Sat, Apr 11, 2026
[Archive]
Volume 23, Issue 9 (September 2025)
IJRM 2025, 23(9): 671-688 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Tjahyadi D, Sudono Riyadi A, Dewi Nugrahani A, Amarullah Ritonga M, Rachmawati A, Husnitawati Madjid T. Potency of oral gonadotropin-releasing hormone antagonist as endometriosis-associated pain novel treatments: An updated meta-analysis of randomized controlled trials. IJRM 2025; 23 (9) :671-688
URL: http://ijrm.ir/article-1-3513-en.html
URL: http://ijrm.ir/article-1-3513-en.html
Dian Tjahyadi1 

 , Anthony Sudono Riyadi *2
, Anthony Sudono Riyadi *2 

 , Annisa Dewi Nugrahani1
, Annisa Dewi Nugrahani1 

 , Mulyanusa Amarullah Ritonga1
, Mulyanusa Amarullah Ritonga1 

 , Anita Rachmawati1
, Anita Rachmawati1 

 , Tita Husnitawati Madjid1
, Tita Husnitawati Madjid1 




 , Anthony Sudono Riyadi *2
, Anthony Sudono Riyadi *2 

 , Annisa Dewi Nugrahani1
, Annisa Dewi Nugrahani1 

 , Mulyanusa Amarullah Ritonga1
, Mulyanusa Amarullah Ritonga1 

 , Anita Rachmawati1
, Anita Rachmawati1 

 , Tita Husnitawati Madjid1
, Tita Husnitawati Madjid1 


1- Department of Obstetrics and Gynecology, Faculty of Medicine, Universitas Padjadjaran, Dr. Hasan Sadikin General Hospital, Bandung, West Java, Indonesia.
2- Department of Obstetrics and Gynecology, Faculty of Medicine, Universitas Padjadjaran, Dr. Hasan Sadikin General Hospital, Bandung, West Java, Indonesia. ,Anthony180187@yahoo.com
2- Department of Obstetrics and Gynecology, Faculty of Medicine, Universitas Padjadjaran, Dr. Hasan Sadikin General Hospital, Bandung, West Java, Indonesia. ,
Full-Text [PDF 3725 kb]
(889 Downloads)
| Abstract (HTML) (738 Views)
Full-Text: (173 Views)
1. Introduction
Endometriosis is a gynecological condition that affects up to 10% of women of reproductive age (1, 2). Endometriosis is heterogeneous and has one of the main symptoms, which is pain. Classically, there are 3 types of pain associated with endometriosis: dysmenorrhea, dyspareunia, and non-menstrual chronic pelvic pain (NMCPP) (3-5). Endometriosis is an inflammatory condition driven by estrogen. Studies indicate that estrogen plays a crucial role in promoting the growth and inflammation of endometriotic lesions (6).
Endometriosis pain can cause a considerable burden for women and affect their quality of life. Several scales have been used to assess pain scales including the verbal rating scale (VRS), numeric rating scale (NRS), and modified Biberoglu and Behrman (mB&B) scale. Nowadays, there is no consensus on the best method to evaluate pain in endometriosis, and many scales have been used (7).
There are many established treatments for endometriosis-associated pain, such as combined oral hormonal contraceptives, progestin-only formulations (oral or depot injection), and gonadotropin releasing hormone (GnRH) agonists, frequently prescribed by many clinicians around the world (8). First-line therapy to second-line therapy in the form of depot injections currently still have many side effects. The provision of treatment given to an individual needs to consider a number of aspects, including their efficacy and safety (8-14).
GnRH antagonists have emerged as a potential novel therapy for the treatment of endometriosis-associated pain. They act by blocking GnRH receptors, which leads to immediate luteinizing hormone and estrogen suppression (8-12). As a recent breakthrough to manage estrogen levels, minimize residual symptoms, and maintain efficacy in reducing endometriosis-related pain, oral GnRH antagonists (Elagolix, Linzagolix, and Relugolix) have emerged as potential alternatives to control estradiol (E2) levels (8-14). Elagolix showed effectiveness in managing dysmenorrhea and NMCPP with partial estrogen suppression at 150 mg daily (15). Linzagolix, under development, may provide consistent E2 suppression with once-daily dosing (16). Relugolix demonstrated rapid action and effective suppression of E2 and progesterone, with promising results in phase 1 and phase 2 studies (8-14, 17).
These novel treatment options need further study regarding their role in reducing pain intensity in dysmenorrhea, dyspareunia, and NMCPP (7) due to endometriosis as well as secondary side effects of treatment due to hypoestrogen conditions (decreased bone mineral density [BMD] and E2 levels, hot flushes, and dyslipidemia). Endometriosis associated-pain leads to decreased quality of life (18), using many assessment tools such as endometriosis health profile (EHP) -5 instrument, patient global impression of change (PGIC) score, EHP-30 instrument.
Previous meta-analysis studies (18, 19) that specifically assessed the outcomes of oral GnRH antagonists, did not focus on looking at the estimated pool effect of each pain scale (VRS, NRS, or other scales), evaluate secondary effect comprehensively, include studies published since 2023, or use funnel plots in the risk of bias (Rob) assessment of randomized controlled trial (RCT).
This study aimed to evaluate the primary outcome of pain intensity reduction in endometriosis and secondary outcomes in the form of side effects of treatment and quality of life with a systematic review approach and meta-analysis of various updated RCT studies as the best form of evidence-based medicine (level I).
2. Materials and Methods
2.1. Protocol and registration
This systematic review and meta analysis was conducted in line with the Cochrane Handbook for Systematic Reviews of Interventions and complied with the reporting standards set by PRISMA (20).
2.2. Search strategy
We conducted a systematic search of Scopus, PubMed, and Cochrane from inception to July 30th, 2024, with restriction to English only. The search query includes keywords and search phrases that involve “endometriosis”, “elagolix”, “linzagolix”, “relugolix”, “dysmenorrhea”, “pelvic pain”, and “GnRH receptor antagonist” and their synonyms. We tailored the search keywords to the particular requirements of each database. The PRISMA criteria were implemented in our search process. At the initial screening, we found 179 articles based on keywords from 3 electronic databases (Table I).

2.3. Study selection
In this meta-analysis, we considered studies studying the potency of GnRH antagonists as novel treatments for endometrial-associated pain. Population, intervention, comparison, and outcomes (PICO) from this study are: P) women of reproductive age diagnosed with endometriosis based on clinical findings, magnetic resonance imaging (MRI), or intraoperative (laparotomy, laparoscopy) according to European Society of Human Reproduction and Embryology (ESHRE) guidelines; I) administration of oral GnRH antagonists (Elagolix, Linzagolix, and Relugolix); C) placebo; and O) primary outcomes include reduction in pain intensity from dysmenorrhea, dyspareunia, and NMCPP and secondary outcomes include quality of life and hypoestrogenic effects (decreased E2 levels, decreased bone mass density/BMD, hot flushes, dyslipidemia, and other drug side effects).
The detailed inclusion criteria were as follows: 1) RCT studies on the role of oral GnRH antagonists (Elagolix, Linzagolix, and Relugolix) with regular monitoring. 2) Subjects in RCT studies were women of reproductive age diagnosed with endometriosis based on clinical findings, MRI, or intraoperative (laparotomy, laparoscopy) according to ESHRE guidelines (1). 3) Subjects in RCT studies had passed the washout phase and did not experience residual symptoms. 4) Studies in English. 5) Studies published in reputable international journals without period restrictions.
Those studies involving subjects with comorbid chronic diseases such as cardiovascular, liver, kidney, autoimmune, malignancy, history of gynecological surgery, and hematological disorders were excluded. Our meta-analysis also excluded case reports/series, review papers, editorials, comments, meta-analyses, and conference abstracts. Independent evaluations of the pre-selected studies that meet the inclusion criteria were conducted by ASR, ADN, and DT. Any differences of opinion were worked out through conversations with the other authors (THM, AR, and MAR).
2.4. Data extraction and Rob assessment
Data were extracted from the eligible studies, including baseline characteristics such as the first author’s name, the country where the study was conducted, study design, and study participants, treatments, and placebo. Primary outcomes include reduction in pain intensity from dysmenorrhea, dyspareunia, and NMCPP and secondary outcomes include quality of life and hypoestrogenic effects (decreased E2 levels, decreased bone mass density/BMD, hot flushes, dyslipidemia, and other drug side effects). The authors utilized the Rob 2.0 tool to evaluate the potential for bias in each study independently. Any disagreements regarding quality ratings were resolved through discussions among the authors.
2.5. Outcome measures
Endometriosis is diagnosed based on clinical, MRI, or intraoperative findings (laparotomy, laparoscopy) according to ESHRE guidelines. Primary outcomes include a reduction in pain intensity in terms of dysmenorrhea, dyspareunia, and NMCPP. Secondary outcomes include quality of life using several assessment tools such as EHP-5 pain dimension component, PGIC score, and EHP-30 pain domain and hypoestrogenism effects (decreased E2 levels, decreased bone mass density as measured by dual-energy X-Ray absorptiometry, hot flushes, and alterations in lipid profile).
2.6. Ethical Considerations
The protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under the ID: CRD42024579105.
2.7. Statistical Analysis
For statistical analysis, review manager version 5.4 (Revman International, Inc, New York) was used. Dichotomous data were analyzed and pooled as odds ratio (OR) with 95% confidence intervals (95% CI) to measure the effect size. For continuous data, mean difference (MD) was used as an effective measure between the 2 groups. Statistical significance was attained by a p < 0.05 (21-23). The I2 index was used to examine inter-study heterogeneity, and an I2 statistic of < 50% disclosed homogeneity. The OR described in each study is pooled using the generic invariance method with a random effect model. These models were used to pool the overall effect magnitude owing to the fact that the inter-study heterogeneity was low. If inter-study heterogeneity is ³ 50%, the pooled outcomes were computed using a random effect model (21-23).
3. Results
Many of the initially retrieved articles may not have been directly related to the topic of interest, despite meeting the search terms. These were not included in further screening (e.g., in PubMed from 127 articles, 66 of them were not related to the topics, thus total from PubMed was 127-66 = 61).
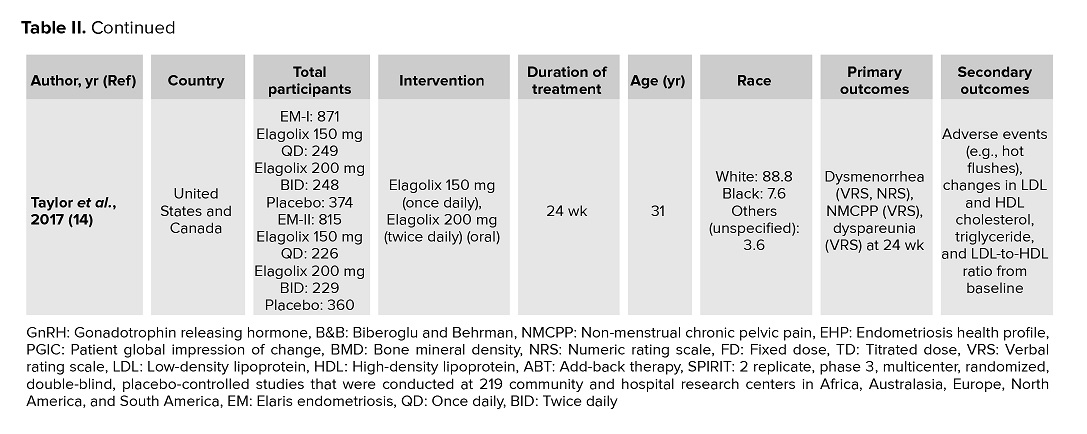
The primary search results yielded a total of 105 articles; after hand exclusion of case reports/series and review papers and removing duplicates, a total of 88 were screened. After screening the titles and abstracts, full papers were identified for further assessment based on the promulgated inclusion criteria. 61 articles were excluded, while 7 others were review articles and 8 articles had wrong comparator from placebo. Only 12 articles passed the step for full-text eligibility assessment. 2 articles had incomplete data, 2 other articles included other gynecological conditions, and one article included placebo < 12 wk. Ultimately, 7 RCT studies were included in our analysis of systematic review and meta-analysis from different countries. Figure 1 depicts the results of the literature search, including the basis for excluding certain publications. The baseline characteristics of the included studies in the final review were presented in table II (8-14).
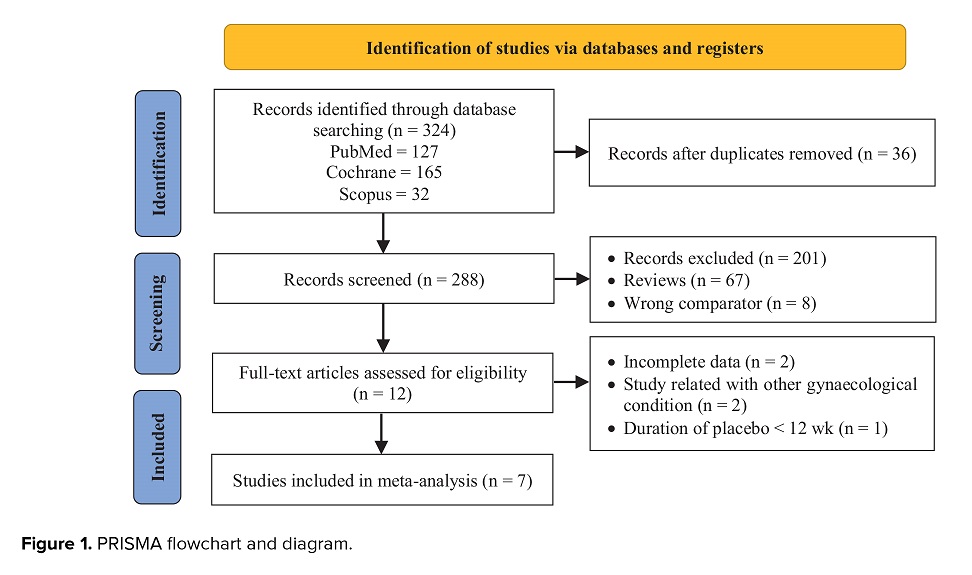
3.1. Study characteristics
In terms of study design and country of origin, the included studies in this article varied widely worldwide (Table II). All of the studies included are RCTs. Based on table II (8-14). The types of oral GnRH antagonist treatments vary in types (Elagolix, Linzagolix, and Relugolix) and dosage. Each experiment also has quite varied inclusion and exclusion criteria with fairly homogeneous primary and secondary outcomes. The majority of studies have a similar follow-up duration.
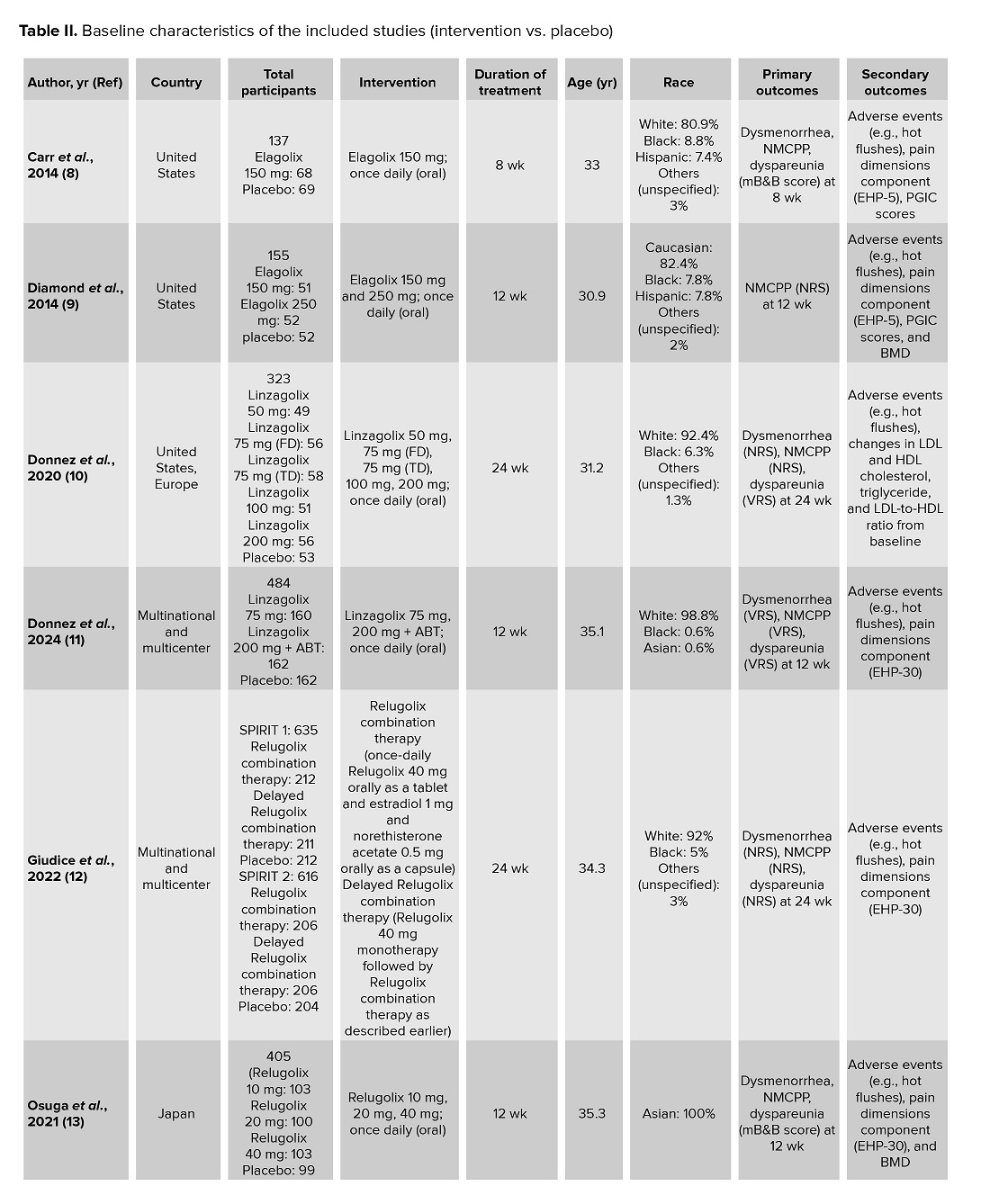
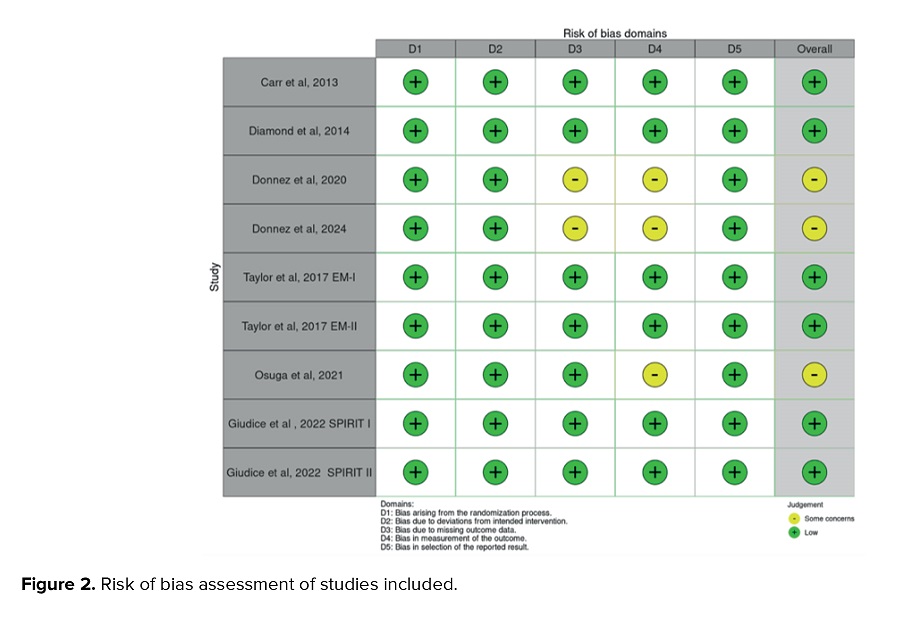
Based on the RoB 2.0 assessment (Figure 2), the majority of the studies were considered to have a low Rob. D4 (bias in measurement outcome data) dominated the moderate Rob domain. The D1 (bias arising from the process of randomization), D2 (bias in deviations of intended intervention), D3 (bias due to missing outcome data), and D5 (bias in the selection of reported results) bias domains were the low-level Rob domains. None of the studies had a significant Rob.
3.2. Publication bias
Publication bias assessed by funnel plot showed an asymmetrical figure; hence, there should be some publication biases in each of the predictors analyzed that should be taken into account.
3.3. Primary outcomes
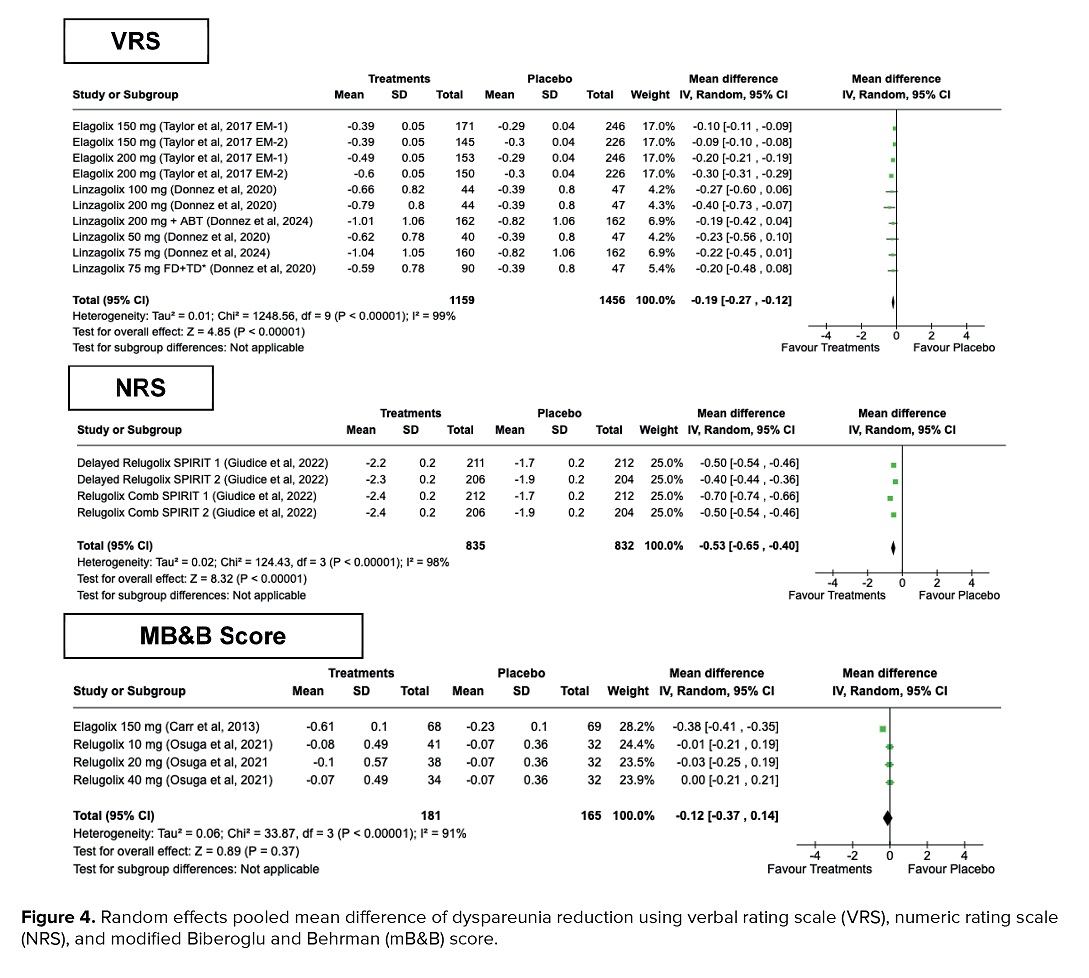
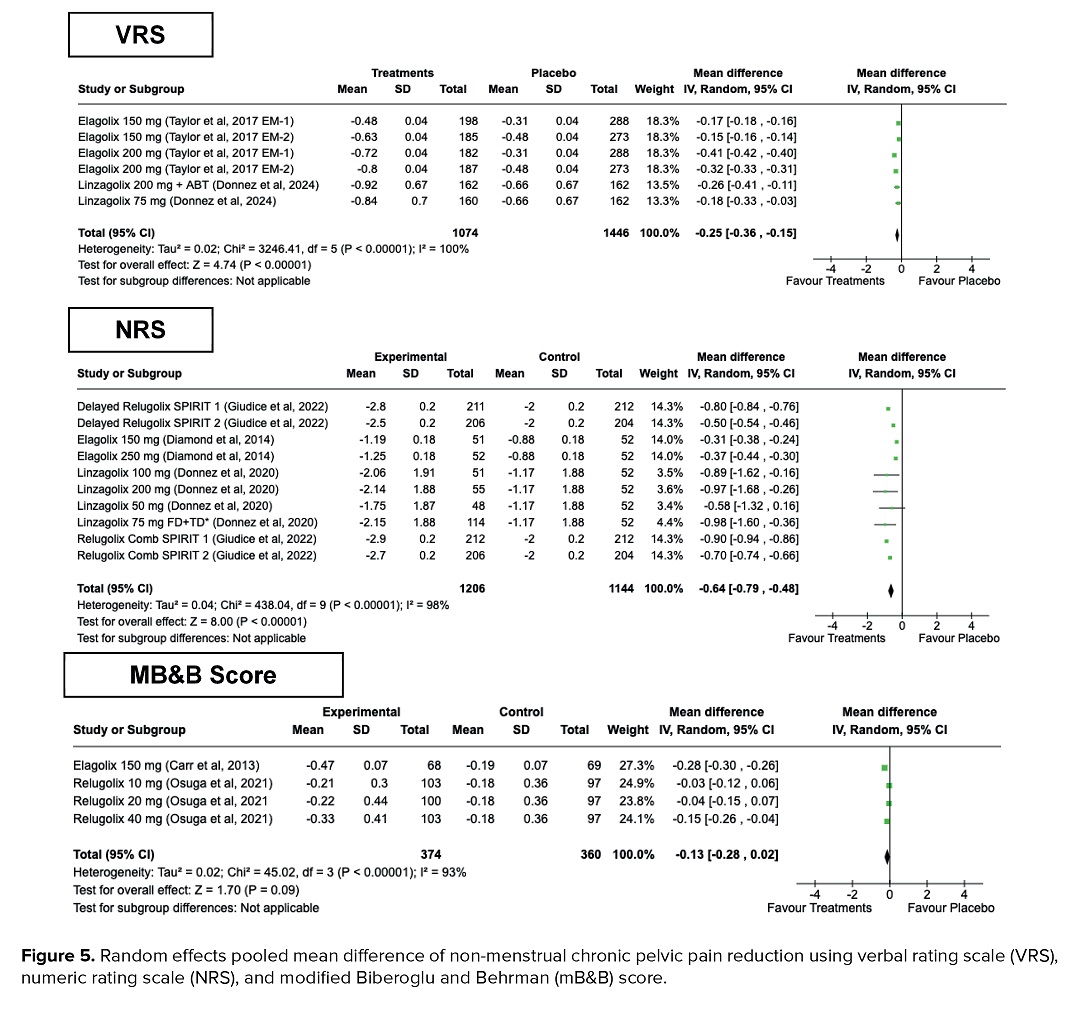
A total of 7 RCTs evaluated the potency of GnRH antagonists for endometriosis-associated pain. Our merged analysis suggested that the population treated with GnRH antagonists had a significant reduction of dysmenorrhea, dyspareunia, and NMCPP using VRS and NRS scales, but not with mB&B scores for dyspareunia and NMCPP (Figures 3-5).
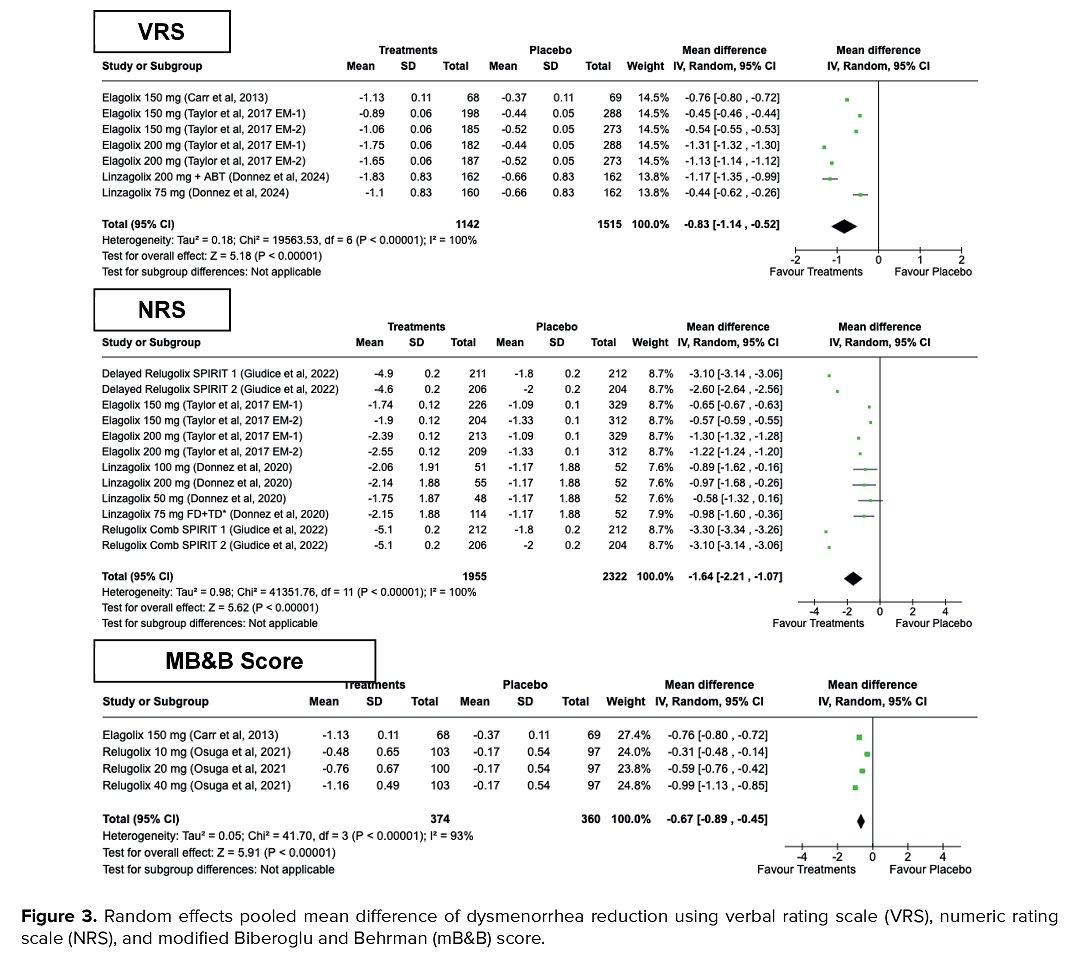
Participants who underwent treatment using GnRH antagonists had a significant reduction of dysmenorrhea using VRS (MD = -0.83, 95% CI = -1.14, -0.52, p < 0.001), NRS (MD = -1.64, 95% CI = -2.21, -1.07, p < 0.001), and mB&B score (MD = -1.64, 95% CI = -2.21, -1.07, p < 0.001) (Figure 3). High heterogeneities were detected in VRS group (I2 = 100%, p < 0.001), NRS group (I2 = 100%, p < 0.001), and mB&B score (I2 = 93%, p < 0.001), respectively.
From the dyspareunia reduction aspect, women who underwent treatment using GnRH antagonists had a significant reduction of dyspareunia using VRS (MD = -0.19, 95% CI = -0.27, -0.12, p < 0.001) and NRS (MD = -0.53, 95% CI = -0.65, -0.40, p < 0.001). Nonetheless, no significant difference was observed in reduction of dyspareunia in participants assessed with MB&B Score (MD = -0.12, 95% CI = -0.37, 0.14, p = 0.37). High heterogeneities were detected in VRS group (I2 = 99%, p < 0.001), NRS group (I2 = 98%, p < 0.001), and MB&B Score (I2 = 91%, p < 0.001), respectively (Figure 4).
Women who underwent treatment using GnRH antagonists had a significant reduction of NMCPP using VRS (MD = -0.25, 95% CI = -0.36, -0.15, p < 0.001) and NRS (MD = -0.64, 95% CI = -0.79, -0.48, p < 0.001). However, for the mB&B score, despite a reduction of -0.13 in MD, the effect size failed to attain statistical significance (95% CI = -0.28, 0.02, p = 0.09). High heterogeneities were detected in VRS group (I2 = 100%, p < 0.001), NRS group (I2 = 98%, p < 0.001), and mB&B score (I2 = 93%, p < 0.001), respectively (Figure 5).
3.4. Secondary outcomes
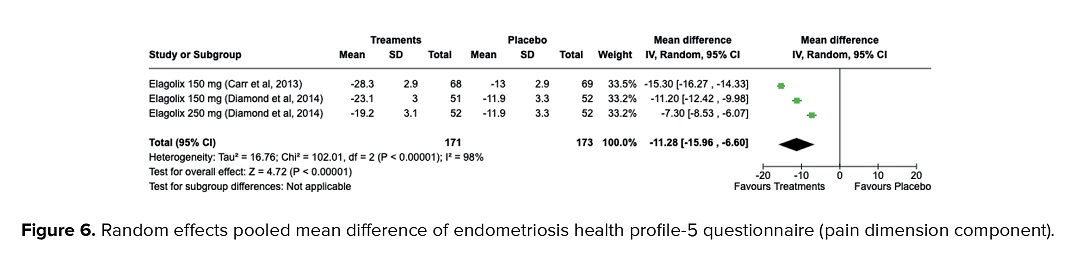
Scores on the EHP-5 score and modular questionnaire are then transformed on a scale of 0 (indicating best possible health status) to 100 (indicating worst possible health status) (18). As secondary outcomes, women who underwent treatment using GnRH antagonists had a significantly higher possibility of health status using the EHP-5 instrument (MD = -11.28, 95% CI = -15.96, -6.60, p < 0.001). High heterogeneities were detected (I2 = 98%, p < 0.001) as presented in figure 6.
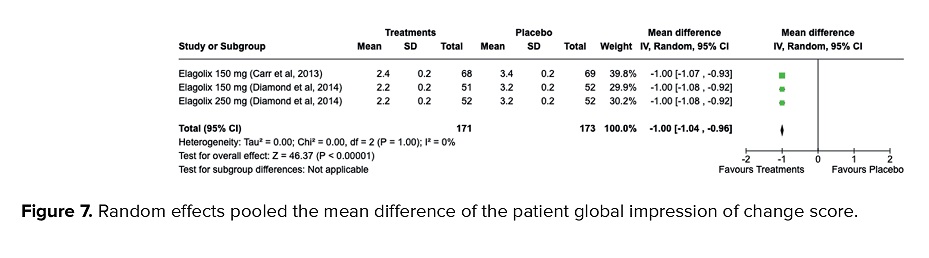
The self-report measures a PGIC and reflects an individual's belief about the efficacy of treatment. Lower levels showed much improvement. In this study, women who underwent treatment using GnRH antagonists had a significant improvement in the treatment arms with PGIC score (MD = -1.00, 95% CI = -1.04, -0.96, p < 0.001). No heterogeneities were detected (I2 = 0%, p = 1.00) as presented in figure 7.
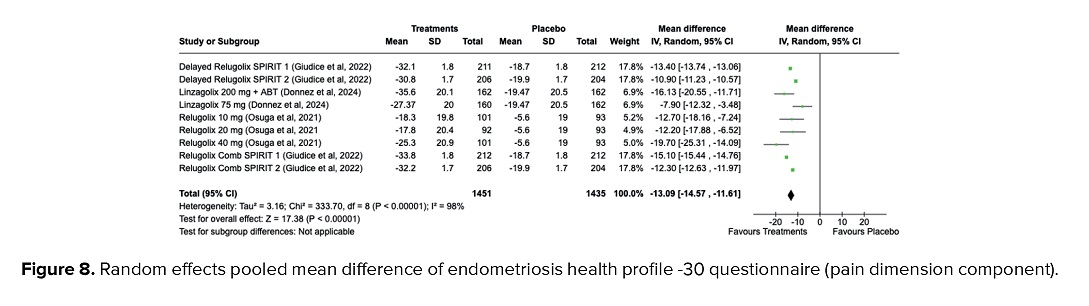
Moreover, women who underwent treatment using GnRH antagonists had a significantly higher possibility of health status using the EHP-30 instrument (MD = -13.09, 95% CI = -14.57, -11.61, p < 0.001). High heterogeneities were detected (I2 = 98%, p < 0.001) as presented in figure 8.
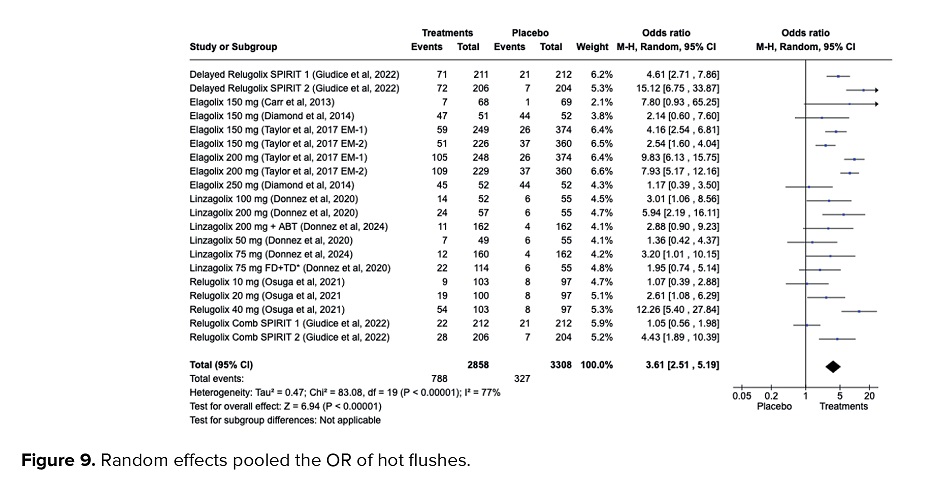
Participants who underwent treatment using GnRH antagonists were more likely to develop hot flushes, 3.61 times higher than placebo groups (OR = 3.61, 95% CI = 2.51, 5.19, p < 0.001). High heterogeneities were detected (I2 = 77%, p < 0.001) as presented in figure 9.
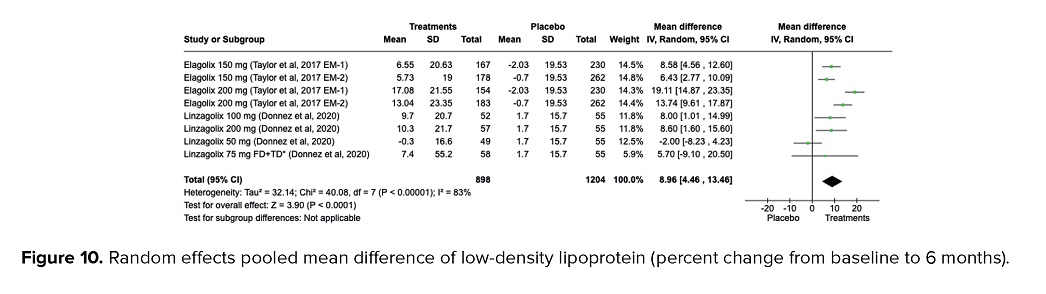
A significant increase of 8.96% was observed in the level of low-density lipoprotein (LDL) after 6 months compared to the baseline in participants receiving treatment using oral GnRH antagonists compared to placebo (MD = 8.96%, 95% CI = 4.46, 13.46, p < 0.001). High heterogeneities were detected (I2 = 83%, p < 0.001) as presented in figure 10.
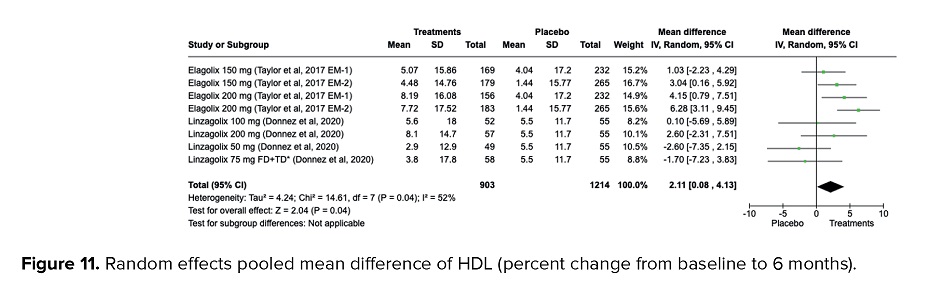
Analyzing the level of high-density lipoprotein (HDL) in participants receiving oral GnRH antagonists, a pooled change of 2.11% increase was observed in the level of HDL compared to placebo after 6 months of treatment (MD = 2.11%, 95% CI = 0.08, 4.13, p = 0.04). High heterogeneities were detected (I2 = 83%, p < 0.001) as presented in figure 11.
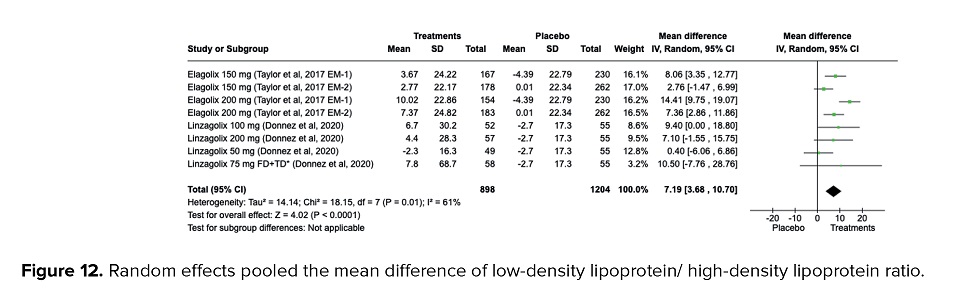
In line with the findings of elevated LDL in the treatment group, there is an expected increase in the ratio of LDL/HDL compared to the baseline after 6 months, since the initiation of therapy (MD = 7.19, 95% CI = 3.68, 10.70, p < 0.001). Moderate heterogeneities were detected (I2 = 61%, p = 0.01) as presented in figure 12.
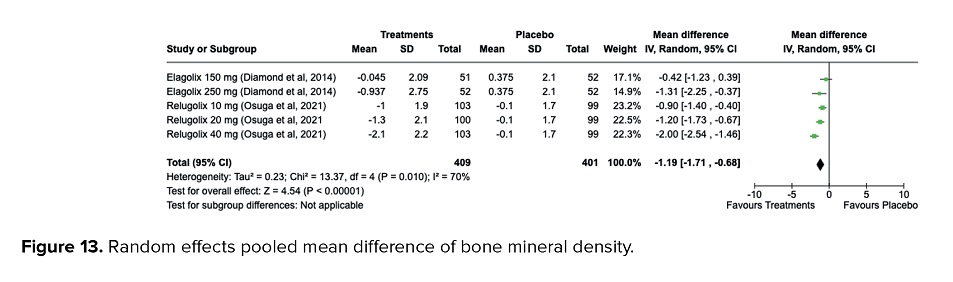
Participants who underwent treatment using GnRH antagonists had significantly lower levels of BMD compared to placebo in spinal assessment (MD = -1.19, 95% CI = -1.71, -0.68, p < 0.001). Moderate heterogeneities were detected in pooled studies assessing spinal BMD (I2 = 70%, p = 0.010), as presented in figure 13.
4. Discussion
4.1. Main findings
Based on a pooled meta-analysis of 7 RCTs, populations treated with GnRH antagonists demonstrated significant reductions in dysmenorrhea, dyspareunia, and NMCPP using VRS and NRS scales, although not with the mB&B score for dyspareunia and NMCPP. As secondary outcomes, women receiving GnRH antagonists reported significantly better health status based on the EHP-5, PGIC, and EHP-30 instruments. However, adverse effects were observed, including a 3.61-fold higher likelihood of hot flushes compared to placebo, an 8.96% increase in LDL after 6 months, a parallel rise in HDL levels, an elevated LDL/HDL ratio from baseline, and significantly lower BMD in spinal assessments.
Endometriosis is a chronic, estrogen-driven inflammatory disorder requiring lifelong management (6). Ongoing research seeks novel therapies that alleviate pain and enhance quality of life. Combined oral contraceptives and progestogens are typically first-line therapies (8, 24, 25), though about one-third of women exhibit progesterone resistance, limiting efficacy. GnRH agonists, used as second-line treatments, are constrained by adverse hypoestrogenic effects and a 6-month duration limit. In contrast, oral GnRH antagonists provide advantages -oral administration, absence of flare effects, dose-dependent E2 suppression, and reversibility- making them promising agents for endometriosis (8-10).
This analysis demonstrates that oral GnRH antagonists significantly alleviate dysmenorrhea across various pain scales. These results align with a prior Cochrane review showing that antagonists are generally safer and associated with fewer side effects than agonists (26). Nonetheless, few direct head-to-head trials exist, as most studies compared various doses against placebo. Notably, the pain reduction in dyspareunia and CNMPP was less pronounced than in dysmenorrhea, possibly due to different underlying mechanisms, including central sensitization, myofascial trigger points, pelvic adhesions, and persistent inflammation (27, 28). Dyspareunia is also affected by individual sexual activity and psychological factors, complicating consistent evaluation (29). Treatment duration may contribute to variability in the mB&B score, as one study had a 12-wk follow-up and another 24 wk (12). Although pain scores declined, results were not always statistically significant. Consistent suppression of serum E2 (20-60 pg/mL) correlated with pain relief (30).
Pain severity remains the key determinant of reduced quality of life in endometriosis. The EHP-5, a validated, concise, participant-reported outcome tool assessing pain, emotional well-being, control, social support, and self-image (31), showed significant improvement among women receiving GnRH antagonists. However, only 2 studies evaluated varying doses of Elagolix across pain domains, indicating a gap in multidimensional assessments. The PGIC, used in trials of Elagolix 150 mg and 250 mg (9), showed participants’ subjective improvements in pain, physical function, and well-being (32, 33). The EHP-30, a broader 30-item instrument evaluating self-image, emotions, pain, social support, and control (34, 35), further confirmed improved health status among women treated with Linzagolix or Relugolix (8-14). Collectively, these outcomes indicate that GnRH antagonists provide effective symptom control with acceptable tolerability, improving quality of life.
Hot flushes were more prevalent among treatment groups, consistent with hypoestrogenic states (36). The Edelweiss 3 trial (11) found a dose-related increase in hot flushes with Linzagolix, which improved with add-back therapy (ABT). Incidences were similar between Linzagolix 75 mg (7.5%) and Linzagolix 200 mg + ABT (6.8%). Comparable patterns were reported with Relugolix (13), showing dose-dependent effects -8.7% at 10 mg and 52.4% at 40 mg- with earlier onset in antagonist groups than leuprorelin users. Similarly, Elaris EM-I and EM-II (14) found significantly higher rates of hot flushes with high-dose Elagolix (200 mg twice daily: 42.3-47.6%) compared to 150 mg once daily (22.6-23.7%).
Lipid metabolism changes, particularly LDL and LDL/HDL ratio elevations, have been noted with oral GnRH antagonists (14). Linzagolix studies (10, 11) confirmed dose-dependent increases in LDL, HDL, and LDL/HDL ratios at 12 and 24 wk. Similar elevations were observed with GnRH agonists, raising concerns about long-term cardiovascular implications. LDL/HDL ratio, a key cardiovascular risk marker, reflects atherogenic balance; persistent elevation suggests increased atherosclerosis potential (30, 37). While the short-term impact remains limited in premenopausal women with low baseline risk, longitudinal data are required to clarify whether these lipid alterations translate into future cardiovascular events or necessitate intervention, especially in patients with metabolic comorbidities.
Reduced BMD in spinal regions among GnRH antagonist users stems from estrogen suppression and subsequent bone resorption. Studies of Elagolix, Linzagolix, and Relugolix consistently report dose- and duration-dependent BMD declines (38, 39). Lumbar spine losses are most pronounced due to high trabecular bone turnover (40). Although ABT mitigates BMD reduction, it cannot entirely prevent bone loss. Recovery occurs partially post-treatment, but long-term effects on fracture risk remain uncertain, emphasizing the need for BMD monitoring, calcium/vitamin D supplementation, and tailored regimens (30, 31).
4.2. Clinical implications
Endometriosis-related pain remains challenging to treat effectively. While GnRH antagonists demonstrate efficacy in pain relief within 12 wk, their hypoestrogenic side effects (hot flushes, BMD reduction, and mood disturbances) can impact quality of life. Nevertheless, their favorable pharmacokinetics and reversibility make them viable for individualized therapy, emphasizing the importance of dose optimization and monitoring.
4.3. Strengths and limitations
This is the first updated meta-analysis since 2024 evaluating RCTs on the efficacy and safety of oral GnRH antagonists for endometriosis-related pain. The inclusion of multiple dosages strengthens reliability. However, several limitations exist. High heterogeneity across studies, stemming from different drug types (Elagolix, Linzagolix, Relugolix), dosages, and treatment durations (8-24 wk); limits comparability. Publication bias was also indicated by an asymmetrical funnel plot. Additionally, data on long-term outcomes and recurrence prevention post-surgery are lacking. Future studies should explore extended therapy effects, recurrence reduction potential, and use advanced analytic methods such as Bayesian network meta-analysis and SUCRA ranking to provide comparative efficacy insights.
5. Conclusion
Oral GnRH antagonists might be effective in managing endometriosis-associated pain, and the efficacy and safety of oral GnRH antagonists were dose-dependent. Moreover, some risks of bias should be considered when interpreting the findings. Future research should investigate the long-term efficacy and safety of oral GnRH antagonists using advanced approaches such as Bayesian network meta-analysis, while also evaluating their role in reducing postoperative endometriosis recurrence beyond pain management outcomes.
Data Availability
The data that support the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
D. Tjahyadi: Conceptualization, study design, methodology, data curation, and manuscript drafting. A. Sudono Riyadi: Methodology, statistical analysis, resources, and critical revision of the manuscript. A. Dewi Nugrahani: Data extraction, project administration, statistical analysis, and manuscript editing. M. Amarullah Ritonga: Validation, data interpretation, and supervision. A. Rachmawati: Validation, data interpretation, and supervision. T. Husnitawati Madjid: Validation, data interpretation, and supervision.
Acknowledgments
No artificial intelligence or assisted technologies were used in the production of the study.
Conflict of Interest
The authors declare that there is no conflict of interest.
Endometriosis is a gynecological condition that affects up to 10% of women of reproductive age (1, 2). Endometriosis is heterogeneous and has one of the main symptoms, which is pain. Classically, there are 3 types of pain associated with endometriosis: dysmenorrhea, dyspareunia, and non-menstrual chronic pelvic pain (NMCPP) (3-5). Endometriosis is an inflammatory condition driven by estrogen. Studies indicate that estrogen plays a crucial role in promoting the growth and inflammation of endometriotic lesions (6).
Endometriosis pain can cause a considerable burden for women and affect their quality of life. Several scales have been used to assess pain scales including the verbal rating scale (VRS), numeric rating scale (NRS), and modified Biberoglu and Behrman (mB&B) scale. Nowadays, there is no consensus on the best method to evaluate pain in endometriosis, and many scales have been used (7).
There are many established treatments for endometriosis-associated pain, such as combined oral hormonal contraceptives, progestin-only formulations (oral or depot injection), and gonadotropin releasing hormone (GnRH) agonists, frequently prescribed by many clinicians around the world (8). First-line therapy to second-line therapy in the form of depot injections currently still have many side effects. The provision of treatment given to an individual needs to consider a number of aspects, including their efficacy and safety (8-14).
GnRH antagonists have emerged as a potential novel therapy for the treatment of endometriosis-associated pain. They act by blocking GnRH receptors, which leads to immediate luteinizing hormone and estrogen suppression (8-12). As a recent breakthrough to manage estrogen levels, minimize residual symptoms, and maintain efficacy in reducing endometriosis-related pain, oral GnRH antagonists (Elagolix, Linzagolix, and Relugolix) have emerged as potential alternatives to control estradiol (E2) levels (8-14). Elagolix showed effectiveness in managing dysmenorrhea and NMCPP with partial estrogen suppression at 150 mg daily (15). Linzagolix, under development, may provide consistent E2 suppression with once-daily dosing (16). Relugolix demonstrated rapid action and effective suppression of E2 and progesterone, with promising results in phase 1 and phase 2 studies (8-14, 17).
These novel treatment options need further study regarding their role in reducing pain intensity in dysmenorrhea, dyspareunia, and NMCPP (7) due to endometriosis as well as secondary side effects of treatment due to hypoestrogen conditions (decreased bone mineral density [BMD] and E2 levels, hot flushes, and dyslipidemia). Endometriosis associated-pain leads to decreased quality of life (18), using many assessment tools such as endometriosis health profile (EHP) -5 instrument, patient global impression of change (PGIC) score, EHP-30 instrument.
Previous meta-analysis studies (18, 19) that specifically assessed the outcomes of oral GnRH antagonists, did not focus on looking at the estimated pool effect of each pain scale (VRS, NRS, or other scales), evaluate secondary effect comprehensively, include studies published since 2023, or use funnel plots in the risk of bias (Rob) assessment of randomized controlled trial (RCT).
This study aimed to evaluate the primary outcome of pain intensity reduction in endometriosis and secondary outcomes in the form of side effects of treatment and quality of life with a systematic review approach and meta-analysis of various updated RCT studies as the best form of evidence-based medicine (level I).
2. Materials and Methods
2.1. Protocol and registration
This systematic review and meta analysis was conducted in line with the Cochrane Handbook for Systematic Reviews of Interventions and complied with the reporting standards set by PRISMA (20).
2.2. Search strategy
We conducted a systematic search of Scopus, PubMed, and Cochrane from inception to July 30th, 2024, with restriction to English only. The search query includes keywords and search phrases that involve “endometriosis”, “elagolix”, “linzagolix”, “relugolix”, “dysmenorrhea”, “pelvic pain”, and “GnRH receptor antagonist” and their synonyms. We tailored the search keywords to the particular requirements of each database. The PRISMA criteria were implemented in our search process. At the initial screening, we found 179 articles based on keywords from 3 electronic databases (Table I).

2.3. Study selection
In this meta-analysis, we considered studies studying the potency of GnRH antagonists as novel treatments for endometrial-associated pain. Population, intervention, comparison, and outcomes (PICO) from this study are: P) women of reproductive age diagnosed with endometriosis based on clinical findings, magnetic resonance imaging (MRI), or intraoperative (laparotomy, laparoscopy) according to European Society of Human Reproduction and Embryology (ESHRE) guidelines; I) administration of oral GnRH antagonists (Elagolix, Linzagolix, and Relugolix); C) placebo; and O) primary outcomes include reduction in pain intensity from dysmenorrhea, dyspareunia, and NMCPP and secondary outcomes include quality of life and hypoestrogenic effects (decreased E2 levels, decreased bone mass density/BMD, hot flushes, dyslipidemia, and other drug side effects).
The detailed inclusion criteria were as follows: 1) RCT studies on the role of oral GnRH antagonists (Elagolix, Linzagolix, and Relugolix) with regular monitoring. 2) Subjects in RCT studies were women of reproductive age diagnosed with endometriosis based on clinical findings, MRI, or intraoperative (laparotomy, laparoscopy) according to ESHRE guidelines (1). 3) Subjects in RCT studies had passed the washout phase and did not experience residual symptoms. 4) Studies in English. 5) Studies published in reputable international journals without period restrictions.
Those studies involving subjects with comorbid chronic diseases such as cardiovascular, liver, kidney, autoimmune, malignancy, history of gynecological surgery, and hematological disorders were excluded. Our meta-analysis also excluded case reports/series, review papers, editorials, comments, meta-analyses, and conference abstracts. Independent evaluations of the pre-selected studies that meet the inclusion criteria were conducted by ASR, ADN, and DT. Any differences of opinion were worked out through conversations with the other authors (THM, AR, and MAR).
2.4. Data extraction and Rob assessment
Data were extracted from the eligible studies, including baseline characteristics such as the first author’s name, the country where the study was conducted, study design, and study participants, treatments, and placebo. Primary outcomes include reduction in pain intensity from dysmenorrhea, dyspareunia, and NMCPP and secondary outcomes include quality of life and hypoestrogenic effects (decreased E2 levels, decreased bone mass density/BMD, hot flushes, dyslipidemia, and other drug side effects). The authors utilized the Rob 2.0 tool to evaluate the potential for bias in each study independently. Any disagreements regarding quality ratings were resolved through discussions among the authors.
2.5. Outcome measures
Endometriosis is diagnosed based on clinical, MRI, or intraoperative findings (laparotomy, laparoscopy) according to ESHRE guidelines. Primary outcomes include a reduction in pain intensity in terms of dysmenorrhea, dyspareunia, and NMCPP. Secondary outcomes include quality of life using several assessment tools such as EHP-5 pain dimension component, PGIC score, and EHP-30 pain domain and hypoestrogenism effects (decreased E2 levels, decreased bone mass density as measured by dual-energy X-Ray absorptiometry, hot flushes, and alterations in lipid profile).
2.6. Ethical Considerations
The protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under the ID: CRD42024579105.
2.7. Statistical Analysis
For statistical analysis, review manager version 5.4 (Revman International, Inc, New York) was used. Dichotomous data were analyzed and pooled as odds ratio (OR) with 95% confidence intervals (95% CI) to measure the effect size. For continuous data, mean difference (MD) was used as an effective measure between the 2 groups. Statistical significance was attained by a p < 0.05 (21-23). The I2 index was used to examine inter-study heterogeneity, and an I2 statistic of < 50% disclosed homogeneity. The OR described in each study is pooled using the generic invariance method with a random effect model. These models were used to pool the overall effect magnitude owing to the fact that the inter-study heterogeneity was low. If inter-study heterogeneity is ³ 50%, the pooled outcomes were computed using a random effect model (21-23).
3. Results
Many of the initially retrieved articles may not have been directly related to the topic of interest, despite meeting the search terms. These were not included in further screening (e.g., in PubMed from 127 articles, 66 of them were not related to the topics, thus total from PubMed was 127-66 = 61).
The primary search results yielded a total of 105 articles; after hand exclusion of case reports/series and review papers and removing duplicates, a total of 88 were screened. After screening the titles and abstracts, full papers were identified for further assessment based on the promulgated inclusion criteria. 61 articles were excluded, while 7 others were review articles and 8 articles had wrong comparator from placebo. Only 12 articles passed the step for full-text eligibility assessment. 2 articles had incomplete data, 2 other articles included other gynecological conditions, and one article included placebo < 12 wk. Ultimately, 7 RCT studies were included in our analysis of systematic review and meta-analysis from different countries. Figure 1 depicts the results of the literature search, including the basis for excluding certain publications. The baseline characteristics of the included studies in the final review were presented in table II (8-14).

3.1. Study characteristics
In terms of study design and country of origin, the included studies in this article varied widely worldwide (Table II). All of the studies included are RCTs. Based on table II (8-14). The types of oral GnRH antagonist treatments vary in types (Elagolix, Linzagolix, and Relugolix) and dosage. Each experiment also has quite varied inclusion and exclusion criteria with fairly homogeneous primary and secondary outcomes. The majority of studies have a similar follow-up duration.


Based on the RoB 2.0 assessment (Figure 2), the majority of the studies were considered to have a low Rob. D4 (bias in measurement outcome data) dominated the moderate Rob domain. The D1 (bias arising from the process of randomization), D2 (bias in deviations of intended intervention), D3 (bias due to missing outcome data), and D5 (bias in the selection of reported results) bias domains were the low-level Rob domains. None of the studies had a significant Rob.

3.2. Publication bias
Publication bias assessed by funnel plot showed an asymmetrical figure; hence, there should be some publication biases in each of the predictors analyzed that should be taken into account.
3.3. Primary outcomes
A total of 7 RCTs evaluated the potency of GnRH antagonists for endometriosis-associated pain. Our merged analysis suggested that the population treated with GnRH antagonists had a significant reduction of dysmenorrhea, dyspareunia, and NMCPP using VRS and NRS scales, but not with mB&B scores for dyspareunia and NMCPP (Figures 3-5).

Participants who underwent treatment using GnRH antagonists had a significant reduction of dysmenorrhea using VRS (MD = -0.83, 95% CI = -1.14, -0.52, p < 0.001), NRS (MD = -1.64, 95% CI = -2.21, -1.07, p < 0.001), and mB&B score (MD = -1.64, 95% CI = -2.21, -1.07, p < 0.001) (Figure 3). High heterogeneities were detected in VRS group (I2 = 100%, p < 0.001), NRS group (I2 = 100%, p < 0.001), and mB&B score (I2 = 93%, p < 0.001), respectively.
From the dyspareunia reduction aspect, women who underwent treatment using GnRH antagonists had a significant reduction of dyspareunia using VRS (MD = -0.19, 95% CI = -0.27, -0.12, p < 0.001) and NRS (MD = -0.53, 95% CI = -0.65, -0.40, p < 0.001). Nonetheless, no significant difference was observed in reduction of dyspareunia in participants assessed with MB&B Score (MD = -0.12, 95% CI = -0.37, 0.14, p = 0.37). High heterogeneities were detected in VRS group (I2 = 99%, p < 0.001), NRS group (I2 = 98%, p < 0.001), and MB&B Score (I2 = 91%, p < 0.001), respectively (Figure 4).

Women who underwent treatment using GnRH antagonists had a significant reduction of NMCPP using VRS (MD = -0.25, 95% CI = -0.36, -0.15, p < 0.001) and NRS (MD = -0.64, 95% CI = -0.79, -0.48, p < 0.001). However, for the mB&B score, despite a reduction of -0.13 in MD, the effect size failed to attain statistical significance (95% CI = -0.28, 0.02, p = 0.09). High heterogeneities were detected in VRS group (I2 = 100%, p < 0.001), NRS group (I2 = 98%, p < 0.001), and mB&B score (I2 = 93%, p < 0.001), respectively (Figure 5).
3.4. Secondary outcomes
Scores on the EHP-5 score and modular questionnaire are then transformed on a scale of 0 (indicating best possible health status) to 100 (indicating worst possible health status) (18). As secondary outcomes, women who underwent treatment using GnRH antagonists had a significantly higher possibility of health status using the EHP-5 instrument (MD = -11.28, 95% CI = -15.96, -6.60, p < 0.001). High heterogeneities were detected (I2 = 98%, p < 0.001) as presented in figure 6.


The self-report measures a PGIC and reflects an individual's belief about the efficacy of treatment. Lower levels showed much improvement. In this study, women who underwent treatment using GnRH antagonists had a significant improvement in the treatment arms with PGIC score (MD = -1.00, 95% CI = -1.04, -0.96, p < 0.001). No heterogeneities were detected (I2 = 0%, p = 1.00) as presented in figure 7.

Moreover, women who underwent treatment using GnRH antagonists had a significantly higher possibility of health status using the EHP-30 instrument (MD = -13.09, 95% CI = -14.57, -11.61, p < 0.001). High heterogeneities were detected (I2 = 98%, p < 0.001) as presented in figure 8.

Participants who underwent treatment using GnRH antagonists were more likely to develop hot flushes, 3.61 times higher than placebo groups (OR = 3.61, 95% CI = 2.51, 5.19, p < 0.001). High heterogeneities were detected (I2 = 77%, p < 0.001) as presented in figure 9.
A significant increase of 8.96% was observed in the level of low-density lipoprotein (LDL) after 6 months compared to the baseline in participants receiving treatment using oral GnRH antagonists compared to placebo (MD = 8.96%, 95% CI = 4.46, 13.46, p < 0.001). High heterogeneities were detected (I2 = 83%, p < 0.001) as presented in figure 10.
Analyzing the level of high-density lipoprotein (HDL) in participants receiving oral GnRH antagonists, a pooled change of 2.11% increase was observed in the level of HDL compared to placebo after 6 months of treatment (MD = 2.11%, 95% CI = 0.08, 4.13, p = 0.04). High heterogeneities were detected (I2 = 83%, p < 0.001) as presented in figure 11.
In line with the findings of elevated LDL in the treatment group, there is an expected increase in the ratio of LDL/HDL compared to the baseline after 6 months, since the initiation of therapy (MD = 7.19, 95% CI = 3.68, 10.70, p < 0.001). Moderate heterogeneities were detected (I2 = 61%, p = 0.01) as presented in figure 12.




Participants who underwent treatment using GnRH antagonists had significantly lower levels of BMD compared to placebo in spinal assessment (MD = -1.19, 95% CI = -1.71, -0.68, p < 0.001). Moderate heterogeneities were detected in pooled studies assessing spinal BMD (I2 = 70%, p = 0.010), as presented in figure 13.

4. Discussion
4.1. Main findings
Based on a pooled meta-analysis of 7 RCTs, populations treated with GnRH antagonists demonstrated significant reductions in dysmenorrhea, dyspareunia, and NMCPP using VRS and NRS scales, although not with the mB&B score for dyspareunia and NMCPP. As secondary outcomes, women receiving GnRH antagonists reported significantly better health status based on the EHP-5, PGIC, and EHP-30 instruments. However, adverse effects were observed, including a 3.61-fold higher likelihood of hot flushes compared to placebo, an 8.96% increase in LDL after 6 months, a parallel rise in HDL levels, an elevated LDL/HDL ratio from baseline, and significantly lower BMD in spinal assessments.
Endometriosis is a chronic, estrogen-driven inflammatory disorder requiring lifelong management (6). Ongoing research seeks novel therapies that alleviate pain and enhance quality of life. Combined oral contraceptives and progestogens are typically first-line therapies (8, 24, 25), though about one-third of women exhibit progesterone resistance, limiting efficacy. GnRH agonists, used as second-line treatments, are constrained by adverse hypoestrogenic effects and a 6-month duration limit. In contrast, oral GnRH antagonists provide advantages -oral administration, absence of flare effects, dose-dependent E2 suppression, and reversibility- making them promising agents for endometriosis (8-10).
This analysis demonstrates that oral GnRH antagonists significantly alleviate dysmenorrhea across various pain scales. These results align with a prior Cochrane review showing that antagonists are generally safer and associated with fewer side effects than agonists (26). Nonetheless, few direct head-to-head trials exist, as most studies compared various doses against placebo. Notably, the pain reduction in dyspareunia and CNMPP was less pronounced than in dysmenorrhea, possibly due to different underlying mechanisms, including central sensitization, myofascial trigger points, pelvic adhesions, and persistent inflammation (27, 28). Dyspareunia is also affected by individual sexual activity and psychological factors, complicating consistent evaluation (29). Treatment duration may contribute to variability in the mB&B score, as one study had a 12-wk follow-up and another 24 wk (12). Although pain scores declined, results were not always statistically significant. Consistent suppression of serum E2 (20-60 pg/mL) correlated with pain relief (30).
Pain severity remains the key determinant of reduced quality of life in endometriosis. The EHP-5, a validated, concise, participant-reported outcome tool assessing pain, emotional well-being, control, social support, and self-image (31), showed significant improvement among women receiving GnRH antagonists. However, only 2 studies evaluated varying doses of Elagolix across pain domains, indicating a gap in multidimensional assessments. The PGIC, used in trials of Elagolix 150 mg and 250 mg (9), showed participants’ subjective improvements in pain, physical function, and well-being (32, 33). The EHP-30, a broader 30-item instrument evaluating self-image, emotions, pain, social support, and control (34, 35), further confirmed improved health status among women treated with Linzagolix or Relugolix (8-14). Collectively, these outcomes indicate that GnRH antagonists provide effective symptom control with acceptable tolerability, improving quality of life.
Hot flushes were more prevalent among treatment groups, consistent with hypoestrogenic states (36). The Edelweiss 3 trial (11) found a dose-related increase in hot flushes with Linzagolix, which improved with add-back therapy (ABT). Incidences were similar between Linzagolix 75 mg (7.5%) and Linzagolix 200 mg + ABT (6.8%). Comparable patterns were reported with Relugolix (13), showing dose-dependent effects -8.7% at 10 mg and 52.4% at 40 mg- with earlier onset in antagonist groups than leuprorelin users. Similarly, Elaris EM-I and EM-II (14) found significantly higher rates of hot flushes with high-dose Elagolix (200 mg twice daily: 42.3-47.6%) compared to 150 mg once daily (22.6-23.7%).
Lipid metabolism changes, particularly LDL and LDL/HDL ratio elevations, have been noted with oral GnRH antagonists (14). Linzagolix studies (10, 11) confirmed dose-dependent increases in LDL, HDL, and LDL/HDL ratios at 12 and 24 wk. Similar elevations were observed with GnRH agonists, raising concerns about long-term cardiovascular implications. LDL/HDL ratio, a key cardiovascular risk marker, reflects atherogenic balance; persistent elevation suggests increased atherosclerosis potential (30, 37). While the short-term impact remains limited in premenopausal women with low baseline risk, longitudinal data are required to clarify whether these lipid alterations translate into future cardiovascular events or necessitate intervention, especially in patients with metabolic comorbidities.
Reduced BMD in spinal regions among GnRH antagonist users stems from estrogen suppression and subsequent bone resorption. Studies of Elagolix, Linzagolix, and Relugolix consistently report dose- and duration-dependent BMD declines (38, 39). Lumbar spine losses are most pronounced due to high trabecular bone turnover (40). Although ABT mitigates BMD reduction, it cannot entirely prevent bone loss. Recovery occurs partially post-treatment, but long-term effects on fracture risk remain uncertain, emphasizing the need for BMD monitoring, calcium/vitamin D supplementation, and tailored regimens (30, 31).
4.2. Clinical implications
Endometriosis-related pain remains challenging to treat effectively. While GnRH antagonists demonstrate efficacy in pain relief within 12 wk, their hypoestrogenic side effects (hot flushes, BMD reduction, and mood disturbances) can impact quality of life. Nevertheless, their favorable pharmacokinetics and reversibility make them viable for individualized therapy, emphasizing the importance of dose optimization and monitoring.
4.3. Strengths and limitations
This is the first updated meta-analysis since 2024 evaluating RCTs on the efficacy and safety of oral GnRH antagonists for endometriosis-related pain. The inclusion of multiple dosages strengthens reliability. However, several limitations exist. High heterogeneity across studies, stemming from different drug types (Elagolix, Linzagolix, Relugolix), dosages, and treatment durations (8-24 wk); limits comparability. Publication bias was also indicated by an asymmetrical funnel plot. Additionally, data on long-term outcomes and recurrence prevention post-surgery are lacking. Future studies should explore extended therapy effects, recurrence reduction potential, and use advanced analytic methods such as Bayesian network meta-analysis and SUCRA ranking to provide comparative efficacy insights.
5. Conclusion
Oral GnRH antagonists might be effective in managing endometriosis-associated pain, and the efficacy and safety of oral GnRH antagonists were dose-dependent. Moreover, some risks of bias should be considered when interpreting the findings. Future research should investigate the long-term efficacy and safety of oral GnRH antagonists using advanced approaches such as Bayesian network meta-analysis, while also evaluating their role in reducing postoperative endometriosis recurrence beyond pain management outcomes.
Data Availability
The data that support the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
D. Tjahyadi: Conceptualization, study design, methodology, data curation, and manuscript drafting. A. Sudono Riyadi: Methodology, statistical analysis, resources, and critical revision of the manuscript. A. Dewi Nugrahani: Data extraction, project administration, statistical analysis, and manuscript editing. M. Amarullah Ritonga: Validation, data interpretation, and supervision. A. Rachmawati: Validation, data interpretation, and supervision. T. Husnitawati Madjid: Validation, data interpretation, and supervision.
Acknowledgments
No artificial intelligence or assisted technologies were used in the production of the study.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Review Article |
Subject:
Reproductive Endocrinology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





