Tue, May 26, 2026
[Archive]
Volume 23, Issue 12 (December 2025)
IJRM 2025, 23(12): 1021-1030 |
Back to browse issues page
Ethics code: IR.TUMS.IKHC.REC.1403.427
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Farzipour S, Kashani L, Khodarahmian M, Jafari K, Moini A. Comparison of intracytoplasmic sperm injection and in vitro fertilization outcomes in double stimulation with conventional protocol in participants with poor ovarian response with minimal injection: An RCT. IJRM 2025; 23 (12) :1021-1030
URL: http://ijrm.ir/article-1-3702-en.html
URL: http://ijrm.ir/article-1-3702-en.html
1- Department of Obstetrics and Gynecology, School of Medicine, Arash Women's Hospital, Vali-E-Asr Reproductive Health Research Center, Tehran University of Medical Sciences, Tehran, Iran. & Ardabil University of Medical Sciences, Ardabil, Iran.
2- Female Infertility Unit, Arash Women's Hospital, Tehran University of Medical Sciences, Tehran, Iran. ,kashani_ladan@tums.ac.ir
3- Department of Female Infertility, Arash Women's Hospital, Department of Anatomy, School of Medicine, Tehran University of Medical Sciences, Tehran, Iran.
4- Research Development Center, Arash Women's Hospital, Tehran University of Medical Sciences, Tehran, Iran. & Department of Epidemiology, School of Health, Iran University of Medical Sciences, Tehran, Iran.
5- Department of Obstetrics and Gynecology, Endocrinology and Female Infertility Unit, Arash Women’s Hospital, Tehran University of Medical Sciences, Tehran, Iran. & Breast Disease Research Center (BDRC), Tehran University of Medical Sciences, Tehran, Iran., Department of Endocrinology and Female Infertility, Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine, ACECR, Tehran, Iran.
2- Female Infertility Unit, Arash Women's Hospital, Tehran University of Medical Sciences, Tehran, Iran. ,
3- Department of Female Infertility, Arash Women's Hospital, Department of Anatomy, School of Medicine, Tehran University of Medical Sciences, Tehran, Iran.
4- Research Development Center, Arash Women's Hospital, Tehran University of Medical Sciences, Tehran, Iran. & Department of Epidemiology, School of Health, Iran University of Medical Sciences, Tehran, Iran.
5- Department of Obstetrics and Gynecology, Endocrinology and Female Infertility Unit, Arash Women’s Hospital, Tehran University of Medical Sciences, Tehran, Iran. & Breast Disease Research Center (BDRC), Tehran University of Medical Sciences, Tehran, Iran., Department of Endocrinology and Female Infertility, Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine, ACECR, Tehran, Iran.
Keywords: Complementary therapy, Oocyte retrieval, Stimulation, Ovarian follicle, In vitro fertilization.
Full-Text [PDF 425 kb]
(319 Downloads)
| Abstract (HTML) (336 Views)
Full-Text: (68 Views)
1. Introduction
Many women affected by infertility require assisted reproductive technologies (ART), such as in vitro fertilization (IVF). One of the most significant and ongoing challenges in ART is managing poor ovarian response (POR). These women have a diminished ovarian reserve, which leads to a reduced number of oocytes retrieved after controlled ovarian stimulation, resulting in lower clinical pregnancy and live birth rates (1-4).
In POR cases, both the quantity and quality of oocytes decline, producing embryos of lower quality. The goal of ovarian stimulation is to increase the number of oocytes and embryos, thereby improving ART success rates (5). Although various treatment protocols have been developed to enhance IVF outcomes (3), there is no standardized regimen specifically designed for managing POR. Protocols often vary based on fertility center practices and physician preference, with the long agonist and gonadotropin-releasing hormone (GnRH) antagonist protocols being the most commonly used.
POR to exogenous gonadotropins remains a major obstacle in ART and can lead to cycle cancellation, fewer retrieved oocytes and embryos, and ultimately reduced pregnancy rates (6, 7). Contributing factors include advanced maternal age, prior ovarian surgery, pelvic adhesions, and elevated body mass index; notably, poor response can also occur in younger women (8).
A novel approach called dual stimulation (DuoStim), involving 2 rounds of controlled ovarian stimulation within a single menstrual cycle, has recently been proposed. This method benefits women with a poor prognosis who aim to maximize ovarian yield in a limited timeframe. DuoStim combines stimulation during both the follicular and luteal phases, making it a promising option for women with diminished ovarian reserve and advanced age (9-11).
A study demonstrated that DuoStim, by combining follicular and luteal phase stimulations, can produce oocytes with sufficient developmental potential (12). When applied in both phases, this method offers a promising alternative for POR. Importantly, antral follicle counts after the initial retrieval were comparable to baseline follicular phase counts, suggesting an opportunity to further enhance ovarian stimulation and increase oocyte yield (13).
2. Materials and Methods
2.1. Study design and participants
This randomized, double-blind, clinical trial was conducted on women aged between 24 and 44 diagnosed with POR undergoing IVF treatment at female infertility unit, Arash Women's hospital, Tehran University of Medical Sciences, Tehran, Iran from February-July 2025 (6-month duration).
Inclusion criteria were anti-Müllerian hormone ≤ 1.2 ng/mL or an AFC < 5 on day 5 of the menstrual cycle or fewer than 5 eggs retrieved in the previous cycle. Exclusion criteria included endometriosis higher than grade 3, contraindications for the use of gonadotropins, and couples with severe male factor infertility.
Participants were divided into 2 groups: group A (conventional) received the conventional antagonist protocol in 2 cycles, and group B received the DuoStim involving ovarian stimulation in both follicular and luteal phases. In this study, to ensure that the same number of participants will be allocated to both groups, a block randomization with a block size of 4 in a ratio of 1:1:1:1 was used. A list of blocks of 4 people will be randomly created using Stata 17.0 software. By this, we ensure that the number of people in each group is the same. Both doctors and the studied women were blinded to group assignments to minimize bias during treatment administration and outcome assessment. We consider poor ovarian responder based on bologna criteria.
Both groups underwent transvaginal ultrasound (HONDA 2200 KOREA) on days 1-3 of the menstrual cycle. In the DuoStim group, pre-treatment involved 4 mg estradiol from day 21 of the previous cycle until day 2 of menstruation, and ElonvA (Corifolitropin alfa 150/0/5 µg/ml, CO. ORGANON) was administered. Both Elonva and Cinnal-F 600 IU vial are highly effective follicle-stimulating hormone medications for stimulating the ovaries to produce multiple eggs. Although, there is no clear evidence that one is more effective than the other; however, we used Elonva with a defined dose because it was with minimal injection rate vs. daily Glyceryl trinitrate injection. Follow-up transvaginal sonography occurred 5-6 days later and then every 2 days to monitor follicular growth.
When a dominant follicle reached 12-13 mm, Cetrorelix (0.25 mg/day, GnRH antagonist) was started. Upon follicles reaching ≥ 17-18 mm, a GnRH agonist trigger (0.3 mg) was administered, followed by oocyte retrieval 36 hr later (Medical mastec Co., needle). After the first retrieval, medroxyprogesterone acetate (5 mg twice daily) was given to suppress lutenizing hormone surge, and gonadotropins were restarted 2-3 days later until follicles again reached 17-18 mm. A second trigger (GnRH-a 0.2 mg + human chorionic gonadotropin 5000 IU, KARMA Co., Irving, Texas) was then given, with oocyte retrieval performed after 36 hr. In DuoStim group, we started stimulation with Elonva, one ampula for 1 wk, after that we continued with Glyceryl trinitrate. In conventional group we had stimulation and oocyte retrieval in 2 cycles.
In the conventional protocol, stimulation began on day 2 of cycle with human menopausal gonadotropin and recombinant follicle-stimulating hormone for 3 days. The GnRH antagonist protocol was initiated when a follicle reached 14 mm. When follicles reached ≥ 18 mm, human chorionic gonadotropin (10,000 IU) was administered, and oocyte retrieval was performed 36 hr later.
Oocyte retrieval was guided by transvaginal sonography 36 hr post trigger. Retrieved oocytes were incubated for 2-4 hr, after which cumulus and corona radiata cells were removed. Mature oocytes underwent intracytoplasmic sperm injection. Embryos were cultured at 6% CO₂ and 5% O₂ at 37°C, with assessment on day 3 (cleavage stage) and day 5 (blastocyst formation). All embryos were vitrified for future use. Egg retrieval in the control group was performed in 2 separate cycles, and the interval between cycles varied among individuals.
2.2. Sample size
Based on a study by Ghahghayi et al. (14), the mean oocyte yield was 9.2 (DuoStim) vs. 6.9 (conventional) with an standard deviation of 3.8. With α = 5%, power = 80%, and 5% margin of error, 45 participants per group (N = 90) were required. We have 10 sample losses in this study (Figure 1). The used formula is:
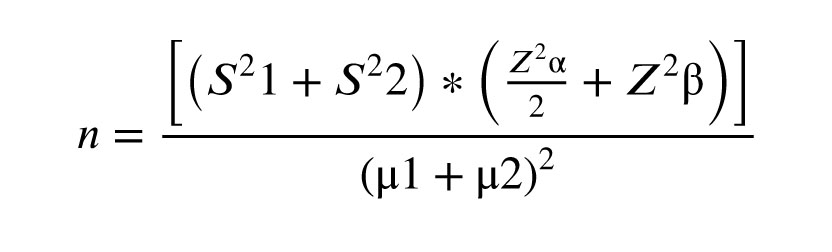
2.3. Outcomes
The primary and secondary outcomes of this study were the number of recovered metaphase II oocytes and the number of oocytes retrieved, and the outcome time point was 36 hr after the intervention. The retrieved oocytes in both groups and each stage were incubated for 2-4 hr, after which the cumulus and corona radiata cells were removed from the eggs with the observation of an inverted microscope by a clinical embryologist. The oocytes are then evaluated. All of them were evaluated by clinical and laboratory methods by an expert embryologist.
2.4. Ethical Considerations
This study was approved by Tehran University of Medical Sciences Ethics Committee, Tehran, Iran (Code: IR.TUMS.IKHC.REC.1403.427) and registration number: IRCT20250107064306N1, date of registration: January 12, 2025; updated date: September 24, 2025. Written informed consent was obtained from all participants, and compliance with the Helsinki Declaration was ensured. Patient privacy and comfort were prioritized during transvaginal ultrasounds.
2.5. Statistical Analysis
All qualitative data presented as frequency and percentage (%), and quantitative data as mean ± standard deviation for normally distributed variables or as median with interquartile range for variables that do not follow a normal distribution. Group comparisons was done by using an independent samples t test when normality assumptions are met and the Mann-Whitney U test when normality is not assumed, with statistical significance set at p < 0.05, and all analyses was conducted on SPSS version 24.
3. Results
In POR cases, both the quantity and quality of oocytes decline, producing embryos of lower quality. The goal of ovarian stimulation is to increase the number of oocytes and embryos, thereby improving ART success rates (5). Although various treatment protocols have been developed to enhance IVF outcomes (3), there is no standardized regimen specifically designed for managing POR. Protocols often vary based on fertility center practices and physician preference, with the long agonist and gonadotropin-releasing hormone (GnRH) antagonist protocols being the most commonly used.
POR to exogenous gonadotropins remains a major obstacle in ART and can lead to cycle cancellation, fewer retrieved oocytes and embryos, and ultimately reduced pregnancy rates (6, 7). Contributing factors include advanced maternal age, prior ovarian surgery, pelvic adhesions, and elevated body mass index; notably, poor response can also occur in younger women (8).
A novel approach called dual stimulation (DuoStim), involving 2 rounds of controlled ovarian stimulation within a single menstrual cycle, has recently been proposed. This method benefits women with a poor prognosis who aim to maximize ovarian yield in a limited timeframe. DuoStim combines stimulation during both the follicular and luteal phases, making it a promising option for women with diminished ovarian reserve and advanced age (9-11).
A study demonstrated that DuoStim, by combining follicular and luteal phase stimulations, can produce oocytes with sufficient developmental potential (12). When applied in both phases, this method offers a promising alternative for POR. Importantly, antral follicle counts after the initial retrieval were comparable to baseline follicular phase counts, suggesting an opportunity to further enhance ovarian stimulation and increase oocyte yield (13).
2. Materials and Methods
2.1. Study design and participants
This randomized, double-blind, clinical trial was conducted on women aged between 24 and 44 diagnosed with POR undergoing IVF treatment at female infertility unit, Arash Women's hospital, Tehran University of Medical Sciences, Tehran, Iran from February-July 2025 (6-month duration).
Inclusion criteria were anti-Müllerian hormone ≤ 1.2 ng/mL or an AFC < 5 on day 5 of the menstrual cycle or fewer than 5 eggs retrieved in the previous cycle. Exclusion criteria included endometriosis higher than grade 3, contraindications for the use of gonadotropins, and couples with severe male factor infertility.
Participants were divided into 2 groups: group A (conventional) received the conventional antagonist protocol in 2 cycles, and group B received the DuoStim involving ovarian stimulation in both follicular and luteal phases. In this study, to ensure that the same number of participants will be allocated to both groups, a block randomization with a block size of 4 in a ratio of 1:1:1:1 was used. A list of blocks of 4 people will be randomly created using Stata 17.0 software. By this, we ensure that the number of people in each group is the same. Both doctors and the studied women were blinded to group assignments to minimize bias during treatment administration and outcome assessment. We consider poor ovarian responder based on bologna criteria.
Both groups underwent transvaginal ultrasound (HONDA 2200 KOREA) on days 1-3 of the menstrual cycle. In the DuoStim group, pre-treatment involved 4 mg estradiol from day 21 of the previous cycle until day 2 of menstruation, and ElonvA (Corifolitropin alfa 150/0/5 µg/ml, CO. ORGANON) was administered. Both Elonva and Cinnal-F 600 IU vial are highly effective follicle-stimulating hormone medications for stimulating the ovaries to produce multiple eggs. Although, there is no clear evidence that one is more effective than the other; however, we used Elonva with a defined dose because it was with minimal injection rate vs. daily Glyceryl trinitrate injection. Follow-up transvaginal sonography occurred 5-6 days later and then every 2 days to monitor follicular growth.
When a dominant follicle reached 12-13 mm, Cetrorelix (0.25 mg/day, GnRH antagonist) was started. Upon follicles reaching ≥ 17-18 mm, a GnRH agonist trigger (0.3 mg) was administered, followed by oocyte retrieval 36 hr later (Medical mastec Co., needle). After the first retrieval, medroxyprogesterone acetate (5 mg twice daily) was given to suppress lutenizing hormone surge, and gonadotropins were restarted 2-3 days later until follicles again reached 17-18 mm. A second trigger (GnRH-a 0.2 mg + human chorionic gonadotropin 5000 IU, KARMA Co., Irving, Texas) was then given, with oocyte retrieval performed after 36 hr. In DuoStim group, we started stimulation with Elonva, one ampula for 1 wk, after that we continued with Glyceryl trinitrate. In conventional group we had stimulation and oocyte retrieval in 2 cycles.
In the conventional protocol, stimulation began on day 2 of cycle with human menopausal gonadotropin and recombinant follicle-stimulating hormone for 3 days. The GnRH antagonist protocol was initiated when a follicle reached 14 mm. When follicles reached ≥ 18 mm, human chorionic gonadotropin (10,000 IU) was administered, and oocyte retrieval was performed 36 hr later.
Oocyte retrieval was guided by transvaginal sonography 36 hr post trigger. Retrieved oocytes were incubated for 2-4 hr, after which cumulus and corona radiata cells were removed. Mature oocytes underwent intracytoplasmic sperm injection. Embryos were cultured at 6% CO₂ and 5% O₂ at 37°C, with assessment on day 3 (cleavage stage) and day 5 (blastocyst formation). All embryos were vitrified for future use. Egg retrieval in the control group was performed in 2 separate cycles, and the interval between cycles varied among individuals.
2.2. Sample size
Based on a study by Ghahghayi et al. (14), the mean oocyte yield was 9.2 (DuoStim) vs. 6.9 (conventional) with an standard deviation of 3.8. With α = 5%, power = 80%, and 5% margin of error, 45 participants per group (N = 90) were required. We have 10 sample losses in this study (Figure 1). The used formula is:
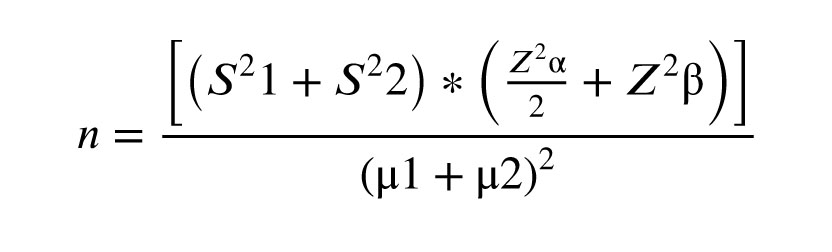
2.3. Outcomes
The primary and secondary outcomes of this study were the number of recovered metaphase II oocytes and the number of oocytes retrieved, and the outcome time point was 36 hr after the intervention. The retrieved oocytes in both groups and each stage were incubated for 2-4 hr, after which the cumulus and corona radiata cells were removed from the eggs with the observation of an inverted microscope by a clinical embryologist. The oocytes are then evaluated. All of them were evaluated by clinical and laboratory methods by an expert embryologist.

2.4. Ethical Considerations
This study was approved by Tehran University of Medical Sciences Ethics Committee, Tehran, Iran (Code: IR.TUMS.IKHC.REC.1403.427) and registration number: IRCT20250107064306N1, date of registration: January 12, 2025; updated date: September 24, 2025. Written informed consent was obtained from all participants, and compliance with the Helsinki Declaration was ensured. Patient privacy and comfort were prioritized during transvaginal ultrasounds.
2.5. Statistical Analysis
All qualitative data presented as frequency and percentage (%), and quantitative data as mean ± standard deviation for normally distributed variables or as median with interquartile range for variables that do not follow a normal distribution. Group comparisons was done by using an independent samples t test when normality assumptions are met and the Mann-Whitney U test when normality is not assumed, with statistical significance set at p < 0.05, and all analyses was conducted on SPSS version 24.
3. Results
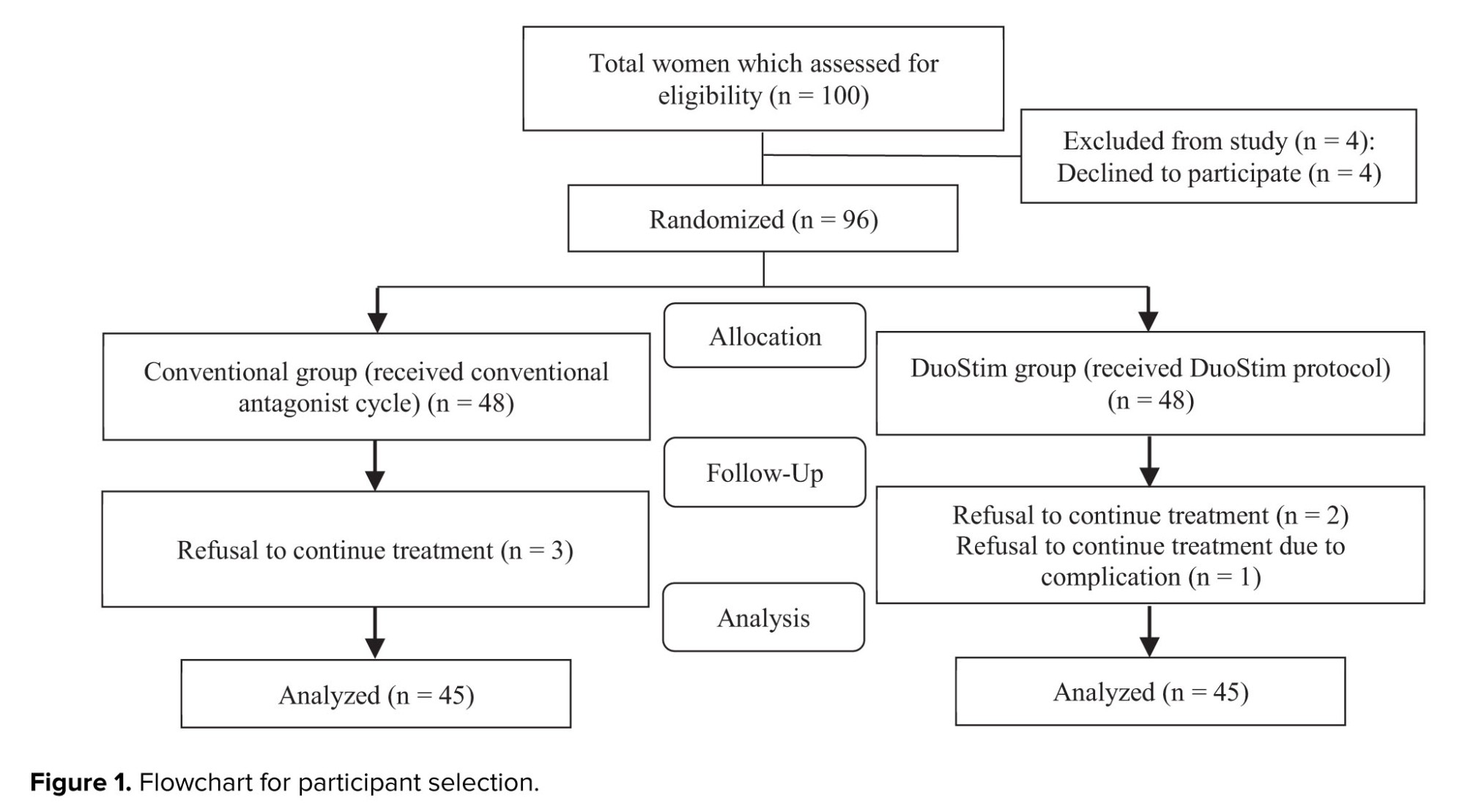
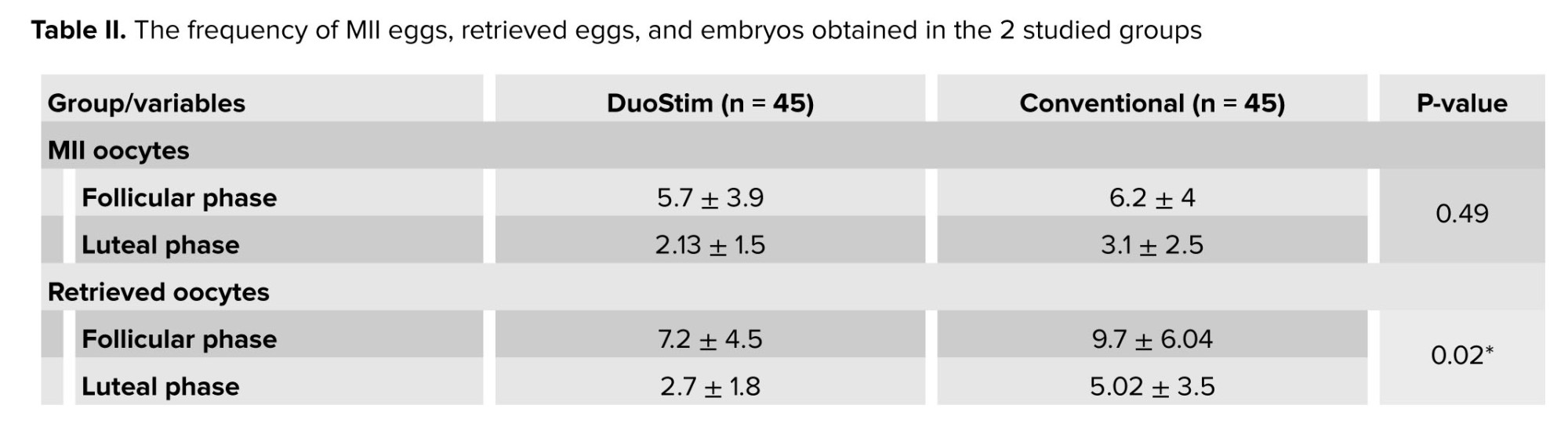
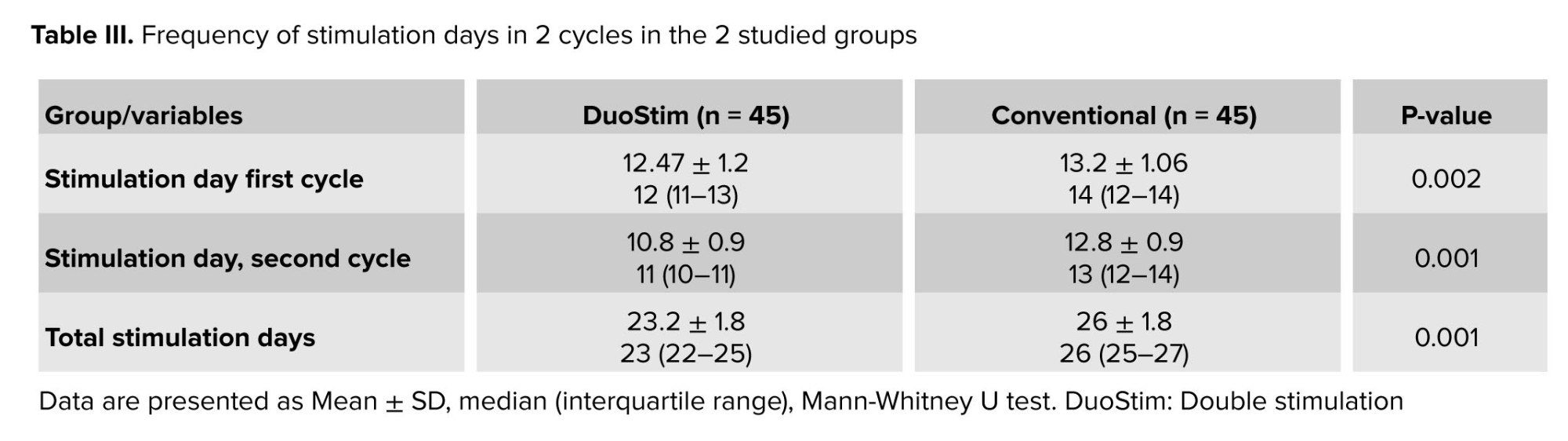
This study involved 100 infertile women with reduced ovarian reserve, participants were randomly assigned to either the DuoStim protocol or the conventional antagonist cycle. After withdrawals, 45 participants remained in each group for analysis. The mean age of all women was 38.6 ± 4.1 yr, and the mean boby mass index was 25.1 ± 2.8 kg/m². The median of the duration of infertility in DuoStim cases was 3, and in the conventional group was 5; no significant differences were observed between the 2 groups (Table I). Anti-Müllerian hormone levels did not differ significantly between groups (p = 0.25) (Table II). The mean number of mature metaphase II oocytes was similar between the DuoStim (5.7 ± 3.9) and control groups (6.2 ± 4, p = 0.53). However, the total number of retrieved eggs was significantly lower in the DuoStim group (7.2 ± 4.5) compared to controls (9.7 ± 6, p = 0.026). Embryo numbers were comparable (4.5 ± 3.5 vs. 4.7 ± 3.1, p = 0.73). In the DuoStim group, second round measures of eggs, mature oocytes, and embryos were significantly higher than the first round (p = 0.035, p = 0.049, p = 0.028). Notably, the duration of ovarian stimulation was significantly shorter in the DuoStim group (p = 0.002). These results suggest that DuoStim can achieve similar mature oocyte and embryo yields in less stimulation time compared to conventional protocols, although with fewer total eggs retrieved (Table III).
The mean of stimulation days in 2 cycles and the total were lower in the DuoStim group than the conventional group, and the difference was significant. The difference between stimulation days comes from data without any explanation (Table III).


4. Discussion
This study compared the DuoStim protocol, which involves dual ovarian stimulation within a single cycle, to the conventional antagonist protocol in POR. Results showed that DuoStim significantly reduced the total number of retrieved oocytes and shortened stimulation duration, while mature oocyte and embryo numbers were similar between groups. These findings align with other research indicating DuoStim improves treatment efficiency but has variable effects on oocyte yield, with some studies reporting higher yields and others comparable or lower.
The findings of a randomized trial directly aligns with and provides specific evidence for the general conclusions in the initial summary text. The study confirms that in POR patients, the DuoStim protocol resulted in a similar number of mature oocytes and high-quality embryos compared to the conventional antagonist protocol, which strongly supports the text’s consistent claim that “embryo quality is preserved across protocols”. Furthermore, it was observed in a study that a lower total oocyte yield in the DuoStim group fits within the text's acknowledgment of “variable effects on oocyte yield”, specifically falling into the category of studies reporting “comparable or lower” numbers (13). By demonstrating that a DuoStim strategy can yield comparable blastocyst rates within a single cycle, the study empirically validates the text’s concept that “combining follicular and luteal phases may maximize oocyte yield within one cycle without compromising embryo viability”. Finally, the use of a randomized design in Ghahghayi’s study directly mirrors the “key strength” highlighted in the text, while its modest sample size reflects the mentioned “limitation” of most current studies, underscoring the text's concluding need for larger trials to firmly establish the role of DuoStim (14).
Consistently, embryo quality is preserved across protocols (5, 10, 14). Differences in outcomes are likely influenced by patient characteristics, stimulation regimens, and embryo transfer strategies, including fresh vs. frozen transfers. Some trials provide a critical piece of evidence that directly addresses and helps explain the variable live birth outcomes observed in other DuoStim studies. Unlike the BISTIM trial, which found no difference in cumulative live birth rates. Demonstrated that a DuoStim followed by a fresh embryo transfer resulted in a significantly higher live birth rate compared to a single stimulation in young poor-prognosis patients. This finding refines the overall picture by strongly supporting the initial text’s assertion that “DuoStim’s efficacy appears more pronounced in younger populations or with fresh transfers”. It suggests that the superior live birth rate seen in another study and this, may be attributable to the fresh transfer strategy. It offers a specific clinical context (younger POR patients undergoing a fresh transfer) where DuoStim's efficiency in oocyte accumulation can most effectively translate into a superior clinical outcome, thereby resolving some of the ambiguity left by other trials (15).
A study in 2022 directly addresses the core conclusion of the initial text by providing crucial evidence on pregnancy outcomes, an area the text identified as needing further research. While the initial text summarized that DuoStim's efficacy is “more pronounced in younger populations or with fresh transfers”. A study offers a specific comparative context: they found that DuoStim resulted in significantly higher cumulative live birth rates compared to 2 consecutive mild stimulations in poor ovarian responders. This finding strongly supports the text’s position that DuoStim improves “treatment efficiency” and “prioritizes cycle efficiency”, as it demonstrates that gathering oocytes from both phases of a single cycle (DuoStim) is more effective for achieving a live birth than spreading the process over 2 separate, milder cycles. This outcome aligns with the text’s suggestion that DuoStim allows for faster oocyte accumulation, and importantly, it begins to answer the text’s call for “larger, standardized studies evaluating pregnancy and live birth outcomes” to clarify DuoStim’s full clinical role (16).
The studies also support the luteal-phase stimulation concept, suggesting that combining follicular and luteal phases may maximize oocyte yield within one cycle without compromising embryo viability (17). Based on the initial summary text, the findings of a trial would directly compare the efficiency and practicality of 2 common POR protocols. The initial text suggests that “DuoStim offers faster oocyte accumulation but may be less tolerable or cost-effective for some women” compared to minimal stimulation. Therefore, the results of this 2018 study would provide critical evidence to either support or refute this trade off. If the trial found that DuoStim yielded more oocytes or embryos in a shorter timeframe but had higher medication costs or patient dropout rates, it would perfectly align with the text's conclusion. Conversely, if it showed no significant difference in efficiency or found minimal stimulation to be non-inferior, it would challenge the notion that DuoStim is the superior strategy for rapid oocyte accumulation, reinforcing the text's point that the optimal protocol is context dependent (18).
Key strengths include the randomized design and consistent application of protocols, allowing controlled comparison. Limitations include modest sample size, restricting subgroup analyses, exclusive use of vitrified embryos, which limits fresh transfer comparisons, and heterogeneity in POR definitions complicating comparisons (17-19).
Overall, this study provides evidence that DuoStim reduces stimulation time and maintains embryo quality, but its impact on oocyte yield is context dependent. It supports DuoStim as a treatment option for POR women, prioritizing cycle efficiency. Larger, standardized studies evaluating pregnancy and live birth outcomes are needed to clarify the full clinical role of DuoStim (16-20).
The Massin study directly validates and refines several key conclusions from the initial text. The finding that DuoStim yielded a significantly higher cumulative number of oocytes and embryos per single initiated cycle, strongly supports the text’s concept that “combining follicular and luteal phases may maximize oocyte yield within one cycle”. Furthermore, by demonstrating that embryo aneuploidy rates and quality were similar between groups, it provides robust, randomized evidence for the text’s consistent claim that “embryo quality is preserved across protocols”. However, the BISTIM study's critical finding (that despite more embryos, DuoStim did not lead to a higher cumulative live birth rate) adds a crucial layer of nuance. This outcome underscores the text’s assertion that differences are influenced by “embryo transfer strategies” and directly addresses its final call for studies “evaluating pregnancy and live birth outcomes”, suggesting that for some POR patients, the efficiency of DuoStim may not automatically translate into superior clinical success (21).
5. Conclusion
The results of this study demonstrated that the DuoStim protocol is a favorable and time-efficient strategy for poor ovarian responders, significantly reducing the duration of stimulation and the number of oocyte retrieval procedures compared to conventional methods. However, the advantages of DuoStim appear to be primarily related to increasing oocyte quantity, as no significant differences were observed in the number of mature oocytes or embryos produced, nor in pregnancy outcomes.
Although in our study, the total number of oocytes was lower in DuoStim, no difference was observed in MII and the number of high-quality embryos. The number of days were shorter and, as a result, the amount of gonadotropin used was less, which is cost-effective. In terms of time, we reached the same number of mature oocytes and high-quality embryos in a shorter time despite lower oocyte retrieval in the DuoStim group and stimulation days, and number of injections were lower in the DuoStim group. In this study, the number of oocytes had decreased in the DuoStim group than in the conventional group, but due to the non-change of MII oocytes and embryos, the final result of IVF was not impacted by these results.
Future research should focus on evaluating the genetic and blastocyst development outcomes of embryos derived from DuoStim cycles. Additionally, studies comparing patient satisfaction across different stimulation protocols are warranted to provide a more comprehensive assessment of this approach.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
Sh. Farzipour: Study design, review articles, paper preparation, data collection. L. Kashani: Review articles, manuscript draft, data collection, clinical examination. M. Khodarahmian and A. Moini: Data collection, review articles, writing results, sampling. K. Jafari: Data analysis, review articles, manuscript preparation, data analysis.
Acknowledgements
The authors would like to thank all the individuals who allowed us to use their medical records. We also would like to acknowledge our colleagues at the Arash Women’s hospital, Tehran, Iran. We used Perplexity AI for checking grammar in some sections of this article.
Conflict of Interest
The authors declare that there is no conflict of interest.
The mean of stimulation days in 2 cycles and the total were lower in the DuoStim group than the conventional group, and the difference was significant. The difference between stimulation days comes from data without any explanation (Table III).




4. Discussion
This study compared the DuoStim protocol, which involves dual ovarian stimulation within a single cycle, to the conventional antagonist protocol in POR. Results showed that DuoStim significantly reduced the total number of retrieved oocytes and shortened stimulation duration, while mature oocyte and embryo numbers were similar between groups. These findings align with other research indicating DuoStim improves treatment efficiency but has variable effects on oocyte yield, with some studies reporting higher yields and others comparable or lower.
The findings of a randomized trial directly aligns with and provides specific evidence for the general conclusions in the initial summary text. The study confirms that in POR patients, the DuoStim protocol resulted in a similar number of mature oocytes and high-quality embryos compared to the conventional antagonist protocol, which strongly supports the text’s consistent claim that “embryo quality is preserved across protocols”. Furthermore, it was observed in a study that a lower total oocyte yield in the DuoStim group fits within the text's acknowledgment of “variable effects on oocyte yield”, specifically falling into the category of studies reporting “comparable or lower” numbers (13). By demonstrating that a DuoStim strategy can yield comparable blastocyst rates within a single cycle, the study empirically validates the text’s concept that “combining follicular and luteal phases may maximize oocyte yield within one cycle without compromising embryo viability”. Finally, the use of a randomized design in Ghahghayi’s study directly mirrors the “key strength” highlighted in the text, while its modest sample size reflects the mentioned “limitation” of most current studies, underscoring the text's concluding need for larger trials to firmly establish the role of DuoStim (14).
Consistently, embryo quality is preserved across protocols (5, 10, 14). Differences in outcomes are likely influenced by patient characteristics, stimulation regimens, and embryo transfer strategies, including fresh vs. frozen transfers. Some trials provide a critical piece of evidence that directly addresses and helps explain the variable live birth outcomes observed in other DuoStim studies. Unlike the BISTIM trial, which found no difference in cumulative live birth rates. Demonstrated that a DuoStim followed by a fresh embryo transfer resulted in a significantly higher live birth rate compared to a single stimulation in young poor-prognosis patients. This finding refines the overall picture by strongly supporting the initial text’s assertion that “DuoStim’s efficacy appears more pronounced in younger populations or with fresh transfers”. It suggests that the superior live birth rate seen in another study and this, may be attributable to the fresh transfer strategy. It offers a specific clinical context (younger POR patients undergoing a fresh transfer) where DuoStim's efficiency in oocyte accumulation can most effectively translate into a superior clinical outcome, thereby resolving some of the ambiguity left by other trials (15).
A study in 2022 directly addresses the core conclusion of the initial text by providing crucial evidence on pregnancy outcomes, an area the text identified as needing further research. While the initial text summarized that DuoStim's efficacy is “more pronounced in younger populations or with fresh transfers”. A study offers a specific comparative context: they found that DuoStim resulted in significantly higher cumulative live birth rates compared to 2 consecutive mild stimulations in poor ovarian responders. This finding strongly supports the text’s position that DuoStim improves “treatment efficiency” and “prioritizes cycle efficiency”, as it demonstrates that gathering oocytes from both phases of a single cycle (DuoStim) is more effective for achieving a live birth than spreading the process over 2 separate, milder cycles. This outcome aligns with the text’s suggestion that DuoStim allows for faster oocyte accumulation, and importantly, it begins to answer the text’s call for “larger, standardized studies evaluating pregnancy and live birth outcomes” to clarify DuoStim’s full clinical role (16).
The studies also support the luteal-phase stimulation concept, suggesting that combining follicular and luteal phases may maximize oocyte yield within one cycle without compromising embryo viability (17). Based on the initial summary text, the findings of a trial would directly compare the efficiency and practicality of 2 common POR protocols. The initial text suggests that “DuoStim offers faster oocyte accumulation but may be less tolerable or cost-effective for some women” compared to minimal stimulation. Therefore, the results of this 2018 study would provide critical evidence to either support or refute this trade off. If the trial found that DuoStim yielded more oocytes or embryos in a shorter timeframe but had higher medication costs or patient dropout rates, it would perfectly align with the text's conclusion. Conversely, if it showed no significant difference in efficiency or found minimal stimulation to be non-inferior, it would challenge the notion that DuoStim is the superior strategy for rapid oocyte accumulation, reinforcing the text's point that the optimal protocol is context dependent (18).
Key strengths include the randomized design and consistent application of protocols, allowing controlled comparison. Limitations include modest sample size, restricting subgroup analyses, exclusive use of vitrified embryos, which limits fresh transfer comparisons, and heterogeneity in POR definitions complicating comparisons (17-19).
Overall, this study provides evidence that DuoStim reduces stimulation time and maintains embryo quality, but its impact on oocyte yield is context dependent. It supports DuoStim as a treatment option for POR women, prioritizing cycle efficiency. Larger, standardized studies evaluating pregnancy and live birth outcomes are needed to clarify the full clinical role of DuoStim (16-20).
The Massin study directly validates and refines several key conclusions from the initial text. The finding that DuoStim yielded a significantly higher cumulative number of oocytes and embryos per single initiated cycle, strongly supports the text’s concept that “combining follicular and luteal phases may maximize oocyte yield within one cycle”. Furthermore, by demonstrating that embryo aneuploidy rates and quality were similar between groups, it provides robust, randomized evidence for the text’s consistent claim that “embryo quality is preserved across protocols”. However, the BISTIM study's critical finding (that despite more embryos, DuoStim did not lead to a higher cumulative live birth rate) adds a crucial layer of nuance. This outcome underscores the text’s assertion that differences are influenced by “embryo transfer strategies” and directly addresses its final call for studies “evaluating pregnancy and live birth outcomes”, suggesting that for some POR patients, the efficiency of DuoStim may not automatically translate into superior clinical success (21).
5. Conclusion
The results of this study demonstrated that the DuoStim protocol is a favorable and time-efficient strategy for poor ovarian responders, significantly reducing the duration of stimulation and the number of oocyte retrieval procedures compared to conventional methods. However, the advantages of DuoStim appear to be primarily related to increasing oocyte quantity, as no significant differences were observed in the number of mature oocytes or embryos produced, nor in pregnancy outcomes.
Although in our study, the total number of oocytes was lower in DuoStim, no difference was observed in MII and the number of high-quality embryos. The number of days were shorter and, as a result, the amount of gonadotropin used was less, which is cost-effective. In terms of time, we reached the same number of mature oocytes and high-quality embryos in a shorter time despite lower oocyte retrieval in the DuoStim group and stimulation days, and number of injections were lower in the DuoStim group. In this study, the number of oocytes had decreased in the DuoStim group than in the conventional group, but due to the non-change of MII oocytes and embryos, the final result of IVF was not impacted by these results.
Future research should focus on evaluating the genetic and blastocyst development outcomes of embryos derived from DuoStim cycles. Additionally, studies comparing patient satisfaction across different stimulation protocols are warranted to provide a more comprehensive assessment of this approach.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
Sh. Farzipour: Study design, review articles, paper preparation, data collection. L. Kashani: Review articles, manuscript draft, data collection, clinical examination. M. Khodarahmian and A. Moini: Data collection, review articles, writing results, sampling. K. Jafari: Data analysis, review articles, manuscript preparation, data analysis.
Acknowledgements
The authors would like to thank all the individuals who allowed us to use their medical records. We also would like to acknowledge our colleagues at the Arash Women’s hospital, Tehran, Iran. We used Perplexity AI for checking grammar in some sections of this article.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Assisted Reproductive Technologies
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







