analogue used? A systematic review and meta-analysis. Hum Reprod Update 2006; 12: 651-671. [DOI:10.1093/humupd/dml038]
11. [11] Olivennes F, Fanchin R, Bouchard P, de Ziegler D, Taieb J, Selva J, et al. The single or dual administration of the gonadotropin-releasing hormone antagonist Cetrorelix in an in vitro fertilization-embryo transfer program. Fertil Steril 1994; 62: 468-476. [DOI:10.1016/S0015-0282(16)56933-7]
12. [12] Kol S. Individualized treatment from theory to practice: the private case of adding LH during GnRH antagonist-based stimulation protocol. Clin Med Insights Reprod Health 2014; 8: 59-64. [DOI:10.4137/CMRH.S17788]
13. [13] Ferraretti AP, Gianaroli L. The bologna criteria for the definition of poor ovarian responders: is there a need for revision? Hum Reprod 2014: 29: 1842-1845. [DOI:10.1093/humrep/deu139]
14. [14] Younis JS, Izhaki I, Ben-Ami M. The effect of LH supplementation to the GnRH antagonist protocol in advanced reproductive ageing women: a prospective randomized controlled study. Clin Endocrinol 2016; 84: 99-106. [DOI:10.1111/cen.12886]
15. [15] Hull M, Corrigan E, Piazzi A, Loumaye E. Recombinant human luteinising hormone: an effective new gonadotropin preparation. Lancet 1994; 344: 334-335. [DOI:10.1016/S0140-6736(94)91373-0]
16. [16] Howles CM. Role of LH and FSH in ovarian function. Mol Cell Endocrinol 2000; 161: 25-30. [DOI:10.1016/S0303-7207(99)00219-1]
17. [17] Hillier SG. Current concepts of the roles of follicle stimulating hormone and luteinizing hormone in folliculogenesis. Hum Reprod 1994; 9: 188-191. [DOI:10.1093/oxfordjournals.humrep.a138480]
18. [18] Balasch J, Fábregues F. Is luteinizing hormone needed for optimal ovulation induction? Curr Opin Obstet Gynecol 2002; 14: 265-274. [DOI:10.1097/00001703-200206000-00004]
19. [19] Al-Inany HG, Youssef MA, Ayeleke RO, Brown J, Lam WS, Broekmans FJ. Gonadotrophin-releasing hormone antagonists for assisted reproductive technology. Cochrane Database Syst Rev 2016; 4: CD001750. [DOI:10.1002/14651858.CD001750.pub4]
20. [20] Huirne JA, van Loenen AC, Schats R, McDonnell J, Hompes PG, Schoemaker J, et al. Dose-finding study of daily GnRH antagonist for the prevention of premature LH surges in IVF/ICSI patients: optimal changes in LH and progesterone for clinical pregnancy. Hum Reprod 2005; 20: 359-367. [DOI:10.1093/humrep/deh601]
21. [21] Loutradis D, Drakakis P, Milingos S, Stefanidis K, Michalas S. Alternative approaches in the management of poor response in controlled ovarian hyperstimulation (COH). Ann NY Acad Sci 2003; 997: 112-119. [DOI:10.1196/annals.1290.013]
22. [22] Griesinger G, Shapiro DB, Kolibianakis EM, Witjes H, Mannaerts BM. No association between endogenous LH and pregnancy in a GnRH antagonist protocol: part II, recombinant FSH. Reprod Biomed Online 2011; 23: 457- 465. [DOI:10.1016/j.rbmo.2011.06.016]
23. [23] Hill MJ, Levens ED, Levy G, Ryan ME, Csokmay JM, DeCherney AH, et al. The use of recombinant luteinizing hormone in patients undergoing assisted reproductive techniques with advanced reproductive age: a systematic review and meta-analysis. Fertil Steril 2012; 97: 1108-1114. [DOI:10.1016/j.fertnstert.2012.01.130]
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





