Thu, May 21, 2026
[Archive]
Volume 24, Issue 1 (January 2026)
IJRM 2026, 24(1): 45-58 |
Back to browse issues page
Ethics code: 1818/14, July 7, 2022
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Hammood M N, AL-Harbi H J, Alhamed T A, Hammod H J. The effects of different doses of Stanozolol on sperm parameters and testicular histology in male rats: An experimental study. IJRM 2026; 24 (1) :45-58
URL: http://ijrm.ir/article-1-3578-en.html
URL: http://ijrm.ir/article-1-3578-en.html
Mahmood Neamah Hammood *1 

 , Hussein Jasim AL-Harbi2
, Hussein Jasim AL-Harbi2 

 , Thoalffakar Abbas Alhamed3
, Thoalffakar Abbas Alhamed3 

 , Hanan Jassim Hammod4
, Hanan Jassim Hammod4 




 , Hussein Jasim AL-Harbi2
, Hussein Jasim AL-Harbi2 

 , Thoalffakar Abbas Alhamed3
, Thoalffakar Abbas Alhamed3 

 , Hanan Jassim Hammod4
, Hanan Jassim Hammod4 


1- Directorate General of Education, Ministry of Education, Karbala, Iraq. , mahmood_neamah@karbala.edu.iq
2- Department of Forensic Science, College of Sciences, University of Al-Zahrawi, Karbala, Iraq.
3- Department of Biology, College of Education for Pure Sciences, University of Karbala, Karbala, Iraq.
4- Babylon Technical Institute, Al-Furat Al-Awsat Technical University, Babylon, Iraq.
2- Department of Forensic Science, College of Sciences, University of Al-Zahrawi, Karbala, Iraq.
3- Department of Biology, College of Education for Pure Sciences, University of Karbala, Karbala, Iraq.
4- Babylon Technical Institute, Al-Furat Al-Awsat Technical University, Babylon, Iraq.
Full-Text [PDF 3723 kb]
(260 Downloads)
| Abstract (HTML) (211 Views)
Full-Text: (79 Views)
1. Introduction
Anabolic androgenic steroids (AAS) are derivatives of dihydrotestosterone or testosterone, first manufactured in the 1930s. Male hypogonadism and delayed puberty are 2 hormonal conditions for which doctors prescribe these drugs (1). Numerous other illnesses, such as osteoporosis, edema, malignant anemia, and breast cancer, are also treated with them (2). However, some bodybuilders and athletes abuse AAS by applying or injecting dosages up to 100 times higher than those used in medical therapy in an attempt to improve their athletic performance or physical appearance. Despite prevention efforts, 2-7% of youth, both male and female, report having used AAS for non-medical purposes in organized sports, either currently or in the past (3, 4). AAS use has raised significant concern due to its adverse effects on consumers' physiological and psychological health (5). Previous studies have shown that AAS produces significant anabolic effects but also cause various physical and psychological side effects, including acne, hypertension, arrhythmia, myocardial infarction, depression, memory dysfunction, and weakened diastolic function (6, 7). Consequences of AAS impact on the male reproductive system include altered libido, erectile dysfunction, testicular atrophy, oligospermia, infertility, and an increased risk of prostate cancer. In women, misuse of AAS can lead to menstrual irregularities, deepening of the voice, and hirsutism (1, 8).
Stanozolol is among the most commonly misused AAS. While it has recognized medical applications, such as in the treatment of hereditary angioedema and certain muscle-wasting disorders, its potent anabolic effects have made it a substance of abuse in sports and bodybuilding (9, 10). Recent studies indicate that Stanozolol causes oxidative stress that leads to disruption of sperm parameters such as motility, vitality, and morphology, as a result of high levels of reactive oxygen species and a weak antioxidant defense system. The integrity of the seminiferous tubules is essential for maintaining spermatogenesis, while oxidative stress is a major factor in male infertility by causing DNA damage, lipid peroxidation, and impaired sperm function (11-13). It is crucial to comprehend the full extent of Stanozolol's effects on reproductive health due to the potential harm associated with its use, particularly at long-term or high doses.
Given the pharmacological importance and uncontrolled misuse of Stanozolol, this study aims to elucidate the interaction between Stanozolol and fertility by evaluating the effects of different doses on sperm quality, histopathological changes, and testis weight in male rats. The use of rats is justified as they represent a well-established model in male reproductive research due to their physiological and hormonal similarities to humans.
2. Materials and Methods
2.1. Chemicals
Stanozolol is among the most commonly misused AAS. While it has recognized medical applications, such as in the treatment of hereditary angioedema and certain muscle-wasting disorders, its potent anabolic effects have made it a substance of abuse in sports and bodybuilding (9, 10). Recent studies indicate that Stanozolol causes oxidative stress that leads to disruption of sperm parameters such as motility, vitality, and morphology, as a result of high levels of reactive oxygen species and a weak antioxidant defense system. The integrity of the seminiferous tubules is essential for maintaining spermatogenesis, while oxidative stress is a major factor in male infertility by causing DNA damage, lipid peroxidation, and impaired sperm function (11-13). It is crucial to comprehend the full extent of Stanozolol's effects on reproductive health due to the potential harm associated with its use, particularly at long-term or high doses.
Given the pharmacological importance and uncontrolled misuse of Stanozolol, this study aims to elucidate the interaction between Stanozolol and fertility by evaluating the effects of different doses on sperm quality, histopathological changes, and testis weight in male rats. The use of rats is justified as they represent a well-established model in male reproductive research due to their physiological and hormonal similarities to humans.
2. Materials and Methods
2.1. Chemicals
Stanozolol was obtained from a local pharmaceutical supplier (Alpha International Pharma Inc., New Delhi, India). The dosages of 5, 10, and 15 mg/kg were chosen based on earlier rodent experiments that showed comparable systemic effects (14). These dosages were selected to reflect low, moderate, and high exposure ranges so that the dose-dependent effects of Stanozolol on male reproductive parameters could be assessed. In order to approximate the equivalent doses reported for human use in previous studies, the concentrations were also adjusted.
2.2. Animal
2.2. Animal
The experimental study was conducted at the Department of Biology, College of Education for Pure Sciences, University of Karbala, Karbala Iraq, between September and November 2022. 20 male Wistar rats (Rattus norvegicus), were obtained from the Animal House of Babylon University, Babylon, Iraq, and were acclimatized for 1 wk under controlled laboratory conditions before the study. Rats were housed in standard cages in a well-ventilated room maintained at a temperature of 20-25°C and relative humidity of 50-55 with 12-hr light/dark cycle, with free access to water ad libitum and feeding on a standard pellet.
2.3. Experimental protocol
2.3. Experimental protocol
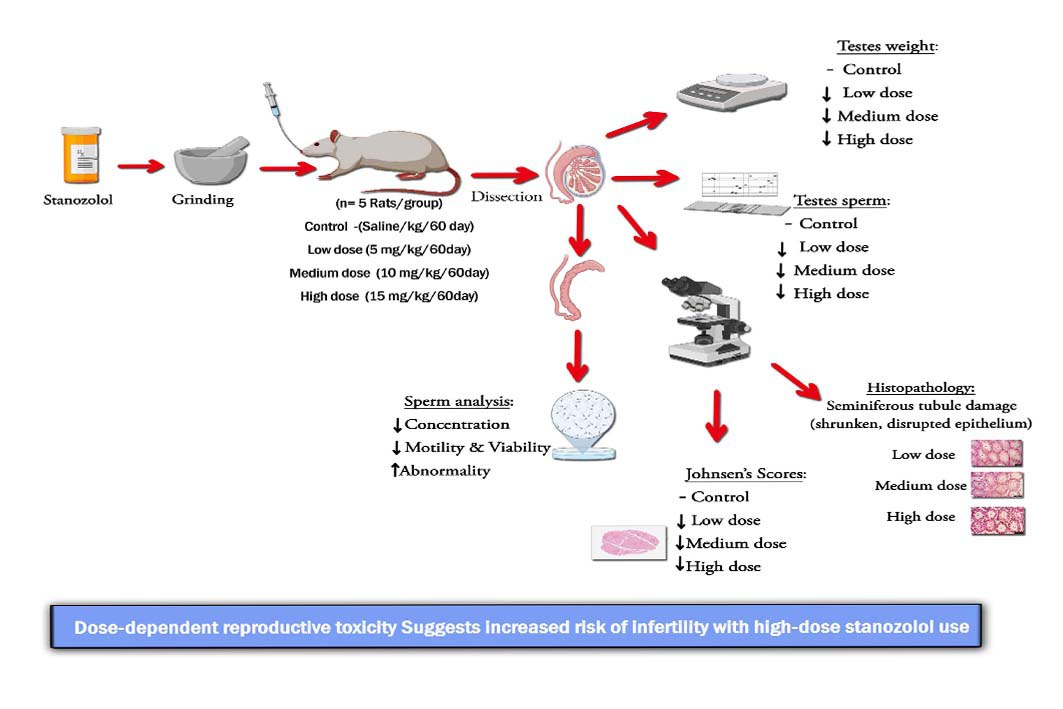
In this experimental study, 20 male Wistar rats (8-10 wk, 250-325 gr) were randomly divided into 4 groups (n = 5/group). The sample size was determined based on a previous study (15). Group 1: animals that were orally gavaged with distilled water served as the control. Group 2, 3, and 4: rats treated with Stanozolol at doses of 5, 10, and 15 mg/kg/day, respectively, by gavage for 60 days. All animals were monitored daily for clinical signs of pain, distress, or illness throughout the experiment. At the end of the experiment, the animals were sacrificed by anesthetizing with chloroform, the testes and epididymis were removed rapidly after the animals had been dissected and placed on the filter paper and then weighed.
2.4. Sperm parameters
2.4.1. Sperm concentration
2.4. Sperm parameters
2.4.1. Sperm concentration
The testes and epididymal sperm concentration were calculated by cutting the right testes into thin slices in a petri-dish, which contains 5 ml of normal saline (0.9% NaCl) to release swim sperm. After 10 min a drop of the homogenate was loaded on the improved neubauer hemocytometer and counted under light microscope (NOVEX, Euromex Co., Holland). The same procedure, with slight modification, was used for epididymal sperm concentration by using the left epididymis (tail) (15).
2.4.2. Sperm motility
2.4.2. Sperm motility
Sperm parameters were evaluated according to standard rodent reproductive assessment protocols previously described in experimental animal studies (15, 16). Immediately after preparation, a drop of the sperm solution was placed on a microscopic slide and covered with a coverslip. For each animal, at least 10 microscopic fields were examined to evaluate sperm motility. The total sperm motility percentage (including progressive and non-progressive motility) and the percentage of immotile sperm were calculated (15).
2.4.3. Sperm viability and morphology
2.4.3. Sperm viability and morphology
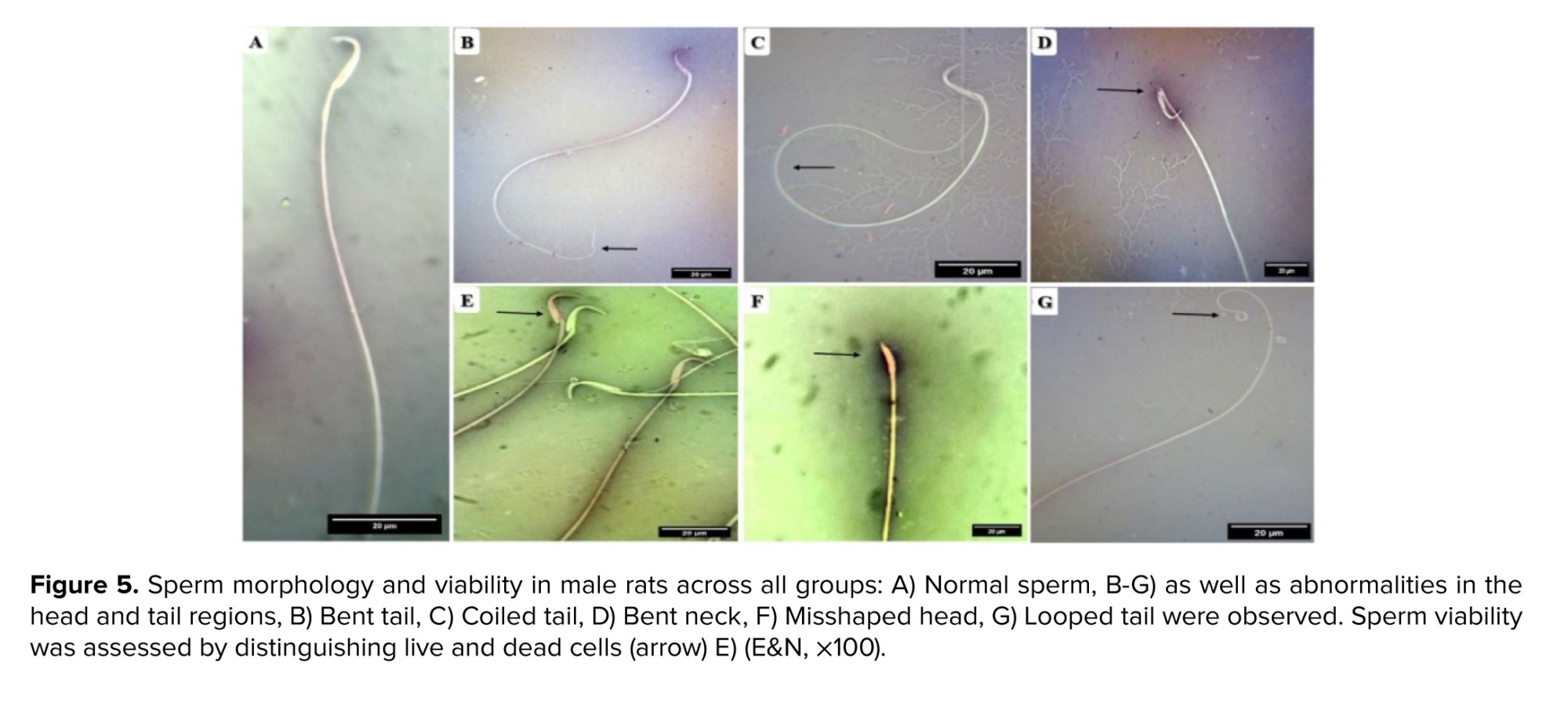
Sperm viability was assessed using eosin (1%) and nigrosin (10%) staining (Merck, Germany). One drop of sperm suspension was mixed with 2 drops of 1% eosin. Subsequently, 2 drops of nigrosin were added to the mixture. Thin smears were then prepared and examined under oil immersion magnification. Live sperm remained colorless, whereas dead sperm were stained. For each sample, a total of 200 spermatozoa were evaluated and characterized as normal or abnormal, with abnormalities including double head, headless, amorphous head, coiled mid-piece, coiled tail, bent tail, and cytoplasmic droplet. Sperm viability and abnormality rates were expressed as percentages (15).
2.5. Histopathological preparations
2.5. Histopathological preparations
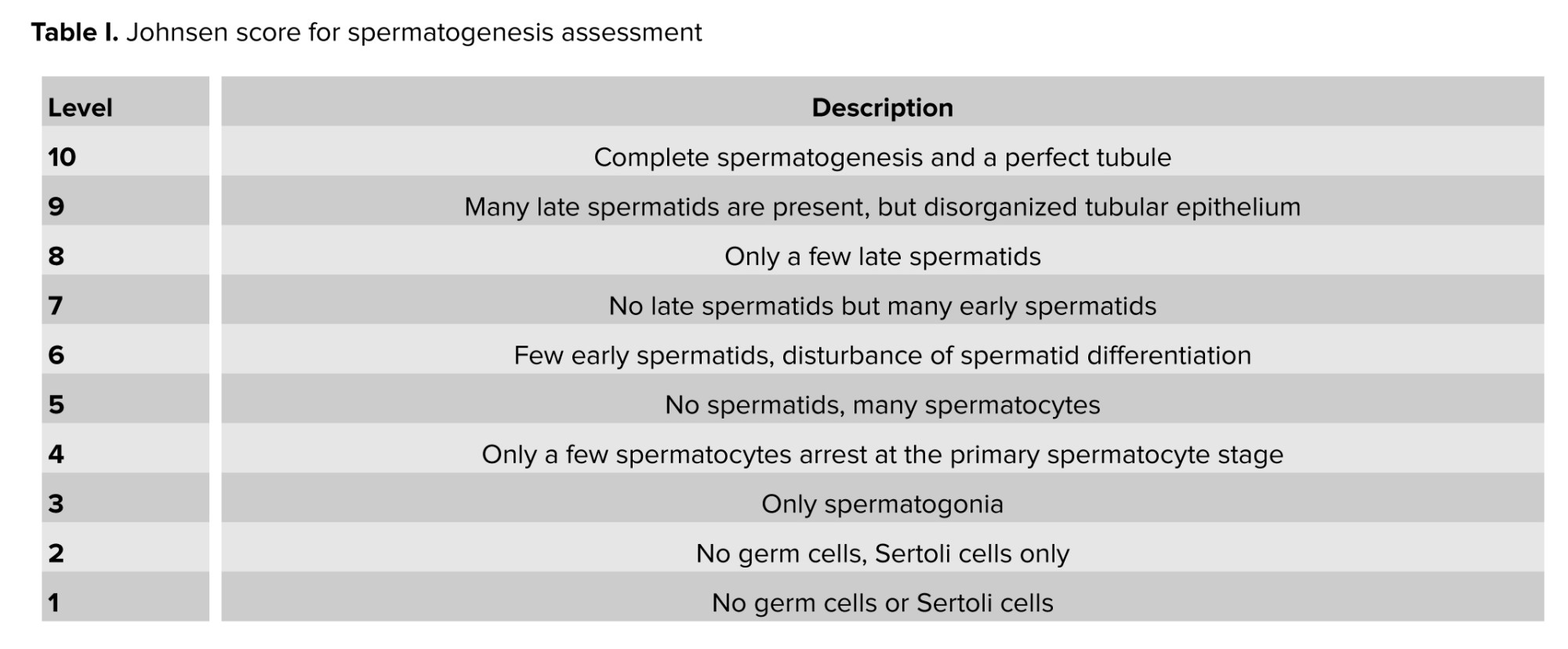
The left testis from each animal was fixed in 10% formalin for 48 hr and then dehydrated through a graded series of ethanol concentrations. The tissues were embedded in paraffin blocks and sectioned at a thickness of 5 µm using a rotary microtome (microTec Laborgeräte GmbH, Germany). Sections were stained with routine hematoxylin and eosin (H&E) (17). The diameter, number of germinal layers, and lumen of 200 seminiferous tubules from each animal were measured using an image analysis system (ImageJ Version 1.52p). Spermatogenesis was evaluated using the Johnsen scoring system according to the criteria shown in table I. All histomorphometric evaluations were conducted by a single trained, calibrated, and blinded examiner, who assessed at least 30 seminiferous tubules per sample (16).
2.6. Ethical Considerations
All animal experimental protocols were approved by the National Institutes of Health (NIH Publication No. 8023, revised 1978) for the care and use of laboratory animals. The study has been conducted with approval from the local Ethics Committee of the College of Education for Pure Sciences, University of Karbala, Karbala, Iraq (Code: 1818/14, July 7, 2022).
2.7. Statistical Analysis

2.6. Ethical Considerations
All animal experimental protocols were approved by the National Institutes of Health (NIH Publication No. 8023, revised 1978) for the care and use of laboratory animals. The study has been conducted with approval from the local Ethics Committee of the College of Education for Pure Sciences, University of Karbala, Karbala, Iraq (Code: 1818/14, July 7, 2022).
2.7. Statistical Analysis
Data normality was verified using the Shapiro-Wilk test, while homogeneity of variances was assessed with Levene's test. As both assumptions were satisfied, parametric tests were applied. The data were expressed as mean ± standard Error, and analyzed by using GraphPad Prism Version 9.5 (CA, USA). The variable was analyzed according to one-way ANOVA. Comparison was done between means by using revised least significant differences in probability level (p < 0.05).
3. Results
3.1. Testes weight
3. Results
3.1. Testes weight
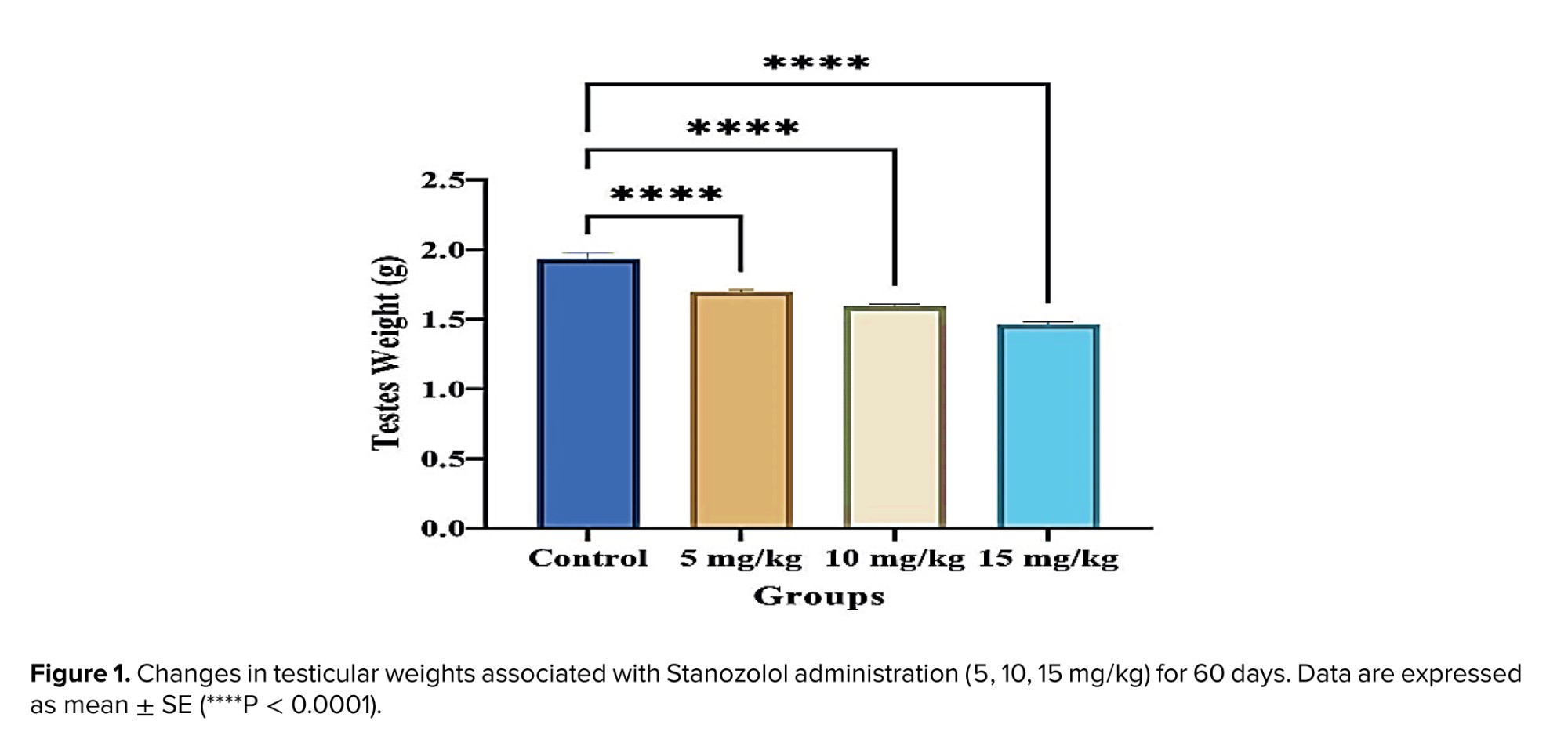
The data indicate a clear reduction in testicular weight following Stanozolol treatment at all administered doses. Testicular weight was significantly decreased in the 5 mg/kg group (p < 0.001), the 10 mg/kg group (p < 0.001), and the 15 mg/kg group (p < 0.001) compared with the control group (Figure 1). The reduction was dose-dependent, with higher doses producing more pronounced effects.
3.2. Sperm analysis
3.2. Sperm analysis
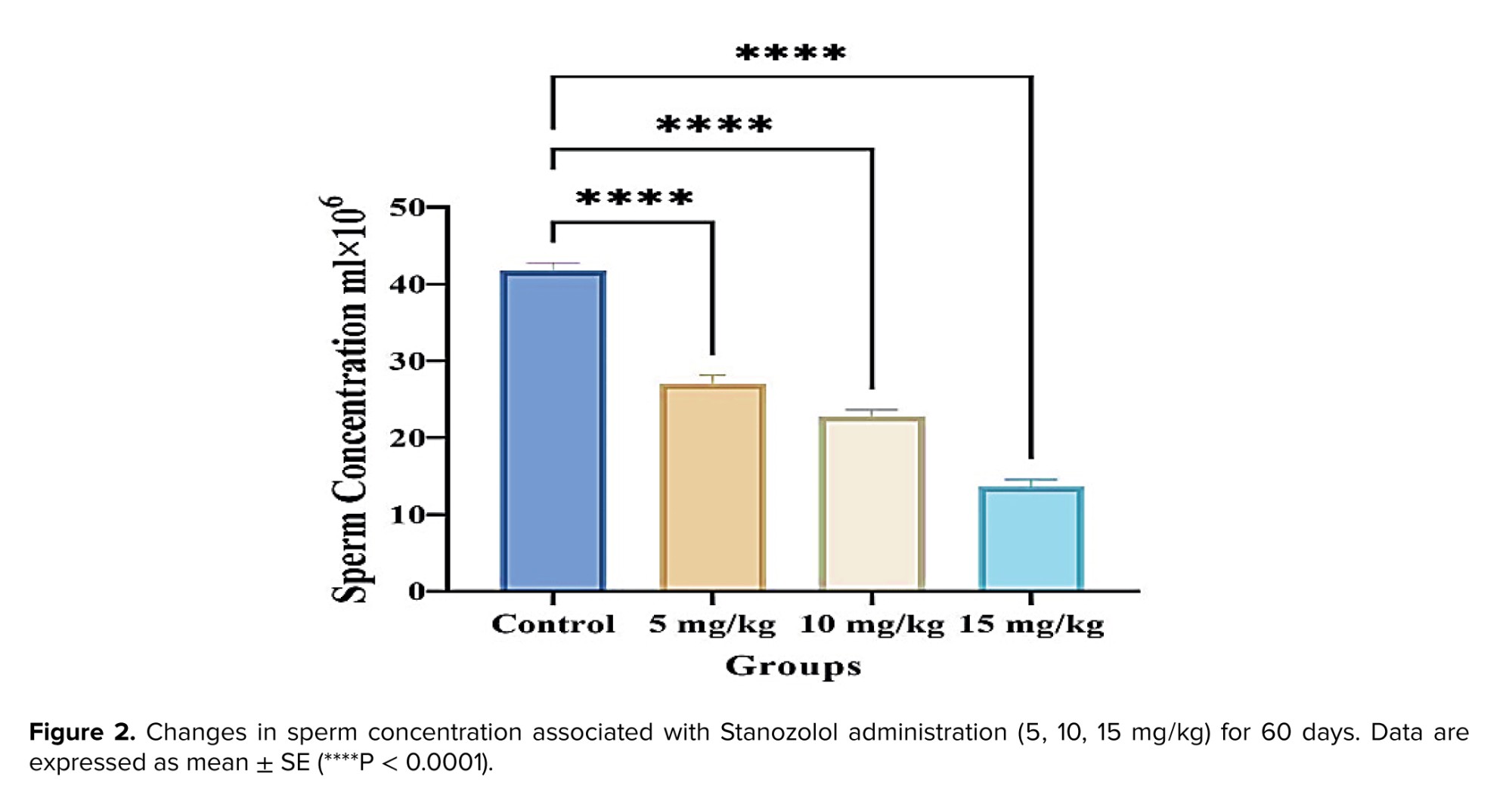
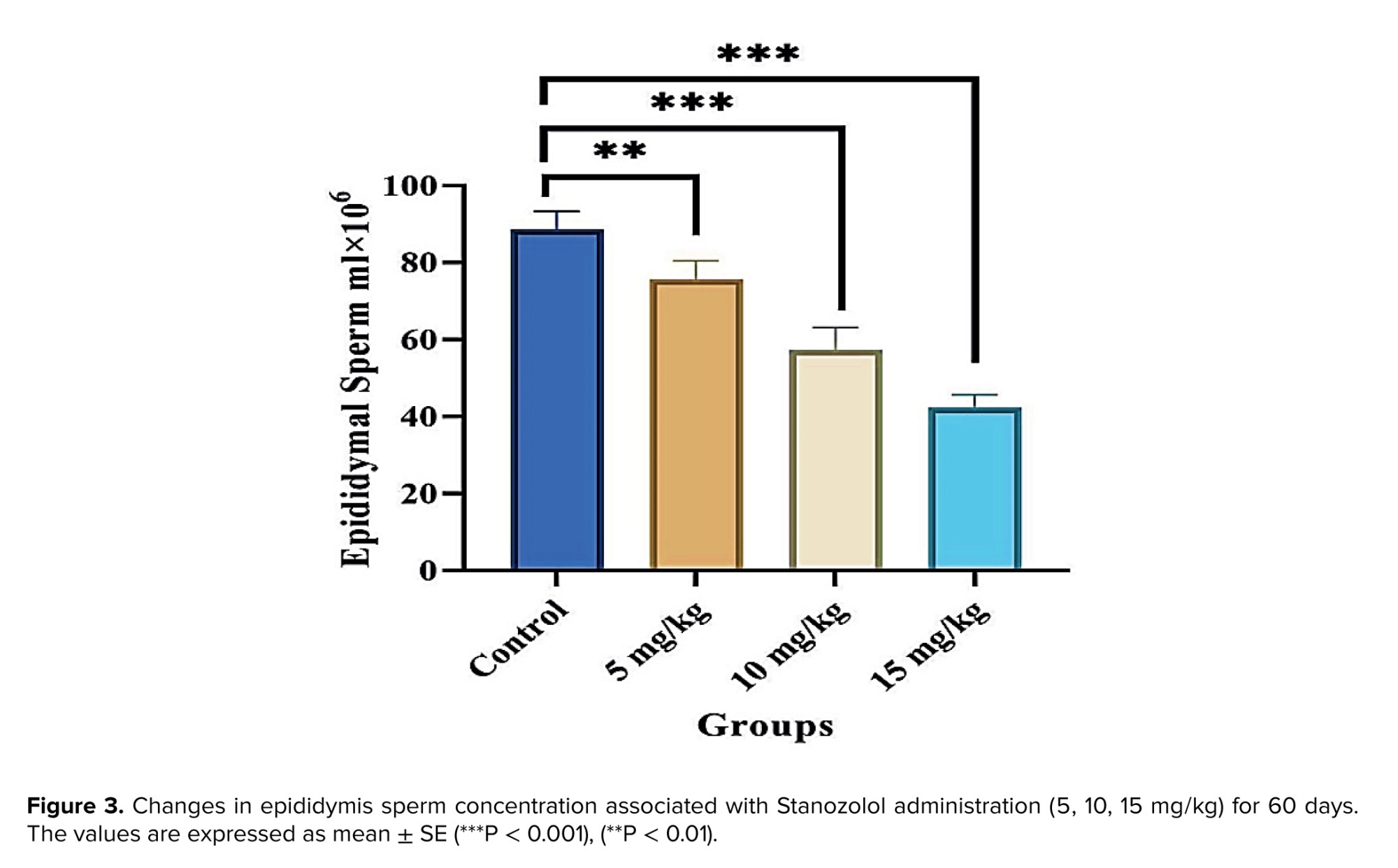
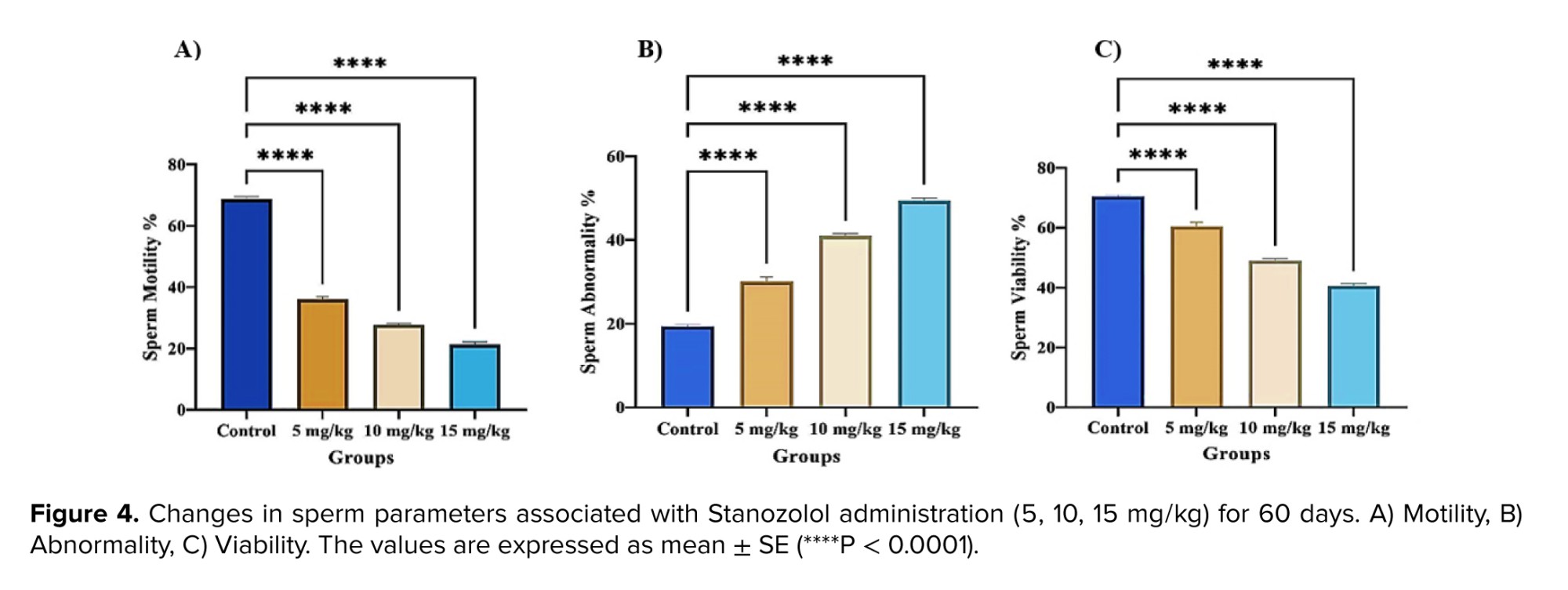
The sperm concentration in both the testes and epididymis was significantly decreased in rats treated with different doses of Stanozolol compared to the control group (p < 0.001). In the epididymis, sperm concentration showed a significant reduction at 5 mg/kg (p < 0.01) and an even greater reduction at 10 mg/kg (p < 0.001). At the highest dose (15 mg/kg), the decline was also highly significant (p < 0.001). Moreover, comparisons between treated groups revealed a dose-dependent reduction, particularly evident in the higher dose groups (Figures 2 and 3). In addition, figures 4 and 5 demonstrate significant decreases in sperm viability and motility (p < 0.001) and a marked increase in abnormal morphology (p < 0.001) in all treated groups compared to controls.
3.3. Histopathological findings
3.3.1. The Johnsen scores
3.3. Histopathological findings
3.3.1. The Johnsen scores
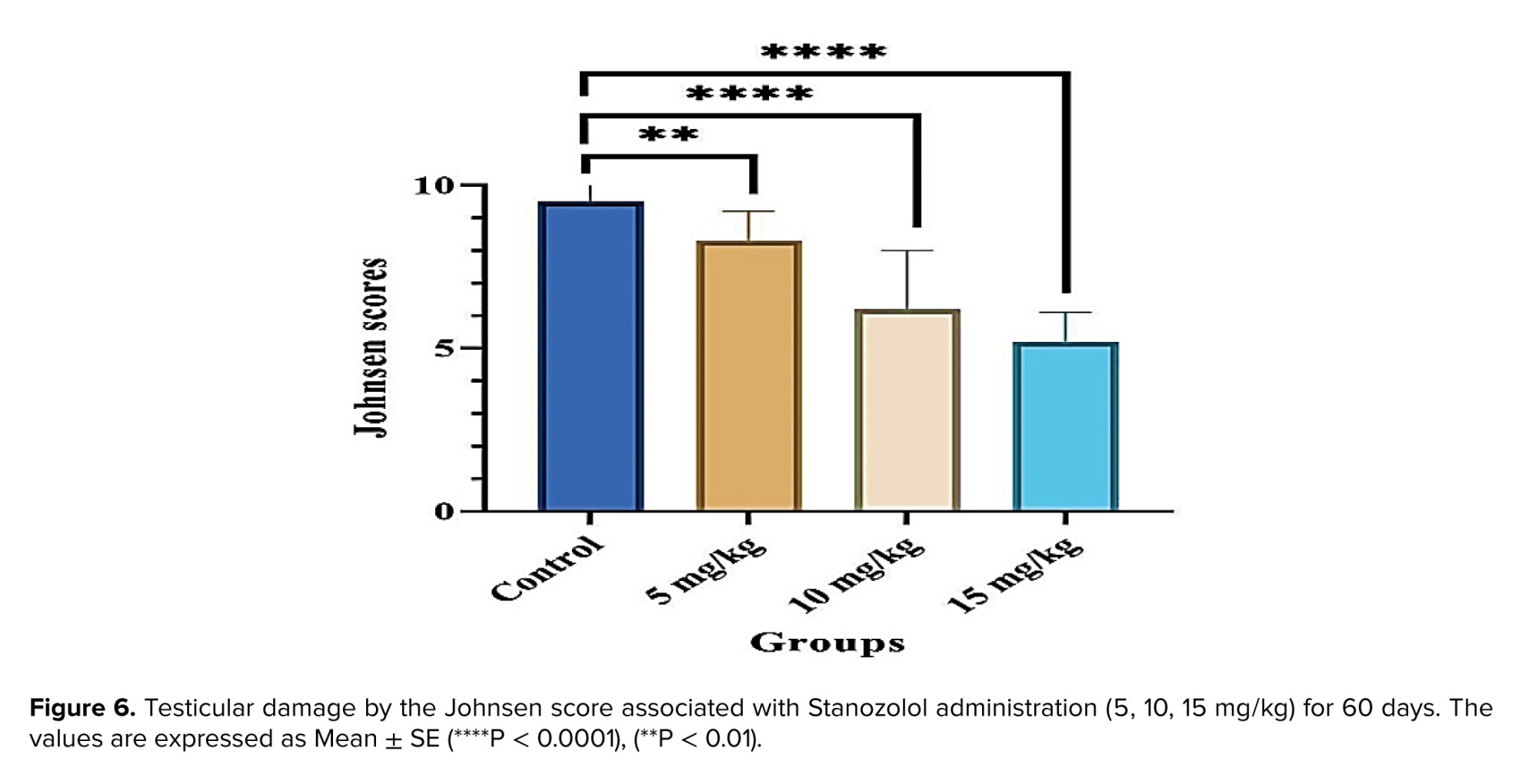
Figure 6 illustrates marked histological seminiferous tubule damage in Stanozolol-exposed rats for 60 days. The group receiving 5 mg/kg showed considerable destruction (p < 0.01), while the 10 mg/kg and 15 mg/kg groups exhibited even more significant damage (p < 0.0001). This histological disturbance was reflected in substantially reduced spermatogenesis, as denoted by lower Johnsen's scores in the treated groups.
3.3.2. Histological features
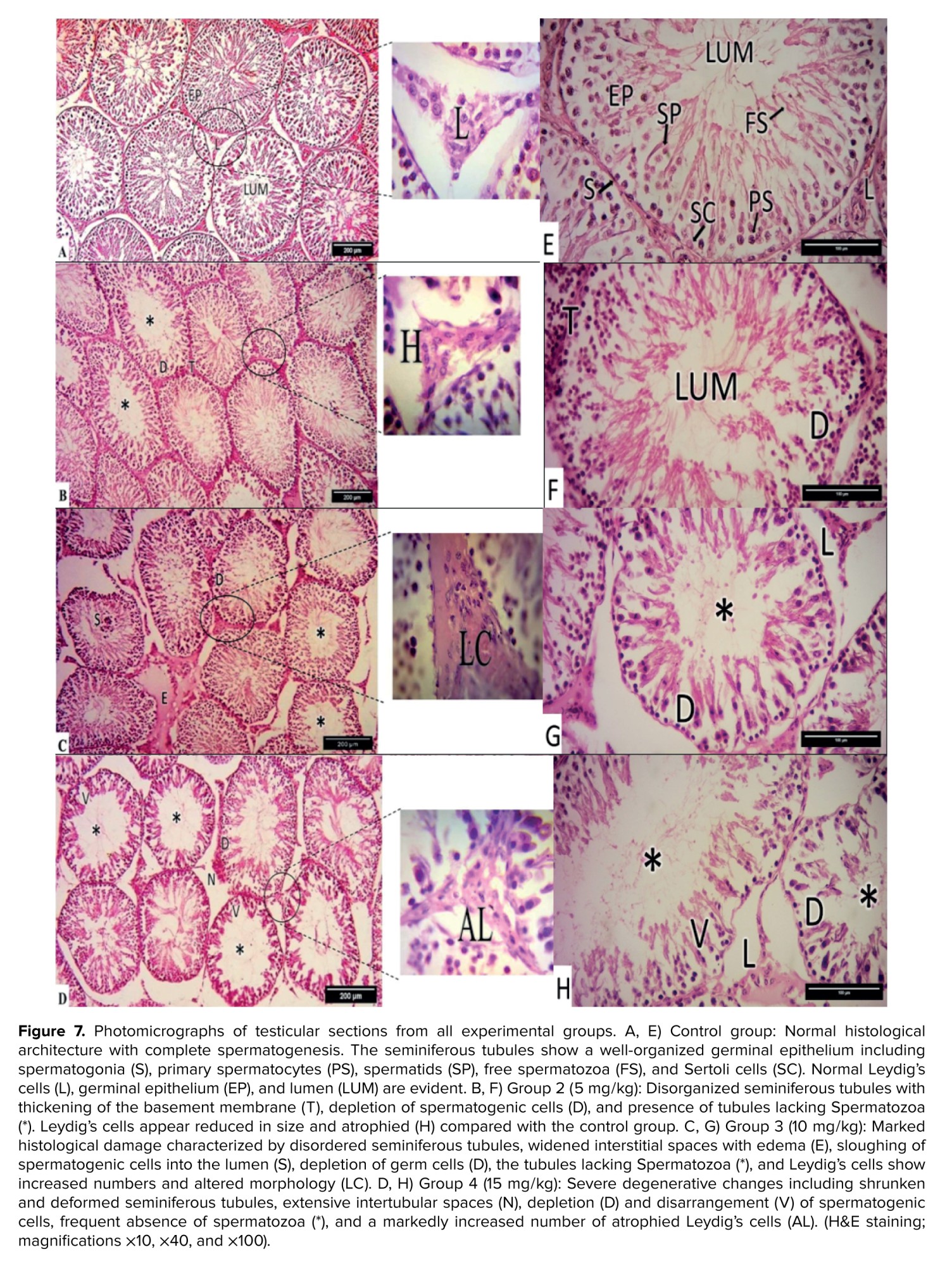
The testes of the control animals were histologically normal and exhibited complete spermatogenesis, with a well-organized spermatogenic series from spermatogonia to spermatozoa. The lumen of the seminiferous tubules contained sperm with long tails (Figure 7). Figure 7E shows details of the germinal epithelium, including the Lumen, Leydig's cells, and germinal epithelium cells with spermatogonia, primary spermatocyte, spermatids, free spermatozoa, and Sertoli cells.
In group 2 (5 mg/kg), the testicular sections showed seminiferous tubules with a variably disorganized arrangement; some appeared normal while others were disorganized (Figure 7B). The majority of Leydig cells were reduced in size (atrophied) compared to the control group, as visible in figure 7F.
Testicular sections from rats that received the higher doses of Stanozolol (10 and 15 mg/kg/BW) showed destructive effects. These included shrunken and deformed tubules, a disordered architecture, and extensive interstitial spaces between seminiferous tubules (Figures 7C, D). Some tubules appeared empty, while others were filled with cellular debris from spermatogenic cells. Edema and cellular infiltration were observed between the tubules, along with an increase in atrophied Leydig cells. The tubules contained disarranged spermatogenic cells, exhibited significant depletion, and showed sloughing of the germinal epithelium (Figures 7G, H).
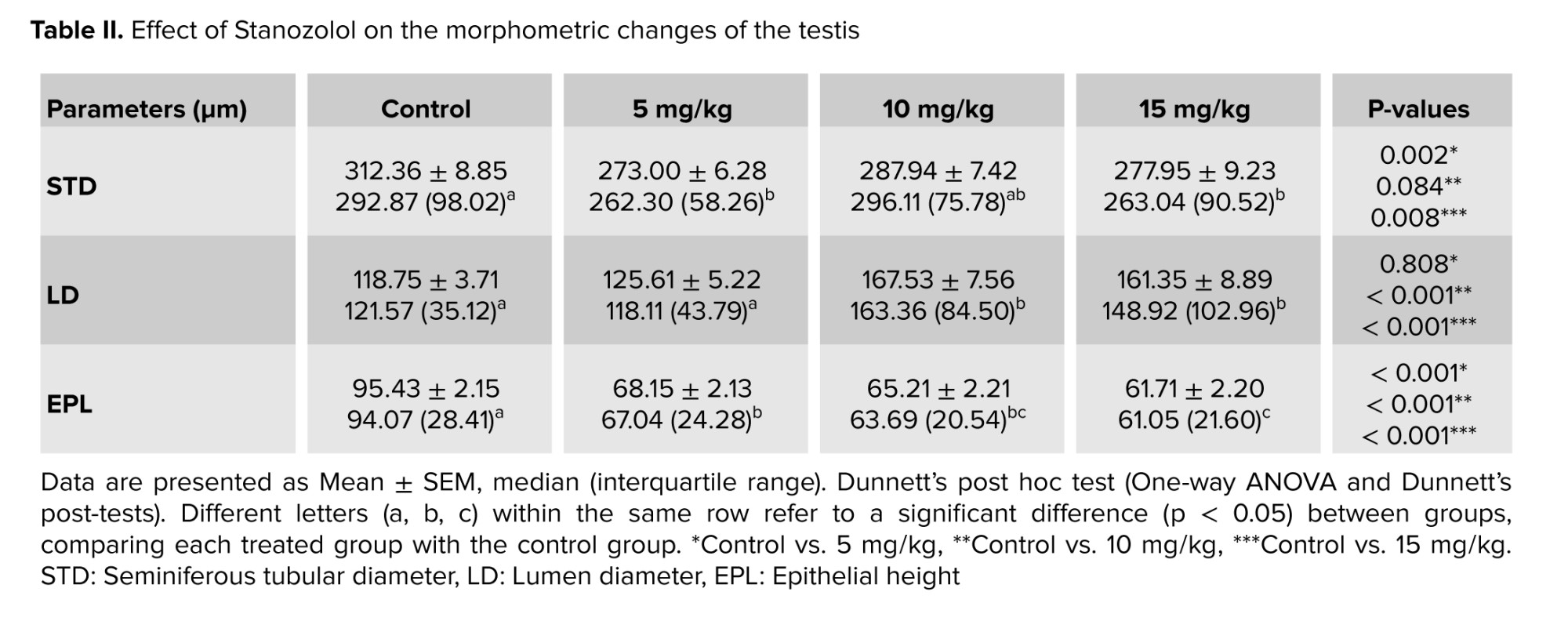
Histomorphometric analysis revealed a significant decrease in the mean seminiferous tubule diameter (STD) in the 5 mg/kg (p = 0.002) and 15 mg/kg (p = 0.008) groups compared to the control. The decrease in the 10 mg/kg group was not significant (p = 0.084). Epithelial height (EPL) was significantly reduced in all treated groups (5, 10, and 15 mg/kg; p < 0.001). Conversely, the lumen diameter was significantly increased in the 10 mg/kg and 15 mg/kg groups (p < 0.001), while the change in the 5 mg/kg group was not significant (p = 0.808) (Table II).
4. Discussion
3.3.2. Histological features
The testes of the control animals were histologically normal and exhibited complete spermatogenesis, with a well-organized spermatogenic series from spermatogonia to spermatozoa. The lumen of the seminiferous tubules contained sperm with long tails (Figure 7). Figure 7E shows details of the germinal epithelium, including the Lumen, Leydig's cells, and germinal epithelium cells with spermatogonia, primary spermatocyte, spermatids, free spermatozoa, and Sertoli cells.
In group 2 (5 mg/kg), the testicular sections showed seminiferous tubules with a variably disorganized arrangement; some appeared normal while others were disorganized (Figure 7B). The majority of Leydig cells were reduced in size (atrophied) compared to the control group, as visible in figure 7F.
Testicular sections from rats that received the higher doses of Stanozolol (10 and 15 mg/kg/BW) showed destructive effects. These included shrunken and deformed tubules, a disordered architecture, and extensive interstitial spaces between seminiferous tubules (Figures 7C, D). Some tubules appeared empty, while others were filled with cellular debris from spermatogenic cells. Edema and cellular infiltration were observed between the tubules, along with an increase in atrophied Leydig cells. The tubules contained disarranged spermatogenic cells, exhibited significant depletion, and showed sloughing of the germinal epithelium (Figures 7G, H).
Histomorphometric analysis revealed a significant decrease in the mean seminiferous tubule diameter (STD) in the 5 mg/kg (p = 0.002) and 15 mg/kg (p = 0.008) groups compared to the control. The decrease in the 10 mg/kg group was not significant (p = 0.084). Epithelial height (EPL) was significantly reduced in all treated groups (5, 10, and 15 mg/kg; p < 0.001). Conversely, the lumen diameter was significantly increased in the 10 mg/kg and 15 mg/kg groups (p < 0.001), while the change in the 5 mg/kg group was not significant (p = 0.808) (Table II).








4. Discussion
The present study demonstrated that the administration of Stanozolol at different doses led to a marked reduction in testicular weight, significant alterations in histomorphometric indices, and a decline in sperm quality, including concentration, motility, viability, and morphology, accompanied by an increase in sperm abnormalities. Histological evaluation further revealed damage to the seminiferous tubules and reduced Johnsen’s scores, reflecting impaired spermatogenesis. These results are consistent with earlier research showing that anabolic-androgenic steroids (AAS), particularly Stanozolol, significantly decrease sperm concentration, vitality, and motility (11). Similar studies have reported that high doses of AAS reduce sperm motility and count while increasing abnormalities and DNA damage compared with untreated controls (18). In addition, synthetic steroids have been shown to alter the activity of sperm-specific channels such as CatSper, which regulates intracellular Ca²⁺ concentration and plays a crucial role in sperm function, potentially compromising fertilization (19).
The high content of polyunsaturated fatty acids in the sperm membrane increases its susceptibility to free radical damage. Oxidative stress compromises sperm membrane integrity, thereby reducing viability, concentration, normal morphology, and motility (13). Anabolic steroids such as boldenone, nandrolone decanoate, and Stanozolol induce oxidative stress, which contributes significantly to testicular damage in male rats. These steroids have been shown to increase reactive oxygen species and reduce antioxidant defenses, leading to sperm damage. Infertility may result from this oxidative imbalance, particularly with prolonged steroid use (20, 21). Earlier studies report that approximately 51% of steroid abusers suffer from testicular atrophy (22). These findings underscore the importance of healthcare providers, including doctors and pharmacists, educating and counseling athletes, adolescents, parents, instructors, and coaches on the risks associated with AAS use.
In the current study, 3 different doses of the long-acting Stanozolol were used to investigate its morphological and histopathological effects on the testes. The results showed a significant decrease in testicular weight, which may be attributed to the deterioration of seminiferous tubules and abnormal spermatogenesis, as confirmed by histological analysis. The extent of testicular atrophy was directly related to the degradation of seminiferous tubules. Previous studies in rats have shown that AAS exposure reduces testicular weight, decreases serum testosterone and estradiol levels, and impairs mating behavior and fertility (8, 23, 24).
Histological analysis of testes from Stanozolol-treated rats revealed deleterious effects on seminiferous tubules. As indicated by Johnsen’s scores, spermatogenesis was significantly impaired, and testicular damage, including interstitial edema, congestion, sloughing, and disorganization, was observed across treatment groups. These findings align with previous reports indicating that Stanozolol treatment results in incomplete spermatogenesis, absence of sperm in the lumen, and degenerative changes such as germinal epithelial depletion, which worsen with prolonged exposure (11, 20). Consistent histopathological alterations, including seminiferous tubule diminution, widened interstitial spaces, and suppressed spermatogenesis, were observed across dose groups (23). The non-significant decrease in STD at the intermediate dose (10 mg/kg), despite the marked decreases at 5 and 15 mg/kg, suggests a possible nonlinear dose-response. This finding may be attributed to compensatory or adaptive physiological mechanisms in testicular tissue that temporarily limit damage at intermediate doses. This pattern of nonlinear response has been previously reported in studies investigating the effects of anabolic steroids and other hormonally active substances such as phthalates and polycyclic aromatic hydrocarbons on fertility. Hormonal analyses and oxidative stress markers are recommended to elucidate the potential mechanism underlying this behavior at intermediate doses (25, 26). Studies also indicate that Stanozolol and nandrolone decanoate cause disordered spermatogonia, vacuolation, multinucleated cells, and Leydig cell hyperplasia, with Stanozolol often producing more pronounced damage (11). Furthermore, AAS exposure has been associated with Leydig cell tumors. Although Leydig cell numbers may increase, many cells appear atrophied due to reduced luteinizing hormone levels (27). This compensatory increase in Leydig cell production may be disrupted, where androgen treatment reduces steroid-synthesizing organelles, suggesting that atrophy may result from diminished hormonal stimulation (28, 29).
Previous studies have reported that exogenous steroids such as Stanozolol inhibit reproductive endocrine function. Although these steroids minimally affect certain hormonal levels, they promote substantial protein synthesis and nitrogen retention (6). Stanozolol suppresses endogenous testosterone production and exhibits estrogen-antagonistic effects. It disrupts homeostasis by inhibiting or attenuating the hypothalamic-pituitary-gonadal axis, which regulates reproductive function through hormones such as luteinizing hormone and follicle-stimulating hormone. These hormonal disruptions can directly damage testicular tissue and reduce sperm quality, leading to male infertility (30, 22).
Although the present study did not investigate the precise roles of oxidative stress and hormonal mechanisms in Stanozolol-induced testicular damage, our findings clearly demonstrate that Stanozolol administration significantly impairs sperm quality and testicular histology in rats.
5. Conclusion
The high content of polyunsaturated fatty acids in the sperm membrane increases its susceptibility to free radical damage. Oxidative stress compromises sperm membrane integrity, thereby reducing viability, concentration, normal morphology, and motility (13). Anabolic steroids such as boldenone, nandrolone decanoate, and Stanozolol induce oxidative stress, which contributes significantly to testicular damage in male rats. These steroids have been shown to increase reactive oxygen species and reduce antioxidant defenses, leading to sperm damage. Infertility may result from this oxidative imbalance, particularly with prolonged steroid use (20, 21). Earlier studies report that approximately 51% of steroid abusers suffer from testicular atrophy (22). These findings underscore the importance of healthcare providers, including doctors and pharmacists, educating and counseling athletes, adolescents, parents, instructors, and coaches on the risks associated with AAS use.
In the current study, 3 different doses of the long-acting Stanozolol were used to investigate its morphological and histopathological effects on the testes. The results showed a significant decrease in testicular weight, which may be attributed to the deterioration of seminiferous tubules and abnormal spermatogenesis, as confirmed by histological analysis. The extent of testicular atrophy was directly related to the degradation of seminiferous tubules. Previous studies in rats have shown that AAS exposure reduces testicular weight, decreases serum testosterone and estradiol levels, and impairs mating behavior and fertility (8, 23, 24).
Histological analysis of testes from Stanozolol-treated rats revealed deleterious effects on seminiferous tubules. As indicated by Johnsen’s scores, spermatogenesis was significantly impaired, and testicular damage, including interstitial edema, congestion, sloughing, and disorganization, was observed across treatment groups. These findings align with previous reports indicating that Stanozolol treatment results in incomplete spermatogenesis, absence of sperm in the lumen, and degenerative changes such as germinal epithelial depletion, which worsen with prolonged exposure (11, 20). Consistent histopathological alterations, including seminiferous tubule diminution, widened interstitial spaces, and suppressed spermatogenesis, were observed across dose groups (23). The non-significant decrease in STD at the intermediate dose (10 mg/kg), despite the marked decreases at 5 and 15 mg/kg, suggests a possible nonlinear dose-response. This finding may be attributed to compensatory or adaptive physiological mechanisms in testicular tissue that temporarily limit damage at intermediate doses. This pattern of nonlinear response has been previously reported in studies investigating the effects of anabolic steroids and other hormonally active substances such as phthalates and polycyclic aromatic hydrocarbons on fertility. Hormonal analyses and oxidative stress markers are recommended to elucidate the potential mechanism underlying this behavior at intermediate doses (25, 26). Studies also indicate that Stanozolol and nandrolone decanoate cause disordered spermatogonia, vacuolation, multinucleated cells, and Leydig cell hyperplasia, with Stanozolol often producing more pronounced damage (11). Furthermore, AAS exposure has been associated with Leydig cell tumors. Although Leydig cell numbers may increase, many cells appear atrophied due to reduced luteinizing hormone levels (27). This compensatory increase in Leydig cell production may be disrupted, where androgen treatment reduces steroid-synthesizing organelles, suggesting that atrophy may result from diminished hormonal stimulation (28, 29).
Previous studies have reported that exogenous steroids such as Stanozolol inhibit reproductive endocrine function. Although these steroids minimally affect certain hormonal levels, they promote substantial protein synthesis and nitrogen retention (6). Stanozolol suppresses endogenous testosterone production and exhibits estrogen-antagonistic effects. It disrupts homeostasis by inhibiting or attenuating the hypothalamic-pituitary-gonadal axis, which regulates reproductive function through hormones such as luteinizing hormone and follicle-stimulating hormone. These hormonal disruptions can directly damage testicular tissue and reduce sperm quality, leading to male infertility (30, 22).
Although the present study did not investigate the precise roles of oxidative stress and hormonal mechanisms in Stanozolol-induced testicular damage, our findings clearly demonstrate that Stanozolol administration significantly impairs sperm quality and testicular histology in rats.
5. Conclusion
The current study suggests that the usage of high-dose Stanozolol significantly impairs testicular tissue and sperm quality, adversely affecting male reproductive health and increasing infertility risk. Individuals considering anabolic steroid use should be aware of these dangers and prioritize long-term health over short-term benefits. Avoiding non-medical use is strongly advised to prevent reproductive health complications, and to seek safer, medically-supervised alternatives for performance enhancement. Further research remains necessary to understand mechanisms, inform clinical practice, and support public health measures against steroid misuse.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
H.J. Hammod and H.J. Al-Harbi designed the study and contributed to the conception, planning, and interpretation of the research. H.J. Hammod and M.N. Hammood prepared the drug solutions, collected the biological specimens, and performed the histopathological examinations. M.N. Hammood evaluated sperm parameters and prepared the initial draft of the manuscript. T.A. Alhamed was responsible for animal housing, daily care, and overall experimental supervision. M.N. Hammood and T.A. Alhamed performed the statistical analyses. H.J. Hammod and H.J. Al-Harbi supervised all stages of the study to ensure methodological accuracy. All authors reviewed the manuscript, critically revised it for important intellectual content, approved the final version for submission, and agreed to be accountable for the integrity and accuracy of the data.
Acknowledgments
This study did not use any grant or financial support. The study was entirely self-funded by the authors. Artificial intelligence tools (Grammarly) were used for language grammar checking to improve the quality of the manuscript. However, all scientific content, data interpretation, and conclusions were entirely prepared by the authors.
Conflict of Interest
The authors declare that there is no conflict of interest.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
H.J. Hammod and H.J. Al-Harbi designed the study and contributed to the conception, planning, and interpretation of the research. H.J. Hammod and M.N. Hammood prepared the drug solutions, collected the biological specimens, and performed the histopathological examinations. M.N. Hammood evaluated sperm parameters and prepared the initial draft of the manuscript. T.A. Alhamed was responsible for animal housing, daily care, and overall experimental supervision. M.N. Hammood and T.A. Alhamed performed the statistical analyses. H.J. Hammod and H.J. Al-Harbi supervised all stages of the study to ensure methodological accuracy. All authors reviewed the manuscript, critically revised it for important intellectual content, approved the final version for submission, and agreed to be accountable for the integrity and accuracy of the data.
Acknowledgments
This study did not use any grant or financial support. The study was entirely self-funded by the authors. Artificial intelligence tools (Grammarly) were used for language grammar checking to improve the quality of the manuscript. However, all scientific content, data interpretation, and conclusions were entirely prepared by the authors.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Biology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |