Thu, May 21, 2026
[Archive]
Volume 24, Issue 1 (January 2026)
IJRM 2026, 24(1): 59-70 |
Back to browse issues page
Ethics code: IR.TBZMED.AEC.1401.087
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Bilabari M, Mohammadnejad D, Rahbarghazi R, Shafaei H. Comparative effects of adipose-derived mesenchymal stem cells and their exosomes on interleukin-1 beta-induced testicular inflammation in rats: An experimental study. IJRM 2026; 24 (1) :59-70
URL: http://ijrm.ir/article-1-3690-en.html
URL: http://ijrm.ir/article-1-3690-en.html
1- Stem Cell Research Center, Tabriz University of Medical Sciences, Tabriz, Iran. & Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran., Department of Anatomical Sciences, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran.
2- Department of Anatomical Sciences, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran.
3- Stem Cell Research Center, Tabriz University of Medical Sciences, Tabriz, Iran.
4- Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran. & Department of Anatomical Sciences, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran. ,shafaei49@gmail.com; shafaeih@tbzmed.ac.ir
2- Department of Anatomical Sciences, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran.
3- Stem Cell Research Center, Tabriz University of Medical Sciences, Tabriz, Iran.
4- Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran. & Department of Anatomical Sciences, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran. ,
Full-Text [PDF 3096 kb]
(222 Downloads)
| Abstract (HTML) (224 Views)
Full-Text: (39 Views)
1. Introduction
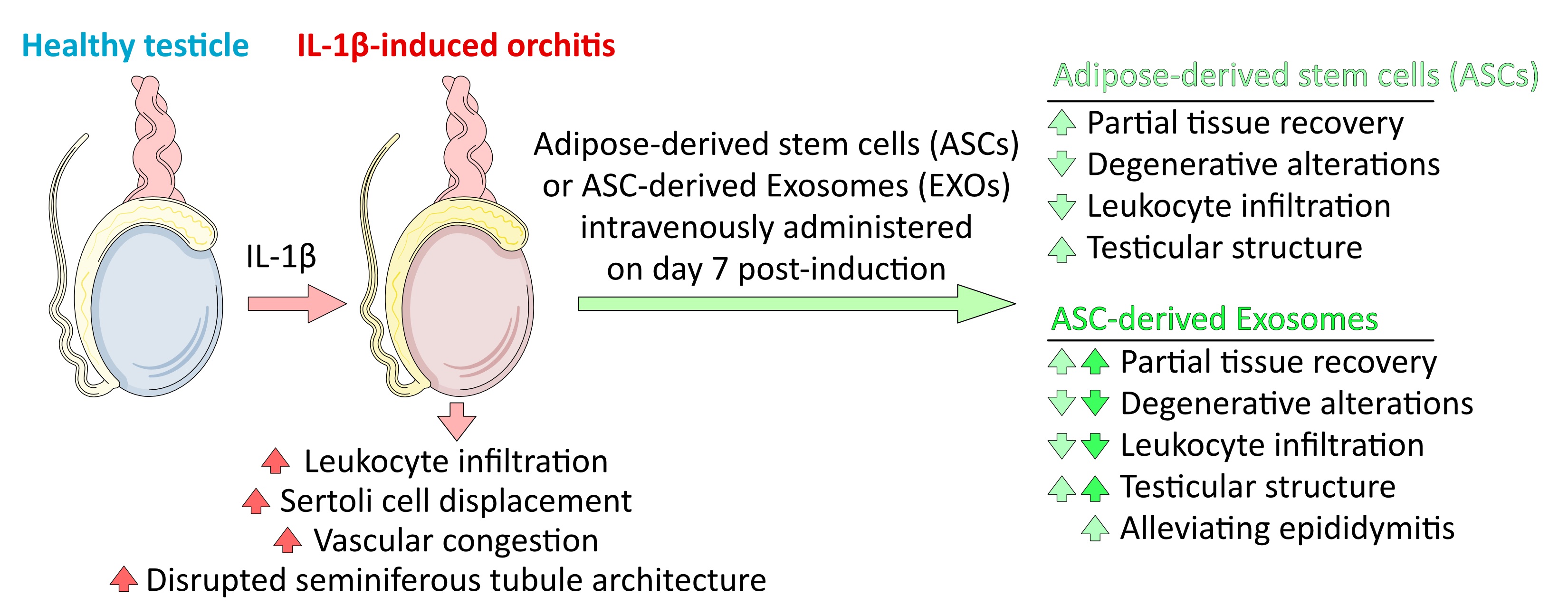
The testis serves as an ideal model for studying inflammation due to its immune-privileged status, where dysregulated immune responses have severe clinical consequences. Unlike systemic organs, testicular inflammation (orchitis) often leads to permanent spermatogenic impairment, blood-testis barrier disruption, and prolonged infertility, yet remains inadequately treated (1). Orchitis arises from various causes, including infection, trauma, autoimmunity, and systemic illness, and can result in irreversible testicular damage (2). As a master regulator of innate immunity, interleukin-1 beta (IL-1β) drives pro-inflammatory cascades through neutrophil recruitment and NLR family pyrin domain containing 3 (NLRP3) inflammasome activation, directly undermining immune-privileged sites (3, 4).
In the susceptible testicular microenvironment, IL-1β disrupts the blood-testis barrier, induces leukocyte infiltration, and impairs spermatogenesis, mirroring the pathophysiology of sterile orchitis (5). Using IL-1β to induce inflammation allows precise, reproducible control while avoiding endotoxin confounders. Its clinical relevance is underscored by the efficacy of IL-1-targeted therapies in human disorders, making this a translatable platform for evaluating novel interventions like adipose-derived stem cells (ASCs) and their exosomes (EXO) (6, 7).
ASCs and EXO harbor therapeutic potential against IL-1β-driven inflammation via immunomodulatory properties that disrupt NLRP3 inflammasome activation and NF-κB signaling (8). They secrete paracrine factors that suppress pro-inflammatory cytokines like tumor necrosis factor alpha and IL-6 (9). Notably, EXO delivers bioactive cargo (e.g., miR-21) that permeates testicular tissue effectively, quenches IL-1β amplification loops, polarizes macrophages, and mitigates fibrosis (10, 11). These nanovesicles function as intercellular messengers, modulating NF-κB/MAPK/STAT3 pathways to reduce cytokines and promote regeneration (8-10, 12, 13).
This study introduces a novel comparative approach, directly evaluating the efficacy of ASCs versus their isolated EXO in IL-1β-induced orchitis. While ASCs' regenerative potential is established, the superior capacity of EXO to mitigate testicular inflammation remains less explored. Our aim was to determine which intervention more effectively reduces leukocyte infiltration and restores testicular histoarchitecture, highlighting a promising cell-free therapeutic strategy. Here, we compare ASC and EXO efficacy via correlative histopathological analysis.
2. Materials and Methods
2.1. Animal care and ethical statement
The experiment commenced in July 2023 at the Animal House of Tabriz University of Medical Sciences Research Center, Tabriz, Iran. 32 healthy male Wistar rats (6-7 wk old; 250-300 gr) obtained from the Laboratory Animal Breeding Center of Tabriz University of Medical Sciences, Tabriz, Iran were housed under controlled conditions (22 ± 2°C, 55 ± 5% humidity, 12 hr light/dark cycle) with free access to food and water. Upon arrival, the animals were allowed to acclimate to the new housing environment for 7 days under standard laboratory conditions before initiating any experimental procedures. Animal health was monitored daily. All animals were wild-type with no prior procedures. Rats that developed parasitic infections during the project were excluded prior to group allocation. Predefined criteria were used to remove data points resulting from technical errors or outliers (> 3 SD). Personnel were blinded during critical experimental phases, and cage positions as well as procedure timing were standardized. Following euthanasia, carcasses were placed in special biological waste bags and sent to a medical waste disposal center for incineration.
2.2. Adipose tissue extraction and cell culture
Adipose tissue was extracted from a single 11-wk-old rat under ketamine/xylazine anesthesia. Perirenal adipose tissue was harvested under aseptic conditions, transferred to phosphate-buffered saline (PBS, synthesized by the authors) containing 1% antibiotics, and processed under a laminar flow hood. Tissue was chopped, rinsed with PBS, and subjected to digestion with 0.5 mg/gr collagenase type I (Sigma-Aldrich, United States) for 1 hr. The digest was neutralized with Dulbecco's Modified Eagle Medium (Gibco, New York) containing 10% fetal bovine serum (FBS, Gibco, New York) and centrifuged (1600 rpm, 5 min). Pelleted cells were cultured in T25 flasks at 37°C/5% CO2. Medium was refreshed every 3 days, and cells were passaged at 70-80% confluency (11).
2.3. Flow cytometry
To confirm the expression of surface antigen, the cell suspension was washed with PBS containing 1% bovine serum albumin (BSA, Sigma-Aldrich, United States) and then incubated with 3 µl of the following fluorochrome-conjugated monoclonal antibodies: CD105-PE (CD105 conjugated with phycoerythrin), CD45-FITC (CD45 labeled with fluorescein isothiocyanate), and CD14-PE (CD14 conjugated with phycoerythrin) for 30 min in a dark room. After staining, the cells were washed with PBS containing 1% BSA, the cell pellet vertices were centrifuged with 500 µl PBS/1% BSA, and evaluated further using a BD (Becton, Dickinson and Company) instrument. The data were analyzed using WIN MDI software (11).
2.4. Immunocytochemistry
ASCs were cultured on coverslips and fixed with 10% formaldehyde for 2 hr. Fixed cells were washed using PBS and then incubated with the primary antibody of CD44 overnight at 4°C. After washing for 5 min, the samples were incubated for 30 min at room temperature in HRP-conjugated secondary (horseradish peroxidase-conjugated secondary antibody). DAB (3,3-diaminobenzidine) staining was used to visualize antigen expression on cells (14).
2.5. Exosome isolation and scanning electron microscopy (SEM) imaging
For EXO isolation, P3 ASCs were incubated for 48 hr in exosome-depleted 1% FBS medium. Conditioned medium underwent sequential centrifugation: 1500 × g (10 min), 10,000 × g (1 hr), and 100,000 × g (1 hr at 4°C) (Figure 1). EXO were characterized by SEM imaging (35,000-65,000×) with size distribution analyzed in MATLAB (15).
2.6. Study groups and experimental procedure
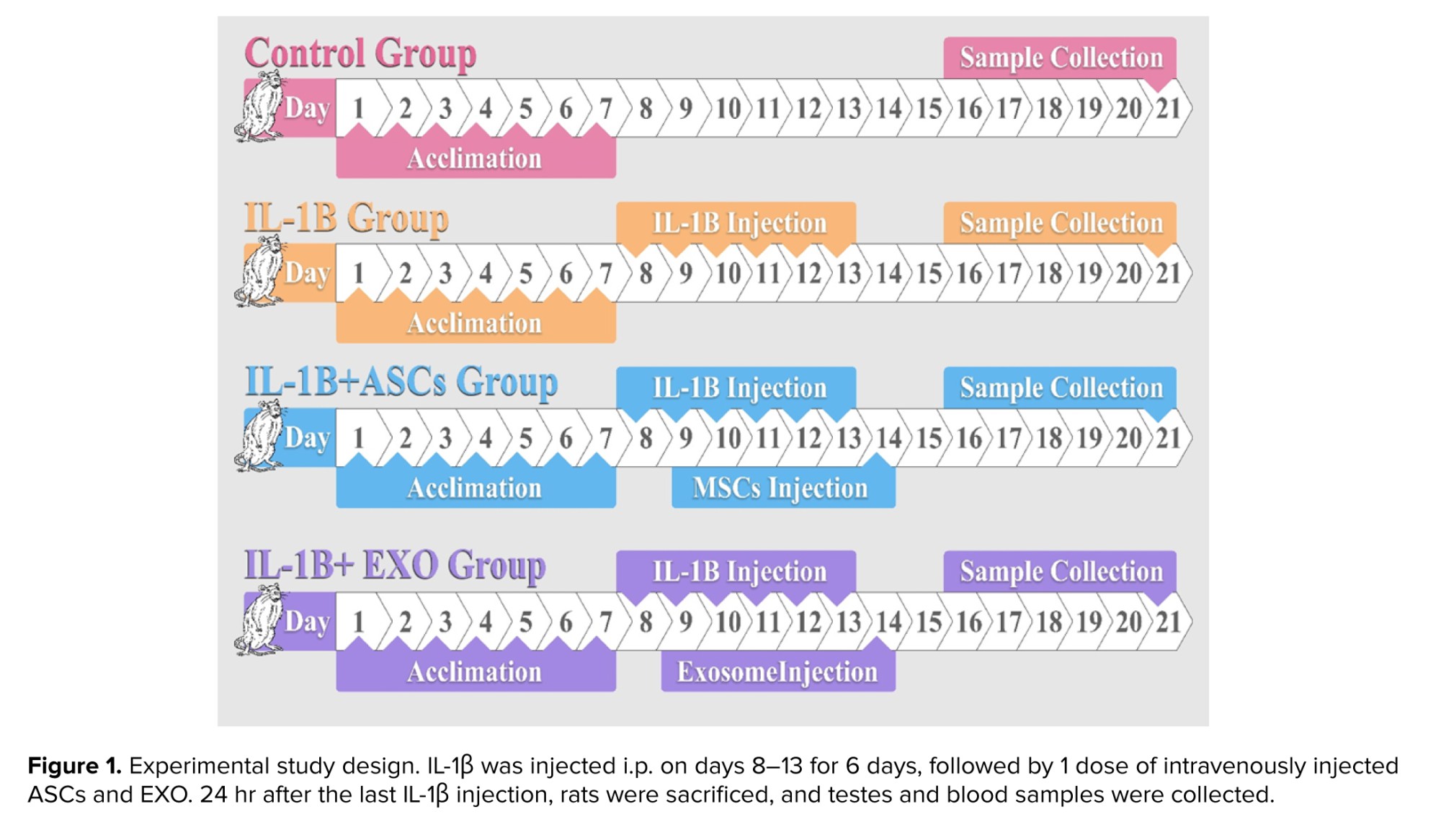
Group allocation was randomized using Excel’s RAND function. Rats (N = 32) were ranked by random numbers and assigned evenly to 4 groups (n = 8/group): 1) control, 2) IL-1β only, 3) IL-1β + ASCs (1 × 10⁶ cells, intravenous injection [IV]), 4) IL-1β + EXO (200 μg, IV). Treatments were administered on the day after the last induction of IL-1β under ketamine/xylazine anesthesia. Inflammation was induced via daily intraperitoneal IL-1β injections (10 μg/kg in 0.2 mL PBS; Sigma-Aldrich, USA) for 6 days (16) (Figure 1).
To determine the appropriate sample size, we performed a power analysis using the following standard formula for ANOVA studies. n = [2 × (1.96 + 0.84)² × 1²] / 1.5² ≈ 7.5 → 8 rats per group.
Based on this calculation with an anticipated effect size (Δ/σ) of 1.5, 80% power, and 5% significance level, we determined that 8 rats per group (total N = 32) would be required to detect statistically significant differences between experimental conditions.
2.7. Histological preparation
Testicular tissues were fixed in buffered formaldehyde (pH 2.7-4.7) for 24 hr, followed by 10-day immersion. Samples were then dehydrated using graded ethanol, cleared with xylene, and then impregnated with dual paraffin. Testis and epididymis tissues were separately embedded in L-shaped molds and sectioned at 5-8 μm thickness using rotary microtomes. Sections were floated in a 45°C water bath, then mounted on alcohol-coated slides and dried at 60°C for 24 hr. After deparaffinization, slides were stained with hematoxylin (5 min) and eosin (3 min) following standard protocols (17). For leukocyte analysis, hematoxylin and eosin (H&E)-stained sections were systematically examined under 400× light microscopy. Leukocytes within testicular vessels were identified by nuclear morphology and cytoplasmic staining characteristics.
2.8. Leukocyte quantification
Leukocytes within clearly identifiable vessels in the testicular interstitium were counted per high-power field (HPF, 400x magnification) across 10 non-overlapping fields per animal. Leukocytes were identified based on characteristic nuclear morphology (prominent, condensed chromatin) and cytoplasmic staining in H&E sections.
2.9. Ethical Considerations
All experimental procedures and animal care protocols were reviewed by the Ethics Committee for Research on Laboratory Animals of Tabriz University of Medical Sciences, Tabriz, Iran (Code: IR.TBZMED.AEC.1401.087), and were performed in strict accordance with the National Institutes of Health guidelines for the care and use of laboratory animals.
2.10. Statistical Analysis
The data collected in this study consisted of both categorical variables (experimental group assignment) and numerical variables (leukocyte count). Numerical data are presented as mean ± standard error of the mean (SEM). Prior to parametric testing, the numerical data were assessed for normality using the Shapiro-Wilk test, and the homogeneity of variances were confirmed via ANOVA's internal tests. Based on the confirmation of these assumptions, the data were analyzed by one-way analysis of variance (ANOVA). When the ANOVA indicated a significant overall effect, post-hoc comparisons between individual group means were performed using the least significant difference test. A p < 0.05 was considered statistically significant for all tests. All statistical analyses were performed using SPSS software (Version 16, IBM Corporation, Armonk, NY, USA) (18).
3. Results
3.1. Characterization of ASCs culture
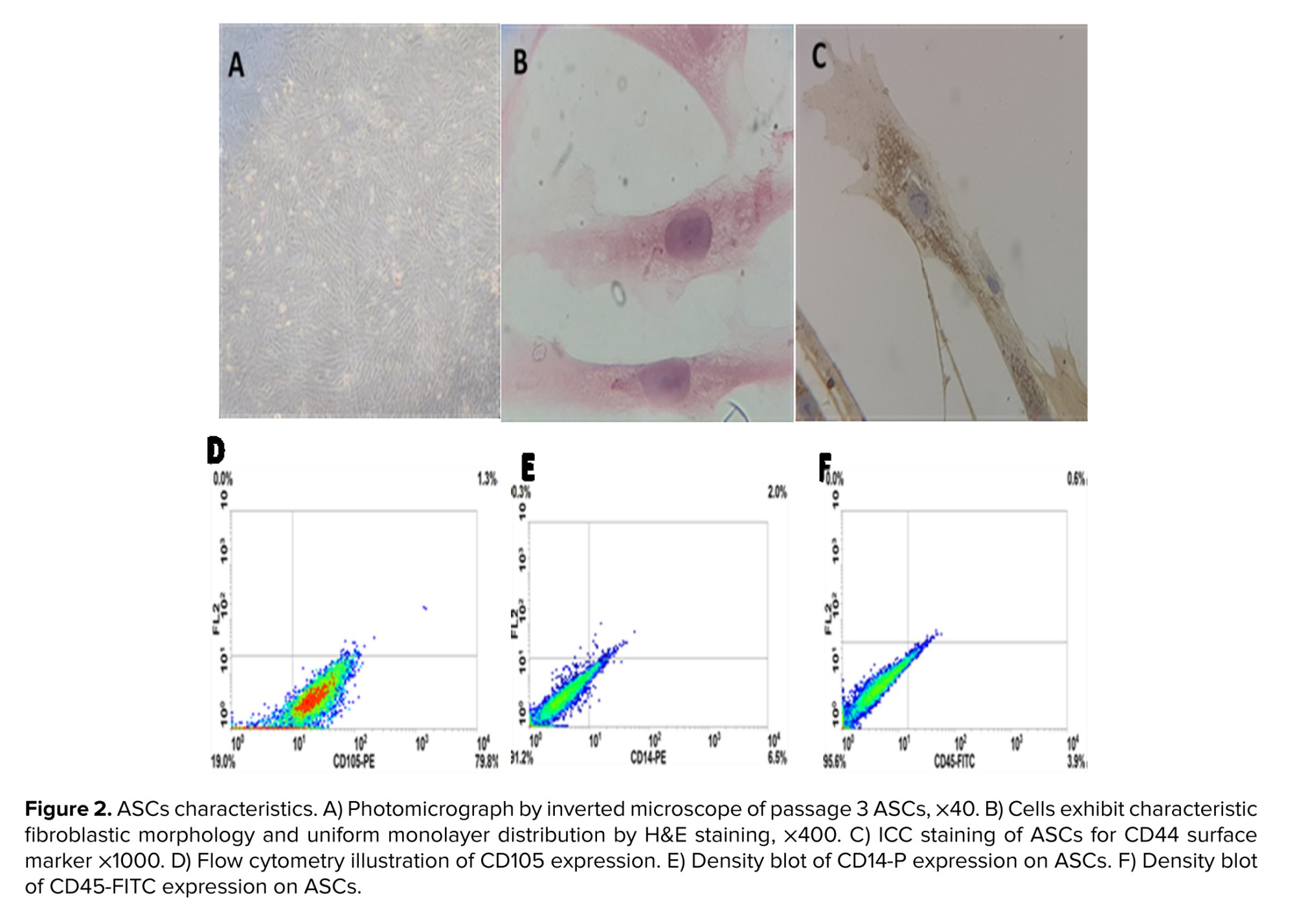
ASCs exhibited typical fibroblastic morphology and formed a homogeneous monolayer. Immunophenotyping confirmed MSC identity, with positive expression of CD44 and CD105 (79.8%) and positive expression of CD14 (91.2%) and CD45 (95.6%) (Figure 2A-F).
3.2. Characterization of ASCs derived EXO
SEM showed the typical cup-shaped morphology of ASC-derived EXO with a particle size distribution between 30 and 150 nm, with a prominent peak at around 150 nm (Figure 3).
3.3. Histopathological changes in testicular tissue
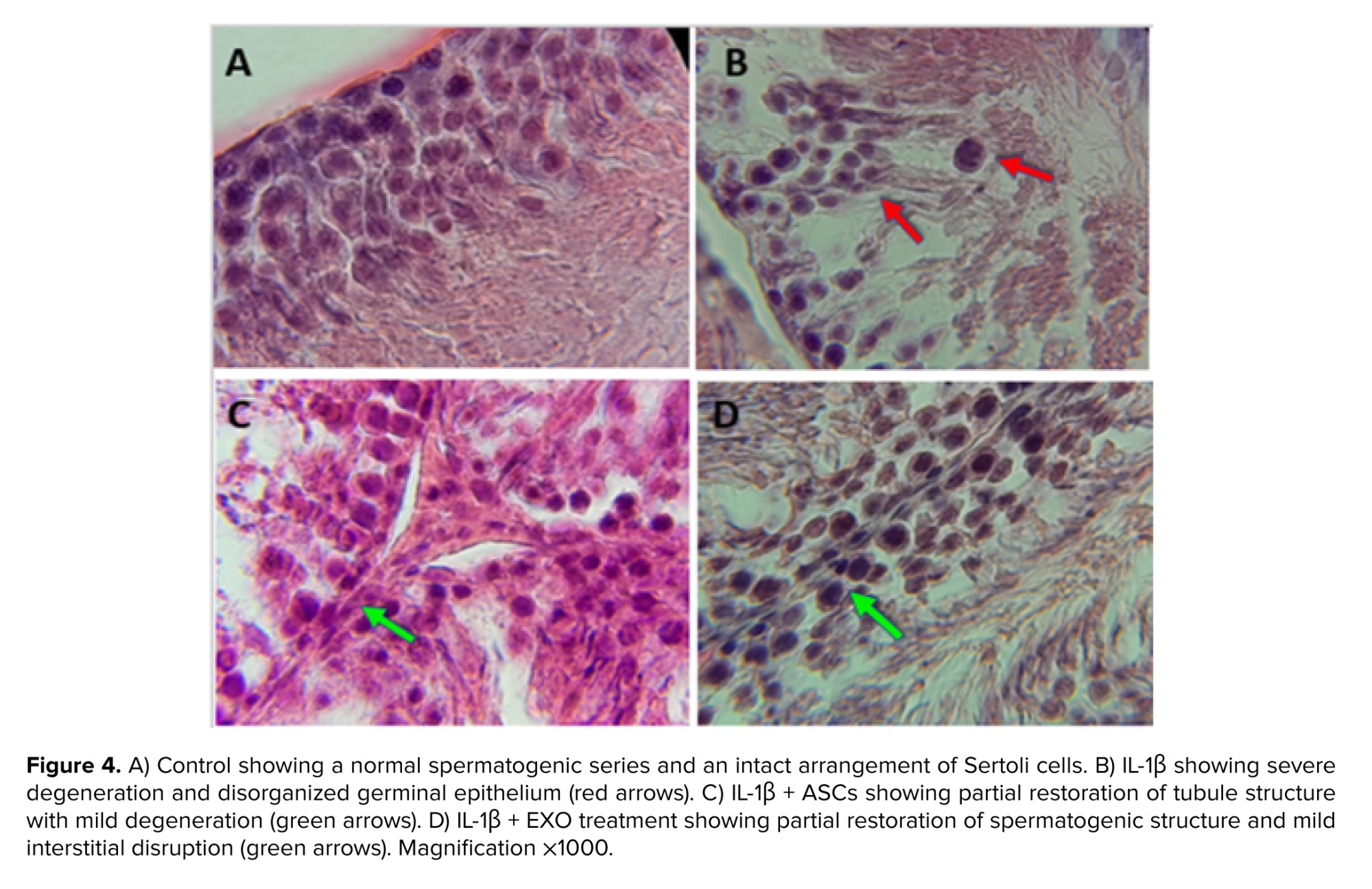
Control seminiferous tubules (Figure 4A) show normal spermatogonia, primary spermatocytes, spermatids near the lumen, and Sertoli cells with triangular nuclei, maintaining regular structure and tissue integrity via cell-cell junctions.
The IL-1β group (Figure 4B) exhibits severe disruption; Sertoli-spermatogenic cell junctions are broken, Sertoli cells retract toward the tubule wall, tissue integrity is lost, cells degrade, and primary spermatocytes appear ectopically in the lumen. Significant interstitial vascular congestion and increased leukocytes (including monocytes) are present, with evident interstitial damage.
The treatment with ASCs (Figure 4C) reduces the harm caused by IL-1β, exhibiting modest degenerative changes in comparison to the control. Vascular obstruction is substantially reduced, interstitial disruption/degeneration is minimal, and tissue integrity is preserved. Sertoli cells partially relocate toward the basal membrane (Figure 4D).
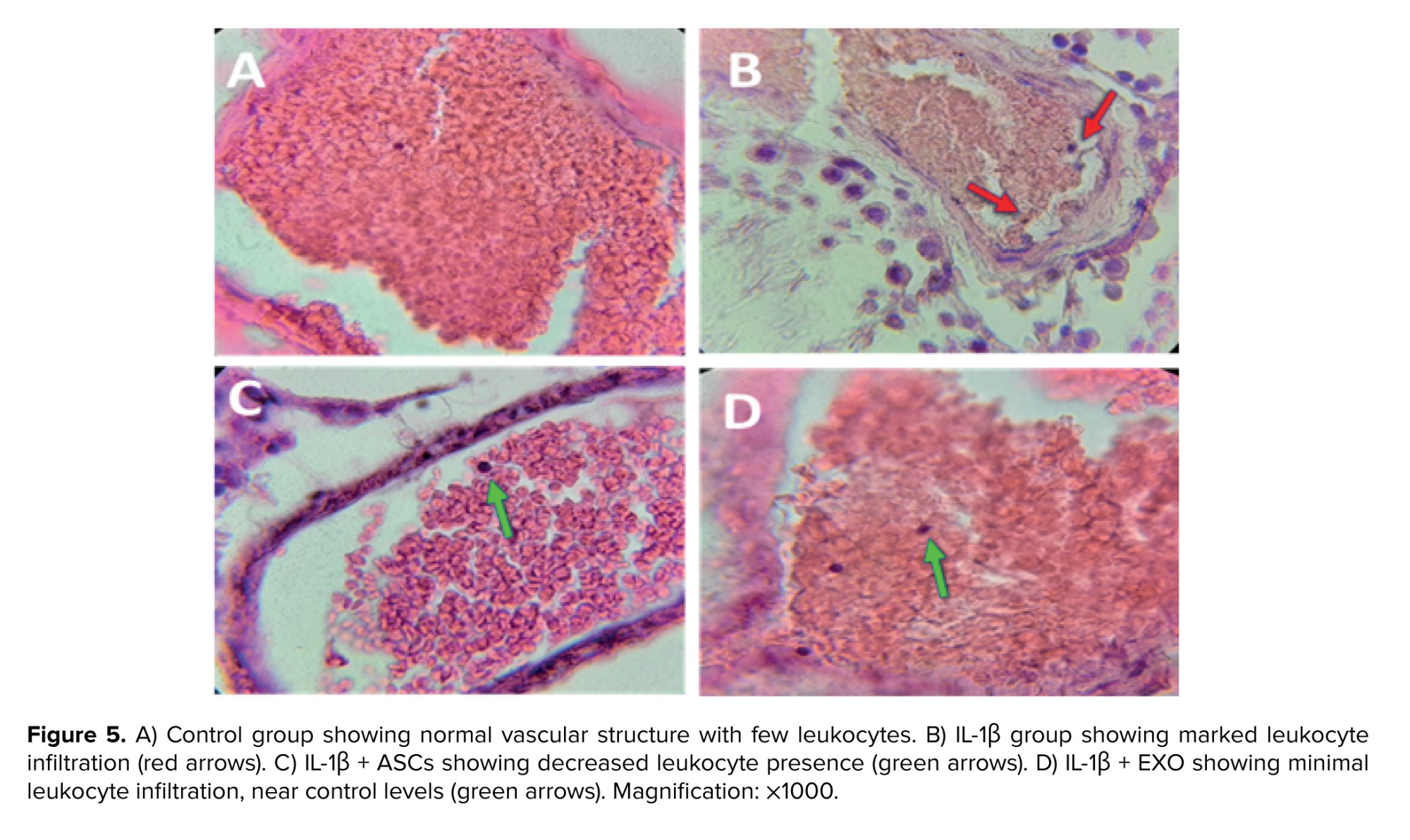
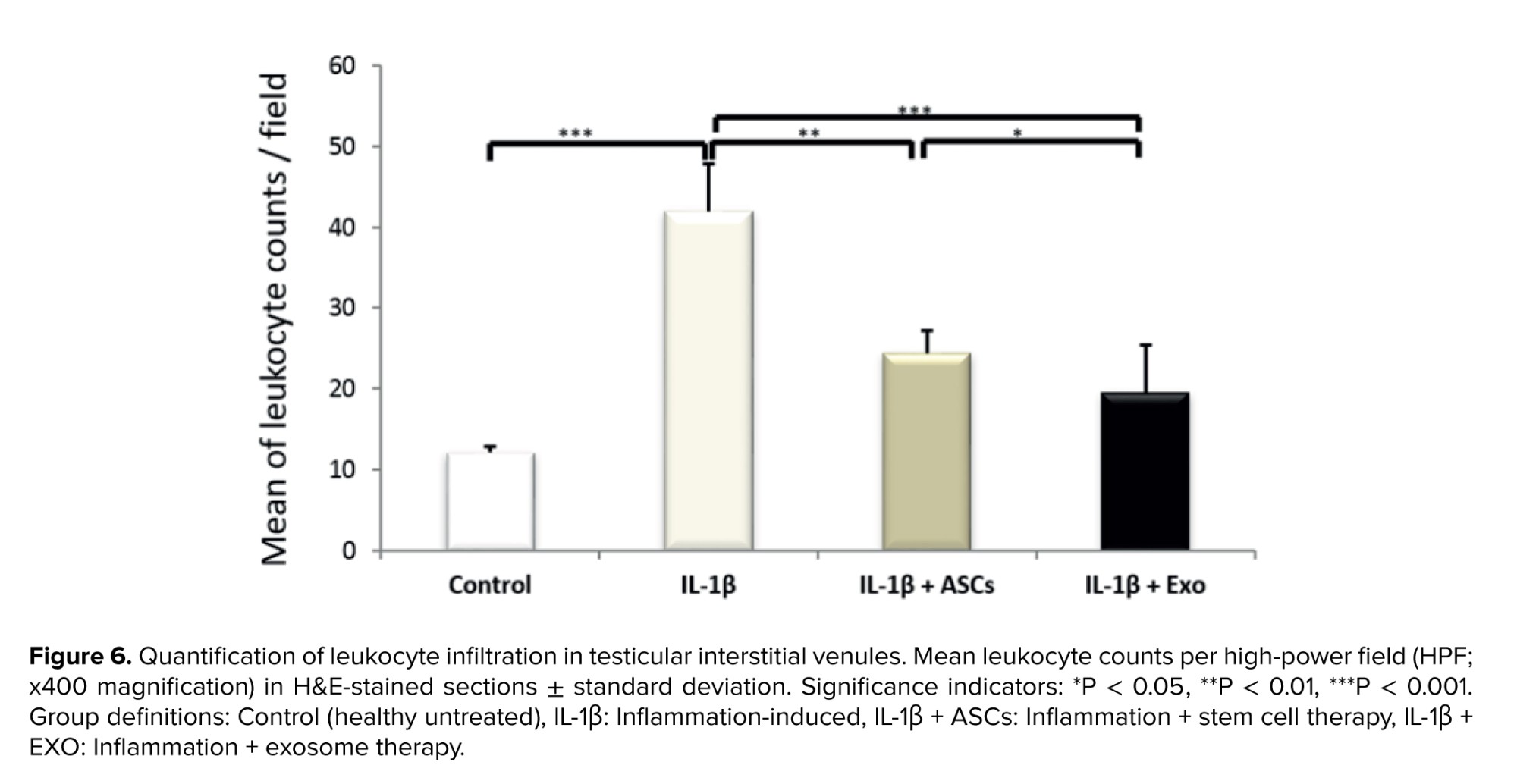
3.4. Leukocyte infiltration in testicular tissue
Leukocyte quantification, characterized by conspicuous and identifiable chromatin nuclei, was conducted in all visible blood vessels on testicular tissue slides across the 4 experimental groups (Figure 5). The IL-1β group showed a significant increase in leukocyte count compared to the control group (p < 0.001) (Figure 6), confirming substantial inflammation induction. Both treatments showed significantly reduced IL-1β-driven leukocytosis. Figure 6 illustrates these comparative leukocyte counts across groups and the superior anti-inflammatory effect of EXO compared to ASCs.
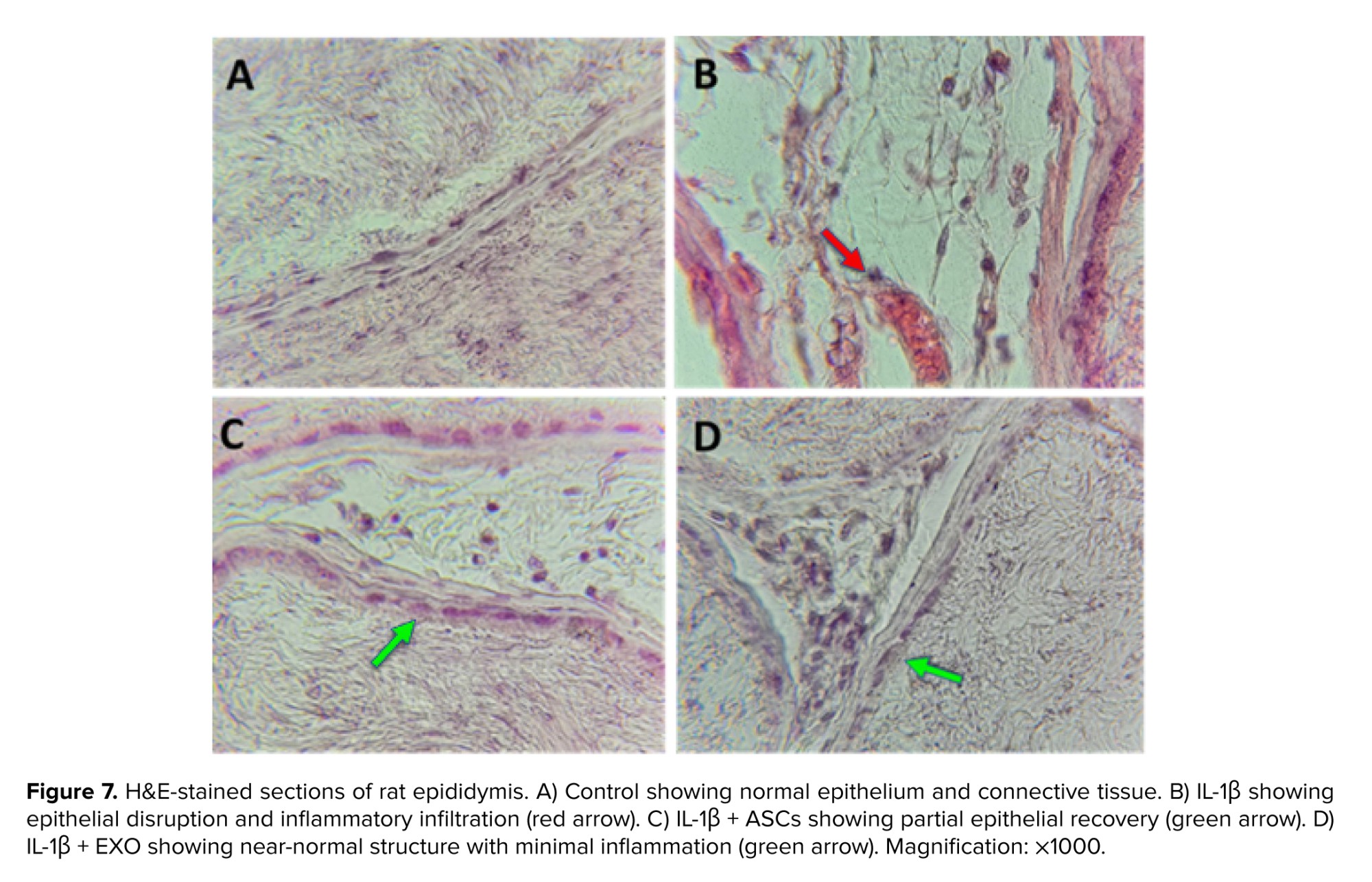
3.5. Histopathological changes in epididymis tissue
Histological examination of the cauda epididymis revealed distinct morphological alterations across the experimental groups (Figure 7). In the control group, the epididymal ducts were lined by an intact epithelium and contained abundant spermatozoa within the lumen, with minimal connective tissue edema (Figure 7A). The IL-1β group exhibited marked pathological changes, including a visible reduction in intraluminal sperm density, the presence of degenerating spermatocytes, foamy transformation and swelling of epithelial cells, and interstitial edema with vascular hyperemia. An apparent increase in interstitial leukocytes and macrophages was also observed.
Treatment with ASCs resulted in an observable improvement in several of these parameters. The ASCs group showed a higher apparent sperm density compared to the IL-1β group, along with reduced epithelial cell swelling and less vascular hyperemia (Figure 7C). In the EXO group, the histoarchitecture appeared more preserved, with features such as epithelial morphology and reduced edema showing a trend toward the control phenotype.

4. Discussion
This study demonstrated that ASCs and their EXO effectively mitigate IL-1β-induced inflammation and tissue damage in rat testicular tissue, with EXO exhibiting superior therapeutic efficacy. The enhanced efficacy of ASCs and their EXO stems from their unique biological properties, including nanoscale bioavailability, targeted cargo delivery, and multi-modal immunomodulatory actions that collectively address the complex pathophysiology of testicular inflammation (19, 20).
The significant histopathological repair seen with exosome treatment (almost total clearance of Sertoli cells displacement, interstitial edema, and vascular congestion) demonstrates their capacity to infiltrate immune-privileged areas and convey regenerative material directly to cellular targets. Unlike whole ASCs, which rely on paracrine signaling limited by vascular entrapment and rapid clearance, EXO (30-150 nm) efficiently cross biological barriers to deliver concentrated payloads of miR-21, miR-146a, and Annexin A1 (21). These molecules collectively suppress NLRP3 inflammasome activation (19), inhibit NF-κB translocation, and promote tissue repair pathways, explaining significantly reduced leukocyte counts (60% reduction vs. IL-1β) and superior architectural preservation compared to ASCs (38% reduction) (17, 20). The cup-shaped morphology of our isolated EXO (Figure 3) further enhances cellular uptake efficiency, enabling precise modulation of inflammatory cascades within the testicular microenvironment (22).
Our findings should be contextualized within clinical orchitis management challenges. Current anti-inflammatory therapies (e.g., corticosteroids) often fail to penetrate the blood-testis barrier and risk exacerbating infertility through germ cell apoptosis (2). EXO provides a potential alternative by combining immunosuppression with tissue regeneration, which is crucial for sustaining spermatogenesis (11). The observed interstitial collagen remodeling in exosome-treated groups suggests additional antifibrotic effects mediated by miR-29c, which targets collagen I/III transcripts (15). This dual action addresses both acute inflammation and chronic fibrotic sequelae that underlie permanent infertility in unresolved orchitis (23).
The significant increase in leukocyte count observed within the testicular interstitium and vasculature following IL-1β induction was histologically consistent with an influx of innate immune cells, primarily neutrophils, identifiable by their characteristic multi-lobed nuclei, along with mononuclear cells such as lymphocytes and monocytes/macrophages. This cellular profile is a standard response to acute inflammation, as IL-1β is a potent mediator for endothelial activation and the recruitment of these specific leukocyte populations. The notable reduction in these infiltrating cells following ASC and EXO treatments underscores their potent immunomodulatory effects, with leukocyte quantification data critically revealing that EXO achieved a 44% greater reduction than ASCs (p = 0.011). This finding underscores the superior capacity of EXO to disrupt chemokine-mediated recruitment pathways and resolve local inflammation within the testicular environment, likely through a more efficient delivery of paracrine factors (21).
The differential reduction in leukocyte infiltration observed between treatments may be attributed to distinct mechanistic pathways. For EXO, their effects likely involve the inhibition of IL-1β-induced CCL2 secretion from Sertoli cells, a key chemotactic signal for monocytes, while simultaneously promoting Treg recruitment via transforming growth factor β1 delivery (24). This coordinated immunomodulation leverages systemic delivery of paracrine factors. In contrast, ASCs may operate through a more localized mechanism, potentially relying on their physical presence and contact-dependent signaling pathways such as PD-1/PD-L1 interaction, which could be limited by their migration and retention within the inflamed tissue (25).
Several limitations warrant consideration. The short-term, 14-day duration of the disease induction and treatment protocol precludes assessment of long-term functional recovery and may not be generalizable to chronic therapeutic regimens; future studies should evaluate spermatogenic restoration through sperm counts and mating trials. Proteomic characterization of exosomal cargo would identify essential therapeutic factors, as the exclusive focus on structural outcomes overlooks molecular events (26). Although IV administration was effective, localized intratesticular delivery has the potential to increase bioavailability and decrease the number of dosing requirements (24). Comparative studies with other MSC sources (e.g., bone marrow) could clarify tissue-specific therapeutic advantages.
The clinical application of these findings faces practical challenges. Large-scale production of EXO remains difficult, and ultracentrifugation fails to yield sufficient quantities for human use. Emerging methods, such as tangential flow filtration or CRISPR-engineered ASCs with increased EXO secretion, might help to bridge the gap. Dose optimization studies are needed to establish therapeutic windows, as our rat dose (200 μg) translates to ~0.5 mg/kg in humans, potentially requiring 30-50 mg per patient. Stability protocols, including lyophilization, must be developed to extend shelf life beyond current cold-chain limitations (25).
Despite these challenges, EXO presents distinct advantages over cellular therapies: reduced immunogenicity, avoidance of ectopic differentiation risks, and lower thrombotic potential (21). Their acellular nature streamlines regulatory approval routes as "non-living" biologics. EXO might be used to treat male infertility in acute orchitis to avoid lasting damage or in chronic situations to reverse existing inflammation-fibrosis cycles (2, 7). The ability to engineer EXO for enhanced targeting (e.g., testicular homing peptides) or cargo loading (e.g., anti-IL-1β siRNA) further expands their therapeutic potential (10, 23).
5. Conclusion
In conclusion, EXO appeared more effective in total, possibly due to better bioavailability and paracrine signaling (18-20). Their ability to concurrently resolve local inflammation (leukocyte reduction), restore tissue architecture, and normalize systemic biomarker positions them as a next-generation therapeutic platform for testicular inflammation and related infertility. Future research should prioritize clinical translation via scalable production, delivery optimization, and validation of functional reproductive recovery.
Our research provides evidence that IL-1β-induced inflammation in rat testicular tissues can be effectively alleviated by both mesenchymal stem cells and their derived EXO. Both treatment approaches demonstrated substantial anti-inflammatory and tissue-protective effects, indicating their therapeutic potential in mitigating testicular inflammation. These findings contribute to the growing body of evidence supporting the feasibility of cell-based and cell-free regenerative strategies, which may open new avenues for managing inflammatory and reproductive disorders.
Data Availability
Data will be available if requested.
Author Contributions
H. Shafaei, D. Mohammadnejad, and M. Bilabari designed the study and conducted the research. M. Bilabari, D. Mohammadnejad, and R. Rahbarghazi monitored, evaluated, and analyzed the results of the study. All authors reviewed and approved the final manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Acknowledgements
The authors acknowledge the use of DeepSeek AI (developed by DeepSeek) for grammatical refinement of the manuscript and summarizing portions of the abstract and introduction. The research design, data interpretation, and scientific conclusions are solely attributable to the authors. This work was funded by the Immunology Research Center of Tabriz University of Medical Sciences, Tabriz, Iran (grant number: 70810).
Conflict of Interest
The author declares that there is no conflict of interest.
In the susceptible testicular microenvironment, IL-1β disrupts the blood-testis barrier, induces leukocyte infiltration, and impairs spermatogenesis, mirroring the pathophysiology of sterile orchitis (5). Using IL-1β to induce inflammation allows precise, reproducible control while avoiding endotoxin confounders. Its clinical relevance is underscored by the efficacy of IL-1-targeted therapies in human disorders, making this a translatable platform for evaluating novel interventions like adipose-derived stem cells (ASCs) and their exosomes (EXO) (6, 7).
ASCs and EXO harbor therapeutic potential against IL-1β-driven inflammation via immunomodulatory properties that disrupt NLRP3 inflammasome activation and NF-κB signaling (8). They secrete paracrine factors that suppress pro-inflammatory cytokines like tumor necrosis factor alpha and IL-6 (9). Notably, EXO delivers bioactive cargo (e.g., miR-21) that permeates testicular tissue effectively, quenches IL-1β amplification loops, polarizes macrophages, and mitigates fibrosis (10, 11). These nanovesicles function as intercellular messengers, modulating NF-κB/MAPK/STAT3 pathways to reduce cytokines and promote regeneration (8-10, 12, 13).
This study introduces a novel comparative approach, directly evaluating the efficacy of ASCs versus their isolated EXO in IL-1β-induced orchitis. While ASCs' regenerative potential is established, the superior capacity of EXO to mitigate testicular inflammation remains less explored. Our aim was to determine which intervention more effectively reduces leukocyte infiltration and restores testicular histoarchitecture, highlighting a promising cell-free therapeutic strategy. Here, we compare ASC and EXO efficacy via correlative histopathological analysis.
2. Materials and Methods
2.1. Animal care and ethical statement
The experiment commenced in July 2023 at the Animal House of Tabriz University of Medical Sciences Research Center, Tabriz, Iran. 32 healthy male Wistar rats (6-7 wk old; 250-300 gr) obtained from the Laboratory Animal Breeding Center of Tabriz University of Medical Sciences, Tabriz, Iran were housed under controlled conditions (22 ± 2°C, 55 ± 5% humidity, 12 hr light/dark cycle) with free access to food and water. Upon arrival, the animals were allowed to acclimate to the new housing environment for 7 days under standard laboratory conditions before initiating any experimental procedures. Animal health was monitored daily. All animals were wild-type with no prior procedures. Rats that developed parasitic infections during the project were excluded prior to group allocation. Predefined criteria were used to remove data points resulting from technical errors or outliers (> 3 SD). Personnel were blinded during critical experimental phases, and cage positions as well as procedure timing were standardized. Following euthanasia, carcasses were placed in special biological waste bags and sent to a medical waste disposal center for incineration.
2.2. Adipose tissue extraction and cell culture
Adipose tissue was extracted from a single 11-wk-old rat under ketamine/xylazine anesthesia. Perirenal adipose tissue was harvested under aseptic conditions, transferred to phosphate-buffered saline (PBS, synthesized by the authors) containing 1% antibiotics, and processed under a laminar flow hood. Tissue was chopped, rinsed with PBS, and subjected to digestion with 0.5 mg/gr collagenase type I (Sigma-Aldrich, United States) for 1 hr. The digest was neutralized with Dulbecco's Modified Eagle Medium (Gibco, New York) containing 10% fetal bovine serum (FBS, Gibco, New York) and centrifuged (1600 rpm, 5 min). Pelleted cells were cultured in T25 flasks at 37°C/5% CO2. Medium was refreshed every 3 days, and cells were passaged at 70-80% confluency (11).
2.3. Flow cytometry
To confirm the expression of surface antigen, the cell suspension was washed with PBS containing 1% bovine serum albumin (BSA, Sigma-Aldrich, United States) and then incubated with 3 µl of the following fluorochrome-conjugated monoclonal antibodies: CD105-PE (CD105 conjugated with phycoerythrin), CD45-FITC (CD45 labeled with fluorescein isothiocyanate), and CD14-PE (CD14 conjugated with phycoerythrin) for 30 min in a dark room. After staining, the cells were washed with PBS containing 1% BSA, the cell pellet vertices were centrifuged with 500 µl PBS/1% BSA, and evaluated further using a BD (Becton, Dickinson and Company) instrument. The data were analyzed using WIN MDI software (11).
2.4. Immunocytochemistry
ASCs were cultured on coverslips and fixed with 10% formaldehyde for 2 hr. Fixed cells were washed using PBS and then incubated with the primary antibody of CD44 overnight at 4°C. After washing for 5 min, the samples were incubated for 30 min at room temperature in HRP-conjugated secondary (horseradish peroxidase-conjugated secondary antibody). DAB (3,3-diaminobenzidine) staining was used to visualize antigen expression on cells (14).
2.5. Exosome isolation and scanning electron microscopy (SEM) imaging
For EXO isolation, P3 ASCs were incubated for 48 hr in exosome-depleted 1% FBS medium. Conditioned medium underwent sequential centrifugation: 1500 × g (10 min), 10,000 × g (1 hr), and 100,000 × g (1 hr at 4°C) (Figure 1). EXO were characterized by SEM imaging (35,000-65,000×) with size distribution analyzed in MATLAB (15).
2.6. Study groups and experimental procedure
Group allocation was randomized using Excel’s RAND function. Rats (N = 32) were ranked by random numbers and assigned evenly to 4 groups (n = 8/group): 1) control, 2) IL-1β only, 3) IL-1β + ASCs (1 × 10⁶ cells, intravenous injection [IV]), 4) IL-1β + EXO (200 μg, IV). Treatments were administered on the day after the last induction of IL-1β under ketamine/xylazine anesthesia. Inflammation was induced via daily intraperitoneal IL-1β injections (10 μg/kg in 0.2 mL PBS; Sigma-Aldrich, USA) for 6 days (16) (Figure 1).
To determine the appropriate sample size, we performed a power analysis using the following standard formula for ANOVA studies. n = [2 × (1.96 + 0.84)² × 1²] / 1.5² ≈ 7.5 → 8 rats per group.
Based on this calculation with an anticipated effect size (Δ/σ) of 1.5, 80% power, and 5% significance level, we determined that 8 rats per group (total N = 32) would be required to detect statistically significant differences between experimental conditions.
2.7. Histological preparation
Testicular tissues were fixed in buffered formaldehyde (pH 2.7-4.7) for 24 hr, followed by 10-day immersion. Samples were then dehydrated using graded ethanol, cleared with xylene, and then impregnated with dual paraffin. Testis and epididymis tissues were separately embedded in L-shaped molds and sectioned at 5-8 μm thickness using rotary microtomes. Sections were floated in a 45°C water bath, then mounted on alcohol-coated slides and dried at 60°C for 24 hr. After deparaffinization, slides were stained with hematoxylin (5 min) and eosin (3 min) following standard protocols (17). For leukocyte analysis, hematoxylin and eosin (H&E)-stained sections were systematically examined under 400× light microscopy. Leukocytes within testicular vessels were identified by nuclear morphology and cytoplasmic staining characteristics.
2.8. Leukocyte quantification
Leukocytes within clearly identifiable vessels in the testicular interstitium were counted per high-power field (HPF, 400x magnification) across 10 non-overlapping fields per animal. Leukocytes were identified based on characteristic nuclear morphology (prominent, condensed chromatin) and cytoplasmic staining in H&E sections.

2.9. Ethical Considerations
All experimental procedures and animal care protocols were reviewed by the Ethics Committee for Research on Laboratory Animals of Tabriz University of Medical Sciences, Tabriz, Iran (Code: IR.TBZMED.AEC.1401.087), and were performed in strict accordance with the National Institutes of Health guidelines for the care and use of laboratory animals.
2.10. Statistical Analysis
The data collected in this study consisted of both categorical variables (experimental group assignment) and numerical variables (leukocyte count). Numerical data are presented as mean ± standard error of the mean (SEM). Prior to parametric testing, the numerical data were assessed for normality using the Shapiro-Wilk test, and the homogeneity of variances were confirmed via ANOVA's internal tests. Based on the confirmation of these assumptions, the data were analyzed by one-way analysis of variance (ANOVA). When the ANOVA indicated a significant overall effect, post-hoc comparisons between individual group means were performed using the least significant difference test. A p < 0.05 was considered statistically significant for all tests. All statistical analyses were performed using SPSS software (Version 16, IBM Corporation, Armonk, NY, USA) (18).
3. Results
3.1. Characterization of ASCs culture
ASCs exhibited typical fibroblastic morphology and formed a homogeneous monolayer. Immunophenotyping confirmed MSC identity, with positive expression of CD44 and CD105 (79.8%) and positive expression of CD14 (91.2%) and CD45 (95.6%) (Figure 2A-F).
3.2. Characterization of ASCs derived EXO
SEM showed the typical cup-shaped morphology of ASC-derived EXO with a particle size distribution between 30 and 150 nm, with a prominent peak at around 150 nm (Figure 3).
3.3. Histopathological changes in testicular tissue
Control seminiferous tubules (Figure 4A) show normal spermatogonia, primary spermatocytes, spermatids near the lumen, and Sertoli cells with triangular nuclei, maintaining regular structure and tissue integrity via cell-cell junctions.
The IL-1β group (Figure 4B) exhibits severe disruption; Sertoli-spermatogenic cell junctions are broken, Sertoli cells retract toward the tubule wall, tissue integrity is lost, cells degrade, and primary spermatocytes appear ectopically in the lumen. Significant interstitial vascular congestion and increased leukocytes (including monocytes) are present, with evident interstitial damage.
The treatment with ASCs (Figure 4C) reduces the harm caused by IL-1β, exhibiting modest degenerative changes in comparison to the control. Vascular obstruction is substantially reduced, interstitial disruption/degeneration is minimal, and tissue integrity is preserved. Sertoli cells partially relocate toward the basal membrane (Figure 4D).
3.4. Leukocyte infiltration in testicular tissue
Leukocyte quantification, characterized by conspicuous and identifiable chromatin nuclei, was conducted in all visible blood vessels on testicular tissue slides across the 4 experimental groups (Figure 5). The IL-1β group showed a significant increase in leukocyte count compared to the control group (p < 0.001) (Figure 6), confirming substantial inflammation induction. Both treatments showed significantly reduced IL-1β-driven leukocytosis. Figure 6 illustrates these comparative leukocyte counts across groups and the superior anti-inflammatory effect of EXO compared to ASCs.
3.5. Histopathological changes in epididymis tissue
Histological examination of the cauda epididymis revealed distinct morphological alterations across the experimental groups (Figure 7). In the control group, the epididymal ducts were lined by an intact epithelium and contained abundant spermatozoa within the lumen, with minimal connective tissue edema (Figure 7A). The IL-1β group exhibited marked pathological changes, including a visible reduction in intraluminal sperm density, the presence of degenerating spermatocytes, foamy transformation and swelling of epithelial cells, and interstitial edema with vascular hyperemia. An apparent increase in interstitial leukocytes and macrophages was also observed.
Treatment with ASCs resulted in an observable improvement in several of these parameters. The ASCs group showed a higher apparent sperm density compared to the IL-1β group, along with reduced epithelial cell swelling and less vascular hyperemia (Figure 7C). In the EXO group, the histoarchitecture appeared more preserved, with features such as epithelial morphology and reduced edema showing a trend toward the control phenotype.






4. Discussion
This study demonstrated that ASCs and their EXO effectively mitigate IL-1β-induced inflammation and tissue damage in rat testicular tissue, with EXO exhibiting superior therapeutic efficacy. The enhanced efficacy of ASCs and their EXO stems from their unique biological properties, including nanoscale bioavailability, targeted cargo delivery, and multi-modal immunomodulatory actions that collectively address the complex pathophysiology of testicular inflammation (19, 20).
The significant histopathological repair seen with exosome treatment (almost total clearance of Sertoli cells displacement, interstitial edema, and vascular congestion) demonstrates their capacity to infiltrate immune-privileged areas and convey regenerative material directly to cellular targets. Unlike whole ASCs, which rely on paracrine signaling limited by vascular entrapment and rapid clearance, EXO (30-150 nm) efficiently cross biological barriers to deliver concentrated payloads of miR-21, miR-146a, and Annexin A1 (21). These molecules collectively suppress NLRP3 inflammasome activation (19), inhibit NF-κB translocation, and promote tissue repair pathways, explaining significantly reduced leukocyte counts (60% reduction vs. IL-1β) and superior architectural preservation compared to ASCs (38% reduction) (17, 20). The cup-shaped morphology of our isolated EXO (Figure 3) further enhances cellular uptake efficiency, enabling precise modulation of inflammatory cascades within the testicular microenvironment (22).
Our findings should be contextualized within clinical orchitis management challenges. Current anti-inflammatory therapies (e.g., corticosteroids) often fail to penetrate the blood-testis barrier and risk exacerbating infertility through germ cell apoptosis (2). EXO provides a potential alternative by combining immunosuppression with tissue regeneration, which is crucial for sustaining spermatogenesis (11). The observed interstitial collagen remodeling in exosome-treated groups suggests additional antifibrotic effects mediated by miR-29c, which targets collagen I/III transcripts (15). This dual action addresses both acute inflammation and chronic fibrotic sequelae that underlie permanent infertility in unresolved orchitis (23).
The significant increase in leukocyte count observed within the testicular interstitium and vasculature following IL-1β induction was histologically consistent with an influx of innate immune cells, primarily neutrophils, identifiable by their characteristic multi-lobed nuclei, along with mononuclear cells such as lymphocytes and monocytes/macrophages. This cellular profile is a standard response to acute inflammation, as IL-1β is a potent mediator for endothelial activation and the recruitment of these specific leukocyte populations. The notable reduction in these infiltrating cells following ASC and EXO treatments underscores their potent immunomodulatory effects, with leukocyte quantification data critically revealing that EXO achieved a 44% greater reduction than ASCs (p = 0.011). This finding underscores the superior capacity of EXO to disrupt chemokine-mediated recruitment pathways and resolve local inflammation within the testicular environment, likely through a more efficient delivery of paracrine factors (21).
The differential reduction in leukocyte infiltration observed between treatments may be attributed to distinct mechanistic pathways. For EXO, their effects likely involve the inhibition of IL-1β-induced CCL2 secretion from Sertoli cells, a key chemotactic signal for monocytes, while simultaneously promoting Treg recruitment via transforming growth factor β1 delivery (24). This coordinated immunomodulation leverages systemic delivery of paracrine factors. In contrast, ASCs may operate through a more localized mechanism, potentially relying on their physical presence and contact-dependent signaling pathways such as PD-1/PD-L1 interaction, which could be limited by their migration and retention within the inflamed tissue (25).
Several limitations warrant consideration. The short-term, 14-day duration of the disease induction and treatment protocol precludes assessment of long-term functional recovery and may not be generalizable to chronic therapeutic regimens; future studies should evaluate spermatogenic restoration through sperm counts and mating trials. Proteomic characterization of exosomal cargo would identify essential therapeutic factors, as the exclusive focus on structural outcomes overlooks molecular events (26). Although IV administration was effective, localized intratesticular delivery has the potential to increase bioavailability and decrease the number of dosing requirements (24). Comparative studies with other MSC sources (e.g., bone marrow) could clarify tissue-specific therapeutic advantages.
The clinical application of these findings faces practical challenges. Large-scale production of EXO remains difficult, and ultracentrifugation fails to yield sufficient quantities for human use. Emerging methods, such as tangential flow filtration or CRISPR-engineered ASCs with increased EXO secretion, might help to bridge the gap. Dose optimization studies are needed to establish therapeutic windows, as our rat dose (200 μg) translates to ~0.5 mg/kg in humans, potentially requiring 30-50 mg per patient. Stability protocols, including lyophilization, must be developed to extend shelf life beyond current cold-chain limitations (25).
Despite these challenges, EXO presents distinct advantages over cellular therapies: reduced immunogenicity, avoidance of ectopic differentiation risks, and lower thrombotic potential (21). Their acellular nature streamlines regulatory approval routes as "non-living" biologics. EXO might be used to treat male infertility in acute orchitis to avoid lasting damage or in chronic situations to reverse existing inflammation-fibrosis cycles (2, 7). The ability to engineer EXO for enhanced targeting (e.g., testicular homing peptides) or cargo loading (e.g., anti-IL-1β siRNA) further expands their therapeutic potential (10, 23).
5. Conclusion
In conclusion, EXO appeared more effective in total, possibly due to better bioavailability and paracrine signaling (18-20). Their ability to concurrently resolve local inflammation (leukocyte reduction), restore tissue architecture, and normalize systemic biomarker positions them as a next-generation therapeutic platform for testicular inflammation and related infertility. Future research should prioritize clinical translation via scalable production, delivery optimization, and validation of functional reproductive recovery.
Our research provides evidence that IL-1β-induced inflammation in rat testicular tissues can be effectively alleviated by both mesenchymal stem cells and their derived EXO. Both treatment approaches demonstrated substantial anti-inflammatory and tissue-protective effects, indicating their therapeutic potential in mitigating testicular inflammation. These findings contribute to the growing body of evidence supporting the feasibility of cell-based and cell-free regenerative strategies, which may open new avenues for managing inflammatory and reproductive disorders.
Data Availability
Data will be available if requested.
Author Contributions
H. Shafaei, D. Mohammadnejad, and M. Bilabari designed the study and conducted the research. M. Bilabari, D. Mohammadnejad, and R. Rahbarghazi monitored, evaluated, and analyzed the results of the study. All authors reviewed and approved the final manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Acknowledgements
The authors acknowledge the use of DeepSeek AI (developed by DeepSeek) for grammatical refinement of the manuscript and summarizing portions of the abstract and introduction. The research design, data interpretation, and scientific conclusions are solely attributable to the authors. This work was funded by the Immunology Research Center of Tabriz University of Medical Sciences, Tabriz, Iran (grant number: 70810).
Conflict of Interest
The author declares that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Stem Cell & Cloning
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |