Sat, Apr 11, 2026
[Archive]
Volume 23, Issue 9 (September 2025)
IJRM 2025, 23(9): 701-712 |
Back to browse issues page
Ethics code: IR.MAZUMS.IMAMHOSPITAL.REC.1401.038
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Peivandi S, Jafarifard P, Zamaniyan M, Yazdani-charati J. Impact of intrauterine infusion of human chorionic gonadotropin and hyaluronic acid enriched medium on pregnancy outcomes infrozen-thawed cycles with recurrent implantation failure: An RCT. IJRM 2025; 23 (9) :701-712
URL: http://ijrm.ir/article-1-3428-en.html
URL: http://ijrm.ir/article-1-3428-en.html
1- Department of Obstetrics and Gynecology, IVF Ward, Faculty of Medicine, Sexual and Reproductive Health Research Center, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran. , dr_peyvandi@yahoo.com; speivandi@mazums.ac.ir
2- Department of Obstetrics and Gynecology, Faculty of Medicine, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran.
3- Department of Obstetrics and Gynecology, IVF Ward, Faculty of Medicine, Sexual and Reproductive Health Research Center, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran. & Diabetes Research Center, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran.
4- Department of Biostatistics, Faculty of Health, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran.
2- Department of Obstetrics and Gynecology, Faculty of Medicine, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran.
3- Department of Obstetrics and Gynecology, IVF Ward, Faculty of Medicine, Sexual and Reproductive Health Research Center, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran. & Diabetes Research Center, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran.
4- Department of Biostatistics, Faculty of Health, Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran.
Keywords: Embryo transfer, Hyaluronic acid, Chorionic gonadotropin, Embryo implantation, Pregnancy outcome.
Full-Text [PDF 460 kb]
(865 Downloads)
| Abstract (HTML) (641 Views)
Full-Text: (111 Views)
1. Introduction
Recurrent implantation failure (RIF) is generally characterized by the inability to achieve a clinical pregnancy despite transferring at least 4 morphologically high-quality embryos in a minimum of 3 fresh or frozen-thawed embryo transfer (FET) cycles, is a significant challenge in assisted reproductive technology (1, 2). Successful embryo implantation is a complex process that requires synchronized development and interaction between the embryo and a receptive endometrium. Factors contributing to RIF include embryonic, endometrial, genetic, and immunological abnormalities (2, 3).
Various strategies have been explored to improve implantation and clinical pregnancy rate in RIF cases, including intrauterine infusion of human chorionic gonadotropin (hCG) and the use of embryo transfer media supplemented with hyaluronic acid (HA), also known as "embryo glue". Several studies have shown that intrauterine HCG administration prior to embryo transfer can enhance the expression of endometrial receptivity markers, such as leukemia inhibitory factor thereby creating a more favorable environment for implantation. HCG has been shown to enhance endometrial receptivity by promoting angiogenesis, increasing uterine blood flow, and modulating cytokine production, all of which are crucial for successful embryo implantation (1, 4, 5).
HA, a major component of the extracellular matrix, has been hypothesized to improve embryo attachment and implantation by mimicking the natural embryo-endometrial interaction (6, 7). However, some studies have reported improved implantation and pregnancy rates with the use of HA-enriched embryo transfer medium in this population (8-11).
Based on existing evidence, the effectiveness of each approach individually in improving endometrial receptivity and increasing implantation and pregnancy rates is demonstrated. While there is ample evidence supporting the individual use of these methods, we acknowledge the current limitation in the availability of comprehensive studies specifically investigating their combined effect. Our hypothesis is based on the potential synergistic impact of these 2 approaches in enhancing embryo-endometrium interaction and ultimately improving pregnancy outcomes. Taken together, the paracrine roles of hCG are essential in processes such as decidualization, implantation, vascularization, and tissue remodeling. HA is a key component of the extracellular matrix and has been shown to play a crucial role. However, the efficacy of these interventions, especially when used in combination, in improving clinical pregnancy rates in RIF cases undergoing FET cycles, remains inconclusive.
Therefore, this study aims to investigate the effect of intrauterine infusion of hCG and the use of embryo transfer medium containing HA on clinical pregnancy rate in women with RIF undergoing FET cycles.
2. Materials and Methods
2.1. Study design and participants
This single-blind, randomized controlled clinical trial study was conducted at the Infertility and IVF Center of Imam Khomeini hospital in Sari in Iran from December 2022-April 2024. Participants were selected using the block randomization method. The inclusion criteria were: women aged 20-40 yr, with a history of at least 3 unsuccessful embryo transfer cycles and or failed transfer of 4 good quality (grade A or B) embryos, those who were undergoing FET cycles with hormone preparation using estrogen and progesterone, and women who have at least one grade A or B embryo on the day of transfer. The 3rd-day embryo quality was evaluated based on Istanbul Consensus Workshop criteria; grade A embryos were defined as embryos with 6-10 cells (< 10% fragmentation, blastomeres of equal size, and without multinucleation), and grade B was defined as 6-10 cells (10-25% fragmentation, blastomeres of equal size in most cells, and no multinucleation). Embryo quality on the 5th day was evaluated using the Gardner and Schoolcraft scoring system. Expansion grade 1-6, internal cell mass morphology (grade A-C), and trophectoderm morphology (grade A-C) were used to grade embryos on the 5th day. Good-quality embryos on the 5th day were defined as ≥ 3BB (AA, AB, BA, and BB). Exclusion criteria included: use of donated egg, donated embryo, or surrogacy; presence of uterine abnormalities, severe endometriosis and endometrioma, hydrosalpinx in hysterosalpingography or sonography, history of uncontrolled endocrine diseases like diabetes, hypothyroidism, or hyperthyroidism; and male factor infertility with azoospermia or difficult embryo transfer requiring cervical manipulation or anesthesia.
2.2. Sample size
The sample size for this study was calculated based on the results reported in the study by Hafezi et al. (12), assuming a live birth rate of 16.6% in the intervention group (P1) and 41.6% in the control group (P2), with a significance level (α) of 0.05 and a power (1-β) of 90%; the required sample size was determined using the following formula, which yielded 66 participants per group (n = 132). Accounting for a 15% dropout rate, we recruited 79 participants per group (n = 158).
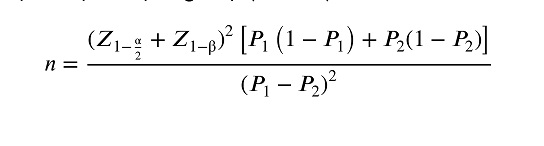
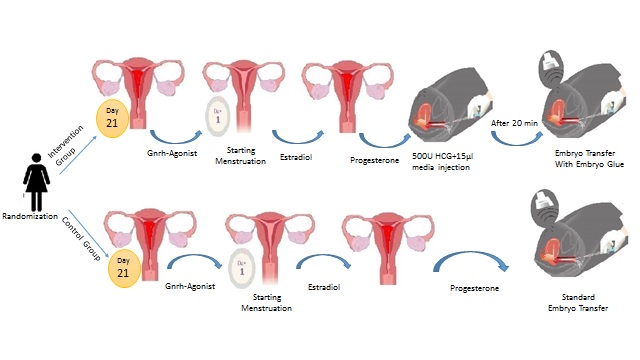
2.3. Endometrial preparation and randomization
In all eligible participants, endometrial preparation was performed artificially by use of estrogen and progesterone hormones. Initially, a gonadotropin releasing hormone agonist was started in the mid-luteal phase of the previous menstrual cycle for all cases and continued through the next cycle. Subsequently, the endometrial growth was supported from the time of menstruation by daily administration of 2-6 mg of oral estradiol valerate (Estraval®, Aburaihan, Tehran, Iran). After 10 days of estradiol administration, vaginal sonography was performed. If the endometrial thickness reached ≥ 8 mm, estradiol valerate was continued at the same dose and 50-100 mg of progesterone (Progestin®, Aburaihan Pharmaceutical Co., Tehran, Iran) was injected intramuscularly for 3-5 days. If the optimal endometrial thickness was not reached, the dose of estradiol was increased up to 8 mg per day, and monitoring of endometrial thickness was continued for another 3 days.
Participants were grouped using a block random allocation by (Random Allocation Software 2.0) and using their admission row number. The participants who met the inclusion criteria were randomly divided into 2 groups: the intervention group and the control group in a 1:1 ratio. 79 women were assigned to each group. The intervention group received an intrauterine infusion of 500 IU of hCG (Amp Choriomon®, Pharmed Daru) diluted with 15 µl of embryo culture medium (SAGE, 1-step culture medium, Origio) by the embryo transfer catheter guided by abdominal ultrasound 20 min before embryo transfer. The thawed embryos were placed in the HA-enriched transfer media (Embryo Glue, Vitrolife) and were transferred into the uterus by the embryo transfer catheter guided by abdominal ultrasound. In the control group, the embryos were transferred into the culture medium without the HA and then transferred into the uterine cavity by transfer catheter guided by abdominal ultrasound. The present study is a single-blind randomized clinical trials in which the physician and participant were aware of the type of intervention, but the embryologist who performed the thawing and transfer and the person collecting the data and the result of the pregnancy outcome for analysis were unaware of the type of intervention. Moreover, double blinding was not possible due to the method of intervention.
2.4. Outcome assessments
The primary outcome was clinical pregnancy rate, defined as the presence of a fetal heartbeat assessed by transvaginal ultrasound at 6 wk after embryo transfer. Secondary outcomes included rates of chemical pregnancy, implantation, miscarriage, and ongoing pregnancy. The chemical pregnancy rate is defined as the positive pregnancy test 3-4 wk after intervention. The implantation rate was evaluated 5 wk after intervention and was calculated as the total number of intrauterine gestational sacs divided by the total number of transferred embryos, multiplied by 100. The abortion rate was evaluated 18 wk after intervention and was defined as the number of clinical pregnancy losses before the 20th wk of pregnancy divided by the total number of clinical pregnancies. Ongoing pregnancy was defined as either a birth at 38 wk after intervention or a pregnancy continued after the 20th wk of gestation.
2.5. Ethical Considerations
This study protocol was approved by the Ethical Committee of Maandaran University of Medical Sciences, Sari, Iran (Code: IR.MAZUMS.IMAMHOSPITAL.REC.1401.038) and the Institutional Review Board of Iran (Code: IR19464). The study was also registered in the Iranian Registry of Clinical Trials with the identification number IRCT20221126056614N1, last updated on July 30, 2024. All participants provided written informed consent prior to enrollment in the study. The confidentiality of participants' personal information was strictly maintained throughout the study. Participants were free to withdraw from the study at any time without any impact on their ongoing medical care. The study procedures were in accordance with the Declaration of Helsinki.
2.6. Statistical Analysis
All statistical analyses were performed using STATA version 17 (StataCorp, College Station, TX, USA), and MedCalc 13.3.3.0 was used to generate forest plots for the estimated effect sizes. Data were presented as mean ± SD, median and interquartile range (IQR), or number (percentage). Comparisons between the intervention and control groups were made using Student's t test, Mann-Whitney U test, and Chi-squared test, as appropriate. The normality of quantitative variables was assessed using histogram plots and the Shapiro-Wilk test. Univariate and multiple logistic regression were employed to explore the intervention's effect and determine the extent of the intervention's effectiveness on the clinical pregnancy rate. Statistical significance was set at a p < 0.05.
3. Results
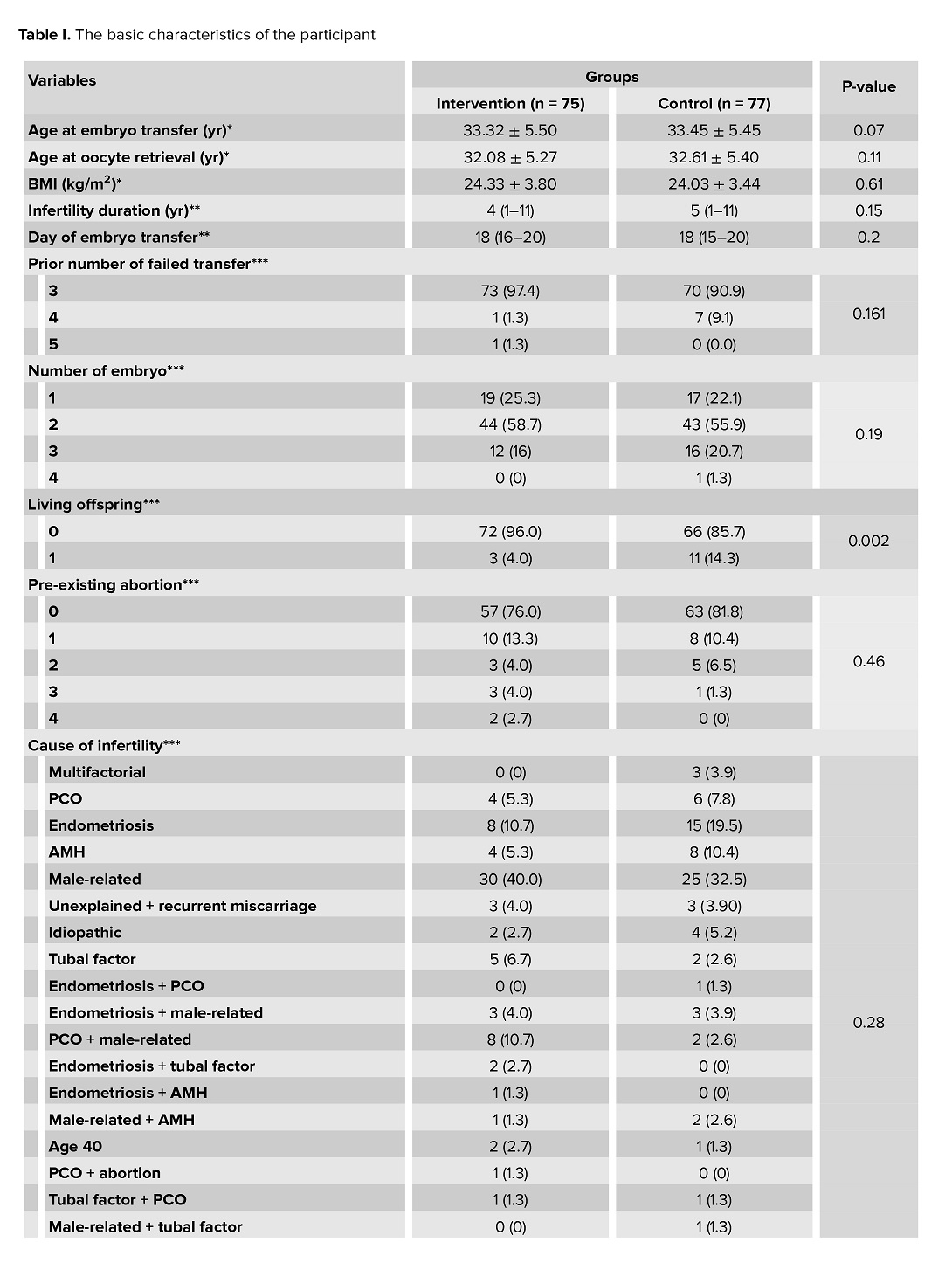
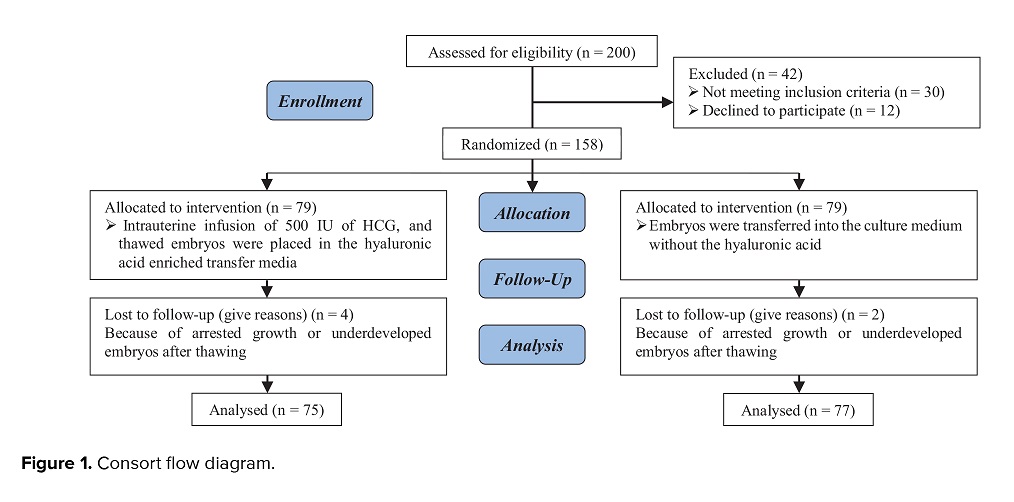
Of 200 cases of infertility assessed for eligibility, 158 participants were enrolled in this study, 79 cases in the intervention group and 79 cases in the control group. During the study, 4 cases in the intervention group and 2 cases in the control group were excluded from analysis because of the arrested growth or underdeveloped embryos after thawing. Finally, 152 women completed the trial. 75 cases were in the intervention group, and 77 cases were in the control group (Figure 1). The mean age of the participants at the time of oocyte retrieval and embryo transfer were 32.8 ± 5.4 and 33.4 ± 5.5 yr old, respectively. The basic characteristics of the included individuals are presented in table I.
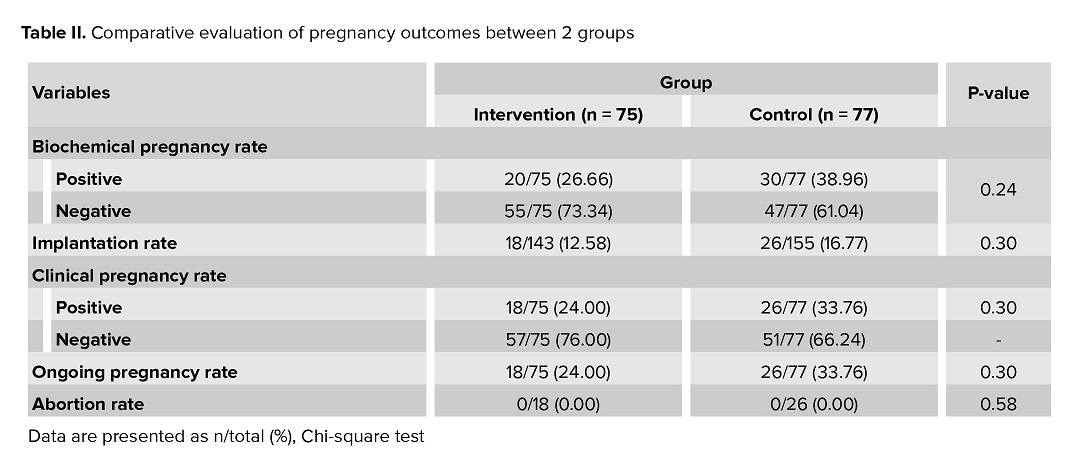
In the intervention group, 20 cases (26.66%) became pregnant, while the control group had 30 cases (38.96%). The results of this study showed that no significant difference was observed in the rate of chemical pregnancy (positive hCG) between the 2 intervention and control groups (p = 0.24).
In the intervention group, 18 cases (24%), and in the control group 26 cases (33.76%) had one gestational sac inside the uterine cavity with fetal pole and heartbeat. The implantation rate was 12.58% in the intervention group and 16.77% in the control group, which was not a significant difference. The rate of clinical and ongoing pregnancy showed no significant difference between 2 study groups (p = 0.30). The pregnancy outcome in participants is presented in table II.
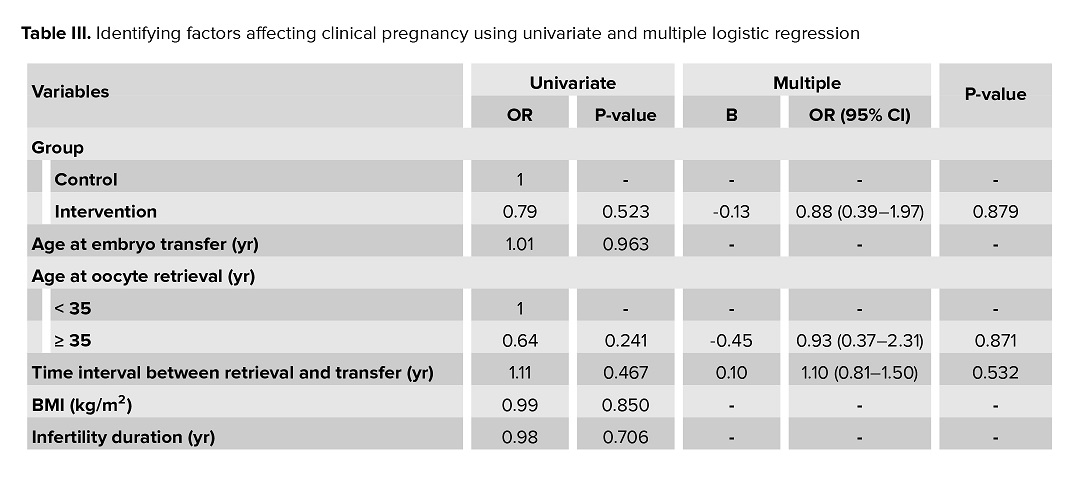
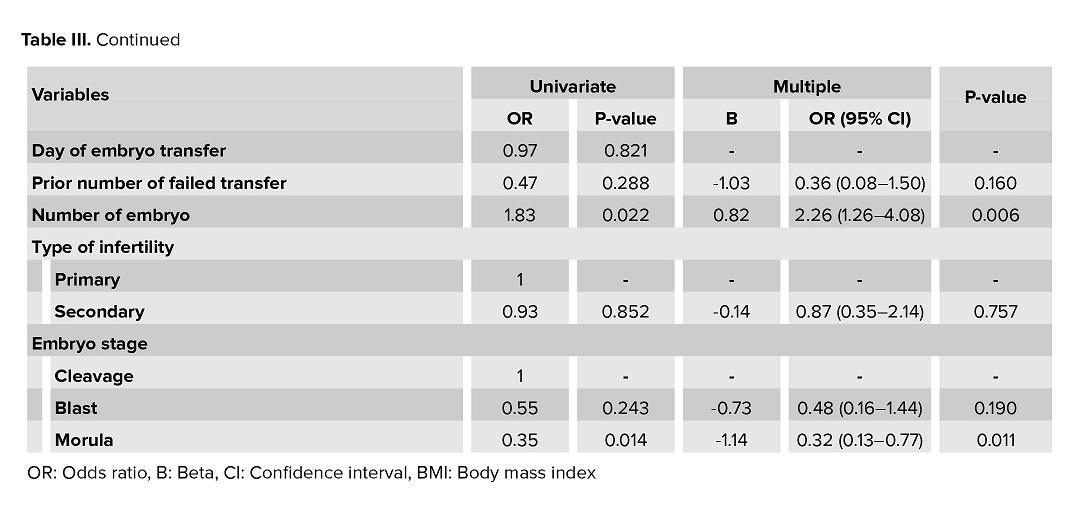
Logistic regression was used to examine the effect of the intervention on clinical pregnancy. Initially, the results of univariate logistic regression showed that the chance of clinical pregnancy in the intervention group was 31% lower than in the control group, but it was not statistically significant (p = 0.523).
Next, to determine potential confounding factors, the effect of the intervention on pregnancy outcome was examined with each variable separately and presented in the univariate column of table III. Then, to examine the factors that affect simultaneously, variables that had a p < 0.5 were entered into multiple regression; the results of which are presented in table III. The results showed that, by controlling variables, the chance of clinical pregnancy in the intervention group was 12% lower than in the control group, although the difference was not statistically significant (p = 0.879).
The variables of the number and stage of transferred embryos were significantly associated with clinical pregnancy. With each increase in the number of embryos, the chance of pregnancy will be 2.26 times higher. Also, the chance of a pregnancy in the morula stage is 68% lower than in the cleavage stage.
4. Discussion
The present study aimed to evaluate the efficacy of the combined treatment of intrauterine hCG infusion and the use of the HA-enriched medium, for embryo transfer on the outcome of pregnancy in women with RIF undergoing FET. Numerous studies have shown that intrauterine hCG infusion or the use of embryo transfer media supplemented with HA improves implantation and clinical pregnancy rate in RIF cases. Our study is the only one that was designed to investigate the effect of synergy and the combination of 2 methods to improve the pregnancy outcome. The findings demonstrated that the overall clinical pregnancy rate did not differ significantly between the intervention and control groups but had a significant effect in specific subgroups, such as cases using more than one embryo at the cleavage stage for transfer.
A recent randomized, double-blind, controlled trial reported comparable live birth rates between the use of a HA-enriched transfer medium and a standard medium for FET (25.5% vs. 25.8%) (13). Similarly, another investigation demonstrated no improvement in pregnancy or implantation outcomes with the use of HA, as clinical pregnancy, implantation, and delivery rates remained similar to controls, even in subgroups with advanced maternal age, prior IVF failures, limited oocyte yield, or poor embryo morphology (14). In contrast, more recent studies have reported positive findings regarding the use of HA in embryo transfer medium. A study looked at the effect of HA-enriched transfer medium on FET outcomes in cases with RIF. They found that the clinical pregnancy rate was significantly higher in the HA group compared to the control group (51.1% vs. 36.8%, p = 0.031), although the live birth rate was not significantly different between the 2 groups (43.8% vs. 33.3%, p = 0.132) (15). By increasing medium viscosity, HA may aid in smoother embryo transfer and help prevent embryo displacement from the uterine cavity. In addition, HA increases cell-cell and cell-matrix adhesion, and this mechanism may improve embryo aggregation and adhesion to the endometrium.
A systematic review and meta-analysis provided a more comprehensive assessment of the impact of adding HA to embryo transfer medium (16). They found that in autologous oocyte IVF cycles, the addition of HA increased live birth (odds ratio [OR] 1.30, 95% confidence interval [CI] 1.09-1.54), clinical pregnancy (OR 1.31, 95% CI 1.13-1.51), and multiple pregnancy rates (OR 1.51, 95% CI 1.13-2.01). However, in donor oocyte cycles, the addition of HA showed little effect on live birth (OR 0.97, 95% CI 0.77-1.22) and clinical pregnancy rates (OR 1.03, 95% CI 0.86-1.24). In another study, the use of hyaluronan-enriched transfer medium (embryo glue) for time-lapse selected elective single embryo transfer in polycystic ovarian syndrome cases resulted in significantly higher implantation rates (39.2% vs. 23.8%, p < 0.001) and live birth rates (35.9% vs. 17.3%, p < 0.001) compared to the use of conventional embryo transfer medium, while also demonstrating lower miscarriage rates (8.3% vs. 28%, p = 0.003) (17). Furthermore, there is report of using hyaluronan-enriched embryo transfer medium, which was associated with significantly higher live birth rates (60.6% vs. 47.5%, p = 0.003) and clinical pregnancy rates (69.5% vs. 57.6%, p = 0.001) compared to a standard transfer medium in frozen embryo transfer cycles (18).
A retrospective study to determine the effect of HA on embryo implantation found higher rate of successful implantation with second-day embryo transfer in cases who used HA. In the HA group, a significant increase in the rate of embryo implantation in cases older than 35 yr and smokers were observed (4). In 2016, a study on the role of HA-enriched medium in fresh embryo transfer cycles, found that the clinical pregnancy rate in the study group was 7% higher than the control group. However, the difference was not statistically significant. A significant difference in clinical pregnancy rate was observed in cases with previous IVF failure (p = 0.04) (19). The reason for this discrepancy could be the conditions of the participants. It should be kept in mind that infertility in people can have different causes. The cause of infertility will be effective in choosing the method of assisted reproduction and its result.
HA is a key component of the extracellular matrix and has been shown to play a crucial role in various reproductive processes, including embryo development, endometrial receptivity, and embryo-endometrial crosstalk (19). Similarly, intrauterine hCG infusion has been proposed to enhance endometrial receptivity and improve implantation outcomes (20). However, the optimal concentration, timing, and method of hCG administration may be critical in determining their effectiveness. Additionally, the presence of other confounding factors, such as the participant’s age, ovarian reserve, endometrial thickness, and underlying infertility diagnoses, may influence the impact of the HA-enriched medium and intrauterine hCG infusion on clinical outcomes. Careful case selection and stratification based on these variables may be necessary to identify the specific subgroups that are most likely to benefit from this approach.
Our study presents a novel approach by combining HA-enriched embryo transfer medium with intrauterine hCG infusion, addressing a critical gap in current reproductive medicine practices. While the overall clinical pregnancy rate did not differ significantly in the present study, the potential benefits of this combination therapy in certain subgroups of cases with RIF were seen and warrant further investigation. The efficacy of HA-enriched media lies in its ability to mimic the natural extracellular matrix, facilitating embryo attachment and migration. This is significant, given that the uterine environment plays a crucial role in implantation. In contrast, intrauterine hCG infusion may provide a supportive hormonal environment, optimizing the uterine lining for implantation (19, 20). By synergistically applying these 2 methods, we aim to create a more conducive environment for embryo implantation. One of the limitations of this study was that the embryos were not considered for PGT-A, the immunological factors that affect implantation were not examined, and all treatment cycles were frozen. Regarding not performing PGT in most centers, and according to the ESHRE guidelines about the evaluation of RIF, immunological evaluation, and PGT-A are not necessary tests. Therefore, this limitation does not have a confounding effect on the results obtained from this study. One of the strong points was the type of study and the matching of the groups in terms of the cause of infertility, which covered most of the factors.
5. Conclusion
The findings of the present study suggest that the efficacy of the combination of HA-enriched embryo transfer medium and intrauterine hCG infusion may be influenced by specific case characteristics. While the overall clinical pregnancy rate did not differ significantly in the current study, the potential benefits of this combined approach were seen in certain subgroups, such as cases using the embryo at the cleavage stage for transfer and cases who received more than one embryo for transfer. Additional large-scale studies are needed to clarify the optimal use of the combination of HA-enriched embryo transfer medium and intrauterine hCG infusion, and its impact on assisted reproductive outcomes in different infertile populations.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
S. Peivandi and P. Jafarifard contributed to the design and conceptualization of the study, selection of methodologies, and drafting of the manuscript. M. Zamaniyan was involved in data collection and editing the draft. J. Yazdani-charati was responsible for data analysis and interpretation of the findings. S. Peivandi played a key role in interpreting the results, editing the manuscript, and providing general feedback throughout all.
Acknowledgments
We would like to acknowledge and appreciate the cooperation of the Research Vice-Chancellor of Mazandaran University of Medical Sciences, Sari, Iran who financially supported this research (grant number: 14202). The authors express gratitude to all women who participated in the study. We did not use artificial intelligence in any way in preparing the manuscript.
Conflict of Interest
The authors declare that there is no conflict of interest.
Recurrent implantation failure (RIF) is generally characterized by the inability to achieve a clinical pregnancy despite transferring at least 4 morphologically high-quality embryos in a minimum of 3 fresh or frozen-thawed embryo transfer (FET) cycles, is a significant challenge in assisted reproductive technology (1, 2). Successful embryo implantation is a complex process that requires synchronized development and interaction between the embryo and a receptive endometrium. Factors contributing to RIF include embryonic, endometrial, genetic, and immunological abnormalities (2, 3).
Various strategies have been explored to improve implantation and clinical pregnancy rate in RIF cases, including intrauterine infusion of human chorionic gonadotropin (hCG) and the use of embryo transfer media supplemented with hyaluronic acid (HA), also known as "embryo glue". Several studies have shown that intrauterine HCG administration prior to embryo transfer can enhance the expression of endometrial receptivity markers, such as leukemia inhibitory factor thereby creating a more favorable environment for implantation. HCG has been shown to enhance endometrial receptivity by promoting angiogenesis, increasing uterine blood flow, and modulating cytokine production, all of which are crucial for successful embryo implantation (1, 4, 5).
HA, a major component of the extracellular matrix, has been hypothesized to improve embryo attachment and implantation by mimicking the natural embryo-endometrial interaction (6, 7). However, some studies have reported improved implantation and pregnancy rates with the use of HA-enriched embryo transfer medium in this population (8-11).
Based on existing evidence, the effectiveness of each approach individually in improving endometrial receptivity and increasing implantation and pregnancy rates is demonstrated. While there is ample evidence supporting the individual use of these methods, we acknowledge the current limitation in the availability of comprehensive studies specifically investigating their combined effect. Our hypothesis is based on the potential synergistic impact of these 2 approaches in enhancing embryo-endometrium interaction and ultimately improving pregnancy outcomes. Taken together, the paracrine roles of hCG are essential in processes such as decidualization, implantation, vascularization, and tissue remodeling. HA is a key component of the extracellular matrix and has been shown to play a crucial role. However, the efficacy of these interventions, especially when used in combination, in improving clinical pregnancy rates in RIF cases undergoing FET cycles, remains inconclusive.
Therefore, this study aims to investigate the effect of intrauterine infusion of hCG and the use of embryo transfer medium containing HA on clinical pregnancy rate in women with RIF undergoing FET cycles.
2. Materials and Methods
2.1. Study design and participants
This single-blind, randomized controlled clinical trial study was conducted at the Infertility and IVF Center of Imam Khomeini hospital in Sari in Iran from December 2022-April 2024. Participants were selected using the block randomization method. The inclusion criteria were: women aged 20-40 yr, with a history of at least 3 unsuccessful embryo transfer cycles and or failed transfer of 4 good quality (grade A or B) embryos, those who were undergoing FET cycles with hormone preparation using estrogen and progesterone, and women who have at least one grade A or B embryo on the day of transfer. The 3rd-day embryo quality was evaluated based on Istanbul Consensus Workshop criteria; grade A embryos were defined as embryos with 6-10 cells (< 10% fragmentation, blastomeres of equal size, and without multinucleation), and grade B was defined as 6-10 cells (10-25% fragmentation, blastomeres of equal size in most cells, and no multinucleation). Embryo quality on the 5th day was evaluated using the Gardner and Schoolcraft scoring system. Expansion grade 1-6, internal cell mass morphology (grade A-C), and trophectoderm morphology (grade A-C) were used to grade embryos on the 5th day. Good-quality embryos on the 5th day were defined as ≥ 3BB (AA, AB, BA, and BB). Exclusion criteria included: use of donated egg, donated embryo, or surrogacy; presence of uterine abnormalities, severe endometriosis and endometrioma, hydrosalpinx in hysterosalpingography or sonography, history of uncontrolled endocrine diseases like diabetes, hypothyroidism, or hyperthyroidism; and male factor infertility with azoospermia or difficult embryo transfer requiring cervical manipulation or anesthesia.
2.2. Sample size
The sample size for this study was calculated based on the results reported in the study by Hafezi et al. (12), assuming a live birth rate of 16.6% in the intervention group (P1) and 41.6% in the control group (P2), with a significance level (α) of 0.05 and a power (1-β) of 90%; the required sample size was determined using the following formula, which yielded 66 participants per group (n = 132). Accounting for a 15% dropout rate, we recruited 79 participants per group (n = 158).
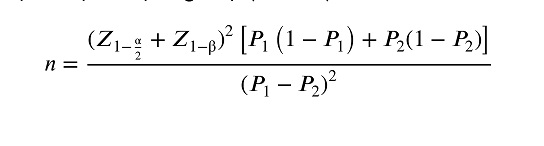
2.3. Endometrial preparation and randomization
In all eligible participants, endometrial preparation was performed artificially by use of estrogen and progesterone hormones. Initially, a gonadotropin releasing hormone agonist was started in the mid-luteal phase of the previous menstrual cycle for all cases and continued through the next cycle. Subsequently, the endometrial growth was supported from the time of menstruation by daily administration of 2-6 mg of oral estradiol valerate (Estraval®, Aburaihan, Tehran, Iran). After 10 days of estradiol administration, vaginal sonography was performed. If the endometrial thickness reached ≥ 8 mm, estradiol valerate was continued at the same dose and 50-100 mg of progesterone (Progestin®, Aburaihan Pharmaceutical Co., Tehran, Iran) was injected intramuscularly for 3-5 days. If the optimal endometrial thickness was not reached, the dose of estradiol was increased up to 8 mg per day, and monitoring of endometrial thickness was continued for another 3 days.
Participants were grouped using a block random allocation by (Random Allocation Software 2.0) and using their admission row number. The participants who met the inclusion criteria were randomly divided into 2 groups: the intervention group and the control group in a 1:1 ratio. 79 women were assigned to each group. The intervention group received an intrauterine infusion of 500 IU of hCG (Amp Choriomon®, Pharmed Daru) diluted with 15 µl of embryo culture medium (SAGE, 1-step culture medium, Origio) by the embryo transfer catheter guided by abdominal ultrasound 20 min before embryo transfer. The thawed embryos were placed in the HA-enriched transfer media (Embryo Glue, Vitrolife) and were transferred into the uterus by the embryo transfer catheter guided by abdominal ultrasound. In the control group, the embryos were transferred into the culture medium without the HA and then transferred into the uterine cavity by transfer catheter guided by abdominal ultrasound. The present study is a single-blind randomized clinical trials in which the physician and participant were aware of the type of intervention, but the embryologist who performed the thawing and transfer and the person collecting the data and the result of the pregnancy outcome for analysis were unaware of the type of intervention. Moreover, double blinding was not possible due to the method of intervention.
2.4. Outcome assessments
The primary outcome was clinical pregnancy rate, defined as the presence of a fetal heartbeat assessed by transvaginal ultrasound at 6 wk after embryo transfer. Secondary outcomes included rates of chemical pregnancy, implantation, miscarriage, and ongoing pregnancy. The chemical pregnancy rate is defined as the positive pregnancy test 3-4 wk after intervention. The implantation rate was evaluated 5 wk after intervention and was calculated as the total number of intrauterine gestational sacs divided by the total number of transferred embryos, multiplied by 100. The abortion rate was evaluated 18 wk after intervention and was defined as the number of clinical pregnancy losses before the 20th wk of pregnancy divided by the total number of clinical pregnancies. Ongoing pregnancy was defined as either a birth at 38 wk after intervention or a pregnancy continued after the 20th wk of gestation.
2.5. Ethical Considerations
This study protocol was approved by the Ethical Committee of Maandaran University of Medical Sciences, Sari, Iran (Code: IR.MAZUMS.IMAMHOSPITAL.REC.1401.038) and the Institutional Review Board of Iran (Code: IR19464). The study was also registered in the Iranian Registry of Clinical Trials with the identification number IRCT20221126056614N1, last updated on July 30, 2024. All participants provided written informed consent prior to enrollment in the study. The confidentiality of participants' personal information was strictly maintained throughout the study. Participants were free to withdraw from the study at any time without any impact on their ongoing medical care. The study procedures were in accordance with the Declaration of Helsinki.
2.6. Statistical Analysis
All statistical analyses were performed using STATA version 17 (StataCorp, College Station, TX, USA), and MedCalc 13.3.3.0 was used to generate forest plots for the estimated effect sizes. Data were presented as mean ± SD, median and interquartile range (IQR), or number (percentage). Comparisons between the intervention and control groups were made using Student's t test, Mann-Whitney U test, and Chi-squared test, as appropriate. The normality of quantitative variables was assessed using histogram plots and the Shapiro-Wilk test. Univariate and multiple logistic regression were employed to explore the intervention's effect and determine the extent of the intervention's effectiveness on the clinical pregnancy rate. Statistical significance was set at a p < 0.05.
3. Results
Of 200 cases of infertility assessed for eligibility, 158 participants were enrolled in this study, 79 cases in the intervention group and 79 cases in the control group. During the study, 4 cases in the intervention group and 2 cases in the control group were excluded from analysis because of the arrested growth or underdeveloped embryos after thawing. Finally, 152 women completed the trial. 75 cases were in the intervention group, and 77 cases were in the control group (Figure 1). The mean age of the participants at the time of oocyte retrieval and embryo transfer were 32.8 ± 5.4 and 33.4 ± 5.5 yr old, respectively. The basic characteristics of the included individuals are presented in table I.
In the intervention group, 20 cases (26.66%) became pregnant, while the control group had 30 cases (38.96%). The results of this study showed that no significant difference was observed in the rate of chemical pregnancy (positive hCG) between the 2 intervention and control groups (p = 0.24).
In the intervention group, 18 cases (24%), and in the control group 26 cases (33.76%) had one gestational sac inside the uterine cavity with fetal pole and heartbeat. The implantation rate was 12.58% in the intervention group and 16.77% in the control group, which was not a significant difference. The rate of clinical and ongoing pregnancy showed no significant difference between 2 study groups (p = 0.30). The pregnancy outcome in participants is presented in table II.
Logistic regression was used to examine the effect of the intervention on clinical pregnancy. Initially, the results of univariate logistic regression showed that the chance of clinical pregnancy in the intervention group was 31% lower than in the control group, but it was not statistically significant (p = 0.523).
Next, to determine potential confounding factors, the effect of the intervention on pregnancy outcome was examined with each variable separately and presented in the univariate column of table III. Then, to examine the factors that affect simultaneously, variables that had a p < 0.5 were entered into multiple regression; the results of which are presented in table III. The results showed that, by controlling variables, the chance of clinical pregnancy in the intervention group was 12% lower than in the control group, although the difference was not statistically significant (p = 0.879).
The variables of the number and stage of transferred embryos were significantly associated with clinical pregnancy. With each increase in the number of embryos, the chance of pregnancy will be 2.26 times higher. Also, the chance of a pregnancy in the morula stage is 68% lower than in the cleavage stage.






4. Discussion
The present study aimed to evaluate the efficacy of the combined treatment of intrauterine hCG infusion and the use of the HA-enriched medium, for embryo transfer on the outcome of pregnancy in women with RIF undergoing FET. Numerous studies have shown that intrauterine hCG infusion or the use of embryo transfer media supplemented with HA improves implantation and clinical pregnancy rate in RIF cases. Our study is the only one that was designed to investigate the effect of synergy and the combination of 2 methods to improve the pregnancy outcome. The findings demonstrated that the overall clinical pregnancy rate did not differ significantly between the intervention and control groups but had a significant effect in specific subgroups, such as cases using more than one embryo at the cleavage stage for transfer.
A recent randomized, double-blind, controlled trial reported comparable live birth rates between the use of a HA-enriched transfer medium and a standard medium for FET (25.5% vs. 25.8%) (13). Similarly, another investigation demonstrated no improvement in pregnancy or implantation outcomes with the use of HA, as clinical pregnancy, implantation, and delivery rates remained similar to controls, even in subgroups with advanced maternal age, prior IVF failures, limited oocyte yield, or poor embryo morphology (14). In contrast, more recent studies have reported positive findings regarding the use of HA in embryo transfer medium. A study looked at the effect of HA-enriched transfer medium on FET outcomes in cases with RIF. They found that the clinical pregnancy rate was significantly higher in the HA group compared to the control group (51.1% vs. 36.8%, p = 0.031), although the live birth rate was not significantly different between the 2 groups (43.8% vs. 33.3%, p = 0.132) (15). By increasing medium viscosity, HA may aid in smoother embryo transfer and help prevent embryo displacement from the uterine cavity. In addition, HA increases cell-cell and cell-matrix adhesion, and this mechanism may improve embryo aggregation and adhesion to the endometrium.
A systematic review and meta-analysis provided a more comprehensive assessment of the impact of adding HA to embryo transfer medium (16). They found that in autologous oocyte IVF cycles, the addition of HA increased live birth (odds ratio [OR] 1.30, 95% confidence interval [CI] 1.09-1.54), clinical pregnancy (OR 1.31, 95% CI 1.13-1.51), and multiple pregnancy rates (OR 1.51, 95% CI 1.13-2.01). However, in donor oocyte cycles, the addition of HA showed little effect on live birth (OR 0.97, 95% CI 0.77-1.22) and clinical pregnancy rates (OR 1.03, 95% CI 0.86-1.24). In another study, the use of hyaluronan-enriched transfer medium (embryo glue) for time-lapse selected elective single embryo transfer in polycystic ovarian syndrome cases resulted in significantly higher implantation rates (39.2% vs. 23.8%, p < 0.001) and live birth rates (35.9% vs. 17.3%, p < 0.001) compared to the use of conventional embryo transfer medium, while also demonstrating lower miscarriage rates (8.3% vs. 28%, p = 0.003) (17). Furthermore, there is report of using hyaluronan-enriched embryo transfer medium, which was associated with significantly higher live birth rates (60.6% vs. 47.5%, p = 0.003) and clinical pregnancy rates (69.5% vs. 57.6%, p = 0.001) compared to a standard transfer medium in frozen embryo transfer cycles (18).
A retrospective study to determine the effect of HA on embryo implantation found higher rate of successful implantation with second-day embryo transfer in cases who used HA. In the HA group, a significant increase in the rate of embryo implantation in cases older than 35 yr and smokers were observed (4). In 2016, a study on the role of HA-enriched medium in fresh embryo transfer cycles, found that the clinical pregnancy rate in the study group was 7% higher than the control group. However, the difference was not statistically significant. A significant difference in clinical pregnancy rate was observed in cases with previous IVF failure (p = 0.04) (19). The reason for this discrepancy could be the conditions of the participants. It should be kept in mind that infertility in people can have different causes. The cause of infertility will be effective in choosing the method of assisted reproduction and its result.
HA is a key component of the extracellular matrix and has been shown to play a crucial role in various reproductive processes, including embryo development, endometrial receptivity, and embryo-endometrial crosstalk (19). Similarly, intrauterine hCG infusion has been proposed to enhance endometrial receptivity and improve implantation outcomes (20). However, the optimal concentration, timing, and method of hCG administration may be critical in determining their effectiveness. Additionally, the presence of other confounding factors, such as the participant’s age, ovarian reserve, endometrial thickness, and underlying infertility diagnoses, may influence the impact of the HA-enriched medium and intrauterine hCG infusion on clinical outcomes. Careful case selection and stratification based on these variables may be necessary to identify the specific subgroups that are most likely to benefit from this approach.
Our study presents a novel approach by combining HA-enriched embryo transfer medium with intrauterine hCG infusion, addressing a critical gap in current reproductive medicine practices. While the overall clinical pregnancy rate did not differ significantly in the present study, the potential benefits of this combination therapy in certain subgroups of cases with RIF were seen and warrant further investigation. The efficacy of HA-enriched media lies in its ability to mimic the natural extracellular matrix, facilitating embryo attachment and migration. This is significant, given that the uterine environment plays a crucial role in implantation. In contrast, intrauterine hCG infusion may provide a supportive hormonal environment, optimizing the uterine lining for implantation (19, 20). By synergistically applying these 2 methods, we aim to create a more conducive environment for embryo implantation. One of the limitations of this study was that the embryos were not considered for PGT-A, the immunological factors that affect implantation were not examined, and all treatment cycles were frozen. Regarding not performing PGT in most centers, and according to the ESHRE guidelines about the evaluation of RIF, immunological evaluation, and PGT-A are not necessary tests. Therefore, this limitation does not have a confounding effect on the results obtained from this study. One of the strong points was the type of study and the matching of the groups in terms of the cause of infertility, which covered most of the factors.
5. Conclusion
The findings of the present study suggest that the efficacy of the combination of HA-enriched embryo transfer medium and intrauterine hCG infusion may be influenced by specific case characteristics. While the overall clinical pregnancy rate did not differ significantly in the current study, the potential benefits of this combined approach were seen in certain subgroups, such as cases using the embryo at the cleavage stage for transfer and cases who received more than one embryo for transfer. Additional large-scale studies are needed to clarify the optimal use of the combination of HA-enriched embryo transfer medium and intrauterine hCG infusion, and its impact on assisted reproductive outcomes in different infertile populations.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
S. Peivandi and P. Jafarifard contributed to the design and conceptualization of the study, selection of methodologies, and drafting of the manuscript. M. Zamaniyan was involved in data collection and editing the draft. J. Yazdani-charati was responsible for data analysis and interpretation of the findings. S. Peivandi played a key role in interpreting the results, editing the manuscript, and providing general feedback throughout all.
Acknowledgments
We would like to acknowledge and appreciate the cooperation of the Research Vice-Chancellor of Mazandaran University of Medical Sciences, Sari, Iran who financially supported this research (grant number: 14202). The authors express gratitude to all women who participated in the study. We did not use artificial intelligence in any way in preparing the manuscript.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Fertility & Infertility
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |