Thu, Apr 23, 2026
[Archive]
Volume 24, Issue 2 (February 2026)
IJRM 2026, 24(2): 117-126 |
Back to browse issues page
Ethics code: IR.IAU.QOM.REC.1402.208
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Hedayatnia H, Kalhor N. In vitro exploration of the therapeutic potential of mesenchymal stem cell conditioned medium in endometriosis treatment by targeting apoptosis and cellular migration. IJRM 2026; 24 (2) :117-126
URL: http://ijrm.ir/article-1-3718-en.html
URL: http://ijrm.ir/article-1-3718-en.html
1- Department of Biology, Faculty of Basic Sciences, Islamic Azad University, Qom Branch, Qom, Iran. , hassanhedayatnia_3@yahoo.com; Naserkalhor@acecr.ir; naserkalhor@acecr.ir
2- Department of Mesenchymal Stem Cells, Research Center, Academic Center for Education, Culture and Research (ACECR), Qom Branch, Qom, Iran.
2- Department of Mesenchymal Stem Cells, Research Center, Academic Center for Education, Culture and Research (ACECR), Qom Branch, Qom, Iran.
Keywords: Endometriosis, Mesenchymal stem cells, Cell migration, Apoptosis, Conditioned culture media.
Full-Text [PDF 2367 kb]
(91 Downloads)
| Abstract (HTML) (254 Views)
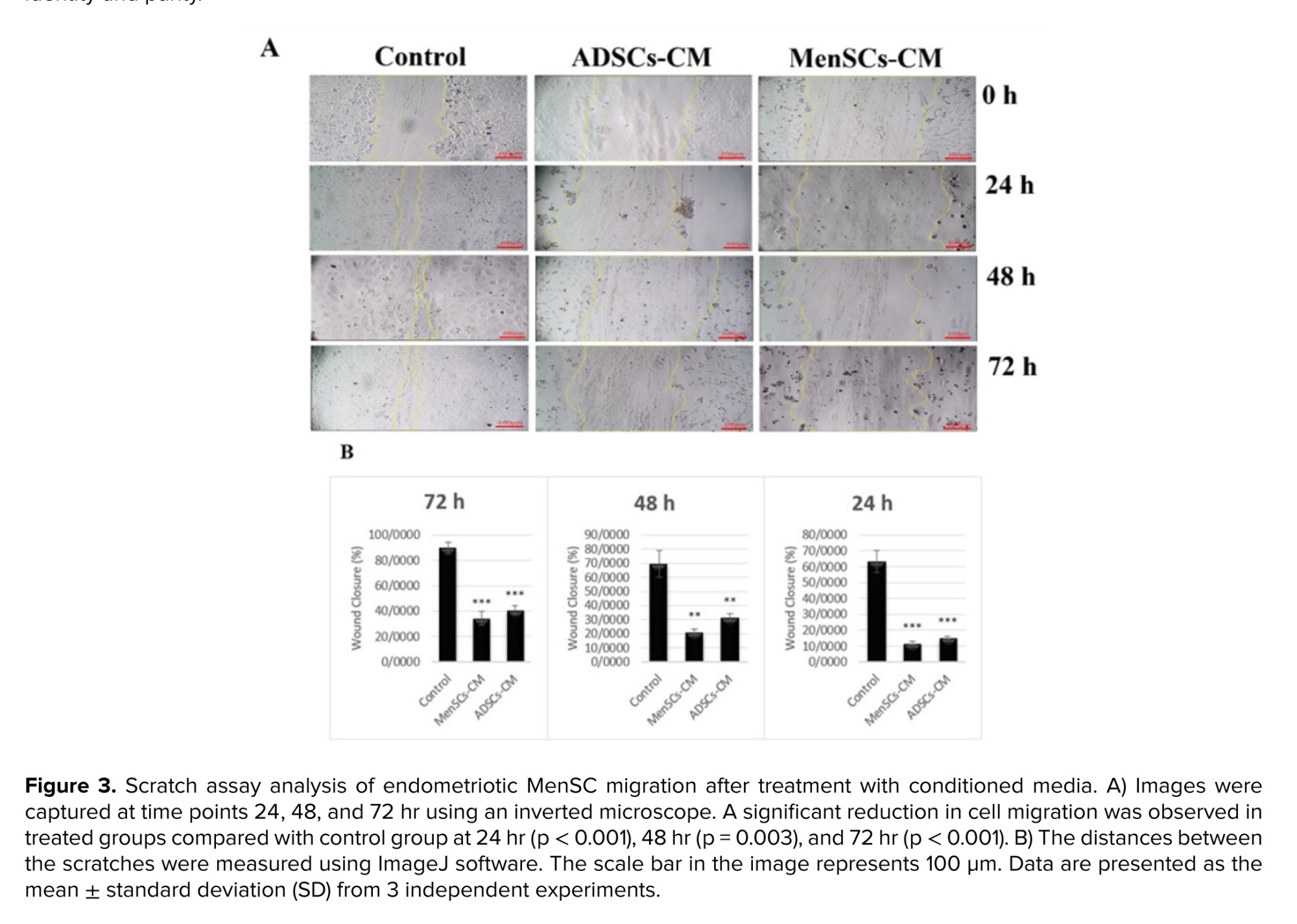
3.2. Scratch assay showing reduced migration
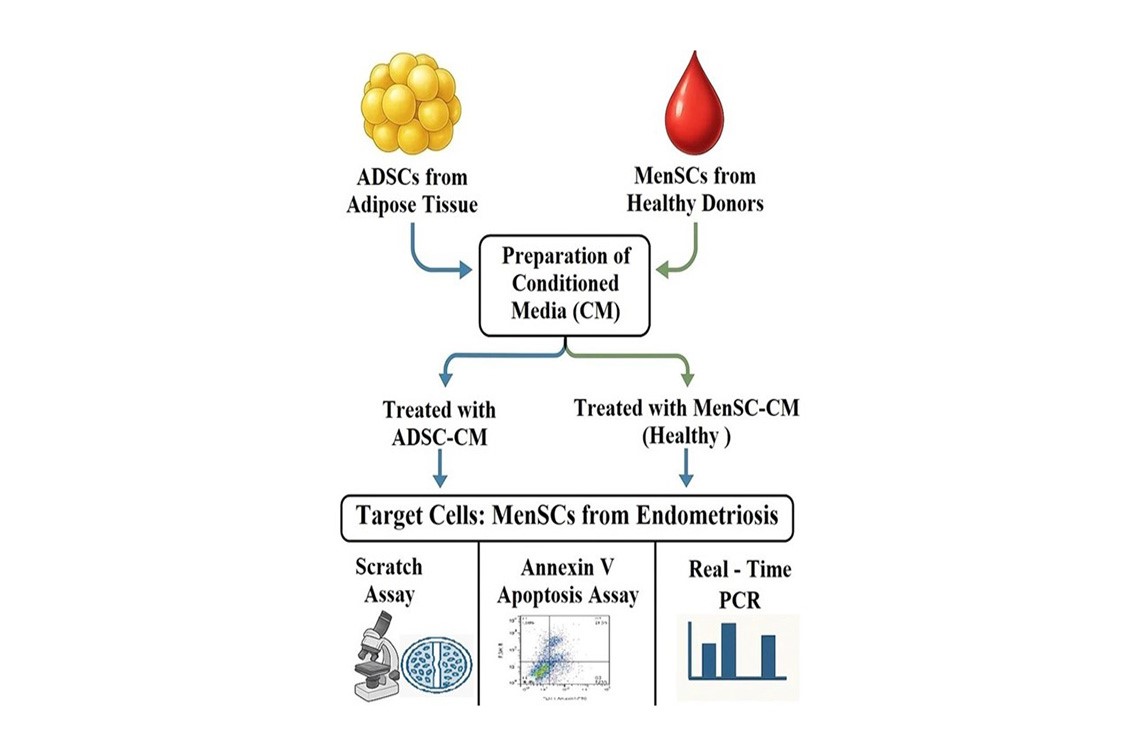
Following treatment with CM, we investigated the effect of different media on the migration of MenSCs from women with endometriosis. 3 experimental groups were analyzed: one group of MenSCs from women with endometriosis treated with ADSCs-CM, another group treated with MenSCs-CM from healthy women, and a control group receiving standard cell culture medium (DMEM-LG) supplemented with 10% FBS. A significant difference was observed in migration of MenSCs regarding initial scratch width at time points 24, 48, and 72 hr post-scratch. Both the ADSCs-CM and MenSCs-CM groups exhibited a significant decrease in the scratch area compared to the control group. These findings indicate that both conditioning media effectively inhibited the migration of MenSCs in women with endometriosis. Results of the scratch assay are summarized in figure 3.
Full-Text: (10 Views)
1. Introduction
Endometriosis is defined as the presence of endometrial-like tissue outside the uterine cavity (1). Women with endometriosis often experience pelvic pain manifested as dysmenorrhea, dyspareunia, and non-menstrual chronic pelvic pain, though some remain asymptomatic. Infertility affects about 30% of individuals, and many also suffer from fatigue, back pain, and urinary or gastrointestinal symptoms that impair quality of life, work, and relationships. Emotional consequences such as depression and anxiety are also common (2).
Although found in the pelvic cavity, endometriosis may occur at sites outside the pelvis (3). Pelvic endometriosis involves lesions near the uterus, including the ovaries, fallopian tubes, uterine ligaments, and pelvic peritoneum. Extra pelvic endometriosis affects other parts of the body, such as the vagina, vulva, cervix, and perineum (4). The main theory of its pathogenesis proposes implantation of endometrial tissue into the peritoneal cavity via retrograde menstruation, whereby shed endometrial cells travel backward through the fallopian tubes (5).
This theory highlights the importance of cellular components in menstrual fluid in the development of endometriosis. Endometrial-derived mesenchymal stem cells (MSCs) and menstrual blood-derived stem cells (MenSCs) may play direct roles in the formation and maintenance of ectopic lesions (6). Apoptosis maintains normal endometrial homeostasis by removing senescent cells before necrosis, thereby preventing migration and aggregation of endometrial cells. In women with endometriosis, the apoptotic rate in eutopic endometrial cells is markedly reduced (7). Existing treatments for endometriosis are frequently associated with disease and pain recurrence and are further limited by multiple side effects (8). Cell therapy, a key strategy in regenerative medicine, offers promising potential for treating or possibly curing multiple diseases (9). Adipose tissue-derived mesenchymal stem cells (ADSCs) have attracted significant attention for their therapeutic potential in regenerative medicine, meeting key criteria for effective cell therapy (10). Their low immunogenicity, self-renewal, multilineage differentiation, migration to injury sites, and autocrine/paracrine actions contribute to tissue repair. Similarly, MenSCs obtained from menstrual shedding offer a non-invasive and ethically acceptable stem cell source (11, 12). Both ADSCs and MenSCs underscore the variety of stem cell sources that can advance tissue‑repair strategies. MSCs derived conditioned medium (CM) also provides strong immunological compatibility and practical advantages for regenerative medicine (13). This study investigates the cell-free effects of MSC-CM derived from healthy MenSCs and ADSCs, on invasion and apoptosis in endometriosis.
2. Materials and Methods
Although found in the pelvic cavity, endometriosis may occur at sites outside the pelvis (3). Pelvic endometriosis involves lesions near the uterus, including the ovaries, fallopian tubes, uterine ligaments, and pelvic peritoneum. Extra pelvic endometriosis affects other parts of the body, such as the vagina, vulva, cervix, and perineum (4). The main theory of its pathogenesis proposes implantation of endometrial tissue into the peritoneal cavity via retrograde menstruation, whereby shed endometrial cells travel backward through the fallopian tubes (5).
This theory highlights the importance of cellular components in menstrual fluid in the development of endometriosis. Endometrial-derived mesenchymal stem cells (MSCs) and menstrual blood-derived stem cells (MenSCs) may play direct roles in the formation and maintenance of ectopic lesions (6). Apoptosis maintains normal endometrial homeostasis by removing senescent cells before necrosis, thereby preventing migration and aggregation of endometrial cells. In women with endometriosis, the apoptotic rate in eutopic endometrial cells is markedly reduced (7). Existing treatments for endometriosis are frequently associated with disease and pain recurrence and are further limited by multiple side effects (8). Cell therapy, a key strategy in regenerative medicine, offers promising potential for treating or possibly curing multiple diseases (9). Adipose tissue-derived mesenchymal stem cells (ADSCs) have attracted significant attention for their therapeutic potential in regenerative medicine, meeting key criteria for effective cell therapy (10). Their low immunogenicity, self-renewal, multilineage differentiation, migration to injury sites, and autocrine/paracrine actions contribute to tissue repair. Similarly, MenSCs obtained from menstrual shedding offer a non-invasive and ethically acceptable stem cell source (11, 12). Both ADSCs and MenSCs underscore the variety of stem cell sources that can advance tissue‑repair strategies. MSCs derived conditioned medium (CM) also provides strong immunological compatibility and practical advantages for regenerative medicine (13). This study investigates the cell-free effects of MSC-CM derived from healthy MenSCs and ADSCs, on invasion and apoptosis in endometriosis.
2. Materials and Methods
2.1. Human specimens
Women without endometriosis were designated as donors for CM preparation (n=3); these healthy women were recruited from individuals whose husbands presented with male factor infertility at the center. women with stage III-IV endometriosis (n=3) were recruited as donors for MenSC isolation. The MenSCs obtained from these patients were used as the primary cells subjected to the experimental treatments. All individuals in both groups underwent laparoscopy for infertility (donor group) and pelvic pain (endometriosis group), respectively, for the collection of tissue sample.
The inclusion criteria were:
1. No history of malignancy or autoimmune disease.
2. No hormone treatment for at least 3 months prior to the study.
3. No prior surgical interventions in women diagnosed with endometriosis.
4. Confirmation of deep endometriosis via transvaginal ultrasound and magnetic resonance imaging.
2.2. MenSCs isolation and primary culture
About 2 mL of menstrual blood was collected using an intrauterine insemination catheter on days 2 and 3 of the menstrual cycle from infertile women aged between 25 and 35 yr. Mononuclear cells were separated, and the cell pellet was resuspended in Dulbecco’s modified Eagle’s (DMEM) low glucose (LG) medium (Invitrogen, Life Technologies, Germany) supplemented with 100 mg/mL penicillin-streptomycin (Gibco, USA) and 10% fetal bovine serum (FBS) (Gibco, USA). Cells were seeded in a T25 flask and incubated at 37°C in a humidified atmosphere with 5% carbon dioxide. The medium was replaced after 24 hr to remove detached cells, followed by regular exchange of culture medium every 3 days.
2.3. Immunophenotyping analysis of MenSCs
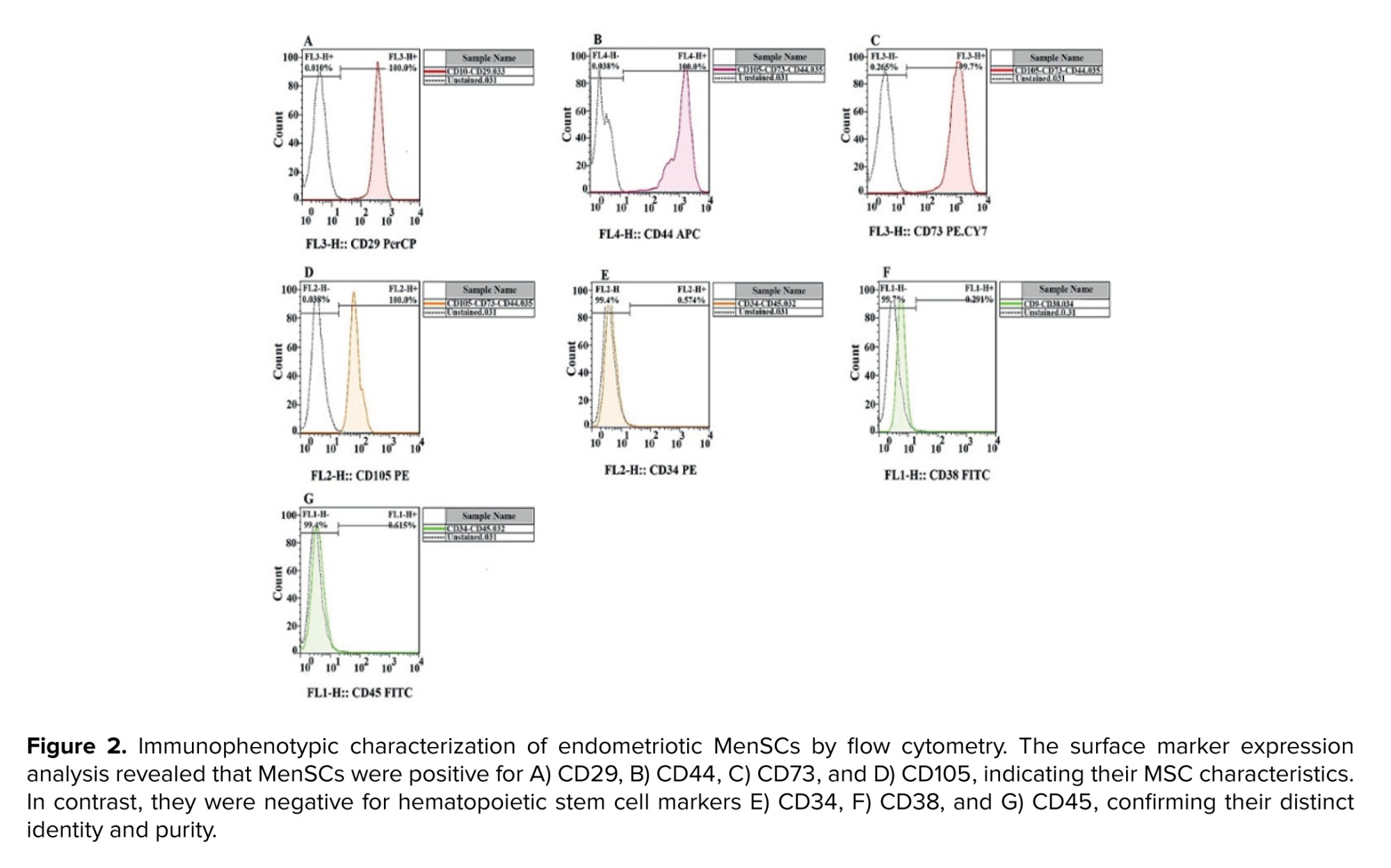
To confirm the identity of the isolated cells as MenSCs, the expression of cell surface markers was analyzed using flow cytometry. After 3 passages, cells were evaluated for the presence of positive markers (CD29, CD44, CD73, and CD105) and the absence of negative markers (CD34, CD38, and CD45). Flow cytometry was performed using an FC500 flow cytometer (Beckman Coulter, Fullerton, USA). Monoclonal antibodies for flow cytometry were obtained from BioLegend Co. (USA) and used according to the manufacturer’s recommendations. Data acquisition and analysis were performed using Beckman Coulter CXP software.
2.4. Preparation of CM
Following the isolation of ADSCs and MenSCs, the cells were transferred to separate T25 flasks, each containing DMEM culture medium with 10% FBS. At the 3rd passage, after 3 days ofincubation, both ADSCs and MenSCs achieved approximately 80% confluence. The culture medium from each flask was then collected separately, centrifuged at 600 g for 5 min, and the resulting supernatant was passed through a 0.22 µm syringe filter and stored at -80°C for future experimentation.
2.5. Scratch assay
In passage 3, MenSCs derived from women with endometriosis were cultured in 6-well plates. After 24 hr of incubation under standard conditions, the control group's culture medium was replaced with DMEM-LG medium supplemented with 10% FBS. 2 treatment groups were established: one receiving CM from ADSCs, and another receiving CM from MenSCs derived from healthy women. All groups were incubated for an additional 72 hr. To evaluate the impact of CM on wound closure, a scratch was made in each cell monolayer using a pipette tip, followed by washing with phosphate‑buffered saline to remove suspended cells. Fresh culture medium was then added, and the cells were incubated. Imaging assessments were conducted at 0, 24, 48, and 72 hr.
2.6. Annexin V apoptosis assay
Data analysis was performed using the fluorescein isothiocyanate (FITC) Annexin V apoptosis detection kit (BD Biosciences, USA) following the manufacturer’s instructions. In summary, 1 × 106 cells were detached from the flask, washed with phosphate‑buffered saline, and resuspended in 1× binding buffer. Subsequently, 5 µL of Annexin V and 5 µL of propidium iodide (PI) were added according to the manufacturer’s guidelines. After incubation in the dark at room temperature, the percentage of apoptotic cells was assessed using a Becton Dickinson FACSCalibur flow cytometer (BD Biosciences, USA). The experiments were performed in triplicate.
2.7. Gene expression analysis
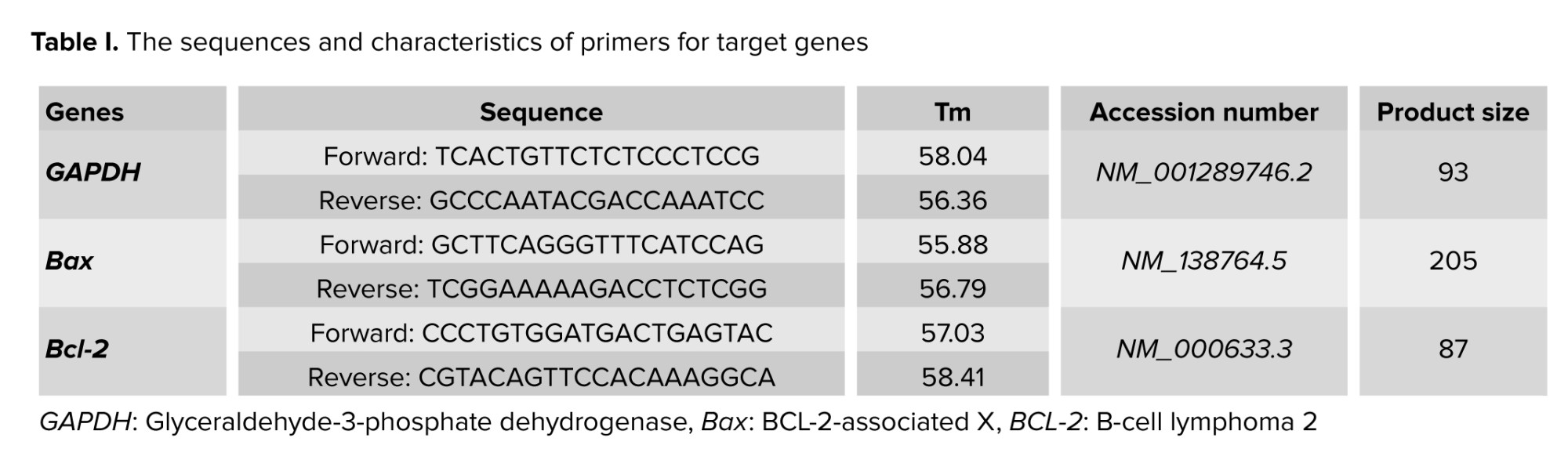
Total RNA was extracted using the RNeasy mini kit (Qiagen, Germany), and its concentration was determined with a NanoDrop instrument (Thermo Fisher Scientific, USA). Subsequently, cDNA synthesis was performed according to the manufacturer’s instructions using the Sambio™ cDNA synthesis kit. Real-time polymerase chain reaction (PCR) was conducted using equipment from Applied Biosystems (CA, USA) to measure intracellular mRNA levels, with glyceraldehyde-3-phosphate dehydrogenase serving as the endogenous control. Fluorescence data obtained during real-time PCR were analyzed using the 2^–ΔΔCt formula. Primer sequences and characteristics for the target genes are summarized in table I.
2.8. Ethical Considerations
This experimental study was conducted at the Infertility Treatment Center of ACECR, Qom, following ethical approval in 2023. Ethical approval was obtained from the ethics committee of Islamic Azad University, Qom Branch, Qom, Iran (Code: IR.IAU.QOM.REC.1402.208). Written informed consent was obtained from all participants, prior to the study.
2.9. Statistical Analysis
The sample size was determined based on previous literature studies. Statistical power analysis indicated that with this volume per participant, we could achieve approximately 80% power at a significance level of α = 0.05 to detect clinically relevant differences between the endometriosis and control groups, considering potential variability in sample quality and an anticipated dropout rate of about 10%. This volume was sufficient for the planned analyses, including scratch assay, real-time PCR, and flow cytometry, enabling the successful isolation and characterization of MenSCs. The results were presented as mean ± standard deviation and evaluated using analysis of variance (ANOVA) followed by Tukey’s post-hoc test for multiple comparisons. Statistical significance was considered at p ≤ 0.05. To ensure technical and biological reproducibility, all experiments were performed in triplicate.
3. Results
Women without endometriosis were designated as donors for CM preparation (n=3); these healthy women were recruited from individuals whose husbands presented with male factor infertility at the center. women with stage III-IV endometriosis (n=3) were recruited as donors for MenSC isolation. The MenSCs obtained from these patients were used as the primary cells subjected to the experimental treatments. All individuals in both groups underwent laparoscopy for infertility (donor group) and pelvic pain (endometriosis group), respectively, for the collection of tissue sample.
The inclusion criteria were:
1. No history of malignancy or autoimmune disease.
2. No hormone treatment for at least 3 months prior to the study.
3. No prior surgical interventions in women diagnosed with endometriosis.
4. Confirmation of deep endometriosis via transvaginal ultrasound and magnetic resonance imaging.
2.2. MenSCs isolation and primary culture
About 2 mL of menstrual blood was collected using an intrauterine insemination catheter on days 2 and 3 of the menstrual cycle from infertile women aged between 25 and 35 yr. Mononuclear cells were separated, and the cell pellet was resuspended in Dulbecco’s modified Eagle’s (DMEM) low glucose (LG) medium (Invitrogen, Life Technologies, Germany) supplemented with 100 mg/mL penicillin-streptomycin (Gibco, USA) and 10% fetal bovine serum (FBS) (Gibco, USA). Cells were seeded in a T25 flask and incubated at 37°C in a humidified atmosphere with 5% carbon dioxide. The medium was replaced after 24 hr to remove detached cells, followed by regular exchange of culture medium every 3 days.
2.3. Immunophenotyping analysis of MenSCs
To confirm the identity of the isolated cells as MenSCs, the expression of cell surface markers was analyzed using flow cytometry. After 3 passages, cells were evaluated for the presence of positive markers (CD29, CD44, CD73, and CD105) and the absence of negative markers (CD34, CD38, and CD45). Flow cytometry was performed using an FC500 flow cytometer (Beckman Coulter, Fullerton, USA). Monoclonal antibodies for flow cytometry were obtained from BioLegend Co. (USA) and used according to the manufacturer’s recommendations. Data acquisition and analysis were performed using Beckman Coulter CXP software.
2.4. Preparation of CM
Following the isolation of ADSCs and MenSCs, the cells were transferred to separate T25 flasks, each containing DMEM culture medium with 10% FBS. At the 3rd passage, after 3 days ofincubation, both ADSCs and MenSCs achieved approximately 80% confluence. The culture medium from each flask was then collected separately, centrifuged at 600 g for 5 min, and the resulting supernatant was passed through a 0.22 µm syringe filter and stored at -80°C for future experimentation.
2.5. Scratch assay
In passage 3, MenSCs derived from women with endometriosis were cultured in 6-well plates. After 24 hr of incubation under standard conditions, the control group's culture medium was replaced with DMEM-LG medium supplemented with 10% FBS. 2 treatment groups were established: one receiving CM from ADSCs, and another receiving CM from MenSCs derived from healthy women. All groups were incubated for an additional 72 hr. To evaluate the impact of CM on wound closure, a scratch was made in each cell monolayer using a pipette tip, followed by washing with phosphate‑buffered saline to remove suspended cells. Fresh culture medium was then added, and the cells were incubated. Imaging assessments were conducted at 0, 24, 48, and 72 hr.
2.6. Annexin V apoptosis assay
Data analysis was performed using the fluorescein isothiocyanate (FITC) Annexin V apoptosis detection kit (BD Biosciences, USA) following the manufacturer’s instructions. In summary, 1 × 106 cells were detached from the flask, washed with phosphate‑buffered saline, and resuspended in 1× binding buffer. Subsequently, 5 µL of Annexin V and 5 µL of propidium iodide (PI) were added according to the manufacturer’s guidelines. After incubation in the dark at room temperature, the percentage of apoptotic cells was assessed using a Becton Dickinson FACSCalibur flow cytometer (BD Biosciences, USA). The experiments were performed in triplicate.
2.7. Gene expression analysis
Total RNA was extracted using the RNeasy mini kit (Qiagen, Germany), and its concentration was determined with a NanoDrop instrument (Thermo Fisher Scientific, USA). Subsequently, cDNA synthesis was performed according to the manufacturer’s instructions using the Sambio™ cDNA synthesis kit. Real-time polymerase chain reaction (PCR) was conducted using equipment from Applied Biosystems (CA, USA) to measure intracellular mRNA levels, with glyceraldehyde-3-phosphate dehydrogenase serving as the endogenous control. Fluorescence data obtained during real-time PCR were analyzed using the 2^–ΔΔCt formula. Primer sequences and characteristics for the target genes are summarized in table I.

2.8. Ethical Considerations
This experimental study was conducted at the Infertility Treatment Center of ACECR, Qom, following ethical approval in 2023. Ethical approval was obtained from the ethics committee of Islamic Azad University, Qom Branch, Qom, Iran (Code: IR.IAU.QOM.REC.1402.208). Written informed consent was obtained from all participants, prior to the study.
2.9. Statistical Analysis
The sample size was determined based on previous literature studies. Statistical power analysis indicated that with this volume per participant, we could achieve approximately 80% power at a significance level of α = 0.05 to detect clinically relevant differences between the endometriosis and control groups, considering potential variability in sample quality and an anticipated dropout rate of about 10%. This volume was sufficient for the planned analyses, including scratch assay, real-time PCR, and flow cytometry, enabling the successful isolation and characterization of MenSCs. The results were presented as mean ± standard deviation and evaluated using analysis of variance (ANOVA) followed by Tukey’s post-hoc test for multiple comparisons. Statistical significance was considered at p ≤ 0.05. To ensure technical and biological reproducibility, all experiments were performed in triplicate.
3. Results
3.1. MenSCs identification
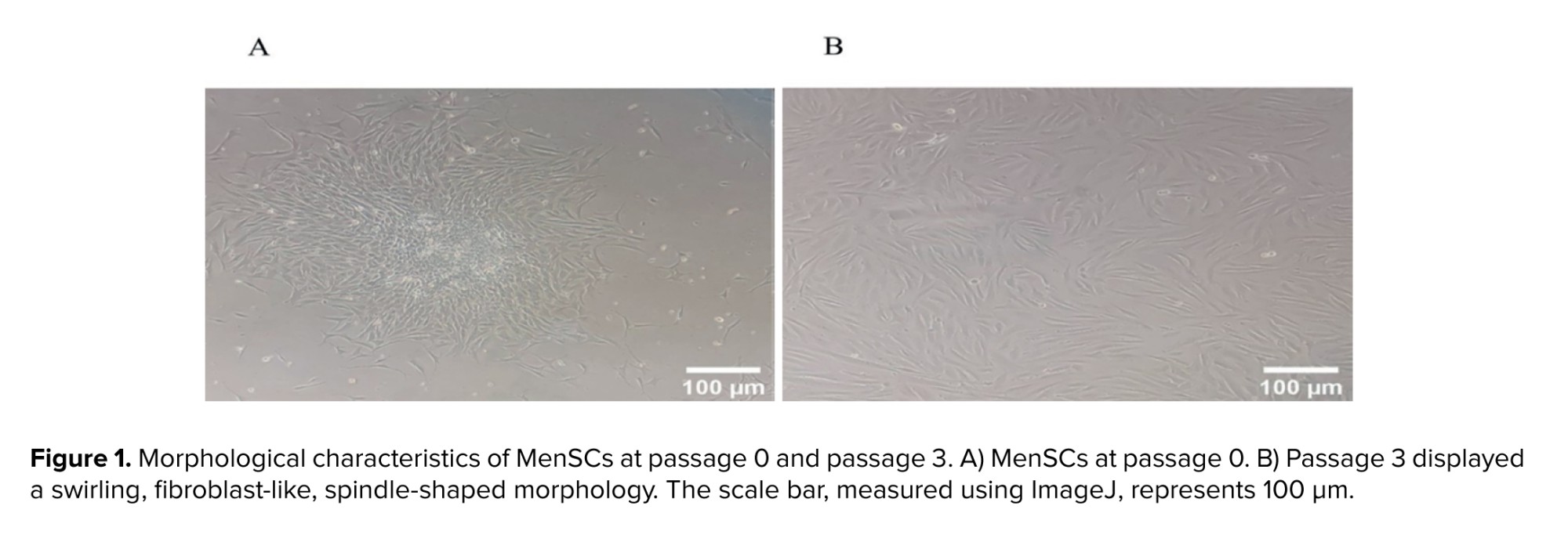
Both primary and passaged cells exhibited a typical fibroblast-like spindle-shaped morphology, confirming the successful isolation of MenSCs (Figure 1a, b). Furthermore, flow cytometric analysis indicated that the isolated cells expressed the specific markers associated with MenSCs, differentiating them from other cell types. The flow cytometry results showed that MenSCs were positive for CD29, CD44, CD73, and CD105. Conversely, these cells did not express hematopoietic markers, as evidenced by the absence of CD34, CD38, and CD45 (Figure 2).
3.2. Scratch assay showing reduced migration
Following treatment with CM, we investigated the effect of different media on the migration of MenSCs from women with endometriosis. 3 experimental groups were analyzed: one group of MenSCs from women with endometriosis treated with ADSCs-CM, another group treated with MenSCs-CM from healthy women, and a control group receiving standard cell culture medium (DMEM-LG) supplemented with 10% FBS. A significant difference was observed in migration of MenSCs regarding initial scratch width at time points 24, 48, and 72 hr post-scratch. Both the ADSCs-CM and MenSCs-CM groups exhibited a significant decrease in the scratch area compared to the control group. These findings indicate that both conditioning media effectively inhibited the migration of MenSCs in women with endometriosis. Results of the scratch assay are summarized in figure 3.
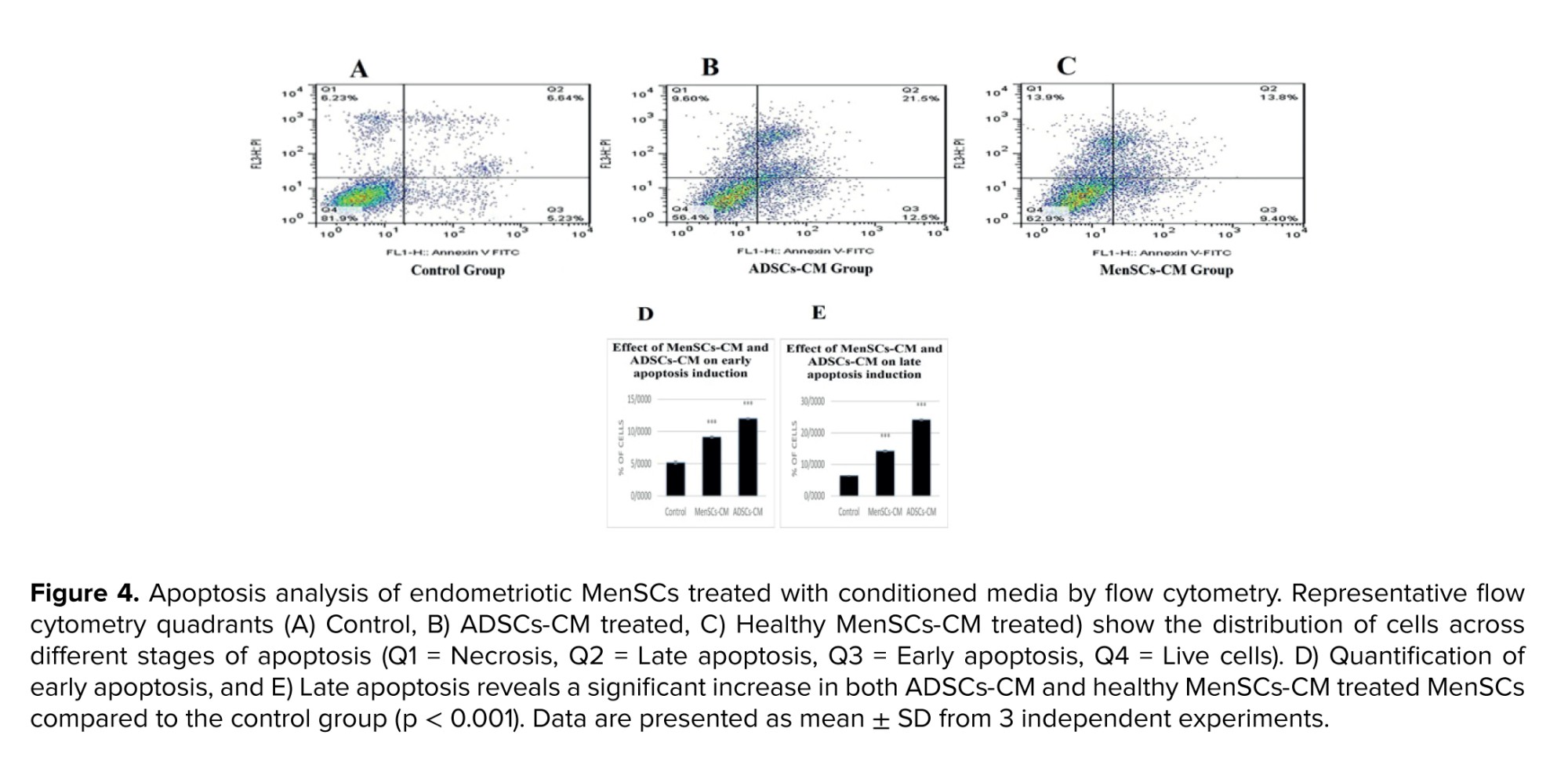
3.3. Flow cytometry analysis of apoptosis after CM treatments
Following treatment with ADSCs-CM and healthy MenSCs-CM, late apoptosis and early apoptosis significantly increased in comparison with the control group (Figure 4).
Following treatment with ADSCs-CM and healthy MenSCs-CM, late apoptosis and early apoptosis significantly increased in comparison with the control group (Figure 4).
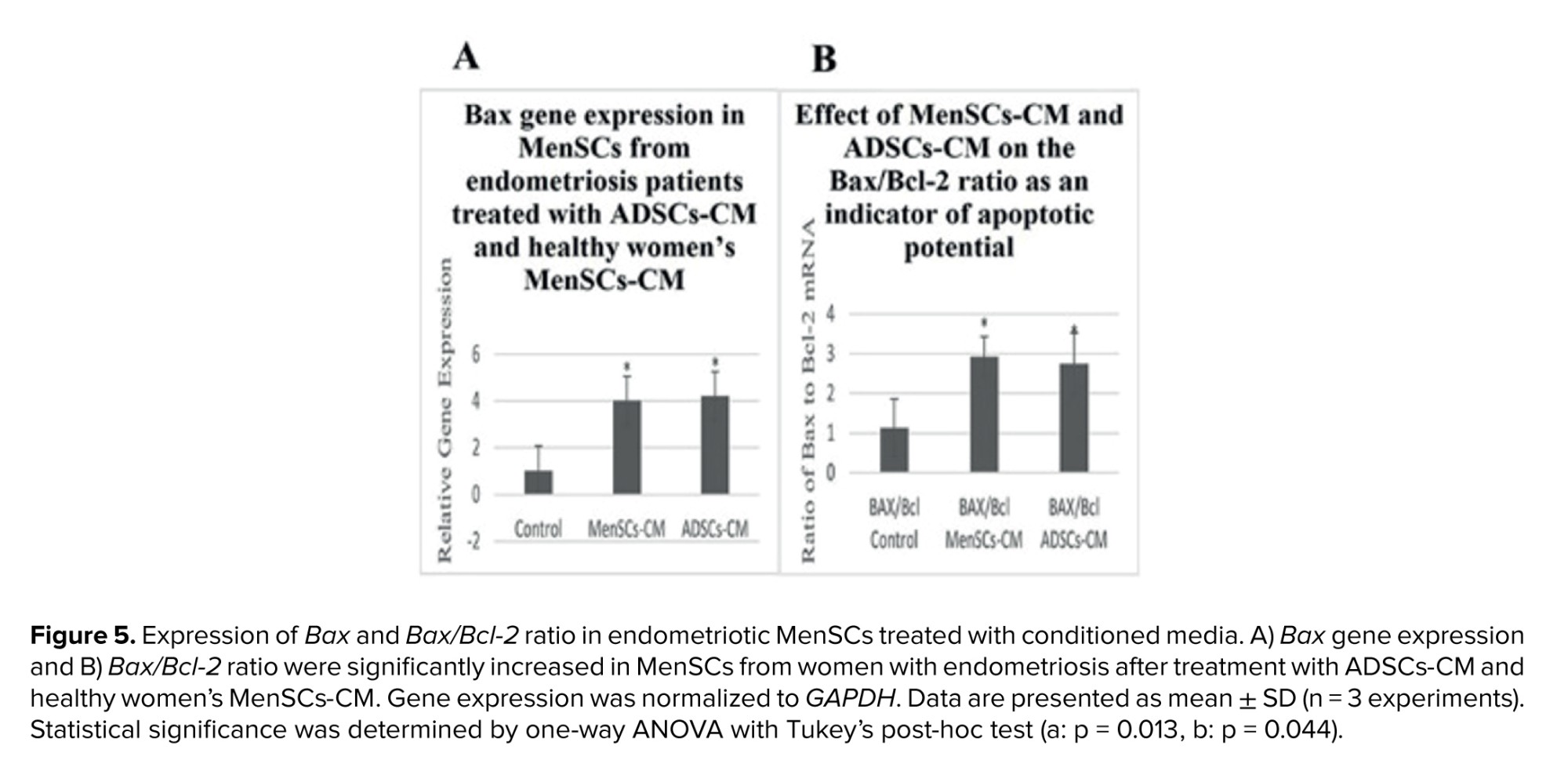
3.4. Bax expression and Bax/Bcl-2 ratio after CM treatments
The mRNA expression of Bax was significantly increased (p < 0.05) in MenSCs treated with ADSCs-CM and healthy women’s MenSCs-CM compared to untreated MenSCs. Additionally, a significant increase (p < 0.05) was observed in Bax/Bcl-2 ratio of the treated group (Figure 5).
4. Discussion
4.1. Pathophysiology of endometriosis and therapeutic potential of MSC-derived CM
Endometriosis is characterized by the benign proliferation of functioning endometrial glands and occurs in areas where endometrial tissue is located outside the uterine cavity (14). Among the several theories proposed to explain the origin of endometriosis, the most widely accepted is the retrograde menstruation-based implantation theory. This concept states that endometrial fragments carried through the fallopian tubes during backward menstrual flow can adhere and develop into ectopic lesions. It further suggests that the cellular and molecular components of menstrual blood, including the functional properties of MenSCs, play critical roles in initiating and sustaining these ectopic endometrial lesions (15). Accumulating evidence indicates that impaired regulation of apoptosis plays a pivotal role in the pathogenesis of endometriosis. In this context, eutopic endometrium of affected women shows increased expression of antiapoptotic factors and reduced expression of pro‑apoptotic mediators compared with healthy controls (16). Despite these advances in understanding disease mechanisms, current medical therapies primarily focus on estrogen suppression or symptom management rather than definitive disease eradication, and high recurrence rates persist following both medical and surgical interventions (17). Accordingly, stem cell-based therapies hold promise as versatile, cell-free approaches suitable for large groups of individuals. The CM consists of growth stem cells, and a complex mixture of secreted cytokines, growth factors, and other bioactive molecules. Among its key components are exosomes, nanosized (30-100 nm) extracellular vesicles formed via fusion of multivesicular bodies with the plasma membrane. These vesicles carry cell-specific cargos of proteins, lipids, and genetic material, and can be selectively internalized by nearby or distant cells, reprogramming recipient cells through their bioactive compounds (18). Exosomes are capable of conveying microRNAs that can inhibit cellular invasion and migration, thereby contributing to the therapeutic potential of CM in disease contexts such as endometriosis (19).
4.2. Future directions and therapeutic implications of conditioned media in endometriosis
Studies show that apoptosis in the eutopic endometrium of women with endometriosis is lower than that observed in non-endometriotic individuals. Furthermore, the ectopic endometrium of women with endometriosis exhibits reduced apoptosis compared to their own eutopic tissue. In addition, early investigations have reported inconsistent findings regarding, differences in gene expression of the anti-apoptotic Bcl-2 and the pro‑apoptotic Bax between normal endometrial tissue and eutopic and/or ectopic endometrium in women with endometriosis (20). This suggests that endometriosis-derived MenSCs might exhibit increased viability and survival compared to those from healthy women. This imbalance further highlights the distinct biological characteristics of MenSCs obtained from these 2 populations. The contrasting apoptotic behaviors of MenSCs derived from healthy women and those with endometriosis provide a strong rationale for comparing the effects of both ADSCs-CM and MenSCs-CM on these cells.
4.3. Annexin V-FITC/PI, Bax expression, Bax/Bcl-2 ratio, and migration analyses reveal the therapeutic potential of CM in endometriosis
Apoptosis in MenSCs from women with endometriosis was assessed using Annexin V-FITC/PI staining. Treatment with ADSCs-CM and MenSCs-CM from healthy donors significantly increased apoptotic cell populations compared with untreated controls, confirming the pro-apoptotic effect of both conditioned media. To investigate the molecular basis of this effect, mRNA expression of Bax and the Bax/Bcl-2 ratio were measured.
Both parameters showed marked elevations following CM treatment. In addition to apoptosis, the present study examined the migration of endometriotic cells. Although endometriosis is a benign condition, it shares cancer-like traits such as enhanced cellular migration (21). Liu et al. demonstrated that ectopic endometrial stem cells possess superior migratory capacity compared with eutopic cells from healthy donors (22). In our scratch assay experiments, both ADSC‑CM and healthy MenSC-CM treatments significantly reduced migration relative to controls, indicating that CM not only triggers apoptosis but also suppresses migratory potential in endometriotic MenSCs.
These findings underscore the multifaceted therapeutic potential of CM in targeting key pathogenic processes of endometriosis. Nevertheless, translating these promising in vitro outcomes into clinically applicable interventions requires careful consideration of several limitations. Despite the promising nature of our findings, it is imperative to acknowledge certain limitations inherent in our study. The experimental design was primarily in vitro and may not fully recapitulate the complex biological and immunological milieu of the in vivo environment. Additionally, the long-term stability of the bioactive components within CM under physiological conditions remains largely uncharacterized.
This uncertainty raises valid concerns regarding the potential for diminished therapeutic potency following in vivo administration. Future research should meticulously investigate dose-response relationships, aim to isolate and identify the key active constituents responsible for the observed effects (e.g., specific proteins, miRNAs, or lipids), and conduct direct comparative analyses between MenSCs and ADSCs concerning their distinct secretory profiles.
5. Conclusion
In conclusion, treatment with ADSCs-CM and MenSCs-CM is effective in decreasing the progression of endometriosis by promoting apoptosis and reducing migration, which inhibits invasiveness in endometriosis. However, to precisely elucidate the effects of ADSCs-CM and MenSCs-CM on the pathophysiology of endometriosis, additional research is necessary. This study will advance the field of endometriosis research by identifying which source of MSC therapy is most effective for treating the condition, thereby serving as a foundation for future investigations. Additionally, our findings support Samson’s theory and highlight the potential contribution of MenSCs to the pathogenesis of endometriosis, offering further evidence for both the retrograde menstruation and stem cell theories. To further substantiate these findings, future studies should employ Western blotting to evaluate Bax, Bcl-2, caspase-3, and caspase-7 protein levels and activation states, thereby providing deeper mechanistic insights into the balance of pro- and anti-apoptotic signals.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
Both authors designed the study and conducted the research. Also monitored, evaluated, and analyzed the results of the study. Further, they reviewed the article. Both authors approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgments
The authors gratefully acknowledge all volunteers who generously provided samples for this study. We also extend sincere thanks to the gynecology specialists and laboratory staff for their valuable assistance in sample collection and processing. This study did not receive any specific financial support. The authors also acknowledge the assistance of Writefull (Amsterdam, The Netherlands) in enhancing the clarity and readability of the manuscript during its preparation.
Conflict of Interest
The authors declare that they have no conflict of interest.
The mRNA expression of Bax was significantly increased (p < 0.05) in MenSCs treated with ADSCs-CM and healthy women’s MenSCs-CM compared to untreated MenSCs. Additionally, a significant increase (p < 0.05) was observed in Bax/Bcl-2 ratio of the treated group (Figure 5).





4. Discussion
4.1. Pathophysiology of endometriosis and therapeutic potential of MSC-derived CM
Endometriosis is characterized by the benign proliferation of functioning endometrial glands and occurs in areas where endometrial tissue is located outside the uterine cavity (14). Among the several theories proposed to explain the origin of endometriosis, the most widely accepted is the retrograde menstruation-based implantation theory. This concept states that endometrial fragments carried through the fallopian tubes during backward menstrual flow can adhere and develop into ectopic lesions. It further suggests that the cellular and molecular components of menstrual blood, including the functional properties of MenSCs, play critical roles in initiating and sustaining these ectopic endometrial lesions (15). Accumulating evidence indicates that impaired regulation of apoptosis plays a pivotal role in the pathogenesis of endometriosis. In this context, eutopic endometrium of affected women shows increased expression of antiapoptotic factors and reduced expression of pro‑apoptotic mediators compared with healthy controls (16). Despite these advances in understanding disease mechanisms, current medical therapies primarily focus on estrogen suppression or symptom management rather than definitive disease eradication, and high recurrence rates persist following both medical and surgical interventions (17). Accordingly, stem cell-based therapies hold promise as versatile, cell-free approaches suitable for large groups of individuals. The CM consists of growth stem cells, and a complex mixture of secreted cytokines, growth factors, and other bioactive molecules. Among its key components are exosomes, nanosized (30-100 nm) extracellular vesicles formed via fusion of multivesicular bodies with the plasma membrane. These vesicles carry cell-specific cargos of proteins, lipids, and genetic material, and can be selectively internalized by nearby or distant cells, reprogramming recipient cells through their bioactive compounds (18). Exosomes are capable of conveying microRNAs that can inhibit cellular invasion and migration, thereby contributing to the therapeutic potential of CM in disease contexts such as endometriosis (19).
4.2. Future directions and therapeutic implications of conditioned media in endometriosis
Studies show that apoptosis in the eutopic endometrium of women with endometriosis is lower than that observed in non-endometriotic individuals. Furthermore, the ectopic endometrium of women with endometriosis exhibits reduced apoptosis compared to their own eutopic tissue. In addition, early investigations have reported inconsistent findings regarding, differences in gene expression of the anti-apoptotic Bcl-2 and the pro‑apoptotic Bax between normal endometrial tissue and eutopic and/or ectopic endometrium in women with endometriosis (20). This suggests that endometriosis-derived MenSCs might exhibit increased viability and survival compared to those from healthy women. This imbalance further highlights the distinct biological characteristics of MenSCs obtained from these 2 populations. The contrasting apoptotic behaviors of MenSCs derived from healthy women and those with endometriosis provide a strong rationale for comparing the effects of both ADSCs-CM and MenSCs-CM on these cells.
4.3. Annexin V-FITC/PI, Bax expression, Bax/Bcl-2 ratio, and migration analyses reveal the therapeutic potential of CM in endometriosis
Apoptosis in MenSCs from women with endometriosis was assessed using Annexin V-FITC/PI staining. Treatment with ADSCs-CM and MenSCs-CM from healthy donors significantly increased apoptotic cell populations compared with untreated controls, confirming the pro-apoptotic effect of both conditioned media. To investigate the molecular basis of this effect, mRNA expression of Bax and the Bax/Bcl-2 ratio were measured.
Both parameters showed marked elevations following CM treatment. In addition to apoptosis, the present study examined the migration of endometriotic cells. Although endometriosis is a benign condition, it shares cancer-like traits such as enhanced cellular migration (21). Liu et al. demonstrated that ectopic endometrial stem cells possess superior migratory capacity compared with eutopic cells from healthy donors (22). In our scratch assay experiments, both ADSC‑CM and healthy MenSC-CM treatments significantly reduced migration relative to controls, indicating that CM not only triggers apoptosis but also suppresses migratory potential in endometriotic MenSCs.
These findings underscore the multifaceted therapeutic potential of CM in targeting key pathogenic processes of endometriosis. Nevertheless, translating these promising in vitro outcomes into clinically applicable interventions requires careful consideration of several limitations. Despite the promising nature of our findings, it is imperative to acknowledge certain limitations inherent in our study. The experimental design was primarily in vitro and may not fully recapitulate the complex biological and immunological milieu of the in vivo environment. Additionally, the long-term stability of the bioactive components within CM under physiological conditions remains largely uncharacterized.
This uncertainty raises valid concerns regarding the potential for diminished therapeutic potency following in vivo administration. Future research should meticulously investigate dose-response relationships, aim to isolate and identify the key active constituents responsible for the observed effects (e.g., specific proteins, miRNAs, or lipids), and conduct direct comparative analyses between MenSCs and ADSCs concerning their distinct secretory profiles.
5. Conclusion
In conclusion, treatment with ADSCs-CM and MenSCs-CM is effective in decreasing the progression of endometriosis by promoting apoptosis and reducing migration, which inhibits invasiveness in endometriosis. However, to precisely elucidate the effects of ADSCs-CM and MenSCs-CM on the pathophysiology of endometriosis, additional research is necessary. This study will advance the field of endometriosis research by identifying which source of MSC therapy is most effective for treating the condition, thereby serving as a foundation for future investigations. Additionally, our findings support Samson’s theory and highlight the potential contribution of MenSCs to the pathogenesis of endometriosis, offering further evidence for both the retrograde menstruation and stem cell theories. To further substantiate these findings, future studies should employ Western blotting to evaluate Bax, Bcl-2, caspase-3, and caspase-7 protein levels and activation states, thereby providing deeper mechanistic insights into the balance of pro- and anti-apoptotic signals.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
Both authors designed the study and conducted the research. Also monitored, evaluated, and analyzed the results of the study. Further, they reviewed the article. Both authors approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgments
The authors gratefully acknowledge all volunteers who generously provided samples for this study. We also extend sincere thanks to the gynecology specialists and laboratory staff for their valuable assistance in sample collection and processing. This study did not receive any specific financial support. The authors also acknowledge the assistance of Writefull (Amsterdam, The Netherlands) in enhancing the clarity and readability of the manuscript during its preparation.
Conflict of Interest
The authors declare that they have no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Genetics
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |