Mon, May 25, 2026
[Archive]
Volume 23, Issue 12 (December 2025)
IJRM 2025, 23(12): 985-994 |
Back to browse issues page
Ethics code: No. 39 of May 22, 2014
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Efremova O, Ponomarenko I, Churnosov M, Efremova O. Association of polymorphic loci of the matrix metalloproteinase genes with the development of fetal growth retardation: A case-control study. IJRM 2025; 23 (12) :985-994
URL: http://ijrm.ir/article-1-3570-en.html
URL: http://ijrm.ir/article-1-3570-en.html
1- Department of Biomedical Disciplines, Belgorod State National Research University, Belgorod, Russia. , efremovaolesya@gmail.com; efremova@bsuedu.ru
2- Department of Biomedical Disciplines, Belgorod State National Research University, Belgorod, Russia.
3- Department of Faculty Therapy, Belgorod State National Research University, Belgorod, Russia.
2- Department of Biomedical Disciplines, Belgorod State National Research University, Belgorod, Russia.
3- Department of Faculty Therapy, Belgorod State National Research University, Belgorod, Russia.
Full-Text [PDF 407 kb]
(337 Downloads)
| Abstract (HTML) (323 Views)
Full-Text: (47 Views)
1. Introduction
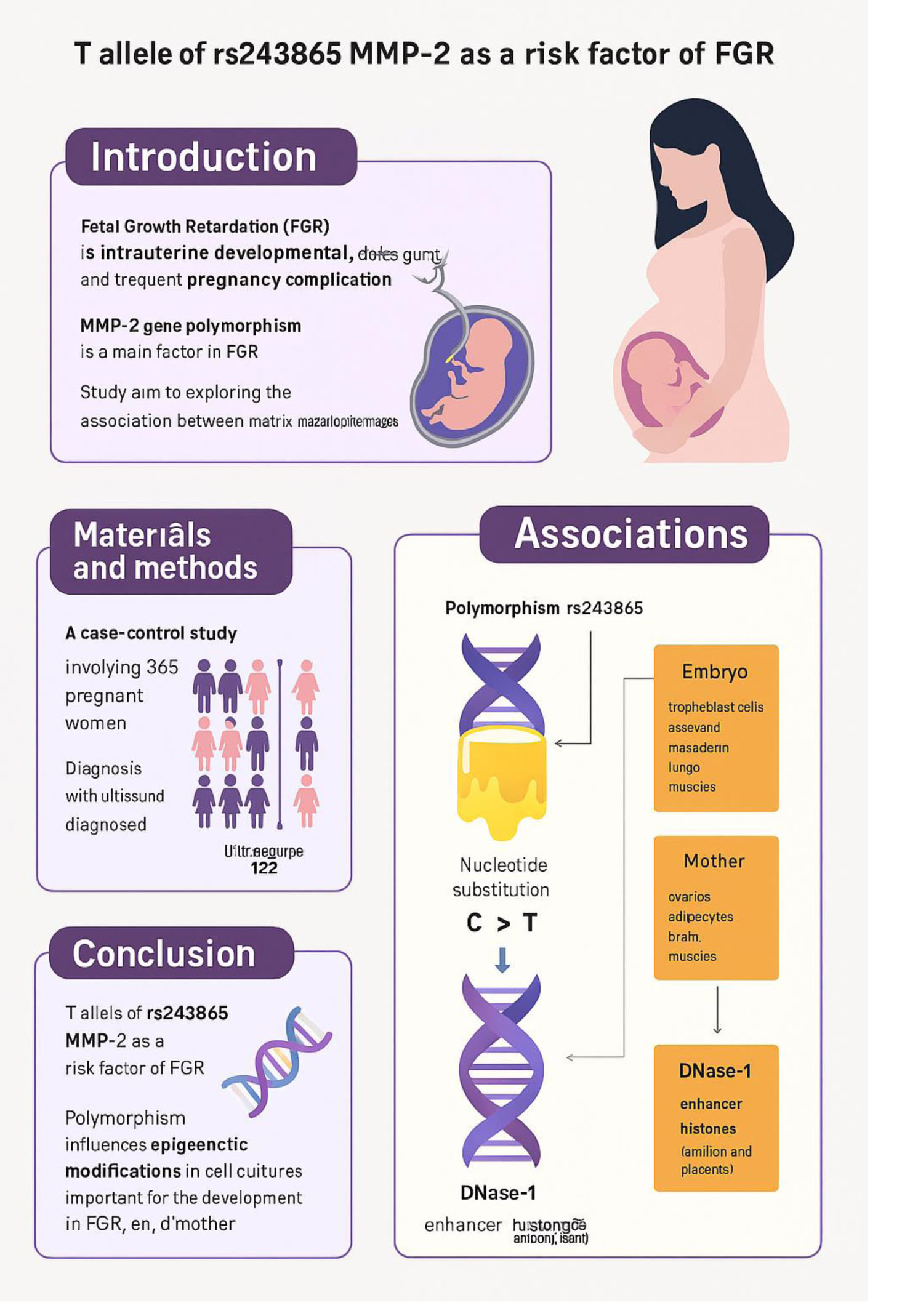
Fetal growth retardation (FGR), also known as intrauterine developmental delay, is a frequent complication of pregnancy that is associated with many adverse perinatal outcomes (1). FGR is generally defined as the failure of the fetus to reach its full genetically determined growth potential and involves a fetal weight less than the 10th percentile for gestational age on prenatal ultrasound assessment. The fetus is hypoxic and/or hypotrophic due to inadequate oxygen and/or nutrition supply due to a variety of causes, one of which is placental insufficiency (2-4). FGR occurs in approximately 10% of pregnancies (3).
In recent years, there has been disagreement about the definition of FGR and fetuses small for gestational age. Nevertheless there is general agreement among the guidelines reviewed about the importance of early universal FGR risk stratification for effective screening, care, prevention, and treatment interventions.
The etiology of FGR is complex, and various maternal, fetal, and placental factors are involved in its development (3-7). One of the important factors affecting fetal growth and development is matrix metalloproteinases (MMPs). The MMPs are a subfamily of zinc-dependent endopeptidases involved in the degradation of the extracellular matrix (5). Although MMPs are mainly characterized in tumor biology, they also play a crucial role during pregnancy (8, 9). Placental MMPs are essential for cytotrophoblast migration and invasion into the uterine wall, as well as for spiral artery remodeling. They are involved in cytotrophoblast fusion to form syncytiotrophoblast, as well as in angiogenesis (9). MMPs expression levels, as well as tissue inhibitors of metalloproteases expression levels, are closely associated with placental insufficiency. Studies have shown that decidual stromal cells expressing cluster of differentiation 82 upregulate tissue inhibitor of metalloproteinases-1 expression in an autocrine manner and inhibit human trophoblast cell invasiveness in the first trimester, partly through the integrin β1/mitogen-activated protein kinase signaling pathway. All these processes are crucial for the establishment and maintenance of a successful pregnancy, and an imbalance in these processes can lead to various abnormalities in the course of pregnancy (9, 10). Disruption of MMPs expression has been shown to be associated with pregnancy complications such as placental insufficiency, pre-eclampsia, FGR, gestational diabetes mellitus, and is also observed in maternal obesity (8, 10, 11). It should be noted that there is little data on the association of MMP polymorphisms with FGR, and thus, further research in this area is warranted.
Hereby, we aimed to investigate the association of MMPs gene polymorphisms with the development of FGR.
2. Materials and Methods
2.1. Design and participants
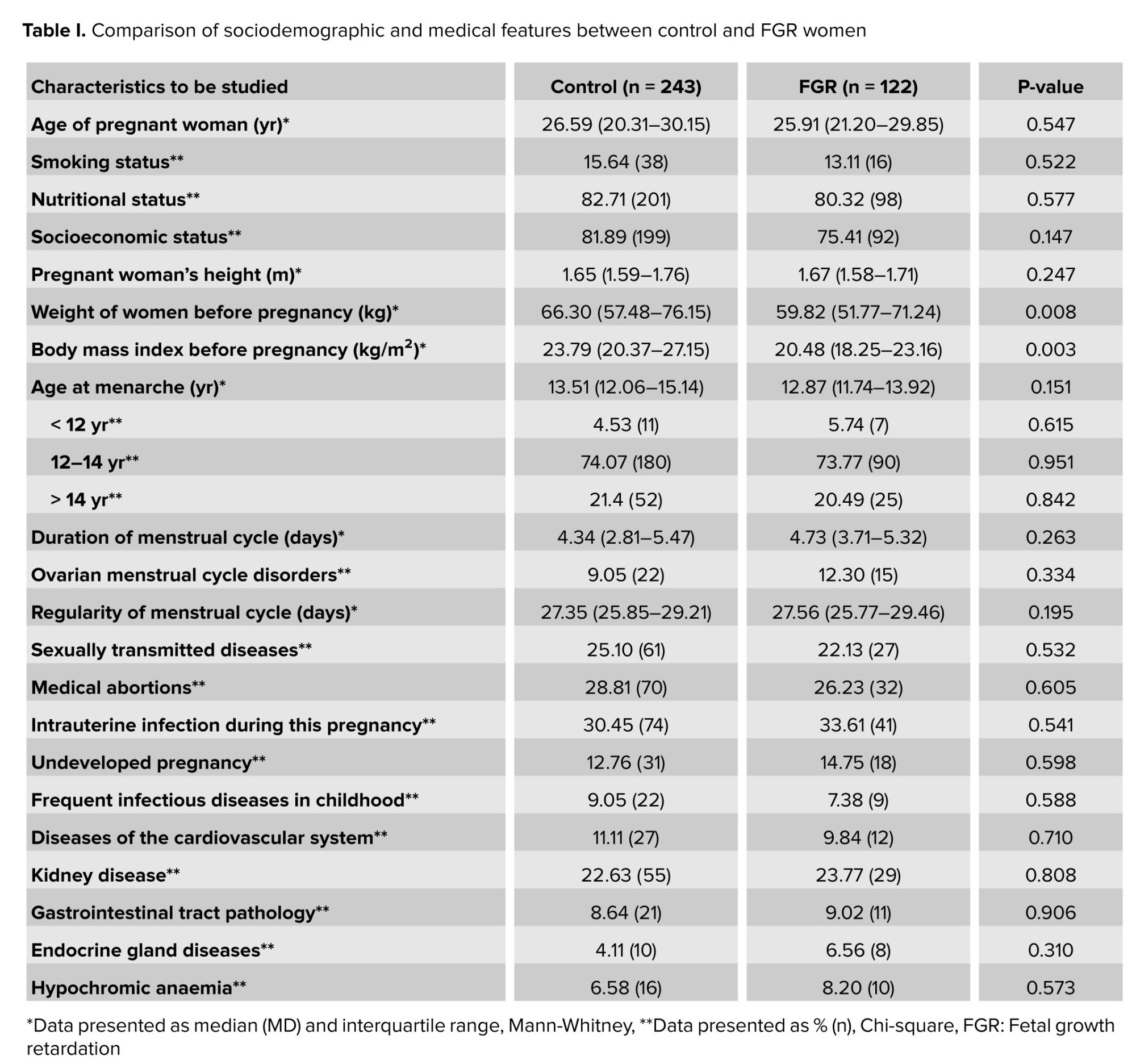
A case-control study was carried out involving a total of 365 pregnant women in the third trimester of pregnancy from February 2015-2022 at the Medical Institute of Belgorod National Research University, Belgorod, Russia. In addition to medical history collection, standard clinical and biochemical parameters were studied in these women. The study was conducted in the Perinatal Center of the Belgorod Regional Clinical hospital of St. Joasaph, Belgorod, Russia. The study consisted of 122 pregnant women with FGR and 243 pregnant women with normal birth weight.
Inclusion criteria: Russian nationality, place of birth and residence in the Central part of Russia (12-15), third trimester of spontaneous singleton pregnancy. The group of pregnant women with FGR included individuals with a confirmed diagnosis of FGR. The diagnosis of FGR was established by obstetricians using ultrasound fetometry of the fetus before birth (TOSHIBA XARIO SSA-660A) and postnatal fetal data (16, 17). The control group consisted of pregnant women with a normally developing fetus. Exclusion criteria: severe chronic diseases, multiple pregnancies, diabetes mellitus, congenital diseases, and fetal malformations, and refusal to participate in the study. The clinical characteristics of pregnant women were based on previously published results (18) and are presented in table I. Venous blood was drawn from all observed women for the isolation of deoxyribonucleic acid (DNA) by the phenol-chloroform method (methodology described previously) (19).
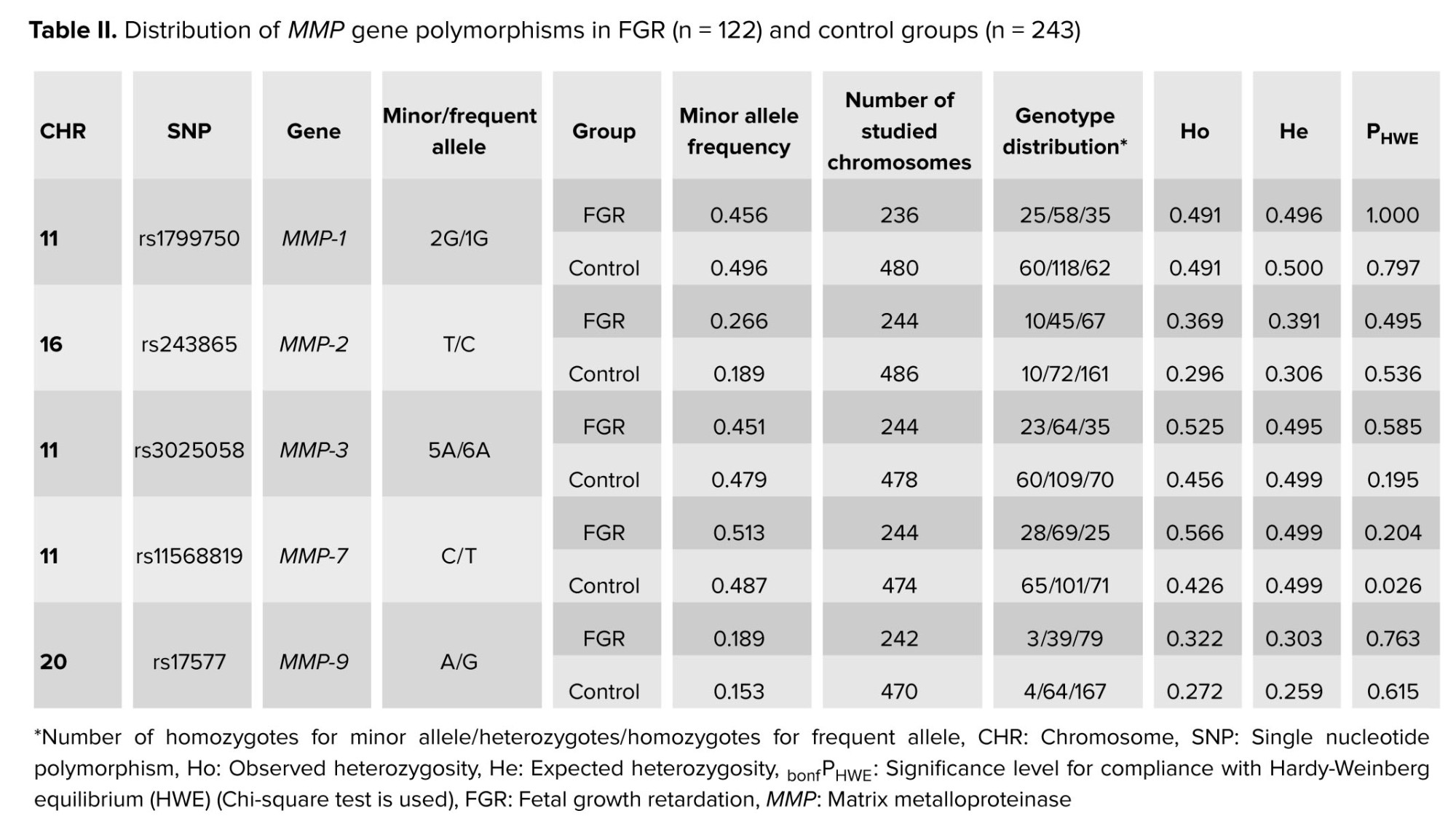
5 single nucleotide polymorphisms (SNPs) in metalloprotease genes with significant regulatory potential were selected for analysis: rs1799750 of the MMP-1 gene, rs243865 of the MMP-2 gene, rs3025058 of the MMP-3 gene, rs11568819 of the MMP-7 gene, and rs17577 of the MMP-9 gene. Genotyping was performed using TaqMan probe-based real-time polymerase chain reaction with specific primers and probes. To ensure accuracy, negative control reactions were included, and 3% of samples were randomly re-genotyped in a blind manner for quality control, following established protocols (20).
2.2. Sample size
The sample size was determined using data on the incidence of FGR in previous studies (1-5) and the available data on frequencies in the European population (1000 Genomes project data), as well as using the Pocock formula. In this case, the percentage of errors (α = 0.05; β = 0.2) taken from the table and the loss of the sample of the main group (P1 = 0.05) and the control group (P2 = 0.2) were taken into account. The minimum sample size in each group was estimated as 106 individuals with a 10% probability of dropout. In our study, the main group included 122 women and the control group 243, which provided a statistical power of 0.80 at the α = 0.05 significance level.
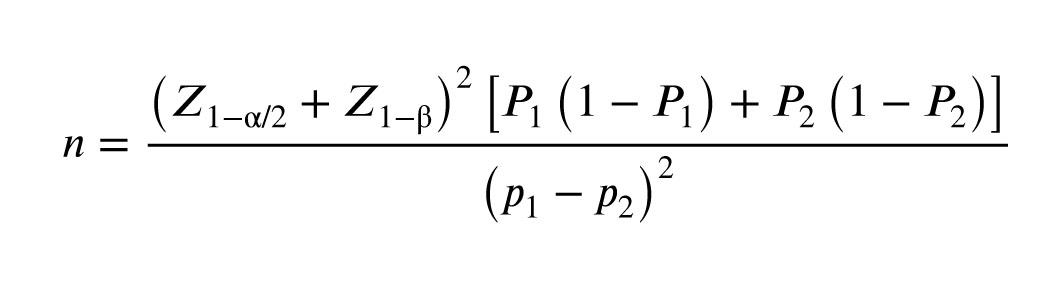
2.3. Ethical Considerations
The study was approved by the Ethical Commission of the Medical Institute of Belgorod National Research University, Belgorod, Russia (Opinion No. 54, Ethical Commission Protocol No. 39 of May 22, 2014). Informed consent was obtained from all the participants, and they were also informed about the purpose of the study.
2.4. Statistical Analysis
The associations of polymorphic loci with FGR within the 4 genetic models were analyzed using a software package (gPLINK v2.050) (21), taking into account covariates (age, pre-pregnancy weight, pre-pregnancy body mass index) and correction for multiple comparisons (permutation testing) (22). The Pperm indicator obtained as a result of permutation testing ≤ 0.050 was taken as statistically significant. When assessing the distribution compliance of the studied loci for Hardy-Weinberg equilibrium (HWE) distribution, a Bonferroni correction was introduced, which was equal to the number of loci studied (n = 5). Thus, the statistically significant level in this analysis (HWE compliance assessment) was bonfPHWE ≤ 0.01 (0.05/5). All analyzed loci, both in the FGR group and in the control, "fulfilled" HWE (bonfPHWE ≥ 0.026) and were included in our association analysis. In logistic regression, odds ratios (OR) and their 95% confidence interval (CI) were calculated. Various bioinformatics resources, HaploReg (v4.1) (23); GTExportal (24), 3′aQTL-atlas (25), were used to assess the functional potential of the polymorphic locus rs243865 of the MMP-2 gene, which showed significant associations with FGR.
3. Results
The analysis of clinical and anamnestic data of the study participants showed that the groups of pregnant women with FGR (n = 122) and the control group (n = 243) did not differ from each other in the vast majority of indicators considered (Table I). Women with FGR had lower weight (p = 0.008) and body mass index before pregnancy (p = 0.003) (Table I), and therefore these parameters (and the woman's age) were used as covariates in genetic calculations. Neonatal birth weight in the main group was 2647.26 ± 621.15 gr, and in the control group was 3463.26 ± 438.26 gr (p = 0.007). The height of newborns in the main group was 41.27 ± 2.41 cm and in the control group, 54.51 ± 2.26 cm (p = 0.081). For the remaining indicators, no statistically significant differences were found between the groups (Table I).
Minor allele frequency was above 5% for all SNPs studied, both in the group of pregnant women with FGR and in the control group. In both study groups, the observed genotype distribution was analyzed, which showed no deviations from the expected HWE distribution for all loci examined, with (observed parameters p [HWE] > 0.05/5 > 0.01 [considered Bonferroni correction for the number of loci examined], Table II).
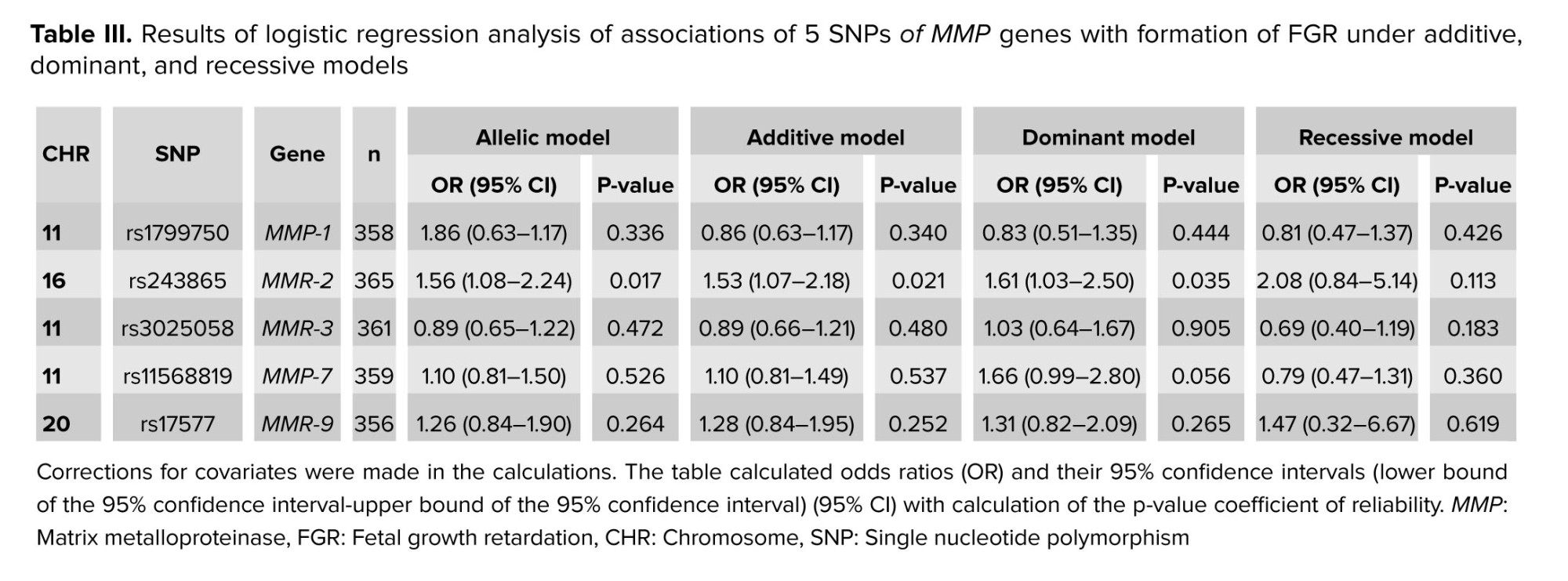
The analysis of associations of alleles of 5 polymorphic loci of MMP genes with the development of FGR showed that the T allele of rs243865 of the MMP-2 gene (OR = 1.56, 95% CI: 1.08-2.24, p = 0.017, pperm = 0.016, Nperm = 1256) was significantly associated with the development of FGR (Table III). The polymorphism rs243865 of the MMP-2 gene was also found to be related to the development of FGR within the additive (OR = 1.53, 95% CI: 1.07-2.18, p = 0.021, pperm = 0.015, Nperm = 1302, power = 78.03%) and dominant (OR = 1.61, 95% CI: 1.03-2.50, p = 0.035, pperm = 0.027, Nperm = 745, power = 72.58%) models (Table III).
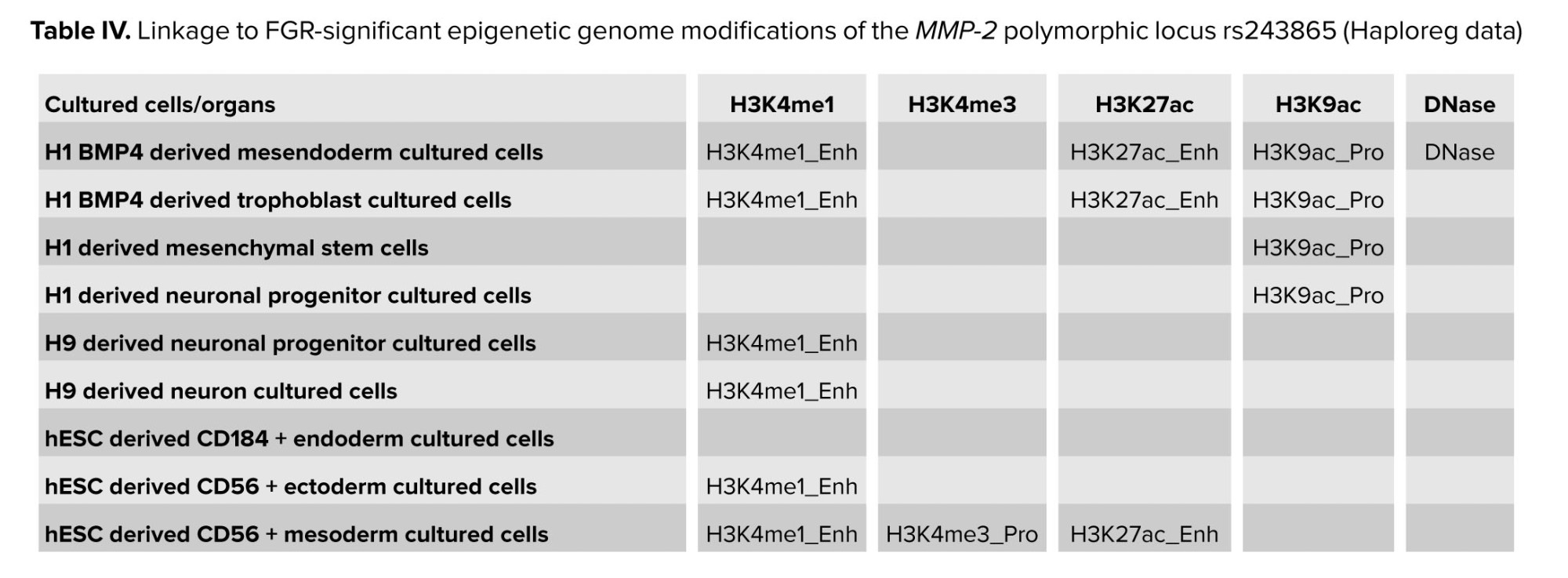
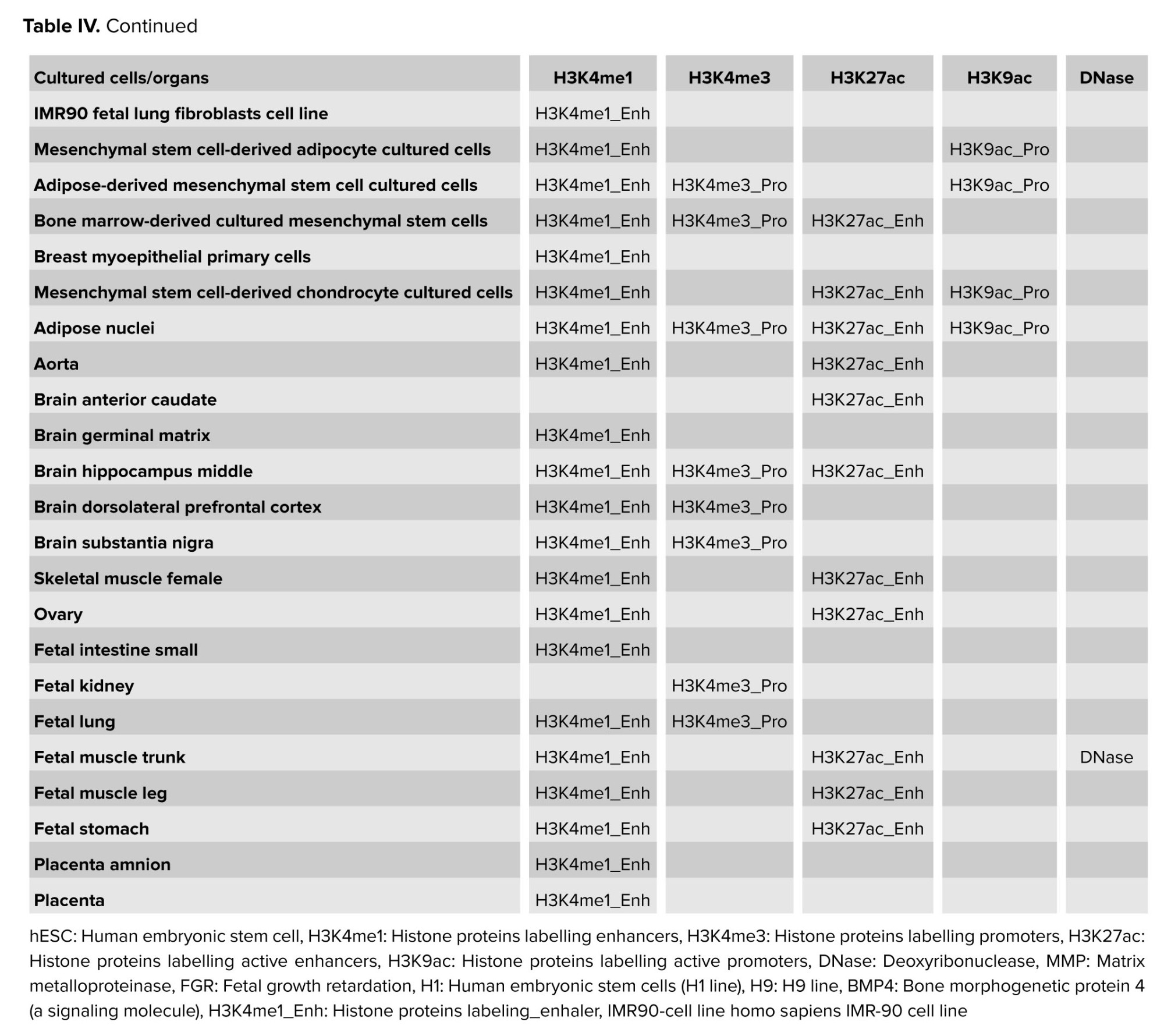
It should be noted that the polymorphic locus rs243865, which shows a major effect in the development of FGR (T allele is a risk factor OR = 1.53-1.61), according to Haploreg data, is located in the region of histone marking promoters and enhancers in many different cell cultures or organs (Table IV). It is located in the deoxyribonuclease-1 (DNase-1) hypersensitivity region and affects the affinity of DNA to myogenic factor (Myf) transcription (nucleotide substitution of C˃T rs243865 leads to decreased affinity of the genome region of the MMP-2 gene to Myf). Polymorphism affects epigenetic modifications in cell cultures and organs, which are important for the development of FGR, both in the embryo/fetus (cultures of trophoblast, ectoderm, mesoderm, etc., and lungs, muscles, etc.) and in the mother's body (ovaries, adipocytes, brain, muscles, etc.). The location of rs243865 in the enhancer region in the amnion and placenta tissues is extremely important for the occurrence of FGR (Table IV).
According to genotype-tissue expression portal data, rs243865 of the MMP-2 gene is associated with the expression level of the RP11-212I21.2 gene in arteries (p < 0.001), with the T allele variant of rs243865 increasing the transcriptional activity of this gene (NES = 0.27). According to the atlas of alternative polyadenylation quantitative trait loci (3′aQTL-atlas) the rs243865 MMP-2 polymorphism is associated with polyadenylation of the lysophosphatidylcholine acyltransferase 2 (LPCAT2) gene in thyroid (p < 0.001) and peripheral blood (p < 0.001).
4. Discussion
The study revealed that the T allele rs243865 of the MMP-2 gene is a risk factor for the development of FGR (OR = 1.53-1.61). This polymorphism is important for fetal development; it influences the development of various organs and systems (25). The location of rs243865 of the MMP-2 gene in the enhancer region in amnion and placental tissues is extremely important for the occurrence of intrauterine growth retardation (26).
MMP2 is an enzyme that can influence the degradation of some components of the extracellular matrix during pregnancy (9). This metalloproteinase is capable of cleaving gelatin, type I and IV collagens, and is an important regulator of uterine and spiral arteries remodeling. A decrease in the number, size, and dilation of uterine spiral arteries has been observed with a decrease in MMP-2, as well as an increase in collagen-IV in the extracellular matrix, which leads to decrease in uterine vascularization and contributes to progressive uteroplacental ischemia (9, 27). “The mechanical force of blood flow is a fundamental determinant of vascular homeostasis. The fetus depends on placental perfusion to meet its circulatory and metabolic needs. Disruption of normal vascular adaptation and the mechanisms that ensure a reciprocal interaction between fetoplacental and maternal circulation can lead to FGR” (26). Experimental studies in rats have demonstrated that MMP-2 is expressed in the endometrial stroma and during the first days of pregnancy. The messenger of ribonucleic acid expression of this gene changes dramatically and is observed mainly in the primary decidual zone and vascular endothelium (9). Thus, MMPs play a vital role in the regulatory during implantation and placentation as well as for successful pregnancy.
The polymorphic variant of MMP2 affects the affinity of DNA to the Myf transcription factor, which is associated with RP11-212I21 gene expression 2 in arteries (FGR-risk allele T rs243865 decreases DNA affinity to Myf transcription factor and increases RP11-212I21.2 gene transcription) and the level of polyadenylation gene LPCAT2 in thyroid and peripheral blood. The protein product of the LPCAT2 gene, LPCAT2, plays an important role in embryonic development. A study by Kondakova et al. showed the involvement of the LPCAT2 gene in estrogen-stimulated cell cycle progression. Estrogens have positive or negative effects on the regulation of gonadotropin secretion, folliculogenesis, and ovulation. Therefore, any changes in the LPCAT2 gene expression may lead to structural changes in the ovaries and altered follicular growth. Additionally, estrogen receptors are interconnected with the ubiquitin-proteasome system, which plays an important role in embryo implantation. It has been shown to be involved in some key processes of trophoblastic invasion, angiogenesis, extracellular matrix degradation, immune tolerance, and glandular secretion (28).
The impact of the rs243865 polymorphism of the MMP2 gene on female reproductive organs has been demonstrated in previous studies. This polymorphism has been associated with primary ovarian failure in Iranian women under 35 yr of age (29), and its association with recurrent miscarriage has been studied in a systematic review, meta-analysis, and bioinformatics analysis (30). Still, no clear associations of this polymorphism with pregnancy loss have been obtained. No evidence has been observed of an association between MMP2 (rs2285053 and rs243865) and recurrent pregnancy loss in the Polish population (31). The association of MMP-2 and MMP-9 polymorphisms with obesity and mammographic features has been proven (32, 33).
Our study demonstrated the clinical significance of the rs243865 polymorphism in the MMP2 gene for gynecologists and obstetricians. We also proved the connection with epigenetic genomic modifications, significant for FGR of the polymorphic locus MMP-2 rs243865 according to Haploreg data. Thus, this locus is responsible for the development of many extracellular matrix cells, derived neuronal, endodermal, and mesodermal cultured cells, organs, and tissues of the developing fetus, and maternal placenta conditions.
4.1. Strengths and limitations
Our study has several limitations: (a) the functional effects of loci associated with FGR, suggested in the work based on in silico analysis, require experimental confirmation in vivo/in vitro; (b) in this study, we did not determine the levels of MMP2 in women with FGR and in controls (the polymorphism of which, according to our data, is associated with FGR), which would have allowed us to demonstrate the association between MMP and FGR.
5. Conclusion
This study suggests that the T allele of rs243865 of the MMP-2 gene is a risk factor for the development of FGR (OR = 1.53-1.61). According to Haploreg data, this polymorphic variant is located in the histone region marking promoters and enhancers in many different cell cultures and organs; is located in the region of hypersensitivity to DNase-1 and affects the affinity of DNA for the transcription factor Myf (nucleotide substitution C˃T rs243865 leads to a decrease in the affinity of the genomic region in the region of the MMP-2 gene for the transcription factor Myf). Further academic studies should confirm these findings and clarify the complex relationship between FGR and maternal metalloproteinase polymorphisms.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
O. Efremova, I. Ponomarenko and M. Churnosov designed the study and conducted the research. O. Efremova and I. Ponomarenko monitored, evaluated, and analyzed the results of the study. Further, O. Efremova and M. Churnosov reviewed the article. All authors approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgments
The work was carried out without external sources of funding. Artificial intelligence was used for graphical visualization of the annotation.
Conflict of Interest
The authors declare that there is no conflict of interest.
Fetal growth retardation (FGR), also known as intrauterine developmental delay, is a frequent complication of pregnancy that is associated with many adverse perinatal outcomes (1). FGR is generally defined as the failure of the fetus to reach its full genetically determined growth potential and involves a fetal weight less than the 10th percentile for gestational age on prenatal ultrasound assessment. The fetus is hypoxic and/or hypotrophic due to inadequate oxygen and/or nutrition supply due to a variety of causes, one of which is placental insufficiency (2-4). FGR occurs in approximately 10% of pregnancies (3).
In recent years, there has been disagreement about the definition of FGR and fetuses small for gestational age. Nevertheless there is general agreement among the guidelines reviewed about the importance of early universal FGR risk stratification for effective screening, care, prevention, and treatment interventions.
The etiology of FGR is complex, and various maternal, fetal, and placental factors are involved in its development (3-7). One of the important factors affecting fetal growth and development is matrix metalloproteinases (MMPs). The MMPs are a subfamily of zinc-dependent endopeptidases involved in the degradation of the extracellular matrix (5). Although MMPs are mainly characterized in tumor biology, they also play a crucial role during pregnancy (8, 9). Placental MMPs are essential for cytotrophoblast migration and invasion into the uterine wall, as well as for spiral artery remodeling. They are involved in cytotrophoblast fusion to form syncytiotrophoblast, as well as in angiogenesis (9). MMPs expression levels, as well as tissue inhibitors of metalloproteases expression levels, are closely associated with placental insufficiency. Studies have shown that decidual stromal cells expressing cluster of differentiation 82 upregulate tissue inhibitor of metalloproteinases-1 expression in an autocrine manner and inhibit human trophoblast cell invasiveness in the first trimester, partly through the integrin β1/mitogen-activated protein kinase signaling pathway. All these processes are crucial for the establishment and maintenance of a successful pregnancy, and an imbalance in these processes can lead to various abnormalities in the course of pregnancy (9, 10). Disruption of MMPs expression has been shown to be associated with pregnancy complications such as placental insufficiency, pre-eclampsia, FGR, gestational diabetes mellitus, and is also observed in maternal obesity (8, 10, 11). It should be noted that there is little data on the association of MMP polymorphisms with FGR, and thus, further research in this area is warranted.
Hereby, we aimed to investigate the association of MMPs gene polymorphisms with the development of FGR.
2. Materials and Methods
2.1. Design and participants
A case-control study was carried out involving a total of 365 pregnant women in the third trimester of pregnancy from February 2015-2022 at the Medical Institute of Belgorod National Research University, Belgorod, Russia. In addition to medical history collection, standard clinical and biochemical parameters were studied in these women. The study was conducted in the Perinatal Center of the Belgorod Regional Clinical hospital of St. Joasaph, Belgorod, Russia. The study consisted of 122 pregnant women with FGR and 243 pregnant women with normal birth weight.
Inclusion criteria: Russian nationality, place of birth and residence in the Central part of Russia (12-15), third trimester of spontaneous singleton pregnancy. The group of pregnant women with FGR included individuals with a confirmed diagnosis of FGR. The diagnosis of FGR was established by obstetricians using ultrasound fetometry of the fetus before birth (TOSHIBA XARIO SSA-660A) and postnatal fetal data (16, 17). The control group consisted of pregnant women with a normally developing fetus. Exclusion criteria: severe chronic diseases, multiple pregnancies, diabetes mellitus, congenital diseases, and fetal malformations, and refusal to participate in the study. The clinical characteristics of pregnant women were based on previously published results (18) and are presented in table I. Venous blood was drawn from all observed women for the isolation of deoxyribonucleic acid (DNA) by the phenol-chloroform method (methodology described previously) (19).
5 single nucleotide polymorphisms (SNPs) in metalloprotease genes with significant regulatory potential were selected for analysis: rs1799750 of the MMP-1 gene, rs243865 of the MMP-2 gene, rs3025058 of the MMP-3 gene, rs11568819 of the MMP-7 gene, and rs17577 of the MMP-9 gene. Genotyping was performed using TaqMan probe-based real-time polymerase chain reaction with specific primers and probes. To ensure accuracy, negative control reactions were included, and 3% of samples were randomly re-genotyped in a blind manner for quality control, following established protocols (20).
2.2. Sample size
The sample size was determined using data on the incidence of FGR in previous studies (1-5) and the available data on frequencies in the European population (1000 Genomes project data), as well as using the Pocock formula. In this case, the percentage of errors (α = 0.05; β = 0.2) taken from the table and the loss of the sample of the main group (P1 = 0.05) and the control group (P2 = 0.2) were taken into account. The minimum sample size in each group was estimated as 106 individuals with a 10% probability of dropout. In our study, the main group included 122 women and the control group 243, which provided a statistical power of 0.80 at the α = 0.05 significance level.
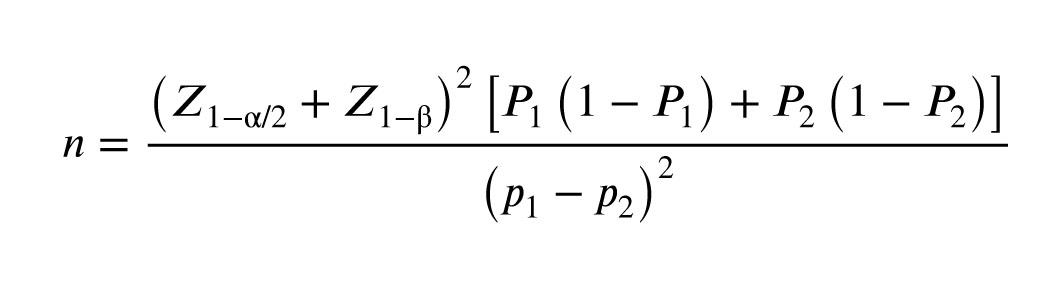
2.3. Ethical Considerations
The study was approved by the Ethical Commission of the Medical Institute of Belgorod National Research University, Belgorod, Russia (Opinion No. 54, Ethical Commission Protocol No. 39 of May 22, 2014). Informed consent was obtained from all the participants, and they were also informed about the purpose of the study.
2.4. Statistical Analysis
The associations of polymorphic loci with FGR within the 4 genetic models were analyzed using a software package (gPLINK v2.050) (21), taking into account covariates (age, pre-pregnancy weight, pre-pregnancy body mass index) and correction for multiple comparisons (permutation testing) (22). The Pperm indicator obtained as a result of permutation testing ≤ 0.050 was taken as statistically significant. When assessing the distribution compliance of the studied loci for Hardy-Weinberg equilibrium (HWE) distribution, a Bonferroni correction was introduced, which was equal to the number of loci studied (n = 5). Thus, the statistically significant level in this analysis (HWE compliance assessment) was bonfPHWE ≤ 0.01 (0.05/5). All analyzed loci, both in the FGR group and in the control, "fulfilled" HWE (bonfPHWE ≥ 0.026) and were included in our association analysis. In logistic regression, odds ratios (OR) and their 95% confidence interval (CI) were calculated. Various bioinformatics resources, HaploReg (v4.1) (23); GTExportal (24), 3′aQTL-atlas (25), were used to assess the functional potential of the polymorphic locus rs243865 of the MMP-2 gene, which showed significant associations with FGR.
3. Results
The analysis of clinical and anamnestic data of the study participants showed that the groups of pregnant women with FGR (n = 122) and the control group (n = 243) did not differ from each other in the vast majority of indicators considered (Table I). Women with FGR had lower weight (p = 0.008) and body mass index before pregnancy (p = 0.003) (Table I), and therefore these parameters (and the woman's age) were used as covariates in genetic calculations. Neonatal birth weight in the main group was 2647.26 ± 621.15 gr, and in the control group was 3463.26 ± 438.26 gr (p = 0.007). The height of newborns in the main group was 41.27 ± 2.41 cm and in the control group, 54.51 ± 2.26 cm (p = 0.081). For the remaining indicators, no statistically significant differences were found between the groups (Table I).
Minor allele frequency was above 5% for all SNPs studied, both in the group of pregnant women with FGR and in the control group. In both study groups, the observed genotype distribution was analyzed, which showed no deviations from the expected HWE distribution for all loci examined, with (observed parameters p [HWE] > 0.05/5 > 0.01 [considered Bonferroni correction for the number of loci examined], Table II).
The analysis of associations of alleles of 5 polymorphic loci of MMP genes with the development of FGR showed that the T allele of rs243865 of the MMP-2 gene (OR = 1.56, 95% CI: 1.08-2.24, p = 0.017, pperm = 0.016, Nperm = 1256) was significantly associated with the development of FGR (Table III). The polymorphism rs243865 of the MMP-2 gene was also found to be related to the development of FGR within the additive (OR = 1.53, 95% CI: 1.07-2.18, p = 0.021, pperm = 0.015, Nperm = 1302, power = 78.03%) and dominant (OR = 1.61, 95% CI: 1.03-2.50, p = 0.035, pperm = 0.027, Nperm = 745, power = 72.58%) models (Table III).
It should be noted that the polymorphic locus rs243865, which shows a major effect in the development of FGR (T allele is a risk factor OR = 1.53-1.61), according to Haploreg data, is located in the region of histone marking promoters and enhancers in many different cell cultures or organs (Table IV). It is located in the deoxyribonuclease-1 (DNase-1) hypersensitivity region and affects the affinity of DNA to myogenic factor (Myf) transcription (nucleotide substitution of C˃T rs243865 leads to decreased affinity of the genome region of the MMP-2 gene to Myf). Polymorphism affects epigenetic modifications in cell cultures and organs, which are important for the development of FGR, both in the embryo/fetus (cultures of trophoblast, ectoderm, mesoderm, etc., and lungs, muscles, etc.) and in the mother's body (ovaries, adipocytes, brain, muscles, etc.). The location of rs243865 in the enhancer region in the amnion and placenta tissues is extremely important for the occurrence of FGR (Table IV).
According to genotype-tissue expression portal data, rs243865 of the MMP-2 gene is associated with the expression level of the RP11-212I21.2 gene in arteries (p < 0.001), with the T allele variant of rs243865 increasing the transcriptional activity of this gene (NES = 0.27). According to the atlas of alternative polyadenylation quantitative trait loci (3′aQTL-atlas) the rs243865 MMP-2 polymorphism is associated with polyadenylation of the lysophosphatidylcholine acyltransferase 2 (LPCAT2) gene in thyroid (p < 0.001) and peripheral blood (p < 0.001).





4. Discussion
The study revealed that the T allele rs243865 of the MMP-2 gene is a risk factor for the development of FGR (OR = 1.53-1.61). This polymorphism is important for fetal development; it influences the development of various organs and systems (25). The location of rs243865 of the MMP-2 gene in the enhancer region in amnion and placental tissues is extremely important for the occurrence of intrauterine growth retardation (26).
MMP2 is an enzyme that can influence the degradation of some components of the extracellular matrix during pregnancy (9). This metalloproteinase is capable of cleaving gelatin, type I and IV collagens, and is an important regulator of uterine and spiral arteries remodeling. A decrease in the number, size, and dilation of uterine spiral arteries has been observed with a decrease in MMP-2, as well as an increase in collagen-IV in the extracellular matrix, which leads to decrease in uterine vascularization and contributes to progressive uteroplacental ischemia (9, 27). “The mechanical force of blood flow is a fundamental determinant of vascular homeostasis. The fetus depends on placental perfusion to meet its circulatory and metabolic needs. Disruption of normal vascular adaptation and the mechanisms that ensure a reciprocal interaction between fetoplacental and maternal circulation can lead to FGR” (26). Experimental studies in rats have demonstrated that MMP-2 is expressed in the endometrial stroma and during the first days of pregnancy. The messenger of ribonucleic acid expression of this gene changes dramatically and is observed mainly in the primary decidual zone and vascular endothelium (9). Thus, MMPs play a vital role in the regulatory during implantation and placentation as well as for successful pregnancy.
The polymorphic variant of MMP2 affects the affinity of DNA to the Myf transcription factor, which is associated with RP11-212I21 gene expression 2 in arteries (FGR-risk allele T rs243865 decreases DNA affinity to Myf transcription factor and increases RP11-212I21.2 gene transcription) and the level of polyadenylation gene LPCAT2 in thyroid and peripheral blood. The protein product of the LPCAT2 gene, LPCAT2, plays an important role in embryonic development. A study by Kondakova et al. showed the involvement of the LPCAT2 gene in estrogen-stimulated cell cycle progression. Estrogens have positive or negative effects on the regulation of gonadotropin secretion, folliculogenesis, and ovulation. Therefore, any changes in the LPCAT2 gene expression may lead to structural changes in the ovaries and altered follicular growth. Additionally, estrogen receptors are interconnected with the ubiquitin-proteasome system, which plays an important role in embryo implantation. It has been shown to be involved in some key processes of trophoblastic invasion, angiogenesis, extracellular matrix degradation, immune tolerance, and glandular secretion (28).
The impact of the rs243865 polymorphism of the MMP2 gene on female reproductive organs has been demonstrated in previous studies. This polymorphism has been associated with primary ovarian failure in Iranian women under 35 yr of age (29), and its association with recurrent miscarriage has been studied in a systematic review, meta-analysis, and bioinformatics analysis (30). Still, no clear associations of this polymorphism with pregnancy loss have been obtained. No evidence has been observed of an association between MMP2 (rs2285053 and rs243865) and recurrent pregnancy loss in the Polish population (31). The association of MMP-2 and MMP-9 polymorphisms with obesity and mammographic features has been proven (32, 33).
Our study demonstrated the clinical significance of the rs243865 polymorphism in the MMP2 gene for gynecologists and obstetricians. We also proved the connection with epigenetic genomic modifications, significant for FGR of the polymorphic locus MMP-2 rs243865 according to Haploreg data. Thus, this locus is responsible for the development of many extracellular matrix cells, derived neuronal, endodermal, and mesodermal cultured cells, organs, and tissues of the developing fetus, and maternal placenta conditions.
4.1. Strengths and limitations
Our study has several limitations: (a) the functional effects of loci associated with FGR, suggested in the work based on in silico analysis, require experimental confirmation in vivo/in vitro; (b) in this study, we did not determine the levels of MMP2 in women with FGR and in controls (the polymorphism of which, according to our data, is associated with FGR), which would have allowed us to demonstrate the association between MMP and FGR.
5. Conclusion
This study suggests that the T allele of rs243865 of the MMP-2 gene is a risk factor for the development of FGR (OR = 1.53-1.61). According to Haploreg data, this polymorphic variant is located in the histone region marking promoters and enhancers in many different cell cultures and organs; is located in the region of hypersensitivity to DNase-1 and affects the affinity of DNA for the transcription factor Myf (nucleotide substitution C˃T rs243865 leads to a decrease in the affinity of the genomic region in the region of the MMP-2 gene for the transcription factor Myf). Further academic studies should confirm these findings and clarify the complex relationship between FGR and maternal metalloproteinase polymorphisms.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
O. Efremova, I. Ponomarenko and M. Churnosov designed the study and conducted the research. O. Efremova and I. Ponomarenko monitored, evaluated, and analyzed the results of the study. Further, O. Efremova and M. Churnosov reviewed the article. All authors approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgments
The work was carried out without external sources of funding. Artificial intelligence was used for graphical visualization of the annotation.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Pregnancy Health
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |