Wed, Jun 10, 2026
[Archive]
Volume 19, Issue 1 (January 2021)
IJRM 2021, 19(1): 87-96 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Bunsueb S, Lapyuneyong N, Tongpan S, Arun S, Iamsaard S. Chronic stress increases the tyrosine phosphorylation in female reproductive organs: An experimental study. IJRM 2021; 19 (1) :87-96
URL: http://ijrm.ir/article-1-1756-en.html
URL: http://ijrm.ir/article-1-1756-en.html
Sudtida Bunsueb1 

 , Natthapol Lapyuneyong1
, Natthapol Lapyuneyong1 

 , Saranya Tongpan1
, Saranya Tongpan1 

 , Supatcharee Arun1
, Supatcharee Arun1 

 , Sitthichai Iamsaard *2
, Sitthichai Iamsaard *2 




 , Natthapol Lapyuneyong1
, Natthapol Lapyuneyong1 

 , Saranya Tongpan1
, Saranya Tongpan1 

 , Supatcharee Arun1
, Supatcharee Arun1 

 , Sitthichai Iamsaard *2
, Sitthichai Iamsaard *2 


1- Department of Anatomy, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand.
2- Department of Anatomy, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand. Research Institute for Human High Performance and Health Promotion (HHP & HP), Khon Kaen, Thailand. ,sittia@kku.ac.th
2- Department of Anatomy, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand. Research Institute for Human High Performance and Health Promotion (HHP & HP), Khon Kaen, Thailand. ,
Full-Text [PDF 15226 kb]
(1268 Downloads)
| Abstract (HTML) (2452 Views)
Full-Text: (547 Views)
- Introduction
Recently female infertility has been reported as the most common problem affecting women of reproductive ages (1). A major cause of female infertility is anovulation, the mechanism of which is unexplained (2). With the present-day modern lifestyle, stress condition is unavoidable for working women who are frequently exposed to many adverse stressors (3). In addition, chronic stress (CS) is known to affect normal physiological ovulation and implantation resulting in no pregnancy (3-5). In experimental animals, stress can significantly increase the corticosterone level, a common marker to determine actual stress conditions (6). Indeed, increased cortisol levels involve the overexpression of steroidogenic acute regulatory (StAR) protein and heat shock protein 70 (Hsp70) in the stress adrenal gland. Various proteins are known to be responsible in regulating the normal functions in the ovary and female reproductive tracts such as oviduct and uterus (7). Additionally, Zangeneh and coworkers reported that cold stress in animal models can promote both ovarian morphological alterations and sex hormonal changes (8). Therefore, many proteins in reproductive organs of stress females may be changed from normal physiology. Tyrosine phosphorylation is a post-transcriptional process and plays essential roles in the regulation of cell proliferation and differentiation (9). In the male reproductive system, tyrosine-phosphorylated (TyrPho) proteins have been localized in the Sertoli cells, seminiferous epithelium, Leydig cells (10), epididymis epithelial cells (11), and seminal vesicle tissue and fluid (12). Previous reports have demonstrated that the patterns of expression of TyrPho proteins in male reproductive tissues were changed by inductions of drugs or other chemical agents (12-15). Those altered expressions of TyrPho proteins are associated with male infertility including induction with restraint stress (16). In females, a TyrPho protein was localized in pig-ovulated oocyte, assumed to be involved in the formation of chromatin in metaphase. Moreover, Richard and coworkers also showed the presence of TyrPho proteins in the uterus of ovariectomized mice (17). Indeed, a previous report has shown that estrogen can affect the phosphorylation protein profiles at different uterine cycles of the oviparous and viviparous lizards (18). Hence, we have already localized the TyrPho proteins in ovarian, oviductal, and uterine tissues of adult rats (19). However, the changes in Tyrpho proteins in stress reproductive organs have never been reported.
This study; aimed to investigate the alterations of TyrPho protein expressions in the ovary, oviduct, and uterus of adult rats induced with chronic restraint stress.
This study; aimed to investigate the alterations of TyrPho protein expressions in the ovary, oviduct, and uterus of adult rats induced with chronic restraint stress.
- Materials and Methods
- 1. Animal and experimental designs
In this experimental study, 16 adult female Sprague-Dawley rats (5 wk: 220-250 gr) were purchased and kept in plastic cages under the experiment room condition (12 hr light/dark cycle, controlled temperature 23 ± 2ºC, humidity 30-60% RH, sound < 85 decibels, and light intensity 350-400 lux) at the Laboratory Animal Unit, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand. The rats were fed commercial pellet food and water ad libitum. After a week of acclimatization, all rats were randomly divided into control and CS groups (n = 8/each). In the control group, the rats were not exposed to any stressor. However, in the CS group, the animals were immobilized within a restraint cage and then forced individually to swim in a plastic tank containing cold water. This was repeated for consecutive 60 days. At the end of the experiment, all animals were anesthetized and euthanatized before the collection of reproductive organs for weighing.
- 2. Stress induction
The rats in the CS group were induced to be stressed by immobilization as described in Arun and coworkers (14) followed by swimming as described previously (17). In brief, the animals were immobilized inside the restraint cage (8 cm long × 6 cm diameter × 14 high) which was developed and modified by Assistant Professor Dr. Supatcharee Arun. Every day, this immobilization was performed for 4 hr continuously. Then, the animals were individually forced to swim in a tank (depth 15.5 cm) of cold water (controlled temperature - approximately 4ºC) for 15 min/day.
- 3. Total protein extraction
After sample collections, the ovary, oviduct, or uterus of all groups kept at -80ºC were mixed with 1X radioimmunoprecipitation assay (RIPA) buffer (10X: Cell Signaling Technology Inc., USA) containing cocktail protease inhibitors (Sigma-Aldrich, Inc., USA) to extract total proteins in such reproductive samples. Subsequently, individual tissue was homogenated and sonicated on ice. The homogenized ovary, oviduct, or uterus tissue was further centrifuged at 12,000 rpm at 4ºC for 10 min to separate the total protein supernatant from tissue pellets. Next, the supernatant of each sample was collected to measure the total protein concentration (µg/µl) at the absorbance with a wavelength of 280 nm using NANO drop ND-100 Spectrophotometer (NanoDrop ND-1000 Spectrophotometer V3.5 User’s Manual, Nano Drop Technologies Inc., USA)
- 4. Western blot analysis
Total proteins (100 ug) of ovary, oviduct, or uterus were loaded and separated on 10% sodium dodecyl sulfate (SDS) polyacrylamide gel by electrophoresis (PAGE). The separated proteins were stained with Coomassie brilliant blue to observe protein profiles between the control and CS groups before transferring onto the nitrocellulose membrane. Subsequently, all membranes were incubated with 5% skim milk in 0.1% TBST (0.1% Tween-20, TBS, pH 7.4) for 1 hr to block. All membranes of the ovary, oviduct, and uterus protein lysates were incubated with primary antibody (mouse anti-phosphotyrosine; 1:2000 dilution, catalog no. 05-321, Merck Millipore Co., USA) at 4ºC overnight followed by washing and incubating with secondary antibody (anti-mouse) (1:2000 dilution, TM catalog no. G-21234, Invitrogen, USA) in 0.1% TBST for 1 hr at room temperature. To confirm the actual stress conditions in the animal model, we have used the adrenal gland proteins to observe the increase of Hsp 70 and StAR which are responsible for cortisol synthesis. Adrenal protein-membrane was incubated with anti-StAR (1:2000 dilution, catalog no. sc-112333, Santa Cruz., USA) or anti-heat shock protein 70 (Hsp70) (1:4000 dilution, catalog no. 05-412, Abcam, USA) followed by washing the excess primary antibody with 0.1% TBST. Then, all membranes were incubated with secondary antibody (anti-mouse) (1:2000 dilution, TM catalog no. G-21234, Invitrogen, USA) in 0.1% TBST for 1 hr at room temperature.
To confirm the equal total protein amount in all sample loading, the beta-actin (probed by anti-actin, 1:2000 dilution, catalog no. Sc-69879, Santa Cruz., USA) was used as an internal control. Next, all membranes were washed with 0.1% TBST before the detection of antigen-antibody complexes for each antibody of all tissues. For negative or positive control of TyrPho protein detections, the epidermal growth factor (EGF; Millipore Co., USA) and bovine serum albumin (BSA; Millipore Co., USA) were used as positive and negative, respectively. To detect the target proteins (StAR, Hsp70, actin, and TyrPho proteins), the enhanced chemiluminescence (ECL) substrate reagent kit (GE Healthcare Life Science, USA) was used to incubate before visualization under gel documentation 4 (ImageQuant 400, GH Healthcare, USA). For quantitative analysis, the relative intensity of TyrPho protein expression was analyzed using the ImageJ program (version 1.49).
To confirm the equal total protein amount in all sample loading, the beta-actin (probed by anti-actin, 1:2000 dilution, catalog no. Sc-69879, Santa Cruz., USA) was used as an internal control. Next, all membranes were washed with 0.1% TBST before the detection of antigen-antibody complexes for each antibody of all tissues. For negative or positive control of TyrPho protein detections, the epidermal growth factor (EGF; Millipore Co., USA) and bovine serum albumin (BSA; Millipore Co., USA) were used as positive and negative, respectively. To detect the target proteins (StAR, Hsp70, actin, and TyrPho proteins), the enhanced chemiluminescence (ECL) substrate reagent kit (GE Healthcare Life Science, USA) was used to incubate before visualization under gel documentation 4 (ImageQuant 400, GH Healthcare, USA). For quantitative analysis, the relative intensity of TyrPho protein expression was analyzed using the ImageJ program (version 1.49).
- 5. Ethical considerations
This study was approved by the Animal Ethics Committee Research of the Faculty of Medicine, Khon Kaen University, based on the Ethics of Animal Experimentation of National Research Council of Thailand (Record No. AEMDKKU 011/2019). This experimental study has followed all animal ethics.
- 6. Statistical analysis
Data were analyzed using the statistical package for the social sciences, version 19.0, SPSS Inc, Armonk, New York, USA (SPSS) and are expressed as means ± standard deviation. In addition, to compare the difference between groups, an independent t-test was used to analyze the normally distributed data and the Mann-Whitney U-test for the not normally distributed data. The P-value < 0.05 was considered as a significant difference between the control and CS groups.
To confirm the actual stress condition of female rats, the results showed that the expression of StAR and Hsp70 in adrenal lysates of CS group were higher than those of the control group using the β-actin as an internal control (Figure 1). This result indicated the increased levels of cortisol in the serum of stress animals.
- 2. Morphology and weight of female reproductive organs
Figure 2 shows the morphology and weights of ovary, oviduct, and uterus compared between the control and CS rats, respectively. The ovary of the CS group was observed to be larger than that of the control (Figure 2A, left panel). However, the absolute weight of ovary in both groups was not significantly different (Figure 2A, right panel). In addition, the morphology and absolute weight of oviduct had no significant difference between the two groups (Figure 2B). Moreover, it was found that the uterus of the CS group was larger than that of the control which corroborated with its absolute weight as shown in Figure 2C.
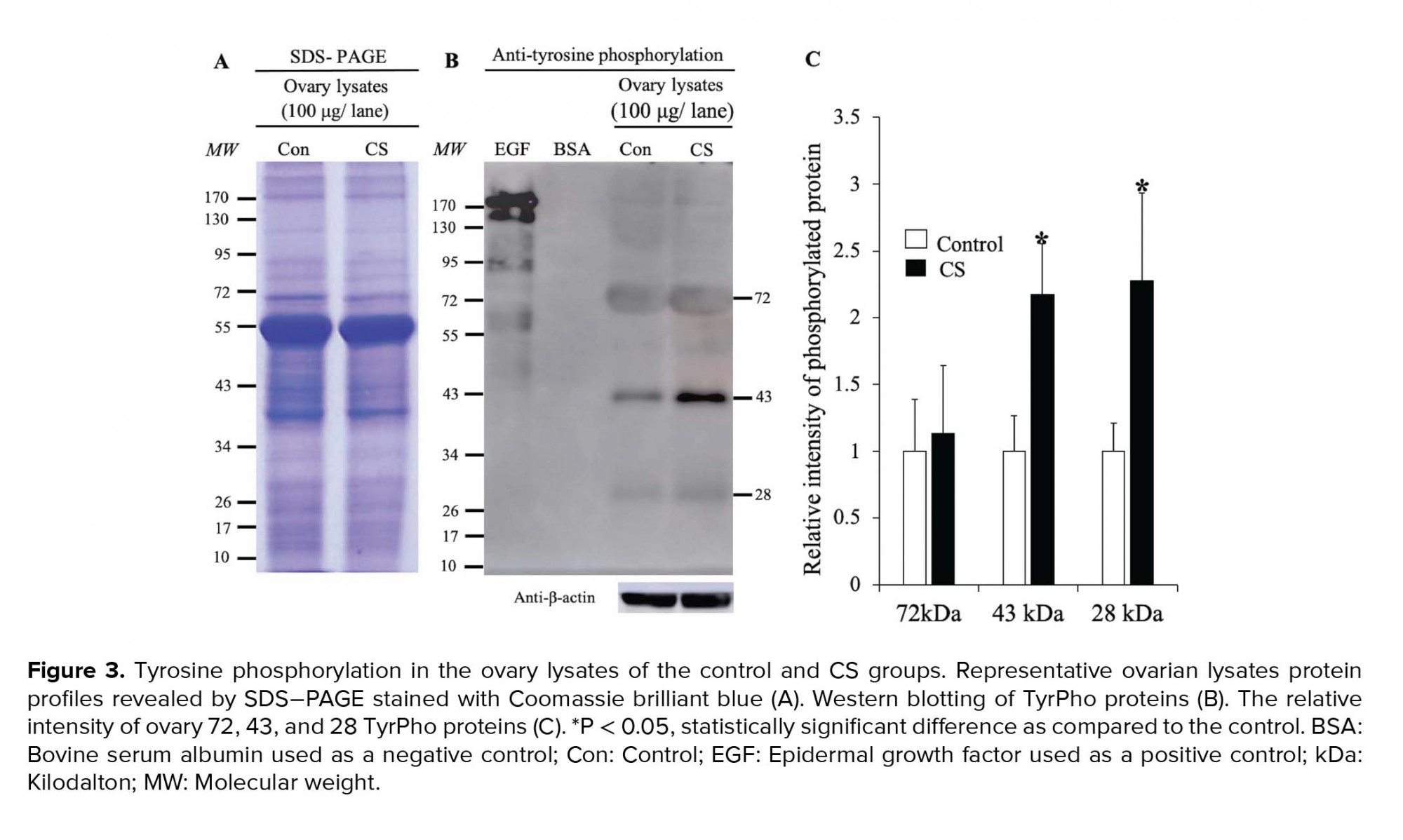
The expression of TyrPho proteins in the ovary, oviduct, and uterus of both groups are shown in figures 3, 4, and 5 respectively. The equal protein loading in control and CS groups. The equal protein loading in the two groups was confirmed by SDS-PAGE profiles and beta-actin used as an internal control (Figures 3-5). In Figure 3A, the results showed that the ovary protein lysate profiles in both groups were similar. The 72, 43, and 28 kDas Tyrpho proteins in ovary were expressed in both groups (Figure 3B). However, it was found that only two major TyrPho proteins (43 and 28 kDas) were significantly overexpressed in the CS group compared to those in the control (Figures 3B and 3C).
In addition, the expression of TyrPho proteins in the oviduct were also investigated as shown in Figure 4. The protein profiles in the oviduct of both groups showed many protein bands and look similar (Figure 4A). For western blotting (Figure 4B), the results demonstrated that the TyrPho proteins were also present in oviduct consisting of three major bands (170, 55, and 43 kDas, respectively). Significantly, the intensity of a 43 kDa oviductal TyrPho protein in the CS group was higher than that of the expression of the control group as shown in Figure 4C.
Moreover, the protein profiles in the uterus of both groups showed many protein bands (Figure 5A). In western blotting, the results indicated that the TyrPho proteins were present in the uterus with three major bands including 55, 54, and 43 kDas, respectively, as shown in Figure 5B. The intensities of 55, 54, and 43 kDas were significantly increased in only the CS group compared with the control (P < 0.05, Figure 5C).




4. Discussion
In addition, the expression of TyrPho proteins in the oviduct were also investigated as shown in Figure 4. The protein profiles in the oviduct of both groups showed many protein bands and look similar (Figure 4A). For western blotting (Figure 4B), the results demonstrated that the TyrPho proteins were also present in oviduct consisting of three major bands (170, 55, and 43 kDas, respectively). Significantly, the intensity of a 43 kDa oviductal TyrPho protein in the CS group was higher than that of the expression of the control group as shown in Figure 4C.
Moreover, the protein profiles in the uterus of both groups showed many protein bands (Figure 5A). In western blotting, the results indicated that the TyrPho proteins were present in the uterus with three major bands including 55, 54, and 43 kDas, respectively, as shown in Figure 5B. The intensities of 55, 54, and 43 kDas were significantly increased in only the CS group compared with the control (P < 0.05, Figure 5C).





4. Discussion
Our results showed a similarity of the morphology and weights of the ovary and oviduct between the control and CS groups. Obviously, the uterine weight of the CS group was significantly increased as compared to the control. Additionally, the expressions of TyrPho proteins in the ovary (72, 43, and 28 kDas), oviduct (170, 55, and 43 kDas), and uterus (55, 54, and 43 kDas) were significantly increased in the CS group. It is known that many stressors including social media, economics, physical, physiological, and psychological factors can affect female reproductive organs, particular ovarian function (20). Previous studies have shown that CS affected the ovarian functions such as ovarian steroid genesis, oocyte maturation, and ovulation (21, 22). Although this study did not show the significantly increased levels of cortisol hormone to determine the actual CS condition in the animal model because the blood serum collected were not enough to be assayed, the high expressions of StAR protein and Hsp70 in the adrenal gland of stress rats could also confirm the stress physiological condition as compared to that of the control (Figure 1). In this study, we found that the profiles of ovarian TyrPho proteins including 72, 43, and 28 kDas of the control and CS groups were not different. CS increased significantly the expression of 43 and 28 kDas as compared to the control rats. It was assumed that such TyrPho proteins may be overexpressed for the compensation to maintain normal ovarian functions such as oogenesis and steroidogenesis, including platelet-derived growth factors, nerve growth factor, angiogenic growth factors like VEGF and TGFbeta1, and transforming growth factors, respectively. Previous proteomic studies have reported that developing ovarian follicles contain 1,401 identified proteins (23).
In addition, the increased protein expressions in growing ovarian follicles are corroborated with the increase of differential global gene expressions in granulosa cells of primordial and primary follicles as reported by Ernst and coworkers (24). It is possible that the ovarian TyrPho proteins observed from this study including their genes could be included in protein profiles from ovarian proteomics as previously described (23). Indeed, the significant overexpression of 43 and 28 kDas TyrPho proteins in CS animals from a recent study may also be involved in stimulating follicular development since stress is a factor that causes the inhibition of follicular growth (25, 26) and anovulation (27). Similar to protein profiles in the ovary, the proteomics of oviduct and uterus from the individual estrous cycle of mammalian animals have been also documented (27-30). Therefore, the proteomic profiles previously reported in the female reproductive tract may include TyrPho proteins that are investigated in our recent study. Particularly, the proteins expressed in oviducts are shown to be essential for early mammalian fertilization (30).
As shown in Figures 4 and 5, CS induced the overexpression of 55, 54, and 43 kDas TyrPho proteins in both the oviduct and uterus when compared to those proteins in non-stress rats. Although the protein profiles in oviduct and uterus between the control and stress groups were relatively similar, some protein expressions were significantly higher in only stress rats. It could also be explained that those proteins are involved to maintain the normal reproductive tract physiology. Such a mechanism is also similar to the changes in ovarian TyrPho proteins in stress conditions. To understand more functions of such increased TyrPho proteins in stress reproductive organs, the proteomics and characterization of those proteins need to be further elucidated to be used as some biomarkers of stress in women of reproductive age.
5. Conclusion
In addition, the increased protein expressions in growing ovarian follicles are corroborated with the increase of differential global gene expressions in granulosa cells of primordial and primary follicles as reported by Ernst and coworkers (24). It is possible that the ovarian TyrPho proteins observed from this study including their genes could be included in protein profiles from ovarian proteomics as previously described (23). Indeed, the significant overexpression of 43 and 28 kDas TyrPho proteins in CS animals from a recent study may also be involved in stimulating follicular development since stress is a factor that causes the inhibition of follicular growth (25, 26) and anovulation (27). Similar to protein profiles in the ovary, the proteomics of oviduct and uterus from the individual estrous cycle of mammalian animals have been also documented (27-30). Therefore, the proteomic profiles previously reported in the female reproductive tract may include TyrPho proteins that are investigated in our recent study. Particularly, the proteins expressed in oviducts are shown to be essential for early mammalian fertilization (30).
As shown in Figures 4 and 5, CS induced the overexpression of 55, 54, and 43 kDas TyrPho proteins in both the oviduct and uterus when compared to those proteins in non-stress rats. Although the protein profiles in oviduct and uterus between the control and stress groups were relatively similar, some protein expressions were significantly higher in only stress rats. It could also be explained that those proteins are involved to maintain the normal reproductive tract physiology. Such a mechanism is also similar to the changes in ovarian TyrPho proteins in stress conditions. To understand more functions of such increased TyrPho proteins in stress reproductive organs, the proteomics and characterization of those proteins need to be further elucidated to be used as some biomarkers of stress in women of reproductive age.
5. Conclusion
A recent study has demonstrated an increase in the TyrPho proteins in the ovary, oviduct, and uterus under CS conditions in adult female rats. This change of such protein was assumed to turn over in normal regulation of fertile female physiology.
Acknowledgements
This study was funded by the Invitation Research (IN. 62336) Grant from the Faculty of Medicine, Khon Kaen University, to SB and SI and by the Research Institute for Human High Performance and Health Promotion (HHP&HP), Khon Kaen, Thailand.
Conflict of Interest
The authors declare no conflict of interest in the present study.
Acknowledgements
This study was funded by the Invitation Research (IN. 62336) Grant from the Faculty of Medicine, Khon Kaen University, to SB and SI and by the Research Institute for Human High Performance and Health Promotion (HHP&HP), Khon Kaen, Thailand.
Conflict of Interest
The authors declare no conflict of interest in the present study.
Type of Study: Original Article |
Subject:
Reproductive Biology
References
1. Vander Borghtb M, Wyns Ch. Fertility and infertility: Definition and epidemiology. Clin Biochem 2018; 62: 2-10. [DOI:10.1016/j.clinbiochem.2018.03.012] [PMID]
2. Barbieri RL. Female infertility. In: Strauss J, Barbieri R. Yen and Jaffe's reproductive endocrinology. 8th Ed. Netherland: Elsevier; 2019. 556-581. [DOI:10.1016/B978-0-323-47912-7.00022-6]
3. Pandey AK, Gupta A, Tiwari M, Prasad Sh, Pandey AN, Yadav PK, et al. Impact of stress on female reproductive health disorders: Possible beneficial effects of shatavari (Asparagus racemosus). Biomed Pharmacother 2018; 103: 46-49. [DOI:10.1016/j.biopha.2018.04.003] [PMID]
4. Farkas J, Rigó A, Demetrovics Z. Psychological aspects of the polycystic ovary syndrome. Gynecol Endocrinol 2013; 30: 95-99. [DOI:10.3109/09513590.2013.852530] [PMID]
5. Rooney KL, Domar AD. The relationship between stress and infertility. Dialogues Clin Neurosci 2018; 20: 41-47. [DOI:10.31887/DCNS.2018.20.1/klrooney] [PMCID]
6. Kala M, Nivsarkar M. Role of cortisol and superoxide dismutase in psychological stress induced anovulation. Gen Comp Endocrinol 2016; 225: 117-124. [DOI:10.1016/j.ygcen.2015.09.010] [PMID]
7. Ouni E, Vertommen D, Amorim ChA. The human ovary and future of fertility assessment in the post-genome Era. Int J Mol Sci 2019; 20: 4209. 1-14. [DOI:10.3390/ijms20174209] [PMID] [PMCID]
8. Zafari Zangeneh F, Abdollahi A, Tavassoli P, Naghizadeh MM. The effect of cold stress on polycystic ovary syndrome in rat: before and during modeling. Arch Gynecol Obstet 2011; 284: 651-657. [DOI:10.1007/s00404-010-1711-y] [PMID]
9. Cerulli RA, Kritzer JA. Phosphotyrosine isosteres: Past, present and future. Org Biomol Chem 2020; 18: 583-605. [DOI:10.1039/C9OB01998G] [PMID] [PMCID]
10. Chaichun A, Arun S, Burawat J, Kanla P, Iamsaard S. Localization and identification of tyrosine phosphorylated proteins in adult Sprague-Dawley rat testis. Int J Morphol 2017; 35: 1322-1327. [DOI:10.4067/S0717-95022017000401322]
11. Sawatpanich T, Arun S, Tongpan S, Chaichun A, Sampannang A, Sukhorum W, et al. Localization and changes of tyrosine phosphorylated proteins and ß actin in epididymis of rats treated with valproic acid. Int J Morphol 2018; 36: 835-840. [DOI:10.4067/S0717-95022018000300835]
12. Tongpan S, Sukhorum W, Arun S, Sawatphanich T, Iamsaard S. Valproic acid changes the expression of tyrosinephosphorylated proteins in rat seminal vesicle. Andrologia 2019; 51: e13303. [DOI:10.1111/and.13303] [PMID]
13. Sukhorum W, Iamsaard S. Changes in testicular function proteins and sperm acrosome status in rats treated with valproic acid. Reprod Fertil Dev 2016; 29: 1585-1592. [DOI:10.1071/RD16205] [PMID]
14. Arun S, Burawat J, Yannasithinon S, Sukhorum W, Limpongsa A, Iamsaard S. Phyllanthus emblica leaf extract ameliorates testicular damage in rats with chronic stress. J Zhejiang Univ Sci 2018; 19: 948-959. [DOI:10.1631/jzus.B1800362] [PMID] [PMCID]
15. Sampannang A, Arun S, Burawat J, Sukhorum W, Iamsaard S. Expression of testicular phosphorylated proteins in types 1 and 2 diabetes mellitus in mice: An experimental study. Int J Reprod BioMed 2019; 17: 567-576. [DOI:10.18502/ijrm.v17i8.4822] [PMID] [PMCID]
16. Arun S, Burawat J, Sukhorum W, Sampannang A, Uabundit N, Iamsaard S. Changes of testicular phosphorylated proteins in response to restraint stress in male rats. J Zhejiang Univ Sci 2016; 17: 21-29. [DOI:10.1631/jzus.B1500174] [PMID] [PMCID]
17. Richards RG, Diaugustine RP, Petrusz P, Clarck GC, Sebastian J. Physiology Estradiol stimulates tyrosine phosphorylation of the insulin-like growth factor-1 receptor and insulin receptor substrate-I in the uterus. Proc Natl Acad Sci USA 1996; 93: 12002-12007. [DOI:10.1073/pnas.93.21.12002] [PMID] [PMCID]
18. homson M, Herbert JF, Thompson MB. Tyrosine phosphorylated proteins in the reproductive tract of the viviparous lizard Eulamprus tympanum and the oviparous lizard Lampropholis guichenoti. Comp Biochem Physiol B Biochem Mol Biol 2006; 144: 382-386. [DOI:10.1016/j.cbpb.2006.04.005] [PMID]
19. Bunsueb S. Presence and changes of tyrosine phosphorylated protein expression in female reproductive organs of chronic stress and polycystic ovary syndrome (PCOS) rats. [Master's thesis] Thailand: Faculty of Medicine, Khon Kaen University; 2019.
20. Petraglia F, Serour GI, Chapron Ch. The changing prevalence of infertility. Int J Gynecol Obstet 2013; 123: S4-S8. [DOI:10.1016/j.ijgo.2013.09.005] [PMID]
21. Sharma RK, Agarwal A. Role of reactive oxygen species in gynecologic diseases. Reprod Med Biol 2004; 3: 177-199. [DOI:10.1111/j.1447-0578.2004.00068.x] [PMID] [PMCID]
22. Breen KM, Oakley AE, Pytiak AV, Tilbrook AJ, Wagenmaker ER, Karsch FJ. Does cortisol acting via the type II glucocorticoid receptor mediate suppression of pulsatile luteinizing hormone secretion in response to psychosocial stress? Endocrinology 2007; 148: 1882-1890. [DOI:10.1210/en.2006-0973] [PMID]
23. Anastácio A, Rodriguez-Wallberg KA, Chardonnet S, Pionneau C, Fédérici Ch, Santos TA, et al. Protein profile of mouse ovarian follicles grown in vitro. Mol Hum Reprod 2017; 23: 827-841. [DOI:10.1093/molehr/gax056] [PMID] [PMCID]
24. Ernst EH, Franks S, Hardy K, Villesen P, Lykke-Hartmann K. Granulosa cells from human primordial and primary follicles show differential global gene expression profiles. Hum Reprod 2018; 33: 666-679. [DOI:10.1093/humrep/dey011] [PMID]
25. Bhat MS, Yajurvedi HN. Stress induced alterations in pre-pubertal ovarian follicular development in rat. Journal of Stress Physiology & Biochemistry 2011; 7: 51-68.
26. Wu LM, Liu YSh, Tong XH, Shen N, Jin RT, Han H, et al. Inhibition of follicular development induced by chronic unpredictable stress is associated with growth and differentiation factor 9 and gonadotropin in mice. Biol Reprod 2012; 86: 1-7. [DOI:10.1095/biolreprod.111.093468]
27. La Y, Tang J, Guo X, Zhang L, Gan Sh, Zhang X, et al. Proteomic analysis of sheep uterus reveals its role in prolificacy. J Proteomics 2020; 210: 103526. [DOI:10.1016/j.jprot.2019.103526] [PMID]
28. Maloney ShE, Khan FA, Chenier TS, Diel de Amorim M, Anthony Hayes M, Scholtz EL. A comparison of the uterine proteome of mares in oestrus and dioestrus. Reprod Domest Anim 2019; 54: 473-479. [DOI:10.1111/rda.13375] [PMID]
29. Soleilhavoup C, Riou C, Tsikis G, Labas V, Harichaux G, Kohnke P, et al. Proteomes of the female genital tract during the oestrous cycle. Mol Cell Proteomic 2016; 15: 93-108. [DOI:10.1074/mcp.M115.052332] [PMID] [PMCID]
30. Coy P, García-Vázquez FA, Visconti PE, Avilés M. Roles of the oviduct in mammalian fertilization. Reproduction 2012; 144: 649-660. [DOI:10.1530/REP-12-0279] [PMID] [PMCID]
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





