Tue, May 26, 2026
[Archive]
Volume 23, Issue 12 (December 2025)
IJRM 2025, 23(12): 1031-1042 |
Back to browse issues page
Ethics code: IR.SSU.MEDICINE.REC.1401.021
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Fakherbaheri M, Shafienia H, Kalantar S M, Montazeri F. Expression of TGFB1 and ERK in cumulus cells: Implications for oocyte maturation in women with polycystic ovary syndrome: A cross-sectional study. IJRM 2025; 23 (12) :1031-1042
URL: http://ijrm.ir/article-1-3605-en.html
URL: http://ijrm.ir/article-1-3605-en.html
1- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Department of Genetics, Faculty of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
2- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Department of Molecular Medicine, School of Advanced Medical Technologies, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
4- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,f.montazeri@ssu.ac.ir
2- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Department of Molecular Medicine, School of Advanced Medical Technologies, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
4- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,
Full-Text [PDF 554 kb]
(300 Downloads)
| Abstract (HTML) (307 Views)
Full-Text: (35 Views)
1. Introduction
Polycystic ovary syndrome (PCOS) is a common endocrine disorder, with a prevalence of 8-13% among reproductive-aged women (1). This condition often leads to infertility due to disrupted oogenesis and poor oocyte quality (2). While in vitro fertilization (IVF) is a standard treatment, PCOS patients face a higher risk of ovarian hyperstimulation, making in vitro maturation (IVM) a safer alternative for promoting oocyte development under controlled laboratory conditions (3). However, in order to enhance the efficiency of IVM, the genes regulating oocyte maturation should be investigated to evaluate oocyte quality (4).
One of the most efficient methods for assessing oocyte quality is analysis of cumulus cells (CCs). Bidirectional communication between the oocyte and CCs has made these cells a non-invasive method to evaluate oocyte quality, embryo competence, and pregnancy outcome (5). Several studies have examined the gene expression profile of CCs during follicular development. Genes expressed in CCs can be valuable for predicting oocyte maturity and embryo development (4). Studies comparing PCOS oocytes with healthy individuals have demonstrated changes in CC gene expression patterns (6-8). By examining the expression profiles of these genes using RNA-Seq and correlating them with clinical data, it has been suggested that genes involved in various signaling pathways, including transforming growth factor-β (TGFB), mitogen-activated protein kinases (MAPK), vascular endothelial growth factor, phosphatidylinositol 3‑kinase/protein kinase B, and steroid hormone synthesis, play roles in CCs during the oocyte maturation process (9).
TGFB1 and extracellular signal-regulated kinase are key regulators of oocyte maturation and follicular development. TGFB1, a member of the TGFB superfamily, influences granulosa and CCs function, promotes follicular growth, and modulates meiotic progression through SMAD-dependent signaling (10). Extracellular signal-regulated kinase (ERK), a central component of the MAPK pathway, plays a critical role in cumulus expansion, resumption of meiosis, and oocyte-cumulus communication (11). Dysregulation of these pathways has been implicated in impaired oocyte competence and abnormal folliculogenesis in PCOS (6). Therefore, evaluating TGFB1 and ERK expression in CCs may provide insight into the molecular mechanisms underlying oocyte quality and developmental potential in PCOS patients undergoing IVM. This study aimed to examine the expression levels of TGFB1 and ERK genes in CCs of immature and mature oocytes obtained from PCOS women to evaluate the relationship between CCs gene expression and the oocyte maturation process.
2. Materials and Methods
2.1. Participants
In this cross-sectional study a total of 30 women with PCOS were selected from individuals referred to Yazd Reproductive Science Institute, Yazd, Iran, between May 2022 and 2023. PCOS was diagnosed based on the presence of at least 2 of the 3 diagnostic criteria established by the Rotterdam Consensus (12), including hyperandrogenism, oligoanovulatory ovarian dysfunction, and polycystic ovarian morphology. Those women aged between 30 and 35 yr, had a number of 10 or more follicular fluid-obtained oocytes after oocyte retrieval, and a normal spermiogram of the partner according to the World Health Organization criteria were included in the study (13). The exclusion criteria included non-PCOS causes of infertility, such as male infertility and hormonal disorders.
2.2. Oocyte retrieval and CCs collection
Controlled ovarian hyperstimulation was conducted for all the women using the antagonist protocol as described previously (14). 36 hr after human chorionic gonadotropin administration, ultrasound-guided oocyte retrieval was performed, and cumulus-oocyte complexes (COCs) were collected in medium for handling and preparation of oocytes (G-IVF, Vitrolife, Sweden), covered with mineral oil (Ovoil, VitroLife). The CCs surrounding each oocyte were mechanically separated and transferred to a labelled sterile 1.5 ml microtube (Eppendorf). Then, CCs were washed twice in phosphate-buffered saline (Sigma-Aldrich, New Zealand) and centrifuged at 5000 g for 1 min. The pellet was stored at -80oC using the appropriate volume of RNAlater preservative solution (Thermo Fisher Scientific, Germany) until RNA extraction for gene expression analysis. The denuded oocytes were placed separately in droplets in G-IVF medium, covered with mineral oil, and incubated at 37oC, with 6% CO2 for 2-3 hr for further assessment of maturation stage.
2.3. Oocyte morphological examination
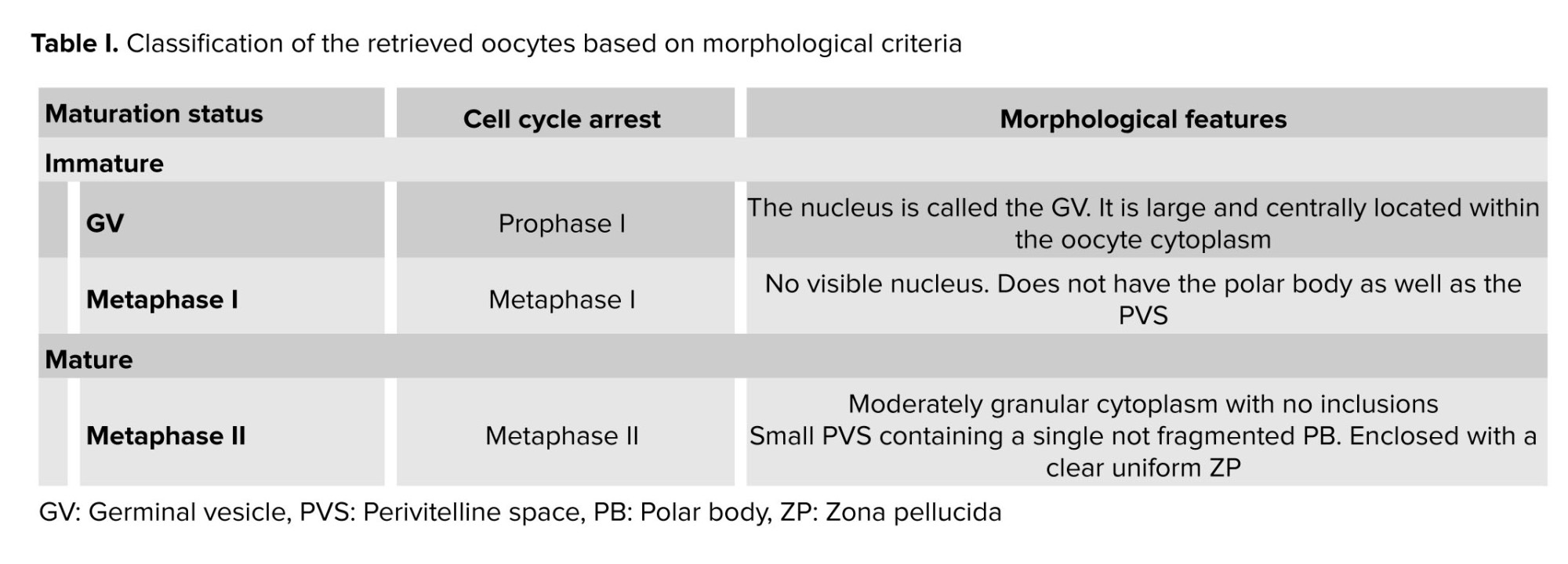
Morphological characteristics of the oocytes were assessed using an inverted microscope (Olympus SZX16, Japan), and their maturation stage was determined. Based on morphological status of the oocytes, the samples were divided into germinal vesicle (GV), Metaphase (M) MI, and MII groups. In this study, immature oocytes in the GV stage and CCs surrounding GV and MII oocytes were utilized for further investigation.
2.4. Oocyte IVM
Immature GV oocytes were cultured in maturation medium (Global Total, Life Global, USA) supplemented with 0.75 IU/mL follicle-stimulating hormone (15) and 0.75 IU/mL luteinizing hormone (16) (Ferring Pharmaceuticals, USA) and incubated at 37°C, 5% CO2, and 95% air with high humidity. After 24 hr, maturation status was assessed using an inverted microscope and the oocytes were divided into immature (I-group) and matured (M-group) groups according to morphological criteria (Table I).
2.5. Intracytoplasmic sperm injection (ICSI) and embryo evaluation
In vitro matured oocytes (M-Group) were used for ICSI to compare the rates of fertilization. The swim-up or density gradient centrifugation techniques were used for sperm preparation. The ICSI procedure was carried out as previously reported (17). Each injected oocyte was then transferred to microdroplets of G1 medium (Vitrolife, Sweden), covered with mineral oil and incubated at 37°C with 6% CO2. 18 hr after ICSI, fertilization was evaluated based on the appearance of 2 pronuclei and 2 phosphate buffered saline. Fertilized oocytes were then cultured in embryo maintenance medium (Global Total LP, Life Global, USA) for 72 hr, and embryo quality was assessed according to the standard embryo grading system (18). Embryos in grades A and B were considered high quality, and those in grades C and D were considered low quality.
2.6. Quantitative expression of TGFB1 and ERK
The CCs surrounding GV and MII oocytes were used to investigate the quantitative expression levels of TGFB1 and ERK. Total RNA was extracted from CCs of each oocyte using TRIzol® Reagent (Invitrogen) following the manufacturer’s protocol. The RNA quality and quantity were assessed using a Nanodrop spectrophotometer (DeNovix, USA).
cDNA was synthesized from 1 µg of purified RNA using a RevertAid First Strand cDNA Synthesis kit (Thermo Fisher Scientific, USA) according to the manufacturer’s instructions. Relative expression levels of TGFB1 and ERK in CCs were evaluated by quantitative reverse transcriptase polymerase chain reaction (qRT-PCR). Amplification reactions were conducted using SYBR Green qPCR Master Mix 2X (Ampliqon, Denmark) on the StepOne™ real-time PCR system (Life Technologies Co., Taiwan) in accordance with the manufacturer's protocol.
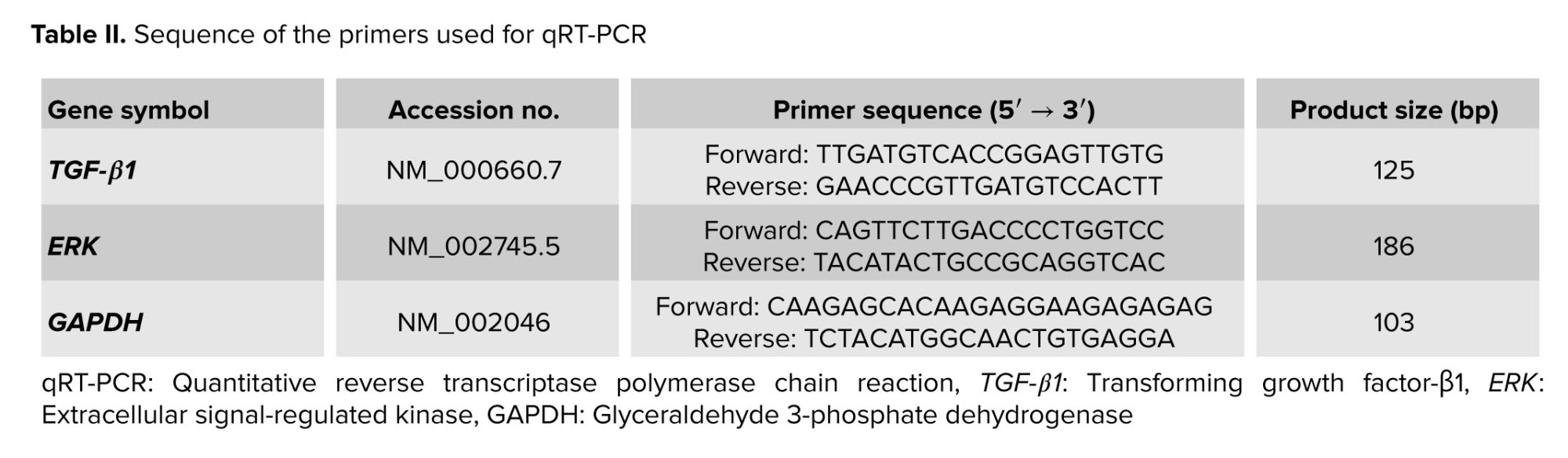
The housekeeping gene GAPDH was used as the reference gene to normalize for RNA input in each sample. The gene-specific qRT-PCR primers used along with their expected product sizes are listed in table II. The PCR thermal cycling conditions were 95°C for 2 min, followed by 40 cycles at 95°C for 10 sec and 60°C for 15 sec. The reaction was continued by a melt curve step at 95°C for 15 sec, 60°C for 1 min, and 95°C for 15 sec to evaluate the specificity of amplification. All PCR reactions were performed in duplicate to minimize the sampling error, and relative quantitation was calculated using the 2−ΔCt method.
2.7. Ethical Considerations
The study was approved by the Ethics Committee of Yazd Reproductive Science Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran (Code: IR.SSU.MEDICINE.REC.1401.021), and informed consent was obtained from all participants.
2.8. Statistical Analysis
In this study, the data analyzed were numerical (quantitative) for gene expression levels of ERK and TGFB1, with any categorical variables noted but not central to the primary endpoints. Results are presented as mean ± SEM for all quantitative variables. Normality of distributions within each group was assessed using the Kolmogorov-Smirnov test. Based on the distribution form, a parametric approach was employed; differences between groups in relative ERK and TGFB1 expression were evaluated with Student’s t test. All statistical analyses were conducted in GraphPad Prism 9 (GraphPad Software, USA).
3. Results
3.1. Clinical characteristics of PCOS women
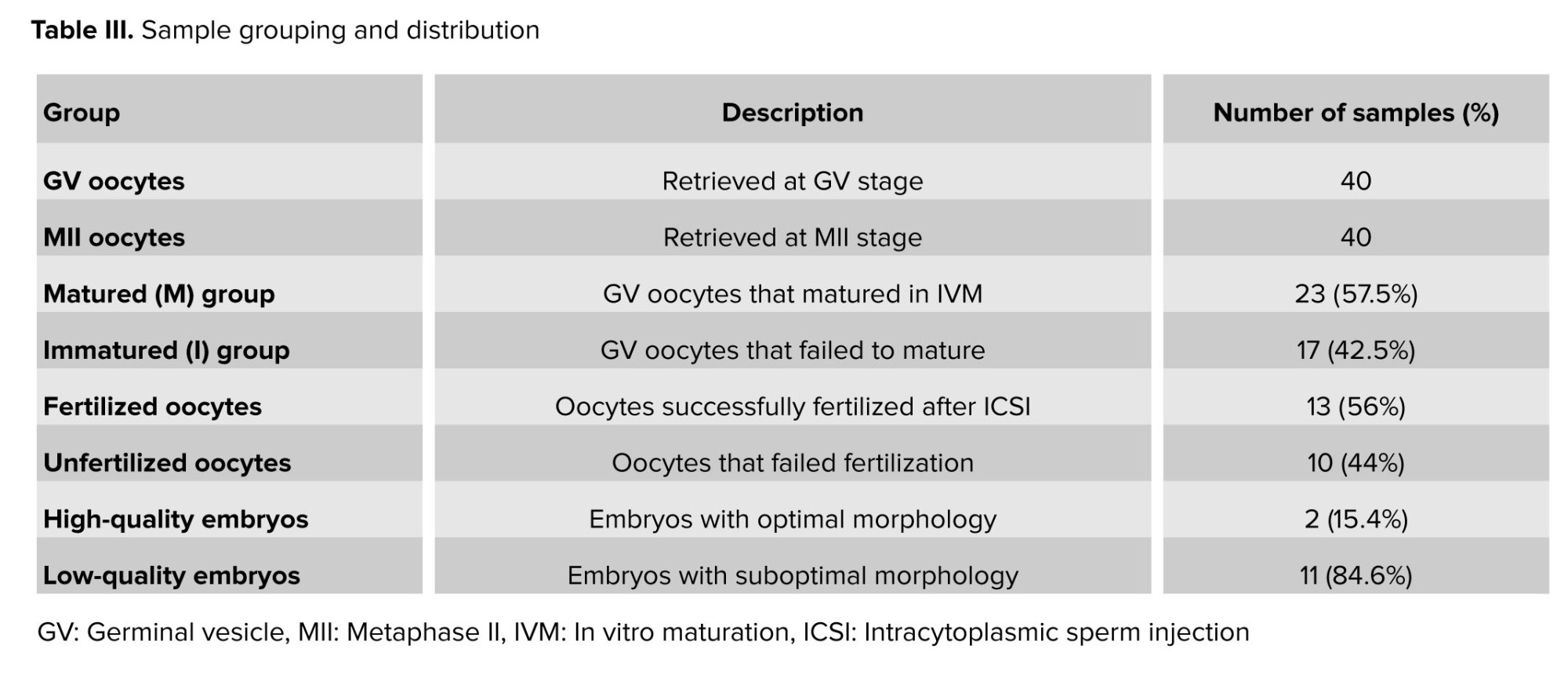
In this study, 40 COCs with GV oocytes and 40 COCs with MII oocytes were collected from 30 women diagnosed with PCOS undergoing IVF treatment. Based on our results, 23 oocytes (57.5%) successfully matured following IVM and were assigned to the M-group, while 17 oocytes (42.5%) remained immature and were categorized into the I-group. Among the 23 matured oocytes subjected to ICSI, 13 (56%) were fertilized, whereas 10 failed to fertilize. Of the resulting embryos, only 2 met the criteria for high quality, and 11 were classified as low quality. CCs corresponding to each oocyte or embryo stage were grouped accordingly, and the expression levels of TGFB1 and ERK genes were analyzed. The number of collected oocytes and organization of groups are summarized in table III.
3.2. Comparison of the expression of TGFB1 and ERK in CCs of GV and MII oocytes
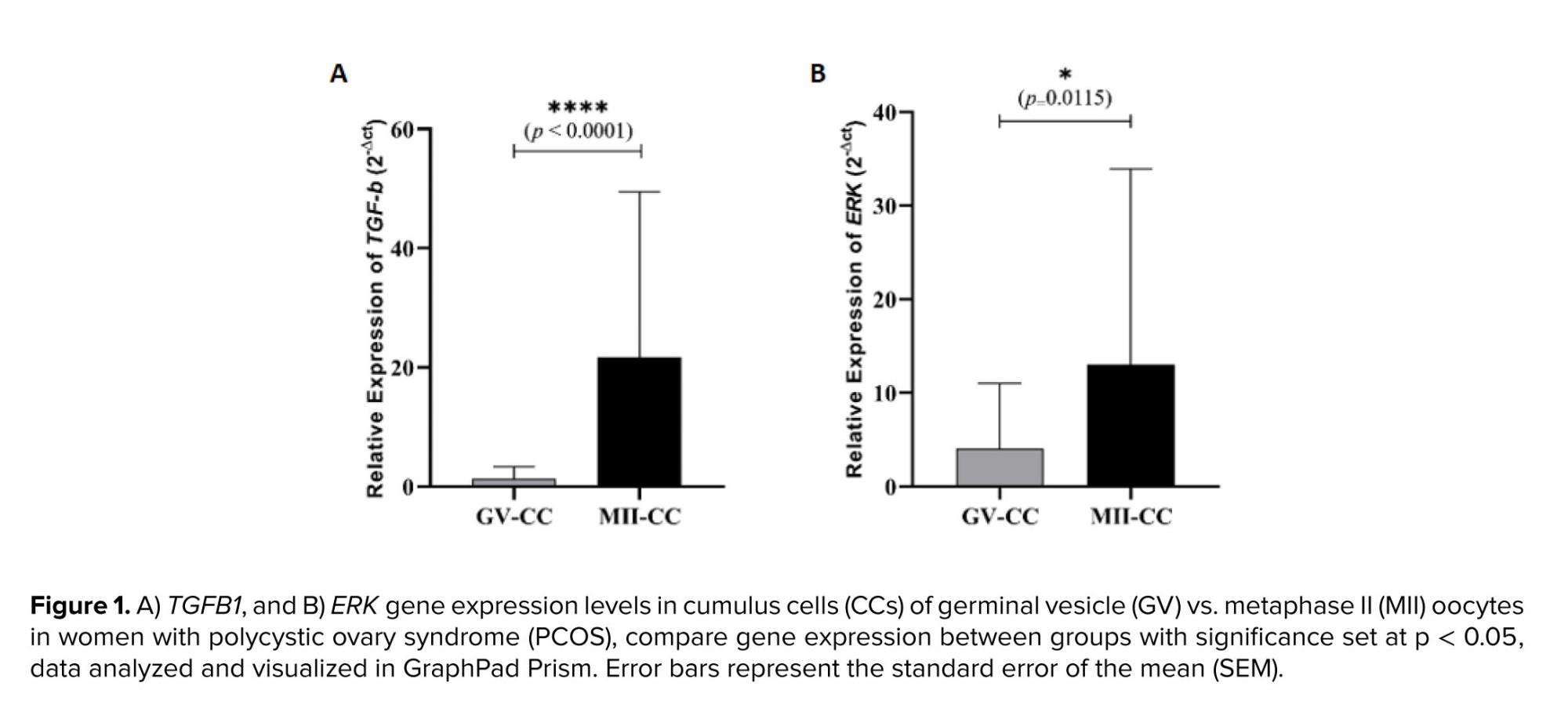
In this study, the relative expression levels of TGFB1 and ERK were evaluated in 40 samples of CCs surrounding GV oocytes and 40 samples of CCs surrounding MII oocytes using qRT-PCR. The comparison between the 2 groups revealed that the mean relative expression of both genes was significantly lower in the GV oocyte group than in the MII oocyte group. Furthermore, the analysis showed that the mean relative expression of TGFB1 in the MII oocyte group was nearly twice as high as that of ERK in the same group (Figure 1).
3.3. Relationship between the expression of TGFB1 and ERK in CCs and IVM outcome
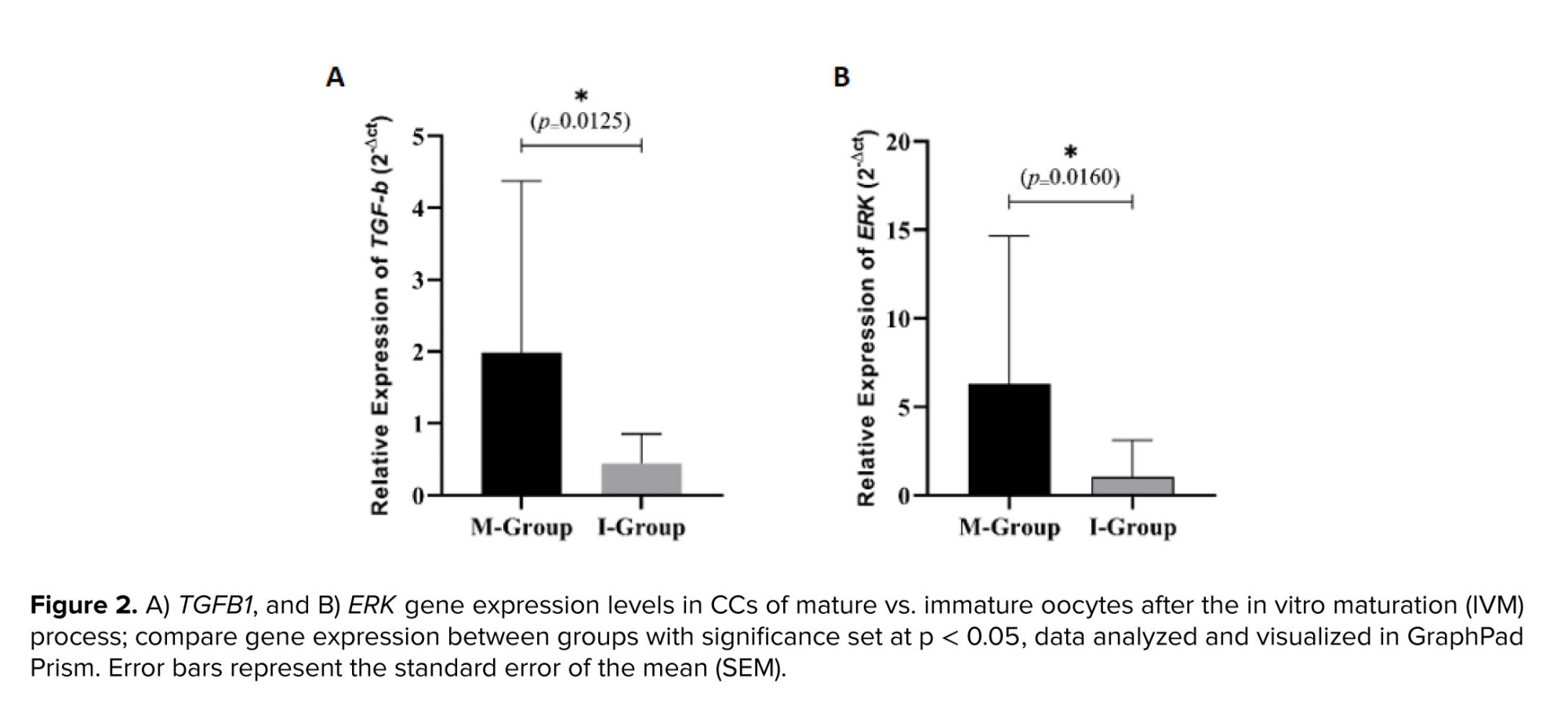
Our findings indicate that out of 40 GV oocytes that underwent the IVM process, 23 matured, while 17 remained immature, resulting in a maturation rate of 57.5%. The comparison of TGFB1 and ERK gene expression between CCs of matured and immature oocytes revealed that the mean relative expression of both genes was significantly higher in the matured group (M-group) than in the immature group (I-group) (Figure 2).
3.4. Relationship between the expression of TGFB1 and ERK in CCs and fertilization rate
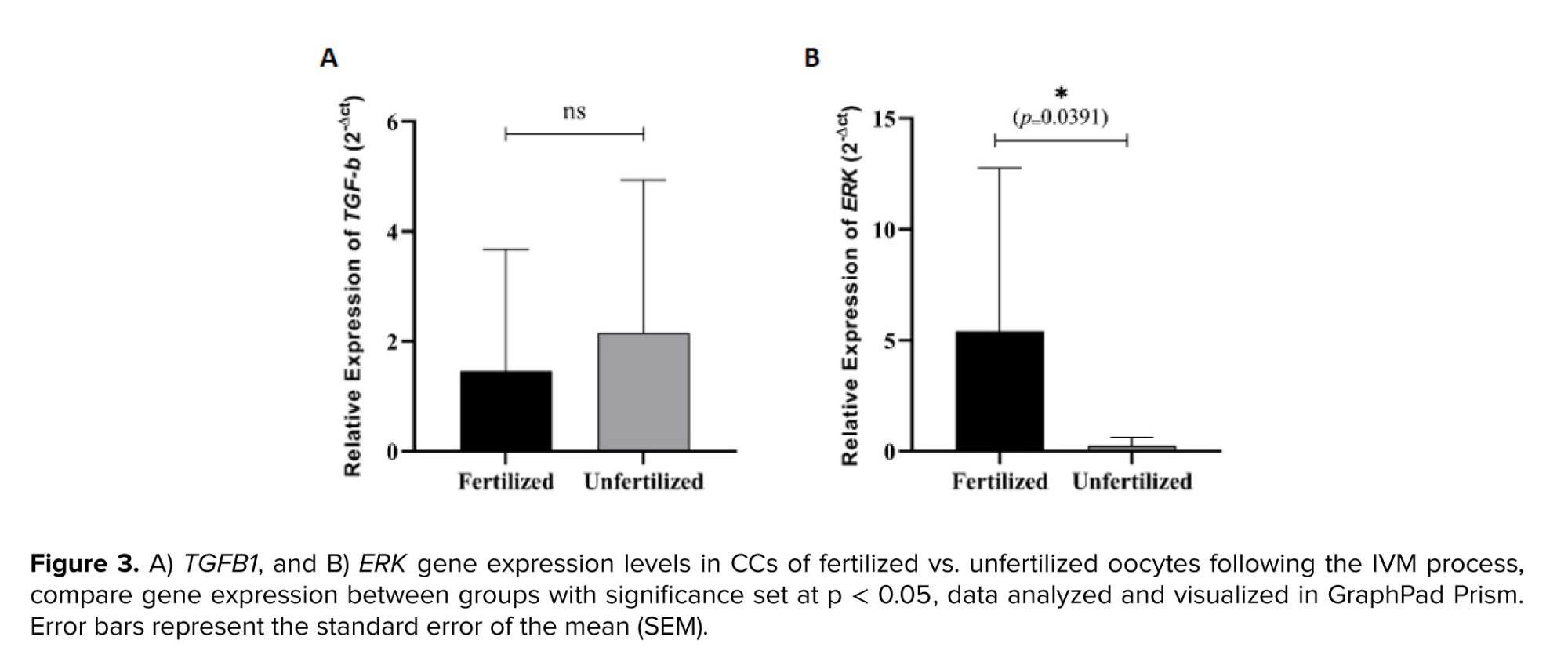
Our findings indicate that out of 23 IVM-matured oocytes that underwent ICSI, 13 were successfully fertilized, resulting in a fertilization rate of 56%. The comparison of TGFB1 and ERK gene expression in CCs between fertilized and unfertilized oocytes revealed that there was no significant difference in the mean relative expression of TGFB1 between fertilized and unfertilized oocytes (Figure 3A). However, a decreasing trend in TGFB1 expression was observed in fertilized oocytes compared to unfertilized ones, though this difference was not statistically significant. In contrast, the mean relative expression of ERK was significantly higher in the fertilized group compared to the unfertilized group (Figure 3B).
3.5. Relationship between the expression of TGFB1 and ERK in CCs and embryo quality
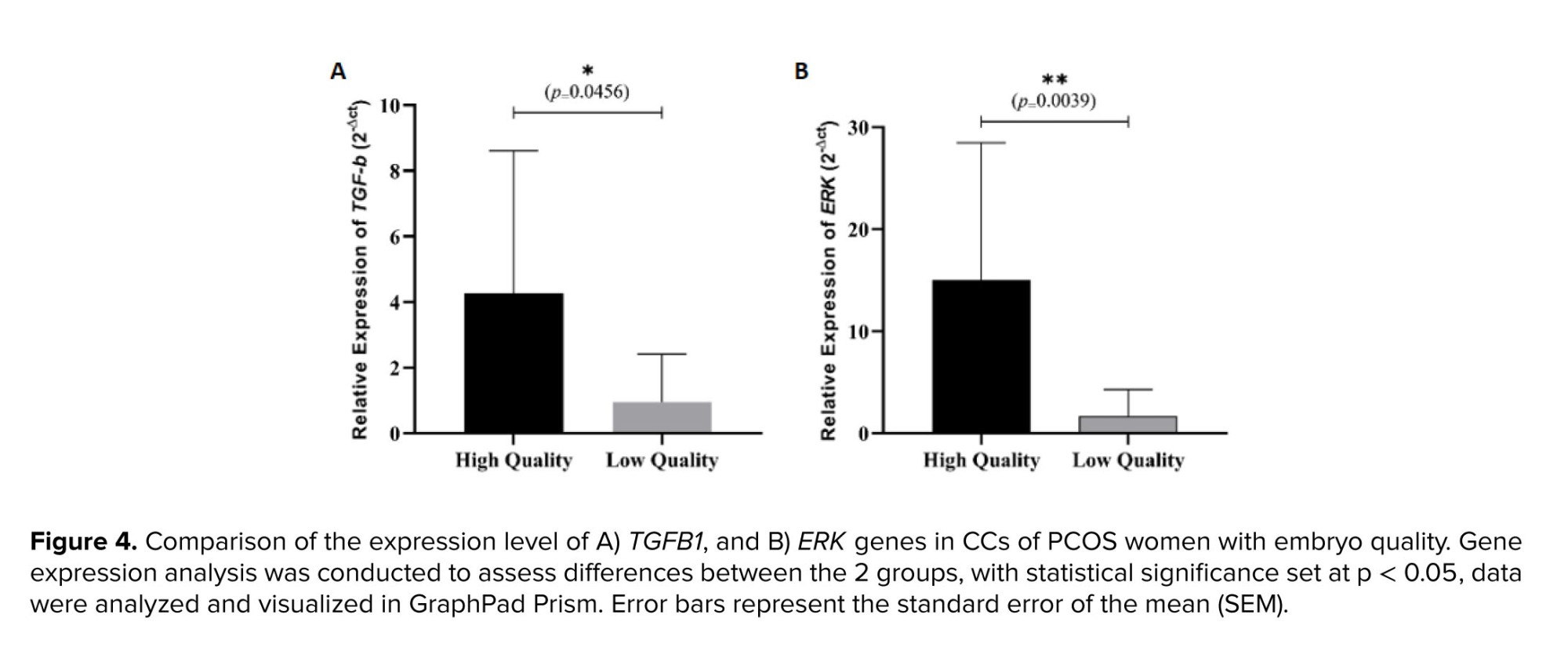
Our findings demonstrate that out of 13 in vitro matured oocytes that successfully fertilized, 11 embryos were classified as low quality. In comparison, only 2 embryos were deemed high quality based on standard embryological assessment criteria (18). A comparative analysis of TGFB1 and ERK gene expression levels in CCs between low and high-quality embryos revealed a notable difference (Figure 4). The mean relative expression of both TGFB1 and ERK was higher in high-quality embryos compared to those classified as low quality. Further statistical analysis confirmed that these differences were significant (p < 0.05).
4. Discussion
In this study, we investigated the expression levels of TGFB1 and ERK genes in CCs associated with different stages of oocyte development in women with PCOS. Previous studies have demonstrated that gene expression profiles in CCs of women with PCOS differ significantly from those of healthy individuals. For example, Sikiru et al. identified over 3200 dysregulated genes in PCOS CCs, involved in reproductive and metabolic pathways (8). While these studies provide valuable context, our research specifically focused on intra-group variations within the PCOS population to explore the role of TGFB1 and ERK in oocyte development process.
4.1. TGFB1 and ERK expression in GV and MII oocytes
Our results showed that the relative expression levels of TGFB1 and ERK were significantly lower in CCs of GV oocytes compared to MII oocytes. TGFB1, a well-known growth factor, is involved in regulating cellular differentiation and follicular development (10). At the same time, ERK, a key component of the MAPK signaling pathway, is crucial for CC-oocyte communication and maturation (19).
Studies suggest that the TGFB pathway plays a crucial role in PCOS pathogenesis. Dysregulation of TGFB1 leads to increased collagen production in the ovaries, contributing to PCOS symptoms and metabolic disturbances (20). TGFB1 also influences granulosa cell proliferation and follicular growth (10). Additionally, it regulates androgen receptor activity, which may affect ovarian function in PCOS (20). Given its pivotal role, TGFB1 may serve as a potential therapeutic target for improving oocyte maturation and fertility outcomes in PCOS women (21).
Emerging studies suggest a potential link between the ERK pathway and PCOS development. Dysregulation of this pathway may increase ovarian cell growth and survival. These changes may lead to the formation of abnormal tissues and ovarian cyst enlargement (22). The abnormal activation of the ERK pathway may also play a significant role in regulating hormonal disturbances in PCOS (23). These studies may provide insights into how these pathways contribute to PCOS onset and progression and help develop more effective therapeutic strategies (24).
4.2. TGFB1 and ERK expression and IVM outcome
Several studies, including our previous works, have explored the relationship between gene expression in CCs and oocyte development (25, 26), supporting our findings that higher expression levels of certain genes are associated with improved maturation rates in PCOS women. Our findings revealed the maturation rate of 57.5% following IVM process. Notably, the expression of both TGFB1 and ERK was significantly higher in CCs of matured oocytes compared to immature oocytes. These results align with previous studies indicating the involvement of TGFB in follicular growth, while ERK activation promotes the resumption of meiosis. A previous study demonstrated that genetic inactivation of the ERK-1 and ERK-2 cascade in mouse granulosa cells leads to sterility, affecting oocyte meiotic maturation, cumulus expansion, and follicle rupture (27). This suggests that the molecular crosstalk between CCs and oocytes, facilitated by these signaling molecules, is critical for successful maturation, particularly in PCOS women who often exhibit dysregulated folliculogenesis.
4.3. TGFB1 and ERK expression and fertilization rate
Based on our ICSI results, the fertilization rate was 56%. Interestingly, the expression of ERK was significantly higher in CCs of fertilized compared to unfertilized oocytes, suggesting the role of ERK-mediated signaling in fertilization success. In contrast, TGFB1 expression showed a decreasing trend in fertilized oocytes, although the difference was not statistically significant. This may reflect the stage-specific role of TGFB1, where its influence is more pronounced during early maturation rather than fertilization success. It may indicate that while TGFB1 plays a crucial role in the early maturation stages, ERK is more directly involved in preparing the oocyte for successful fertilization. Furthermore, the ERK1/2-early growth response 1 pathway was identified as a key downstream mediator in the ovulatory process (28). Dompe et al. provided insights into the role of follicular fluid in bidirectional communication between oocytes and granulosa cells, which is essential for successful fertilization (29). Additionally, another study indicated that farnesyl diphosphate synthase protein in ovarian tissue of PCOS mice could regulate the proliferation of ovarian GCs by modulating the MAPK/ERK pathway (30).
4.4. TGFB and ERK expression and embryo quality
Our analysis of embryo quality revealed higher expression of TGFB1 and ERK in CCs in high-quality compared to low-quality embryos. High expression of TGFB1 might indicate better cellular communication and developmental support from CCs (31). Despite the lack of a significant difference in TGFB1 expression between fertilized and unfertilized oocytes, the gene’s expression was again elevated in CCs of high-quality embryos, suggesting a potential downstream impact on embryonic development. These findings suggest that its role may vary across different stages of oocyte development. At the same time, ERK upregulation could reflect enhanced oocyte competence and metabolic activity (32), both of which are crucial for embryo viability. Faizal et al. found that higher expression of hyaluronic acid synthase 2, cyclooxygenase 2, and gremlin in CCs correlated with better oocyte development (33). Similarly, a systematic review identified increased expression of genes encoding extracellular matrix components and TGF-β family members in CCs as positively correlated with pregnancy rates (34). While these studies did not specifically examine TGFB and ERK expressions, they underscore the significance of gene expression in CCs as indicators of embryo quality. Our findings align with this body of research, suggesting that elevated TGFB1 and ERK expression in CCs may serve as potential biomarkers for selecting high-quality embryos in assisted reproductive technologies. Supporting this, Zhang et al. identified several CC-expressed genes, including TGFB-related pathways, that were predictive of oocyte developmental competence. Their findings reinforce the importance of CC gene signatures in evaluating embryo quality, particularly in PCOS women undergoing IVM (11).
4.5. Clinical implications
Our findings underscore the importance of TGFB and ERK as potential candidate biomarkers for assessing oocyte competence and embryo developmental potential, particularly in women with PCOS undergoing IVM. Given the lower maturation and fertilization rates observed in these women, optimizing conditions that regulate TGFB and ERK expression in CCs may lead to improved clinical outcomes. Further research is needed to determine whether modulating these pathways through growth factor supplementation or targeted molecular interventions could enhance IVM success and overall fertility outcomes in PCOS women.
4.6. Strengths and limitations
This study has several limitations that should be acknowledged. First, the absence of a healthy control group limits the ability to compare gene expression patterns between PCOS and non-PCOS populations. However, we aimed to investigate gene expression dynamics within the PCOS cohort to identify potential biomarkers relevant to IVM outcomes. Second, the analysis of multiple oocytes from the same patient introduces a potential cluster effect, which may violate the assumption of data independence; this was addressed through appropriate statistical adjustments. Third, the relatively small sample size may reduce the statistical power and generalizability of the findings. Lastly, the limited embryo quality observed reflects the clinical challenges of IVM in PCOS patients and may influence the interpretation of biomarker relevance. Therefore, further studies with larger sample size are warranted to validate these findings.
5. Conclusion
This study provides valuable insights into the molecular mechanisms underlying oocyte maturation, fertilization, and embryo quality in PCOS women undergoing IVF and IVM. The observed associations between TGFB1 and ERK expression in CCs and reproductive outcomes highlight their potential as candidate biomarkers for selecting competent oocytes and predicting embryo viability. While TGFB1 and ERK expression showed promising associations with oocyte and embryo quality, larger-scale studies are required to confirm their biomarker potential. Future studies should focus on exploring therapeutic strategies that optimize the activation of these pathways to enhance assisted reproductive technology success rates for women with PCOS.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author Contributions
SM. Kalantar, F. Montazeri, and MH. Fakherbaheri designed the study. MH. Fakherbaheri also conducted the research. F. Montazeri and H. Shafienia monitored, evaluated, and analyzed the results. MH. Fakherbaheri and F. Montazeri contributed to the review of the manuscript. All authors approved the final version and attest to the integrity of the data.
Acknowledgments
This study received no financial support and artificial intelligence tools are not used in this article.
Conflict of Interest
The authors declare that there is no conflict of interest.
Polycystic ovary syndrome (PCOS) is a common endocrine disorder, with a prevalence of 8-13% among reproductive-aged women (1). This condition often leads to infertility due to disrupted oogenesis and poor oocyte quality (2). While in vitro fertilization (IVF) is a standard treatment, PCOS patients face a higher risk of ovarian hyperstimulation, making in vitro maturation (IVM) a safer alternative for promoting oocyte development under controlled laboratory conditions (3). However, in order to enhance the efficiency of IVM, the genes regulating oocyte maturation should be investigated to evaluate oocyte quality (4).
One of the most efficient methods for assessing oocyte quality is analysis of cumulus cells (CCs). Bidirectional communication between the oocyte and CCs has made these cells a non-invasive method to evaluate oocyte quality, embryo competence, and pregnancy outcome (5). Several studies have examined the gene expression profile of CCs during follicular development. Genes expressed in CCs can be valuable for predicting oocyte maturity and embryo development (4). Studies comparing PCOS oocytes with healthy individuals have demonstrated changes in CC gene expression patterns (6-8). By examining the expression profiles of these genes using RNA-Seq and correlating them with clinical data, it has been suggested that genes involved in various signaling pathways, including transforming growth factor-β (TGFB), mitogen-activated protein kinases (MAPK), vascular endothelial growth factor, phosphatidylinositol 3‑kinase/protein kinase B, and steroid hormone synthesis, play roles in CCs during the oocyte maturation process (9).
TGFB1 and extracellular signal-regulated kinase are key regulators of oocyte maturation and follicular development. TGFB1, a member of the TGFB superfamily, influences granulosa and CCs function, promotes follicular growth, and modulates meiotic progression through SMAD-dependent signaling (10). Extracellular signal-regulated kinase (ERK), a central component of the MAPK pathway, plays a critical role in cumulus expansion, resumption of meiosis, and oocyte-cumulus communication (11). Dysregulation of these pathways has been implicated in impaired oocyte competence and abnormal folliculogenesis in PCOS (6). Therefore, evaluating TGFB1 and ERK expression in CCs may provide insight into the molecular mechanisms underlying oocyte quality and developmental potential in PCOS patients undergoing IVM. This study aimed to examine the expression levels of TGFB1 and ERK genes in CCs of immature and mature oocytes obtained from PCOS women to evaluate the relationship between CCs gene expression and the oocyte maturation process.
2. Materials and Methods
2.1. Participants
In this cross-sectional study a total of 30 women with PCOS were selected from individuals referred to Yazd Reproductive Science Institute, Yazd, Iran, between May 2022 and 2023. PCOS was diagnosed based on the presence of at least 2 of the 3 diagnostic criteria established by the Rotterdam Consensus (12), including hyperandrogenism, oligoanovulatory ovarian dysfunction, and polycystic ovarian morphology. Those women aged between 30 and 35 yr, had a number of 10 or more follicular fluid-obtained oocytes after oocyte retrieval, and a normal spermiogram of the partner according to the World Health Organization criteria were included in the study (13). The exclusion criteria included non-PCOS causes of infertility, such as male infertility and hormonal disorders.
2.2. Oocyte retrieval and CCs collection
Controlled ovarian hyperstimulation was conducted for all the women using the antagonist protocol as described previously (14). 36 hr after human chorionic gonadotropin administration, ultrasound-guided oocyte retrieval was performed, and cumulus-oocyte complexes (COCs) were collected in medium for handling and preparation of oocytes (G-IVF, Vitrolife, Sweden), covered with mineral oil (Ovoil, VitroLife). The CCs surrounding each oocyte were mechanically separated and transferred to a labelled sterile 1.5 ml microtube (Eppendorf). Then, CCs were washed twice in phosphate-buffered saline (Sigma-Aldrich, New Zealand) and centrifuged at 5000 g for 1 min. The pellet was stored at -80oC using the appropriate volume of RNAlater preservative solution (Thermo Fisher Scientific, Germany) until RNA extraction for gene expression analysis. The denuded oocytes were placed separately in droplets in G-IVF medium, covered with mineral oil, and incubated at 37oC, with 6% CO2 for 2-3 hr for further assessment of maturation stage.
2.3. Oocyte morphological examination
Morphological characteristics of the oocytes were assessed using an inverted microscope (Olympus SZX16, Japan), and their maturation stage was determined. Based on morphological status of the oocytes, the samples were divided into germinal vesicle (GV), Metaphase (M) MI, and MII groups. In this study, immature oocytes in the GV stage and CCs surrounding GV and MII oocytes were utilized for further investigation.
2.4. Oocyte IVM
Immature GV oocytes were cultured in maturation medium (Global Total, Life Global, USA) supplemented with 0.75 IU/mL follicle-stimulating hormone (15) and 0.75 IU/mL luteinizing hormone (16) (Ferring Pharmaceuticals, USA) and incubated at 37°C, 5% CO2, and 95% air with high humidity. After 24 hr, maturation status was assessed using an inverted microscope and the oocytes were divided into immature (I-group) and matured (M-group) groups according to morphological criteria (Table I).
2.5. Intracytoplasmic sperm injection (ICSI) and embryo evaluation
In vitro matured oocytes (M-Group) were used for ICSI to compare the rates of fertilization. The swim-up or density gradient centrifugation techniques were used for sperm preparation. The ICSI procedure was carried out as previously reported (17). Each injected oocyte was then transferred to microdroplets of G1 medium (Vitrolife, Sweden), covered with mineral oil and incubated at 37°C with 6% CO2. 18 hr after ICSI, fertilization was evaluated based on the appearance of 2 pronuclei and 2 phosphate buffered saline. Fertilized oocytes were then cultured in embryo maintenance medium (Global Total LP, Life Global, USA) for 72 hr, and embryo quality was assessed according to the standard embryo grading system (18). Embryos in grades A and B were considered high quality, and those in grades C and D were considered low quality.
2.6. Quantitative expression of TGFB1 and ERK
The CCs surrounding GV and MII oocytes were used to investigate the quantitative expression levels of TGFB1 and ERK. Total RNA was extracted from CCs of each oocyte using TRIzol® Reagent (Invitrogen) following the manufacturer’s protocol. The RNA quality and quantity were assessed using a Nanodrop spectrophotometer (DeNovix, USA).
cDNA was synthesized from 1 µg of purified RNA using a RevertAid First Strand cDNA Synthesis kit (Thermo Fisher Scientific, USA) according to the manufacturer’s instructions. Relative expression levels of TGFB1 and ERK in CCs were evaluated by quantitative reverse transcriptase polymerase chain reaction (qRT-PCR). Amplification reactions were conducted using SYBR Green qPCR Master Mix 2X (Ampliqon, Denmark) on the StepOne™ real-time PCR system (Life Technologies Co., Taiwan) in accordance with the manufacturer's protocol.
The housekeeping gene GAPDH was used as the reference gene to normalize for RNA input in each sample. The gene-specific qRT-PCR primers used along with their expected product sizes are listed in table II. The PCR thermal cycling conditions were 95°C for 2 min, followed by 40 cycles at 95°C for 10 sec and 60°C for 15 sec. The reaction was continued by a melt curve step at 95°C for 15 sec, 60°C for 1 min, and 95°C for 15 sec to evaluate the specificity of amplification. All PCR reactions were performed in duplicate to minimize the sampling error, and relative quantitation was calculated using the 2−ΔCt method.


2.7. Ethical Considerations
The study was approved by the Ethics Committee of Yazd Reproductive Science Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran (Code: IR.SSU.MEDICINE.REC.1401.021), and informed consent was obtained from all participants.
2.8. Statistical Analysis
In this study, the data analyzed were numerical (quantitative) for gene expression levels of ERK and TGFB1, with any categorical variables noted but not central to the primary endpoints. Results are presented as mean ± SEM for all quantitative variables. Normality of distributions within each group was assessed using the Kolmogorov-Smirnov test. Based on the distribution form, a parametric approach was employed; differences between groups in relative ERK and TGFB1 expression were evaluated with Student’s t test. All statistical analyses were conducted in GraphPad Prism 9 (GraphPad Software, USA).
3. Results
3.1. Clinical characteristics of PCOS women
In this study, 40 COCs with GV oocytes and 40 COCs with MII oocytes were collected from 30 women diagnosed with PCOS undergoing IVF treatment. Based on our results, 23 oocytes (57.5%) successfully matured following IVM and were assigned to the M-group, while 17 oocytes (42.5%) remained immature and were categorized into the I-group. Among the 23 matured oocytes subjected to ICSI, 13 (56%) were fertilized, whereas 10 failed to fertilize. Of the resulting embryos, only 2 met the criteria for high quality, and 11 were classified as low quality. CCs corresponding to each oocyte or embryo stage were grouped accordingly, and the expression levels of TGFB1 and ERK genes were analyzed. The number of collected oocytes and organization of groups are summarized in table III.
3.2. Comparison of the expression of TGFB1 and ERK in CCs of GV and MII oocytes
In this study, the relative expression levels of TGFB1 and ERK were evaluated in 40 samples of CCs surrounding GV oocytes and 40 samples of CCs surrounding MII oocytes using qRT-PCR. The comparison between the 2 groups revealed that the mean relative expression of both genes was significantly lower in the GV oocyte group than in the MII oocyte group. Furthermore, the analysis showed that the mean relative expression of TGFB1 in the MII oocyte group was nearly twice as high as that of ERK in the same group (Figure 1).
3.3. Relationship between the expression of TGFB1 and ERK in CCs and IVM outcome
Our findings indicate that out of 40 GV oocytes that underwent the IVM process, 23 matured, while 17 remained immature, resulting in a maturation rate of 57.5%. The comparison of TGFB1 and ERK gene expression between CCs of matured and immature oocytes revealed that the mean relative expression of both genes was significantly higher in the matured group (M-group) than in the immature group (I-group) (Figure 2).
3.4. Relationship between the expression of TGFB1 and ERK in CCs and fertilization rate
Our findings indicate that out of 23 IVM-matured oocytes that underwent ICSI, 13 were successfully fertilized, resulting in a fertilization rate of 56%. The comparison of TGFB1 and ERK gene expression in CCs between fertilized and unfertilized oocytes revealed that there was no significant difference in the mean relative expression of TGFB1 between fertilized and unfertilized oocytes (Figure 3A). However, a decreasing trend in TGFB1 expression was observed in fertilized oocytes compared to unfertilized ones, though this difference was not statistically significant. In contrast, the mean relative expression of ERK was significantly higher in the fertilized group compared to the unfertilized group (Figure 3B).
3.5. Relationship between the expression of TGFB1 and ERK in CCs and embryo quality
Our findings demonstrate that out of 13 in vitro matured oocytes that successfully fertilized, 11 embryos were classified as low quality. In comparison, only 2 embryos were deemed high quality based on standard embryological assessment criteria (18). A comparative analysis of TGFB1 and ERK gene expression levels in CCs between low and high-quality embryos revealed a notable difference (Figure 4). The mean relative expression of both TGFB1 and ERK was higher in high-quality embryos compared to those classified as low quality. Further statistical analysis confirmed that these differences were significant (p < 0.05).





4. Discussion
In this study, we investigated the expression levels of TGFB1 and ERK genes in CCs associated with different stages of oocyte development in women with PCOS. Previous studies have demonstrated that gene expression profiles in CCs of women with PCOS differ significantly from those of healthy individuals. For example, Sikiru et al. identified over 3200 dysregulated genes in PCOS CCs, involved in reproductive and metabolic pathways (8). While these studies provide valuable context, our research specifically focused on intra-group variations within the PCOS population to explore the role of TGFB1 and ERK in oocyte development process.
4.1. TGFB1 and ERK expression in GV and MII oocytes
Our results showed that the relative expression levels of TGFB1 and ERK were significantly lower in CCs of GV oocytes compared to MII oocytes. TGFB1, a well-known growth factor, is involved in regulating cellular differentiation and follicular development (10). At the same time, ERK, a key component of the MAPK signaling pathway, is crucial for CC-oocyte communication and maturation (19).
Studies suggest that the TGFB pathway plays a crucial role in PCOS pathogenesis. Dysregulation of TGFB1 leads to increased collagen production in the ovaries, contributing to PCOS symptoms and metabolic disturbances (20). TGFB1 also influences granulosa cell proliferation and follicular growth (10). Additionally, it regulates androgen receptor activity, which may affect ovarian function in PCOS (20). Given its pivotal role, TGFB1 may serve as a potential therapeutic target for improving oocyte maturation and fertility outcomes in PCOS women (21).
Emerging studies suggest a potential link between the ERK pathway and PCOS development. Dysregulation of this pathway may increase ovarian cell growth and survival. These changes may lead to the formation of abnormal tissues and ovarian cyst enlargement (22). The abnormal activation of the ERK pathway may also play a significant role in regulating hormonal disturbances in PCOS (23). These studies may provide insights into how these pathways contribute to PCOS onset and progression and help develop more effective therapeutic strategies (24).
4.2. TGFB1 and ERK expression and IVM outcome
Several studies, including our previous works, have explored the relationship between gene expression in CCs and oocyte development (25, 26), supporting our findings that higher expression levels of certain genes are associated with improved maturation rates in PCOS women. Our findings revealed the maturation rate of 57.5% following IVM process. Notably, the expression of both TGFB1 and ERK was significantly higher in CCs of matured oocytes compared to immature oocytes. These results align with previous studies indicating the involvement of TGFB in follicular growth, while ERK activation promotes the resumption of meiosis. A previous study demonstrated that genetic inactivation of the ERK-1 and ERK-2 cascade in mouse granulosa cells leads to sterility, affecting oocyte meiotic maturation, cumulus expansion, and follicle rupture (27). This suggests that the molecular crosstalk between CCs and oocytes, facilitated by these signaling molecules, is critical for successful maturation, particularly in PCOS women who often exhibit dysregulated folliculogenesis.
4.3. TGFB1 and ERK expression and fertilization rate
Based on our ICSI results, the fertilization rate was 56%. Interestingly, the expression of ERK was significantly higher in CCs of fertilized compared to unfertilized oocytes, suggesting the role of ERK-mediated signaling in fertilization success. In contrast, TGFB1 expression showed a decreasing trend in fertilized oocytes, although the difference was not statistically significant. This may reflect the stage-specific role of TGFB1, where its influence is more pronounced during early maturation rather than fertilization success. It may indicate that while TGFB1 plays a crucial role in the early maturation stages, ERK is more directly involved in preparing the oocyte for successful fertilization. Furthermore, the ERK1/2-early growth response 1 pathway was identified as a key downstream mediator in the ovulatory process (28). Dompe et al. provided insights into the role of follicular fluid in bidirectional communication between oocytes and granulosa cells, which is essential for successful fertilization (29). Additionally, another study indicated that farnesyl diphosphate synthase protein in ovarian tissue of PCOS mice could regulate the proliferation of ovarian GCs by modulating the MAPK/ERK pathway (30).
4.4. TGFB and ERK expression and embryo quality
Our analysis of embryo quality revealed higher expression of TGFB1 and ERK in CCs in high-quality compared to low-quality embryos. High expression of TGFB1 might indicate better cellular communication and developmental support from CCs (31). Despite the lack of a significant difference in TGFB1 expression between fertilized and unfertilized oocytes, the gene’s expression was again elevated in CCs of high-quality embryos, suggesting a potential downstream impact on embryonic development. These findings suggest that its role may vary across different stages of oocyte development. At the same time, ERK upregulation could reflect enhanced oocyte competence and metabolic activity (32), both of which are crucial for embryo viability. Faizal et al. found that higher expression of hyaluronic acid synthase 2, cyclooxygenase 2, and gremlin in CCs correlated with better oocyte development (33). Similarly, a systematic review identified increased expression of genes encoding extracellular matrix components and TGF-β family members in CCs as positively correlated with pregnancy rates (34). While these studies did not specifically examine TGFB and ERK expressions, they underscore the significance of gene expression in CCs as indicators of embryo quality. Our findings align with this body of research, suggesting that elevated TGFB1 and ERK expression in CCs may serve as potential biomarkers for selecting high-quality embryos in assisted reproductive technologies. Supporting this, Zhang et al. identified several CC-expressed genes, including TGFB-related pathways, that were predictive of oocyte developmental competence. Their findings reinforce the importance of CC gene signatures in evaluating embryo quality, particularly in PCOS women undergoing IVM (11).
4.5. Clinical implications
Our findings underscore the importance of TGFB and ERK as potential candidate biomarkers for assessing oocyte competence and embryo developmental potential, particularly in women with PCOS undergoing IVM. Given the lower maturation and fertilization rates observed in these women, optimizing conditions that regulate TGFB and ERK expression in CCs may lead to improved clinical outcomes. Further research is needed to determine whether modulating these pathways through growth factor supplementation or targeted molecular interventions could enhance IVM success and overall fertility outcomes in PCOS women.
4.6. Strengths and limitations
This study has several limitations that should be acknowledged. First, the absence of a healthy control group limits the ability to compare gene expression patterns between PCOS and non-PCOS populations. However, we aimed to investigate gene expression dynamics within the PCOS cohort to identify potential biomarkers relevant to IVM outcomes. Second, the analysis of multiple oocytes from the same patient introduces a potential cluster effect, which may violate the assumption of data independence; this was addressed through appropriate statistical adjustments. Third, the relatively small sample size may reduce the statistical power and generalizability of the findings. Lastly, the limited embryo quality observed reflects the clinical challenges of IVM in PCOS patients and may influence the interpretation of biomarker relevance. Therefore, further studies with larger sample size are warranted to validate these findings.
5. Conclusion
This study provides valuable insights into the molecular mechanisms underlying oocyte maturation, fertilization, and embryo quality in PCOS women undergoing IVF and IVM. The observed associations between TGFB1 and ERK expression in CCs and reproductive outcomes highlight their potential as candidate biomarkers for selecting competent oocytes and predicting embryo viability. While TGFB1 and ERK expression showed promising associations with oocyte and embryo quality, larger-scale studies are required to confirm their biomarker potential. Future studies should focus on exploring therapeutic strategies that optimize the activation of these pathways to enhance assisted reproductive technology success rates for women with PCOS.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author Contributions
SM. Kalantar, F. Montazeri, and MH. Fakherbaheri designed the study. MH. Fakherbaheri also conducted the research. F. Montazeri and H. Shafienia monitored, evaluated, and analyzed the results. MH. Fakherbaheri and F. Montazeri contributed to the review of the manuscript. All authors approved the final version and attest to the integrity of the data.
Acknowledgments
This study received no financial support and artificial intelligence tools are not used in this article.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Genetics
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







