Fri, Apr 3, 2026
[Archive]
Volume 23, Issue 12 (December 2025)
IJRM 2025, 23(12): 995-1006 |
Back to browse issues page
Ethics code: IR.TBZMED.REC.1403.492
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Hamdi K, Khakpour S, Ghasemzadeh Darjani A, Farzadi L, Niknafs B, Navali N, et al . Comparison of frozen embryo transfer outcomes in hormonal vs. mild stimulation protocols in polycystic ovary syndrome women: A randomized controlled trial. IJRM 2025; 23 (12) :995-1006
URL: http://ijrm.ir/article-1-3612-en.html
URL: http://ijrm.ir/article-1-3612-en.html
Kobra Hamdi1 

 , Sahar Khakpour *2
, Sahar Khakpour *2 

 , Aliyeh Ghasemzadeh Darjani1
, Aliyeh Ghasemzadeh Darjani1 

 , Laya Farzadi1
, Laya Farzadi1 

 , Behrouz Niknafs3
, Behrouz Niknafs3 

 , Nazli Navali1
, Nazli Navali1 

 , Parvin Hakimi1
, Parvin Hakimi1 

 , Roghayeh Anvari Aliabad1
, Roghayeh Anvari Aliabad1 

 , Hamed Hajipour4
, Hamed Hajipour4 




 , Sahar Khakpour *2
, Sahar Khakpour *2 

 , Aliyeh Ghasemzadeh Darjani1
, Aliyeh Ghasemzadeh Darjani1 

 , Laya Farzadi1
, Laya Farzadi1 

 , Behrouz Niknafs3
, Behrouz Niknafs3 

 , Nazli Navali1
, Nazli Navali1 

 , Parvin Hakimi1
, Parvin Hakimi1 

 , Roghayeh Anvari Aliabad1
, Roghayeh Anvari Aliabad1 

 , Hamed Hajipour4
, Hamed Hajipour4 


1- Women's Reproductive Health Research Center, Tabriz University of Medical Sciences, Tabriz, Iran.
2- Women's Reproductive Health Research Center, Tabriz University of Medical Sciences, Tabriz, Iran. ,Skh55992@gmail.com; saharkhakpour@tbzmed.ac.ir
3- Department of Anatomical Sciences, School of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran.
4- School of Advanced Medical Sciences, Tabriz University of Medical Sciences, Tabriz, Iran.
2- Women's Reproductive Health Research Center, Tabriz University of Medical Sciences, Tabriz, Iran. ,
3- Department of Anatomical Sciences, School of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran.
4- School of Advanced Medical Sciences, Tabriz University of Medical Sciences, Tabriz, Iran.
Keywords: Polycystic ovary syndrome, Frozen embryo, Endometrium, Hormonal therapy, Mild ovarian stimulation protocol.
Full-Text [PDF 438 kb]
(397 Downloads)
| Abstract (HTML) (156 Views)
2.8. Outcomes and data collection
Full-Text: (15 Views)
1. Introduction
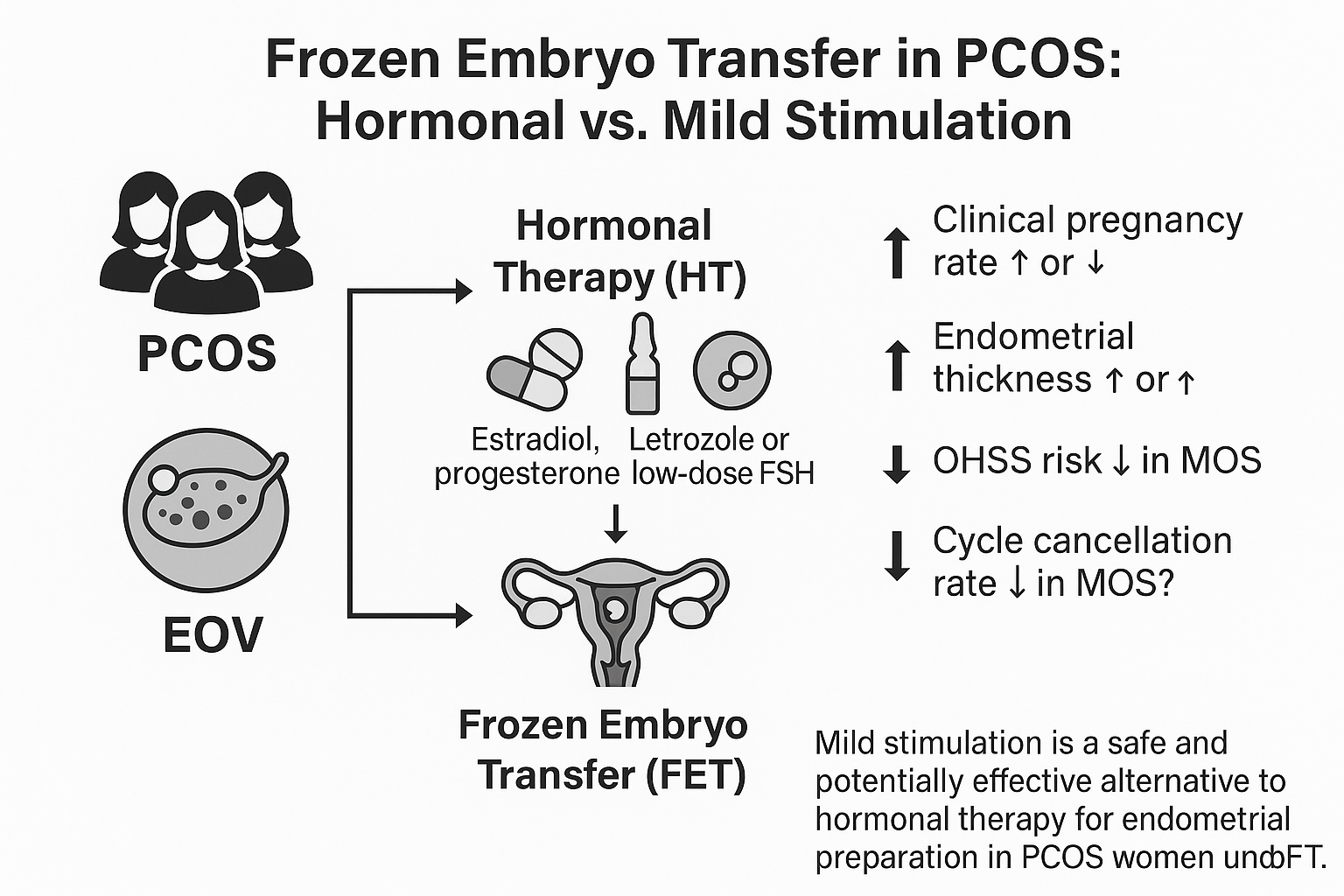
Polycystic ovary syndrome (PCOS) is the most common endocrine disorder among women of reproductive age, with a global prevalence of 20% and 11% in Iran, and accounts for 70% of anovulatory infertility (1). Infertility, defined as the inability to conceive after 1 yr of unprotected intercourse, affects over 80 million people worldwide (2). Assisted reproductive technologies, including in vitro fertilization and frozen embryo transfer (FET), have been developed to address this issue (2, 3). FET reduces the risk of multiple pregnancies and ovarian hyperstimulation syndrome by transferring fewer embryos and allowing multiple transfer cycles without additional ovarian stimulation (3).
Successful pregnancy outcomes in assisted reproductive technologies depend on embryo quality, endometrial receptivity, and proper synchronization between the embryo and endometrium, with success rates largely influenced by endometrial preparation protocols (4). In women with PCOS, endometrial preparation for FET typically involves hormone replacement therapy (HRT) or mild ovarian stimulation (MOS) (3, 5). HRT cycles use estrogen followed by progesterone to prepare the endometrium, offering advantages like less monitoring and timing flexibility, but carry risks such as increased thrombosis. MOS cycles use letrozole and low-dose gonadotropins to stimulate the ovaries, followed by human chorionic gonadotropin (hCG) to trigger ovulation (6-8).
It was showed that in infertile women with ovulatory dysfunction, FET using a letrozole-based regimen resulted in fewer miscarriages compared to HRT (9). A retrospective study in women with PCOS also reported higher live birth rates with letrozole-based FET than with HRT (10). Similarly, in cases of unexplained infertility, letrozole-based FET was associated with improved live birth outcomes compared to HRT (11). While clinical pregnancy rates were similar between letrozole and HRT groups, live birth rates were consistently higher in the letrozole group (12). However, some studies found no significant difference in clinical pregnancy rates or endometrial thickness between the 2 protocols in women with PCOS (13, 14). Estrogen use in HRT increases the risk of thrombosis and thromboembolism (15, 16), which has led national guidelines to recommend MOS protocols as a preventive measure. On a molecular level, letrozole upregulates markers such as leukemia inhibitory factor, dickkopf-related protein 1, leukemia inhibitory factor receptor, and fibroblast growth factor 22 in PCOS participants, which are linked to improved endometrial receptivity and lower miscarriage rates (17).
Additionally, letrozole enhances the expression of integrin ανβ3 in the endometrium, further promoting receptivity and pregnancy success (18). Additionally, studies have shown that the lack of corpus luteum formation is associated with an increased risk of pregnancy complications such as pregnancy-induced hypertension, and human menopausal gonadotropin treatment promotes ovulation and corpus luteum development (19).
This randomized controlled trial uniquely compares 2 endometrial preparation protocols, HRT and MOS, in women with PCOS. It investigates biochemical, clinical, and final pregnancy outcomes, as well as miscarriage within 12th week. Grounded in mechanistic evidence, the study explores how letrozole‑induced upregulation of endometrial αvβ3 integrin and corpus luteum formation may enhance receptivity and reduce pregnancy complications. By addressing existing data gaps, provides new clinical insight into optimizing endometrial preparation in PCOS.
2. Materials and Methods
2.1. Study design
Successful pregnancy outcomes in assisted reproductive technologies depend on embryo quality, endometrial receptivity, and proper synchronization between the embryo and endometrium, with success rates largely influenced by endometrial preparation protocols (4). In women with PCOS, endometrial preparation for FET typically involves hormone replacement therapy (HRT) or mild ovarian stimulation (MOS) (3, 5). HRT cycles use estrogen followed by progesterone to prepare the endometrium, offering advantages like less monitoring and timing flexibility, but carry risks such as increased thrombosis. MOS cycles use letrozole and low-dose gonadotropins to stimulate the ovaries, followed by human chorionic gonadotropin (hCG) to trigger ovulation (6-8).
It was showed that in infertile women with ovulatory dysfunction, FET using a letrozole-based regimen resulted in fewer miscarriages compared to HRT (9). A retrospective study in women with PCOS also reported higher live birth rates with letrozole-based FET than with HRT (10). Similarly, in cases of unexplained infertility, letrozole-based FET was associated with improved live birth outcomes compared to HRT (11). While clinical pregnancy rates were similar between letrozole and HRT groups, live birth rates were consistently higher in the letrozole group (12). However, some studies found no significant difference in clinical pregnancy rates or endometrial thickness between the 2 protocols in women with PCOS (13, 14). Estrogen use in HRT increases the risk of thrombosis and thromboembolism (15, 16), which has led national guidelines to recommend MOS protocols as a preventive measure. On a molecular level, letrozole upregulates markers such as leukemia inhibitory factor, dickkopf-related protein 1, leukemia inhibitory factor receptor, and fibroblast growth factor 22 in PCOS participants, which are linked to improved endometrial receptivity and lower miscarriage rates (17).
Additionally, letrozole enhances the expression of integrin ανβ3 in the endometrium, further promoting receptivity and pregnancy success (18). Additionally, studies have shown that the lack of corpus luteum formation is associated with an increased risk of pregnancy complications such as pregnancy-induced hypertension, and human menopausal gonadotropin treatment promotes ovulation and corpus luteum development (19).
This randomized controlled trial uniquely compares 2 endometrial preparation protocols, HRT and MOS, in women with PCOS. It investigates biochemical, clinical, and final pregnancy outcomes, as well as miscarriage within 12th week. Grounded in mechanistic evidence, the study explores how letrozole‑induced upregulation of endometrial αvβ3 integrin and corpus luteum formation may enhance receptivity and reduce pregnancy complications. By addressing existing data gaps, provides new clinical insight into optimizing endometrial preparation in PCOS.
2. Materials and Methods
2.1. Study design
A randomized clinical trial was conducted from October 2024 and 2025, involving infertile women attending the infertility clinic of Al-Zahra hospital (affiliated with Tabriz University of Medical Sciences, Tabriz, Iran), following inclusion and exclusion criteria.
2.2. Sampling
2.2. Sampling
Based on a previous study, the clinical pregnancy rates for simulated and hormone treatment methods were 47% and 25%, respectively (20). Additionally, based on another meta-analysis the odds ratio for pregnancy in the simulated method was 1.54 times higher than in the hormone treatment method (21). With an effect size of 0.30, a significance level of 5%, and a power of 80%, the minimum sample size per group was estimated to be 38, with a total of 76 participants. However, to increase the validity of the results and to prevent sample loss, 50 participants were included in each group, total 100 participants. These participants were selected using a convenience sampling method.
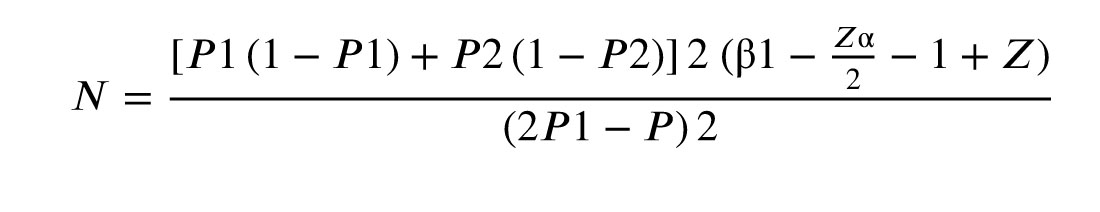
2.3. Inclusion criteria
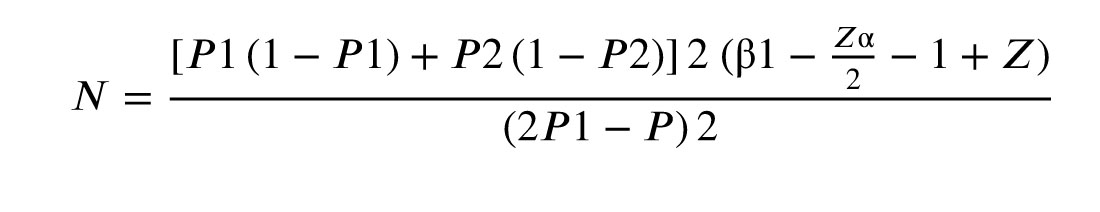
2.3. Inclusion criteria
Eligible participants for this study were infertile women aged between 20 and 40 yr with a diagnosis of PCOS (based on the Rotterdam criteria and confirmed by a specialist physician) as the underlying cause of infertility. Important exclusion criteria prior to randomization included the presence of congenital uterine anomalies, uterine fibroids, severe endometriosis, or a history of > 2 previous unsuccessful embryo transfers.
2.4. Randomization
2.4. Randomization
Participants were randomly allocated to either the HRT or MOS group using block randomization, with numbers generated by the random allocation software. The total number of groups and the sample size were entered into the software, and block randomization was then applied. Participants were assigned to either the intervention or control group based on the random numbers generated.
2.5. Blinding
2.5. Blinding
As the main intervention was performed by the first investigator, blinding was not feasible for this individual. However, the research assistant responsible for recording the outcomes and the individual analyzing the data were blinded to the study groups, ensuring a double-blind design for this study.
2.6. Characteristics
2.6. Characteristics
In this study, a comprehensive range of variables were evaluated to compare outcomes between the MOS and HRT groups. Baseline demographic characteristics included maternal and paternal age, body mass index (BMI), and the presence of consanguinity between spouses (which was recorded because genetic proximity between spouses may influence reproductive outcomes through inherited conditions or subclinical genetic factors), prior gynecologic surgeries, and history of endometriosis. Lifestyle‑related factors, such as cigarette smoking and alcohol consumption, were also recorded for each participant and/or her spouse. Reproductive history variables comprised pre-intervention HCG levels, sperm analysis results, previous embryo transfer attempts, the number of prior cycles, and the type of embryo transfer protocol used. Serum HCG was assessed only to rule out spontaneous pregnancy before cycle initiation and was not included in comparative outcome analyses. Data related to embryo transfer included the number and quality of embryos transferred. Biochemical and hormonal profiles were assessed through measurements of fasting blood sugar, triglycerides, total cholesterol, low-density lipoprotein, high-density lipoprotein, prolactin, follicle-stimulating hormone (FSH), anti-Müllerian hormone, thyroid-stimulating hormone, and vitamin D3 levels. Clinical outcomes included rates of chemical and clinical pregnancy, as well as final pregnancy outcomes such as miscarriage, ectopic pregnancy, and successful ongoing pregnancy. These variables were analyzed to determine any statistically significant differences between the 2 HRT and MOS groups.
2.7. Methods
2.7. Methods
In the HRT group, endometrial preparation was performed using estrogen and progesterone HRT. Oral estradiol 2 mg tablets (Aburaihan Pharmaceutical Co., Iran) were started on day 2 of the menstrual cycle. Initially, one tablet was taken every 12 hr on days 2 and 3, and after that, one tablet was taken every 8 hr. After 10-12 days of estradiol consumption, the next visit was performed, and a transvaginal ultrasound (Esaote-USA) was conducted to measure the endometrial thickness. If the endometrial thickness was > 7.5 mm with a trilaminar pattern, indicating the exclusive effect of estrogen without any space-occupying lesions such as polyps or fibroids, intramuscular progesterone 50 mg (Iran Hormone Co., Iran) was started (one dose on the same day, followed by 2 doses daily). On day 4 after beginning progesterone, the cleavage-stage embryo was transferred to the uterus. Luteal phase support was provided with 400 mg rectal progesterone (Iran Hormone Co., Iran) suppositories every 12 hr and 50 mg intramuscular progesterone every 3 days. If pregnancy occurred, estrogen was continued at the same dose until week 8, then gradually reduced until discontinuation at week 12. Progesterone was continued at the same dose and method until week 12.
If, on days 10-12 of the cycle, the endometrial thickness was < 7.5 mm, estradiol was continued at the same dose, or if necessary, a higher dose was used. A follow-up visit with repeat ultrasound was scheduled 3-4 days later, and this process was repeated until the endometrial thickness reached ≥ 7.5 mm. If no increase was observed in endometrial thickness after 20 days of estradiol use, the treatment cycle was terminated, and the participant was excluded from the study.
In the MOS group, endometrial preparation was performed using MOS. Letrozole 5 mg (2 tablets of 2.5 mg each, Atipharmed Pharmaceutical Co., Iran) was prescribed daily starting from day 2 or 3 of the menstrual cycle for 5 days. Then, FSH (Pars Peyvand, Iran) was administered at doses of 37.5-150 IU daily or every other day (with at least 2 doses and dose escalation if needed). When at least one follicle reached a size of > 17 mm and the endometrial thickness was > 7.5 mm (provided there were no > 2 follicles of 14-16 mm), a 5000 IU injection of HCG was given (Ronak Pharmaceutical Co., Iran). If more than 3 follicles were fully developed, the cycle was canceled to prevent ovarian hyperstimulation syndrome, and embryo transfer did not take place. The next day, 400 mg rectal progesterone suppositories were initiated, and embryo transfer was performed 5-6 days after HCG injection at the cleavage stage. Rectal progesterone continued until the pregnancy test, and if pregnancy occurred, it continued until week 12.
In both groups, only grade 1 or 2 embryos were transferred. Grade 1 embryos were defined as having blastomeres of equal size without cytoplasmic fragments, and grade 2 embryos had blastomeres of equal size with small cytoplasmic fragments. Grade 3 embryos had blastomeres of similar size with small cytoplasmic fragments, while grade 4 embryos had blastomeres of equal or unequal size with substantial cytoplasmic fragments. Finally, grade 5 embryos had blastomeres of different sizes with significant cytoplasmic fragmentation.
14 days after embryo transfer, a β-hCG test (ELISA Kit 96t, USA) was conducted, and the pregnancy results (positive or negative) were recorded and compared between the 2 groups. In participants with positive pregnancy tests, an abdominal ultrasound (Esaote-USA) was performed 2 wk later to confirm the presence of a gestational sac, its number, and the fetal heart rate. The results were compared between the groups. Pregnant women were followed up until week 12 of pregnancy for fetal growth and potential risk of miscarriage.
If, on days 10-12 of the cycle, the endometrial thickness was < 7.5 mm, estradiol was continued at the same dose, or if necessary, a higher dose was used. A follow-up visit with repeat ultrasound was scheduled 3-4 days later, and this process was repeated until the endometrial thickness reached ≥ 7.5 mm. If no increase was observed in endometrial thickness after 20 days of estradiol use, the treatment cycle was terminated, and the participant was excluded from the study.
In the MOS group, endometrial preparation was performed using MOS. Letrozole 5 mg (2 tablets of 2.5 mg each, Atipharmed Pharmaceutical Co., Iran) was prescribed daily starting from day 2 or 3 of the menstrual cycle for 5 days. Then, FSH (Pars Peyvand, Iran) was administered at doses of 37.5-150 IU daily or every other day (with at least 2 doses and dose escalation if needed). When at least one follicle reached a size of > 17 mm and the endometrial thickness was > 7.5 mm (provided there were no > 2 follicles of 14-16 mm), a 5000 IU injection of HCG was given (Ronak Pharmaceutical Co., Iran). If more than 3 follicles were fully developed, the cycle was canceled to prevent ovarian hyperstimulation syndrome, and embryo transfer did not take place. The next day, 400 mg rectal progesterone suppositories were initiated, and embryo transfer was performed 5-6 days after HCG injection at the cleavage stage. Rectal progesterone continued until the pregnancy test, and if pregnancy occurred, it continued until week 12.
In both groups, only grade 1 or 2 embryos were transferred. Grade 1 embryos were defined as having blastomeres of equal size without cytoplasmic fragments, and grade 2 embryos had blastomeres of equal size with small cytoplasmic fragments. Grade 3 embryos had blastomeres of similar size with small cytoplasmic fragments, while grade 4 embryos had blastomeres of equal or unequal size with substantial cytoplasmic fragments. Finally, grade 5 embryos had blastomeres of different sizes with significant cytoplasmic fragmentation.
14 days after embryo transfer, a β-hCG test (ELISA Kit 96t, USA) was conducted, and the pregnancy results (positive or negative) were recorded and compared between the 2 groups. In participants with positive pregnancy tests, an abdominal ultrasound (Esaote-USA) was performed 2 wk later to confirm the presence of a gestational sac, its number, and the fetal heart rate. The results were compared between the groups. Pregnant women were followed up until week 12 of pregnancy for fetal growth and potential risk of miscarriage.
2.8. Outcomes and data collection
In this study, the outcomes were classified into primary and secondary endpoints and are described in detail below:
Primary outcomes: the first primary endpoint was the occurrence of chemical pregnancy, which was measured 14 days after embryo transfer using a serum β-hCG blood test specialists (the first, third, and fourth authors), using an ELISA kit (96-well format). The second primary endpoint was the number of gestational sacs, assessed from the 5th wk of pregnancy by transvaginal ultrasound. Secondary outcome: embryonic development was evaluated from the beginning of the intervention until the end of the 12th wk of pregnancy through ultrasound examinations (Esaote, USA).
2.9. Ethical Considerations
This study was approved by the Ethics Committee of Tabriz University of Medical Sciences, Tabriz, Iran (Code: IR.TBZMED.REC.1403.492). After obtaining ethical approval, the study was registered in the Iranian Clinical Trials Registry (IRCT20130603013566N14; update in: 2025-06-11). The objectives of the study were explained to all participants and their spouses in simple and understandable language, and written informed consent was obtained. Participation in the study was voluntary, and all expenses for the participants were covered by the registered research project. The researchers adhered to all principles outlined in the Helsinki Declaration. Data confidentiality was strictly maintained throughout the study, and all participants' information was protected against unauthorized access and disclosure.
2.10. Statistical Analysis
All data were analyzed using statistical package for the social sciences version 26.0 (SPSS Inc., Chicago, USA). The Shapiro-Wilk test was used to assess the normality of data distribution, confirming that the data followed a normal distribution. Continuous variables were expressed as mean ± SD or median (range) and analyzed using the student’s t test. Categorical variables were expressed as frequency and percentage, and analyzed using the Chi-square test. A p < 0.05 was considered statistically significant for comparing the 2 groups.
3. Results
Primary outcomes: the first primary endpoint was the occurrence of chemical pregnancy, which was measured 14 days after embryo transfer using a serum β-hCG blood test specialists (the first, third, and fourth authors), using an ELISA kit (96-well format). The second primary endpoint was the number of gestational sacs, assessed from the 5th wk of pregnancy by transvaginal ultrasound. Secondary outcome: embryonic development was evaluated from the beginning of the intervention until the end of the 12th wk of pregnancy through ultrasound examinations (Esaote, USA).
2.9. Ethical Considerations
This study was approved by the Ethics Committee of Tabriz University of Medical Sciences, Tabriz, Iran (Code: IR.TBZMED.REC.1403.492). After obtaining ethical approval, the study was registered in the Iranian Clinical Trials Registry (IRCT20130603013566N14; update in: 2025-06-11). The objectives of the study were explained to all participants and their spouses in simple and understandable language, and written informed consent was obtained. Participation in the study was voluntary, and all expenses for the participants were covered by the registered research project. The researchers adhered to all principles outlined in the Helsinki Declaration. Data confidentiality was strictly maintained throughout the study, and all participants' information was protected against unauthorized access and disclosure.
2.10. Statistical Analysis
All data were analyzed using statistical package for the social sciences version 26.0 (SPSS Inc., Chicago, USA). The Shapiro-Wilk test was used to assess the normality of data distribution, confirming that the data followed a normal distribution. Continuous variables were expressed as mean ± SD or median (range) and analyzed using the student’s t test. Categorical variables were expressed as frequency and percentage, and analyzed using the Chi-square test. A p < 0.05 was considered statistically significant for comparing the 2 groups.
3. Results
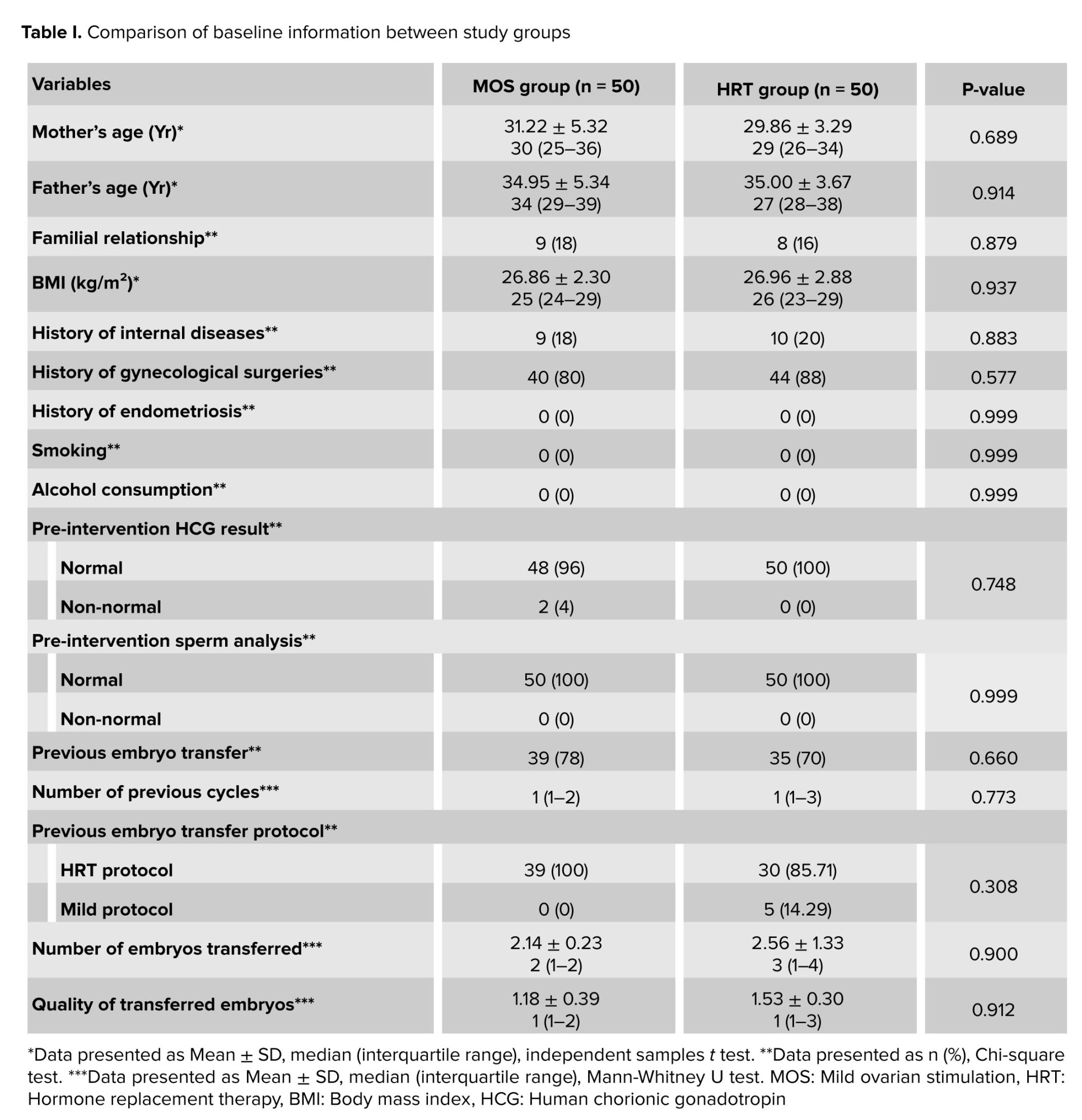
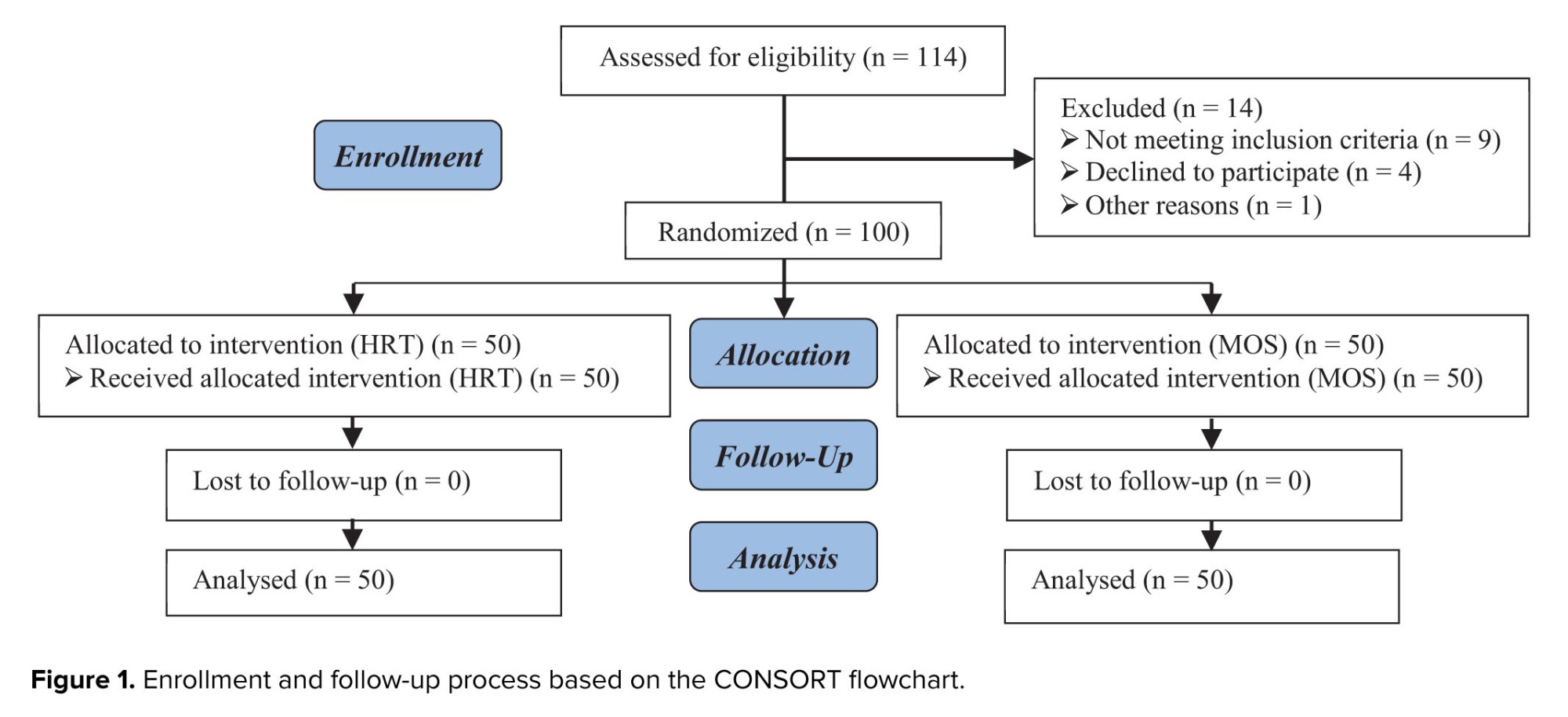
In this study, a total of 100 participants were enrolled, and all participants completed the study, with their data being analyzed. In other words, no sample attrition occurred in this study (Figure 1). We compared baseline data such as maternal age, paternal age, familial relationship between the couple, BMI, history of internal diseases, history of gynecological surgeries, smoking habits, alcohol consumption, pre-intervention HCG results, and sperm analysis between the groups. It was observed that no statistically significant differences were found in these variables (p > 0.05 for all variables). Additionally, no significant differences were observed between the groups in other factors such as pre-intervention sperm analysis results (100% normal in both groups), history of previous embryo transfer, number of prior cycles, previous embryo transfer protocol, number of embryos transferred, and the quality of transferred embryos (p > 0.05 for all variables) (Table I).
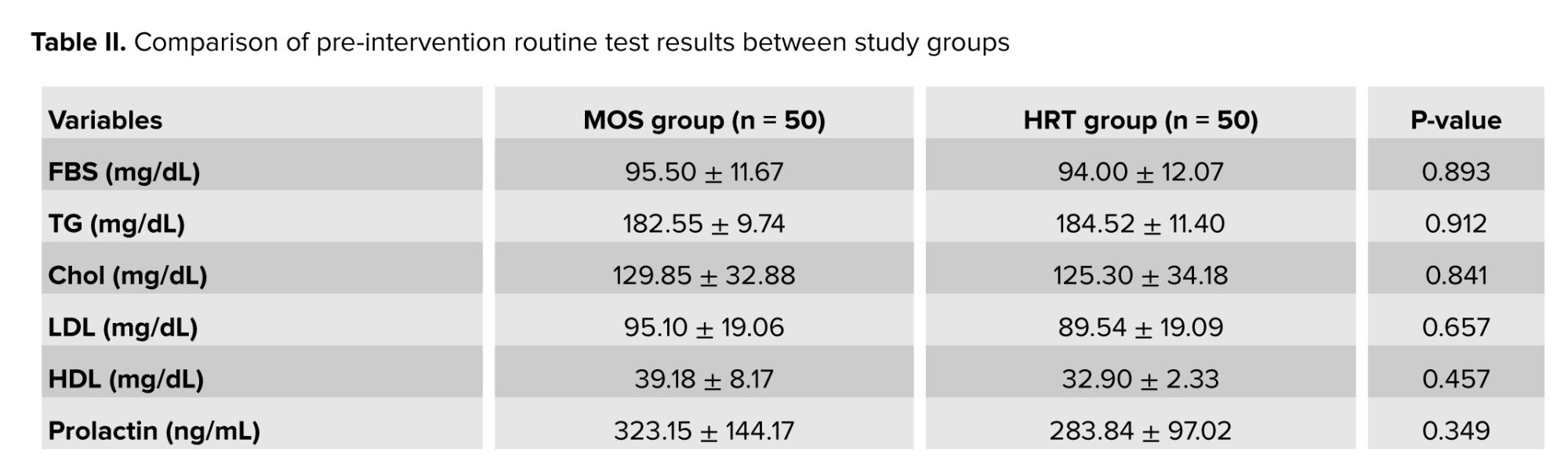
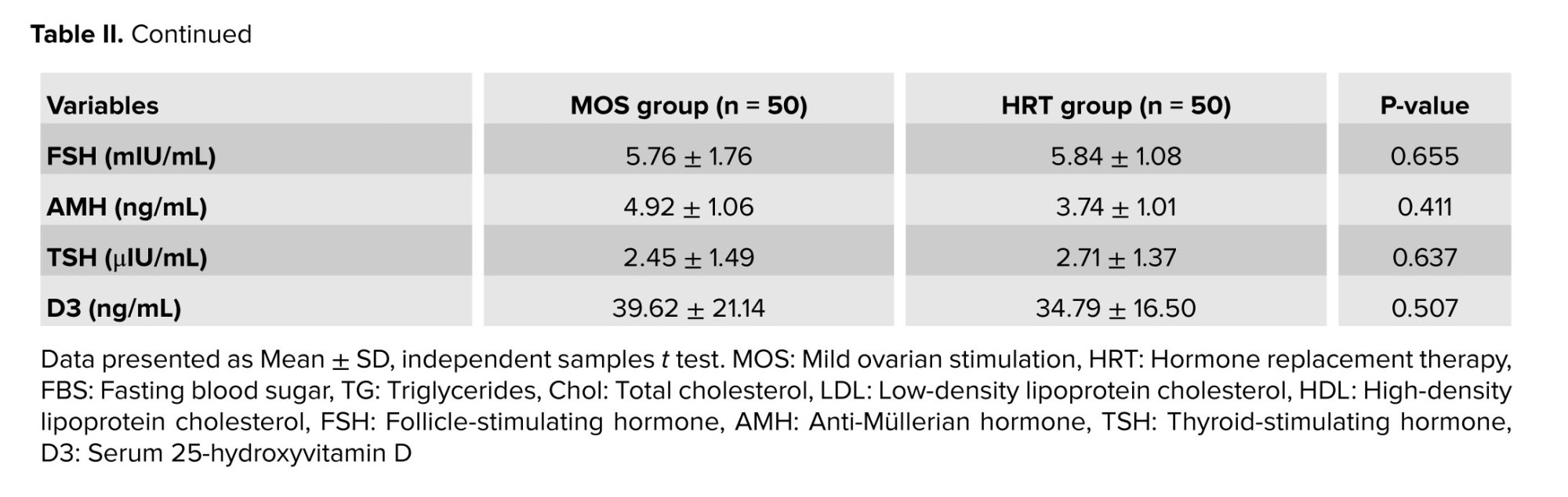
In this study, a comparison of routine pre-intervention tests between the 2 groups, MOS and HRT, revealed no significant differences in several variables. The fasting blood sugar, triglycerides, cholesterol, low‑density lipoprotein, high‑density lipoprotein, prolactin, FSH, anti‑Müllerian hormone, thyroid‑stimulating hormone, and D3 levels did not differ significantly between groups (p > 0.05) (Table II).
Regarding the HRT cycle, the participants in this group received an average of 13.78 ± 63.28 estradiol for 12.40 ± 1.79 days, and the endometrial thickness on the day of progesterone initiation was 7.91 ± 0.38 mm. In contrast, the MOS group received a total gonadotropin dose of 266.66 ± 89.55 mg, with the number of follicles in mid-cycle being 1.0 ± 0.02 and their average size being 18.02 ± 0.90 mm. GCG was administered to 25 participants on day 7, to 15 participants on day 8, and to the remaining participants on day 9.
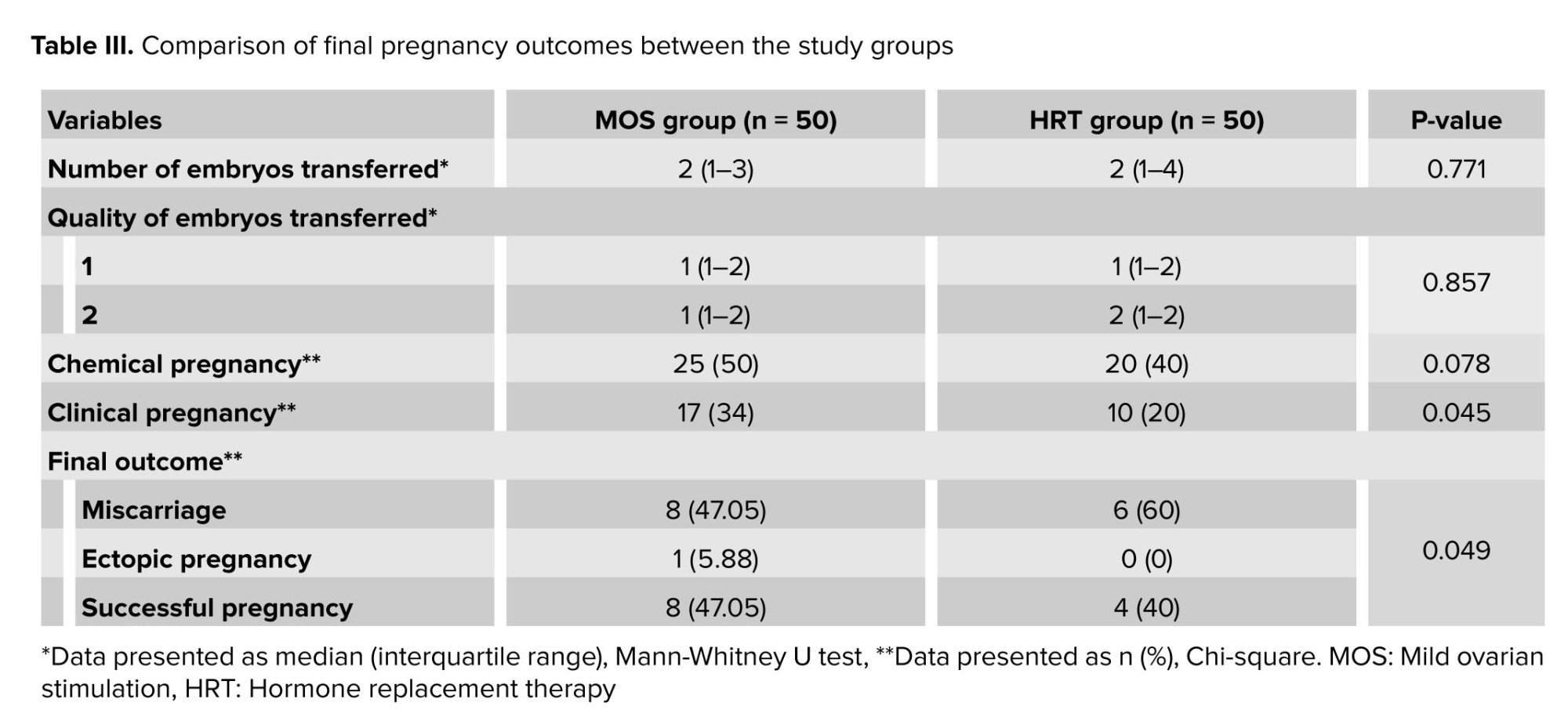
Final study results regarding chemical pregnancy, clinical pregnancy, and final pregnancy outcome indicated that the number of embryos transferred (p = 0.771) and their quality (p = 0.857) were nearly identical between the 2 groups. The chemical pregnancy rate was higher in the MOS group (25 cases) compared to the HRT group (20 cases), but this difference was not statistically significant (p = 0.078). However, the clinical pregnancy rate (p = 0.045) and successful pregnancy rate (p = 0.049) were significantly higher in the MOS group than in the HRT group (Table III).
4. Discussion
In this study, a comparison of routine pre-intervention tests between the 2 groups, MOS and HRT, revealed no significant differences in several variables. The fasting blood sugar, triglycerides, cholesterol, low‑density lipoprotein, high‑density lipoprotein, prolactin, FSH, anti‑Müllerian hormone, thyroid‑stimulating hormone, and D3 levels did not differ significantly between groups (p > 0.05) (Table II).
Regarding the HRT cycle, the participants in this group received an average of 13.78 ± 63.28 estradiol for 12.40 ± 1.79 days, and the endometrial thickness on the day of progesterone initiation was 7.91 ± 0.38 mm. In contrast, the MOS group received a total gonadotropin dose of 266.66 ± 89.55 mg, with the number of follicles in mid-cycle being 1.0 ± 0.02 and their average size being 18.02 ± 0.90 mm. GCG was administered to 25 participants on day 7, to 15 participants on day 8, and to the remaining participants on day 9.
Final study results regarding chemical pregnancy, clinical pregnancy, and final pregnancy outcome indicated that the number of embryos transferred (p = 0.771) and their quality (p = 0.857) were nearly identical between the 2 groups. The chemical pregnancy rate was higher in the MOS group (25 cases) compared to the HRT group (20 cases), but this difference was not statistically significant (p = 0.078). However, the clinical pregnancy rate (p = 0.045) and successful pregnancy rate (p = 0.049) were significantly higher in the MOS group than in the HRT group (Table III).





4. Discussion
The objective of this study was to compare 2 different endometrial preparation methods for FET in participants with PCOS. The 2 methods examined were the HRT protocol and MOS. The study results showed that although the number and quality of transferred embryos were similar between the 2 groups, the clinical and successful pregnancy rates were significantly higher in the MOS group compared to the HRT group. These results suggest that MOS may offer advantages in improving pregnancy outcomes in these participants.
The biological mechanisms underlying these differences may relate to the fundamental differences in how the endometrium is prepared in each group. In the HRT group, endometrial preparation is achieved through oral estrogen followed by progesterone administration (22). This method is often used for participants who are unable to ovulate naturally or in cases where ovulation is impaired (23, 24). In this approach, the endometrium is influenced by synthetic hormones, which may not create the optimal environment for embryo implantation (25). Conversely, in the MOS group, ovarian stimulation is achieved using low doses of FSH and letrozole, which promotes the development of more follicles and naturally increases estrogen levels (26). This natural increase in estrogen levels may enhance endometrial quality and facilitate embryo implantation (27).
Regarding the potential mechanisms through which MOS influences pregnancy outcomes, it is important to highlight its role in improving both endometrial and follicular quality (28, 29). MOS allows participants with PCOS to mature more follicles, leading to a natural rise in estrogen levels (30). This can result in a thicker endometrium and improved conditions for embryo implantation (31, 32). In contrast, the HRT protocol may not be as effective as natural ovarian stimulation and could result in poorer endometrial quality and a lower chance of successful pregnancies (33). Previous studies have shown similar findings when comparing HRT and MOS in infertile participants. A study demonstrated that MOS led to higher clinical and successful pregnancy rates compared to hormonal protocols in infertile participants (34). This study emphasized the importance of natural ovarian stimulation and its impact on improving the conditions for embryo implantation. Additionally, another study compared different hormonal methods in infertile participants and found that MOS yielded better pregnancy outcomes than HRT (28).
The results of the present study are also consistent with those who emphasized the impact of MOS on improving oocyte and endometrial quality in infertile participants (35). These studies suggest that MOS not only increases follicular count but also helps regulate hormone levels and creates more favorable conditions for embryo implantation. This study specifically focused on women with PCOS, who typically face complex hormonal challenges, and showed that MOS could offer additional benefits over hormonal protocols.
However, it is important to note that in this study, the rate of chemical pregnancies was non-significantly higher in the MOS group. This suggests that even though the number of chemical pregnancies was higher in the MOS group, the impact of these pregnancies was less significant compared to the clinically confirmed and successful pregnancies, which were notably higher in the MOS group. Moreover, other variables, such as the participant's age, the quality of the transferred embryos, and the overall health status of the participant, may also influence the results. The inclusion of both women with a history of embryo transfer and those without could be considered a limitation of this study, potentially affecting the homogeneity of the participants. Future studies should address this limitation to ensure a more homogeneous sample.
5. Conclusion
The biological mechanisms underlying these differences may relate to the fundamental differences in how the endometrium is prepared in each group. In the HRT group, endometrial preparation is achieved through oral estrogen followed by progesterone administration (22). This method is often used for participants who are unable to ovulate naturally or in cases where ovulation is impaired (23, 24). In this approach, the endometrium is influenced by synthetic hormones, which may not create the optimal environment for embryo implantation (25). Conversely, in the MOS group, ovarian stimulation is achieved using low doses of FSH and letrozole, which promotes the development of more follicles and naturally increases estrogen levels (26). This natural increase in estrogen levels may enhance endometrial quality and facilitate embryo implantation (27).
Regarding the potential mechanisms through which MOS influences pregnancy outcomes, it is important to highlight its role in improving both endometrial and follicular quality (28, 29). MOS allows participants with PCOS to mature more follicles, leading to a natural rise in estrogen levels (30). This can result in a thicker endometrium and improved conditions for embryo implantation (31, 32). In contrast, the HRT protocol may not be as effective as natural ovarian stimulation and could result in poorer endometrial quality and a lower chance of successful pregnancies (33). Previous studies have shown similar findings when comparing HRT and MOS in infertile participants. A study demonstrated that MOS led to higher clinical and successful pregnancy rates compared to hormonal protocols in infertile participants (34). This study emphasized the importance of natural ovarian stimulation and its impact on improving the conditions for embryo implantation. Additionally, another study compared different hormonal methods in infertile participants and found that MOS yielded better pregnancy outcomes than HRT (28).
The results of the present study are also consistent with those who emphasized the impact of MOS on improving oocyte and endometrial quality in infertile participants (35). These studies suggest that MOS not only increases follicular count but also helps regulate hormone levels and creates more favorable conditions for embryo implantation. This study specifically focused on women with PCOS, who typically face complex hormonal challenges, and showed that MOS could offer additional benefits over hormonal protocols.
However, it is important to note that in this study, the rate of chemical pregnancies was non-significantly higher in the MOS group. This suggests that even though the number of chemical pregnancies was higher in the MOS group, the impact of these pregnancies was less significant compared to the clinically confirmed and successful pregnancies, which were notably higher in the MOS group. Moreover, other variables, such as the participant's age, the quality of the transferred embryos, and the overall health status of the participant, may also influence the results. The inclusion of both women with a history of embryo transfer and those without could be considered a limitation of this study, potentially affecting the homogeneity of the participants. Future studies should address this limitation to ensure a more homogeneous sample.
5. Conclusion
Based on the results of this study, MOS offers significant advantages over HRT in terms of clinical and successful pregnancy rates in participants with PCOS. These findings can serve as a guideline for clinicians in choosing appropriate methods for endometrial preparation in these participants. It is recommended that future studies with larger sample sizes and consideration of additional variables be conducted to investigate these 2 methods further and clarify their advantages and disadvantages under different clinical conditions.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
K. Hamdi contributed to the study concept and design and drafted the manuscript. S. Khakpour was responsible for data acquisition and literature review. A. Ghasemzadeh Darjani performed data analysis and interpretation. L. Farzadi provided clinical supervision and critically revised the manuscript. B. Niknafs conducted the statistical analysis and offered methodological consultation. N. Navali participated in data collection and data management. P. Hakimi contributed to interpretation of the results and manuscript editing. R. Anvari Aliabad assisted in literature search and data analysis. H. Hajipour supervised the study and approved the final version of the manuscript. All authors read and approved the final version of the manuscript.
Acknowledgments
This article is based on the research project (code: 74408) for receiving the sub-specialty degree from Tabriz University of Medical Sciences, Tabri, Iran and the Women's Reproductive Health Research Center at this university. The authors greatly appreciate the financial support provided by this center and the participation of the infertile women involved in the study. The authors acknowledge the use of an artificial intelligence language model (ChatGPT, OpenAI) solely for language editing purposes of the manuscript. No part of the study design, data analysis, interpretation, or scientific content generation involved artificial intelligence tools. The authors reviewed and verified all AI‑edited text for accuracy and integrity.
Conflict of Interest
The authors declare that there is no conflict of interest.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
K. Hamdi contributed to the study concept and design and drafted the manuscript. S. Khakpour was responsible for data acquisition and literature review. A. Ghasemzadeh Darjani performed data analysis and interpretation. L. Farzadi provided clinical supervision and critically revised the manuscript. B. Niknafs conducted the statistical analysis and offered methodological consultation. N. Navali participated in data collection and data management. P. Hakimi contributed to interpretation of the results and manuscript editing. R. Anvari Aliabad assisted in literature search and data analysis. H. Hajipour supervised the study and approved the final version of the manuscript. All authors read and approved the final version of the manuscript.
Acknowledgments
This article is based on the research project (code: 74408) for receiving the sub-specialty degree from Tabriz University of Medical Sciences, Tabri, Iran and the Women's Reproductive Health Research Center at this university. The authors greatly appreciate the financial support provided by this center and the participation of the infertile women involved in the study. The authors acknowledge the use of an artificial intelligence language model (ChatGPT, OpenAI) solely for language editing purposes of the manuscript. No part of the study design, data analysis, interpretation, or scientific content generation involved artificial intelligence tools. The authors reviewed and verified all AI‑edited text for accuracy and integrity.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Fertility & Infertility
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |