Tue, May 19, 2026
[Archive]
Volume 23, Issue 12 (December 2025)
IJRM 2025, 23(12): 1007-1020 |
Back to browse issues page
Ethics code: IR.KMU.REC.1402.071
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Asadollahi Z, Ghaheri A, Haji-Maghsoudi S, Haghdoost A, Ashrafi M, Eftekhari-Yazdi P et al . Determining factors associated with the number of oocytes with appropriate morphology in infertile women: A cross-sectional study. IJRM 2025; 23 (12) :1007-1020
URL: http://ijrm.ir/article-1-3615-en.html
URL: http://ijrm.ir/article-1-3615-en.html
Zahra Asadollahi1 

 , Azadeh Ghaheri2
, Azadeh Ghaheri2 

 , Saiedeh Haji-Maghsoudi3
, Saiedeh Haji-Maghsoudi3 

 , AliAkbar Haghdoost4
, AliAkbar Haghdoost4 

 , Mahnaz Ashrafi5
, Mahnaz Ashrafi5 

 , Poopak Eftekhari-Yazdi6
, Poopak Eftekhari-Yazdi6 

 , Yunes Jahani *7
, Yunes Jahani *7 




 , Azadeh Ghaheri2
, Azadeh Ghaheri2 

 , Saiedeh Haji-Maghsoudi3
, Saiedeh Haji-Maghsoudi3 

 , AliAkbar Haghdoost4
, AliAkbar Haghdoost4 

 , Mahnaz Ashrafi5
, Mahnaz Ashrafi5 

 , Poopak Eftekhari-Yazdi6
, Poopak Eftekhari-Yazdi6 

 , Yunes Jahani *7
, Yunes Jahani *7 


1- Modeling in Health Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran. & Department of Biostatistics and Epidemiology, School of Public Health, Kerman University of Medical Sciences, Kerman, Iran., Department of Epidemiology and Biostatistics, School of Public Health, Rafsanjan University of Medical Sciences, Rafsanjan, Iran.
2- Department of Basic and Population Based Studies in NCD, Reproductive Epidemiology Research Center, Royan Institute, ACECR, Tehran, Iran.
3- Modeling in Health Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran. & Department of Biostatistics and Epidemiology, School of Public Health, Kerman University of Medical Sciences, Kerman, Iran.
4- Medical Informatics Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran. & HIV/STI Surveillance Research Center, WHO Collaborating Center for HIV Surveillance, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran.
5- Department of Endocrinology and Female Infertility, Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine, ACECR, Tehran, Iran.
6- Department of Embryology, Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine, ACECR, Tehran, Iran.
7- Health in Disasters and Emergencies Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran. & Modeling in Health Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran., Department of Biostatistics and Epidemiology, School of Public Health, Kerman University of Medical Sciences, Kerman, Iran. ,yonesjahani@yahoo.com; u.jahani@kmu.ac.ir
2- Department of Basic and Population Based Studies in NCD, Reproductive Epidemiology Research Center, Royan Institute, ACECR, Tehran, Iran.
3- Modeling in Health Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran. & Department of Biostatistics and Epidemiology, School of Public Health, Kerman University of Medical Sciences, Kerman, Iran.
4- Medical Informatics Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran. & HIV/STI Surveillance Research Center, WHO Collaborating Center for HIV Surveillance, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran.
5- Department of Endocrinology and Female Infertility, Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine, ACECR, Tehran, Iran.
6- Department of Embryology, Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine, ACECR, Tehran, Iran.
7- Health in Disasters and Emergencies Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran. & Modeling in Health Research Center, Institute for Futures Studies in Health, Kerman University of Medical Sciences, Kerman, Iran., Department of Biostatistics and Epidemiology, School of Public Health, Kerman University of Medical Sciences, Kerman, Iran. ,
Keywords: Oocytes, Infertility, Female, Assisted reproductive technology, Regression analysis, Statistical models.
Full-Text [PDF 456 kb]
(279 Downloads)
| Abstract (HTML) (287 Views)
2.3.1. Poisson regression
λ i = X i T β λ i = X i T β + offset i
Full-Text: (34 Views)
1. Introduction
Infertility is a significant global health concern, affecting both men and women, and impacting up to 12% of couples worldwide (1, 2). Assisted reproductive technology (ART) includes a range of medical interventions to treat infertility. From 1978-2012, ART has led to the birth of over 5 million babies worldwide (3, 4). In developed countries, approximately 1% of all births result from in vitro fertilization or intracytoplasmic sperm injection (5). Over the past few decades, the success rates of ART have improved significantly, offering hope to many infertile couples (3). However, achieving successful outcomes with ART depends on numerous factors, particularly the quality of embryos. For women undergoing fertility treatments, both the number and quality of oocytes are critical for improving their chances of conception. Studies indicate that the number of morphologically normal oocytes (MNO) plays a key role in enhancing fertility and increasing the likelihood of successful treatment outcomes (2).
The quality of a woman’s oocytes can be influenced by various factors, including age, lifestyle, underlying medical conditions, and health-related behaviors. Age is a major determinant, with both the quantity and quality of oocytes declining over time, particularly after 35 yr. This decline is attributed to accumulated oxidative stress, reduced mitochondrial function, and a marked increase in aneuploidy rates, all of which negatively impact fertility outcomes (3-5).
In addition to age, other factors such as insulin resistance, commonly seen in conditions like polycystic ovary syndrome (PCOS) can also affect oocyte quality. PCOS, which is characterized by hyperinsulinemia, disrupts normal folliculogenesis and oocyte maturation, leading to reduced oocyte quality (4, 6). Smoking, another detrimental factor, impairs ovarian function by increasing oxidative stress and causing DNA damage in oocytes, while alcohol consumption disrupts hormonal balance, further compromising fertility (4, 5). Obesity is similarly associated with metabolic and inflammatory changes that alter the ovarian environment and reduce oocyte competence. Moreover, chronic conditions like endometriosis negatively affect oocyte quality through oxidative stress and disruption of the follicular microenvironment (4, 7).
To assess oocyte competence, morphological evaluation is a standard practice. This involves examining criteria such as cytoplasmic maturation, the structure of the zona pellucida, and the size and shape of oocytes, which are key indicators of reproductive potential (5, 8).
While research on oocyte quality has expanded globally, it still lacks comprehensive studies focusing on the quantification of MNO, particularly in specific populations such as Iranian women. Current literature has largely overlooked systematic evaluations of MNO in this group (3, 8, 9). Given the complexity of MNO data, characterized by a high proportion of zeros, this study employs advanced statistical models, such as zero-inflated Poisson (ZIP) and semiparametric zero-inflated negative binomial (SZINB) regression, to accurately identify factors influencing MNO and provide robust insights into oocyte quality.
This study aims to address this critical gap by investigating the number of MNO and identifying the factors influencing oocyte quality among infertile women referred to the Royan Institute in Tehran, Iran. As one of the first studies of its kind in the region, this research aimed to provide valuable insights into an underexplored area of Iranian reproductive health.
2. Materials and Methods
2.1. Study design and participants
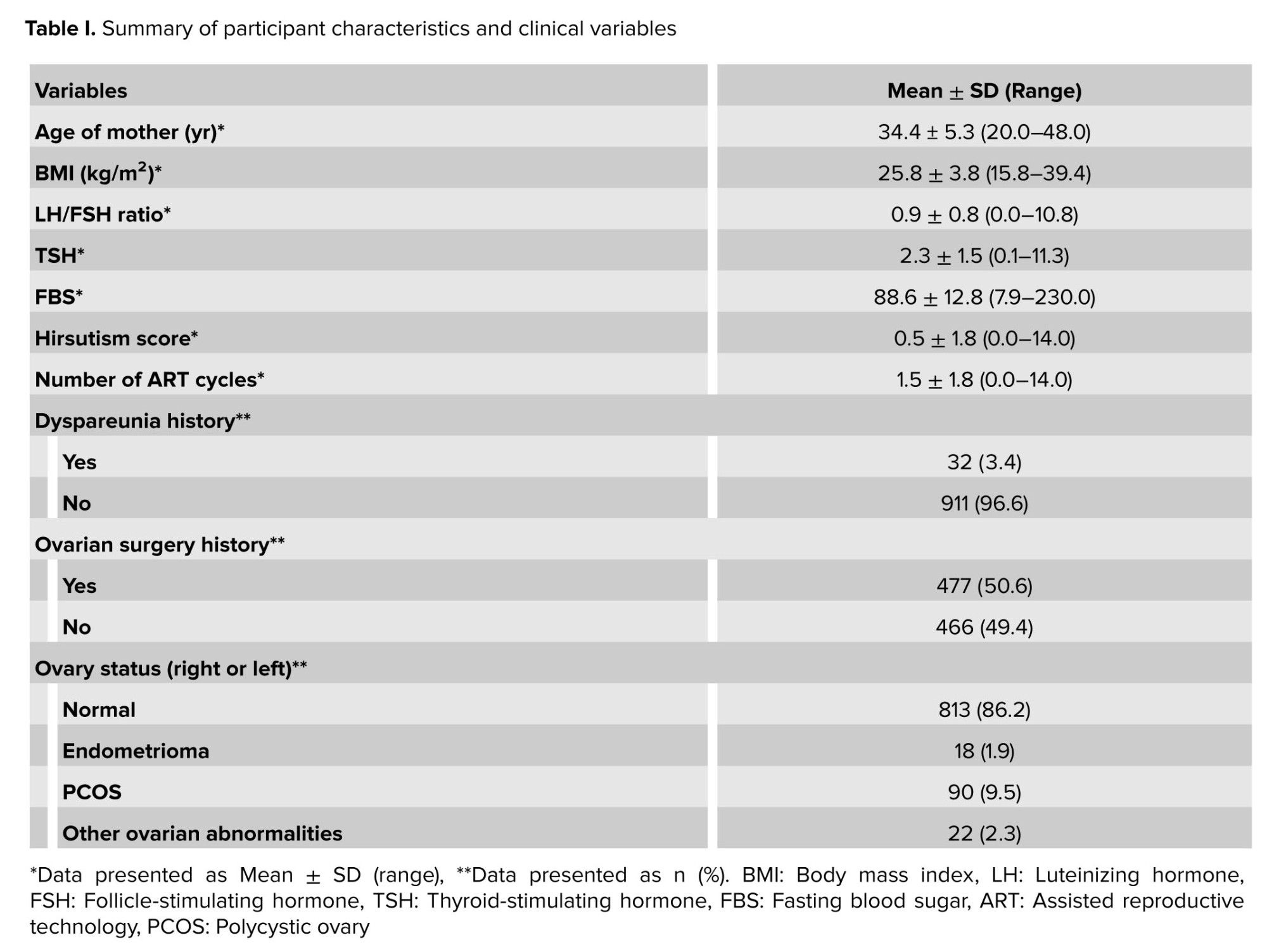
In this cross-sectional study, data of 950 infertile women who referred to the Royan Institute, Tehran, Iran, between January 2012 and December 2013 were extracted from their medical records. This dataset was selected as it represented the most recent non-confidential, high-quality clinical data available at the time of the study, with a large sample size suitable for robust statistical analysis. The study population comprised couples with primary or secondary infertility who had been infertile for at least 1 yr and had referred to the institute, Tehran, Iran for treatment during the study period. Women who did not provide consent or had incomplete oocyte morphology data were not included in the analysis. Medical history data, general physical examinations, and gynecological examinations were extracted from medical records for all participants.
2.2. Variables and measurements
The response variable in this study is the number of MNOs, which is a count variable. An oocyte with appropriate morphology shows uniform cytoplasm, a clear zona, and no visible vacuoles or fragments. The oocyte assessment criteria were established based on international protocols. However, considering the clinical conditions, the variable “rate” was used as the response variable to determine the proportion of MNO. Specifically, this rate was calculated by dividing the number of MNO by the total number of oocytes in the metaphase II (M2) stage. Independent variables include age (yr), body mass index (BMI, kg/m²), dyspareunia history, ovarian surgery history, hirsutism score, ovary status (right or left; normal, endometrioma, PCOS, other abnormalities), luteinizing hormone/follicle-stimulating hormone (LH/FSH, IU/L), thyroid-stimulating hormone (TSH, mIU/L), fasting blood sugar (FBS, mg/dL), and number of ART cycles.
2.3. Statistical modeling approach
In this study, since the response variable is a count variable and contains a large number of zeros, we used Poisson regression, negative binomial (NB) regression, zero-inflated (ZI) models, and ZI semi-parametric models for data analysis. These models were selected due to their ability to handle sparse counts and excess zeros. This study specifically aimed to leverage advanced statistical models, such as ZIP and SZINB regression, to maximize the extraction of insights from high-quality clinical data with a high proportion of zeros (71.2% of cases with zero MNO) and to uncover both linear and non-linear relationships between independent variables and the number of MNO, thereby demonstrating the capability of these models in clinical research. Furthermore, our goal was to explore both linear and non-linear relationships between the independent variables and the response variable. After fitting the models to the data, the best model for both linear and non-linear relationships was chosen and reported.
Infertility is a significant global health concern, affecting both men and women, and impacting up to 12% of couples worldwide (1, 2). Assisted reproductive technology (ART) includes a range of medical interventions to treat infertility. From 1978-2012, ART has led to the birth of over 5 million babies worldwide (3, 4). In developed countries, approximately 1% of all births result from in vitro fertilization or intracytoplasmic sperm injection (5). Over the past few decades, the success rates of ART have improved significantly, offering hope to many infertile couples (3). However, achieving successful outcomes with ART depends on numerous factors, particularly the quality of embryos. For women undergoing fertility treatments, both the number and quality of oocytes are critical for improving their chances of conception. Studies indicate that the number of morphologically normal oocytes (MNO) plays a key role in enhancing fertility and increasing the likelihood of successful treatment outcomes (2).
The quality of a woman’s oocytes can be influenced by various factors, including age, lifestyle, underlying medical conditions, and health-related behaviors. Age is a major determinant, with both the quantity and quality of oocytes declining over time, particularly after 35 yr. This decline is attributed to accumulated oxidative stress, reduced mitochondrial function, and a marked increase in aneuploidy rates, all of which negatively impact fertility outcomes (3-5).
In addition to age, other factors such as insulin resistance, commonly seen in conditions like polycystic ovary syndrome (PCOS) can also affect oocyte quality. PCOS, which is characterized by hyperinsulinemia, disrupts normal folliculogenesis and oocyte maturation, leading to reduced oocyte quality (4, 6). Smoking, another detrimental factor, impairs ovarian function by increasing oxidative stress and causing DNA damage in oocytes, while alcohol consumption disrupts hormonal balance, further compromising fertility (4, 5). Obesity is similarly associated with metabolic and inflammatory changes that alter the ovarian environment and reduce oocyte competence. Moreover, chronic conditions like endometriosis negatively affect oocyte quality through oxidative stress and disruption of the follicular microenvironment (4, 7).
To assess oocyte competence, morphological evaluation is a standard practice. This involves examining criteria such as cytoplasmic maturation, the structure of the zona pellucida, and the size and shape of oocytes, which are key indicators of reproductive potential (5, 8).
While research on oocyte quality has expanded globally, it still lacks comprehensive studies focusing on the quantification of MNO, particularly in specific populations such as Iranian women. Current literature has largely overlooked systematic evaluations of MNO in this group (3, 8, 9). Given the complexity of MNO data, characterized by a high proportion of zeros, this study employs advanced statistical models, such as zero-inflated Poisson (ZIP) and semiparametric zero-inflated negative binomial (SZINB) regression, to accurately identify factors influencing MNO and provide robust insights into oocyte quality.
This study aims to address this critical gap by investigating the number of MNO and identifying the factors influencing oocyte quality among infertile women referred to the Royan Institute in Tehran, Iran. As one of the first studies of its kind in the region, this research aimed to provide valuable insights into an underexplored area of Iranian reproductive health.
2. Materials and Methods
2.1. Study design and participants
In this cross-sectional study, data of 950 infertile women who referred to the Royan Institute, Tehran, Iran, between January 2012 and December 2013 were extracted from their medical records. This dataset was selected as it represented the most recent non-confidential, high-quality clinical data available at the time of the study, with a large sample size suitable for robust statistical analysis. The study population comprised couples with primary or secondary infertility who had been infertile for at least 1 yr and had referred to the institute, Tehran, Iran for treatment during the study period. Women who did not provide consent or had incomplete oocyte morphology data were not included in the analysis. Medical history data, general physical examinations, and gynecological examinations were extracted from medical records for all participants.
2.2. Variables and measurements
The response variable in this study is the number of MNOs, which is a count variable. An oocyte with appropriate morphology shows uniform cytoplasm, a clear zona, and no visible vacuoles or fragments. The oocyte assessment criteria were established based on international protocols. However, considering the clinical conditions, the variable “rate” was used as the response variable to determine the proportion of MNO. Specifically, this rate was calculated by dividing the number of MNO by the total number of oocytes in the metaphase II (M2) stage. Independent variables include age (yr), body mass index (BMI, kg/m²), dyspareunia history, ovarian surgery history, hirsutism score, ovary status (right or left; normal, endometrioma, PCOS, other abnormalities), luteinizing hormone/follicle-stimulating hormone (LH/FSH, IU/L), thyroid-stimulating hormone (TSH, mIU/L), fasting blood sugar (FBS, mg/dL), and number of ART cycles.
2.3. Statistical modeling approach
In this study, since the response variable is a count variable and contains a large number of zeros, we used Poisson regression, negative binomial (NB) regression, zero-inflated (ZI) models, and ZI semi-parametric models for data analysis. These models were selected due to their ability to handle sparse counts and excess zeros. This study specifically aimed to leverage advanced statistical models, such as ZIP and SZINB regression, to maximize the extraction of insights from high-quality clinical data with a high proportion of zeros (71.2% of cases with zero MNO) and to uncover both linear and non-linear relationships between independent variables and the number of MNO, thereby demonstrating the capability of these models in clinical research. Furthermore, our goal was to explore both linear and non-linear relationships between the independent variables and the response variable. After fitting the models to the data, the best model for both linear and non-linear relationships was chosen and reported.
2.3.1. Poisson regression
The Poisson regression model can be modified to include an offset term, defined as the natural logarithm of the exposure. Here, Y is the number of oocytes with good morphology, which is affected by the number of oocytes in the M2. Y is a count variable. The term for inclusion is an offset term:
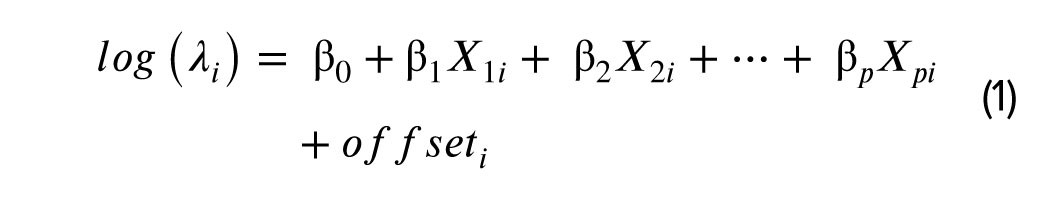
In this model, E(Yi)=Var(Yi)=λi, where λi represents the expected value (mean) of the response variable for the i-th observation (i = 1, 2, …, n). The term offseti=log(mi), where mi is the total number of oocytes in the M2 stage for each individual, is added to the linear predictor (1) to adjust for an exposure. The offset is often used in situations where the response variable represents a rate, and the offset is the denominator of this rate. Here, β0, β1, ..., βp are the coefficients of the model, and X1, X2, …, XP are the explanatory variables.
2.3.2. NB regression
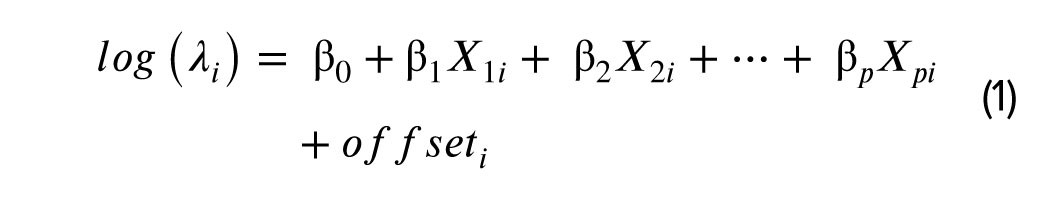
In this model, E(Yi)=Var(Yi)=λi, where λi represents the expected value (mean) of the response variable for the i-th observation (i = 1, 2, …, n). The term offseti=log(mi), where mi is the total number of oocytes in the M2 stage for each individual, is added to the linear predictor (1) to adjust for an exposure. The offset is often used in situations where the response variable represents a rate, and the offset is the denominator of this rate. Here, β0, β1, ..., βp are the coefficients of the model, and X1, X2, …, XP are the explanatory variables.
2.3.2. NB regression
In this model, Y is a count variable that follows NB distribution, where i= 1, 2, ..., n represents the individuals. The probability mass function of the NB distribution is as follows:
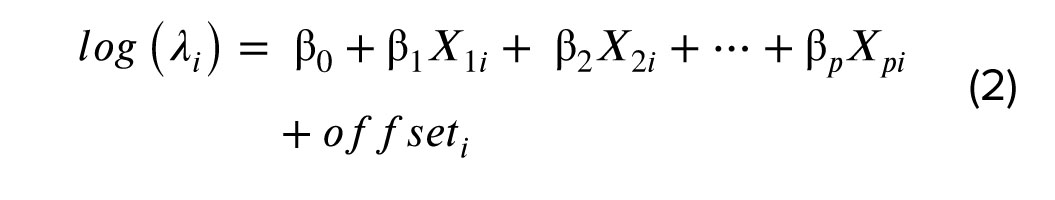
With mean and variance of E(Y) = λ and V(Y) = λ +(αλ2), respectively. The canonical link function of the NB regression is
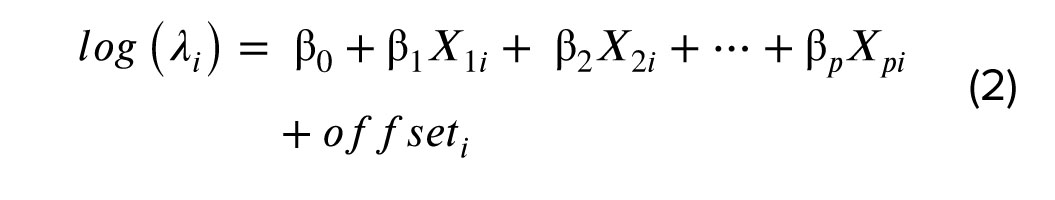
With mean and variance of E(Y) = λ and V(Y) = λ +(αλ2), respectively. The canonical link function of the NB regression is
2.3.3. ZI models
2.3.3. ZI models
At times, datasets contain a significant number of zeros that the standard distributions struggle to accommodate. Poisson and NB distributions can be extended into ZI models, which are mixture models designed to handle this excess of zeros. A ZI model typically uses logistic regression, often with a logit link, to determine the origin of each zero. In this study, where i=1, 2, .., n represents the individuals, given that we use a rate response variable in this study, we suggest modifying the ZIP and Zero-Inflated NB models to account for the different variables at risk in both the count and binary components (12). The revised ZIP and ZINB models are expressed as follows:
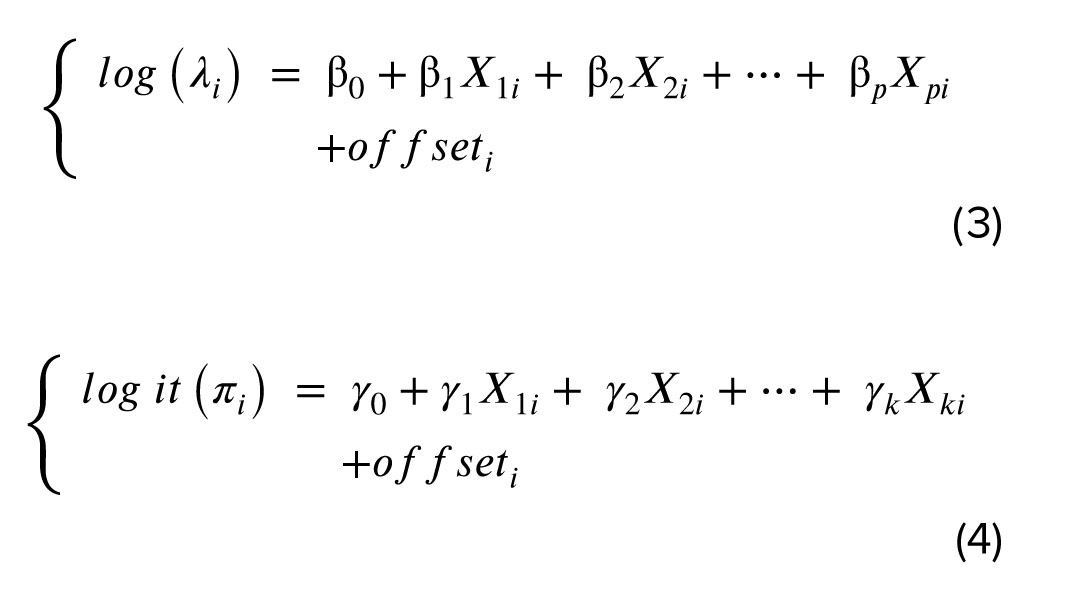
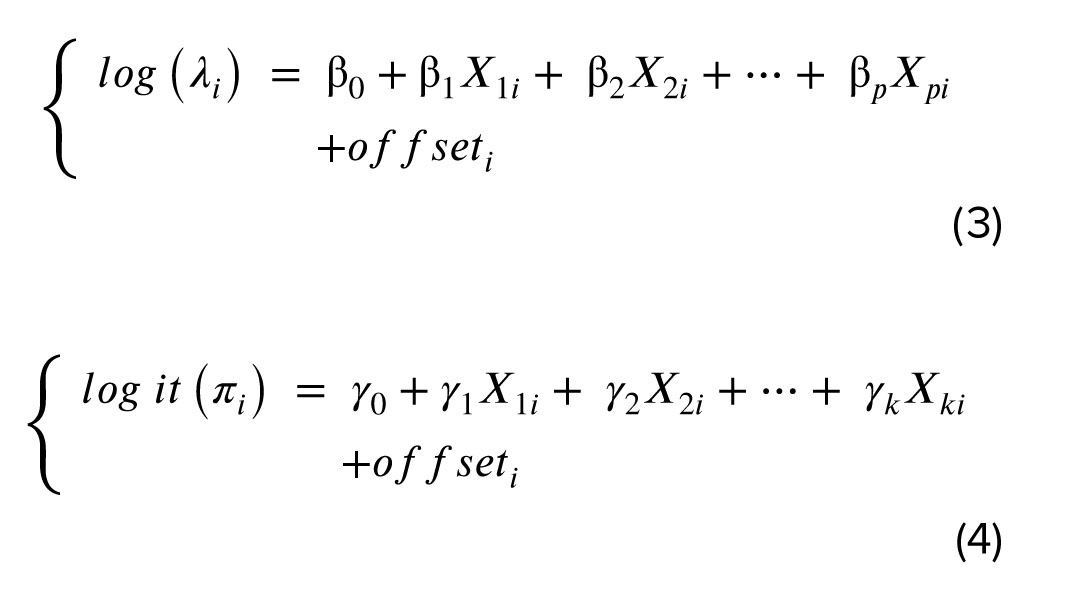
2.3.4. Semiparametric ZI (SZI) regression models
If we have more than one influential predictor variable and the nature of the relationship of some predictors is known, while others remain uncertain in the model, a semi-parametric model is preferred. Additionally, when the data contains a high number of zeros, traditional models may fall short. In such scenarios, semi-parametric models designed to handle zero-inflation are employed to provide a more accurate analysis (13). Poisson and NB distributions can be extended to SZI regression models. These advanced models, which are mixtures of different distributions, are specifically crafted to address complex, nonlinear relationships in data characterized by a high frequency of zero counts (14). Given that we use a rate response variable in this study, we modify the ZIP and ZINB models to account for the different variables at risk in both the count and binary components (15). The revised ZIP and ZINB models are expressed as follows:
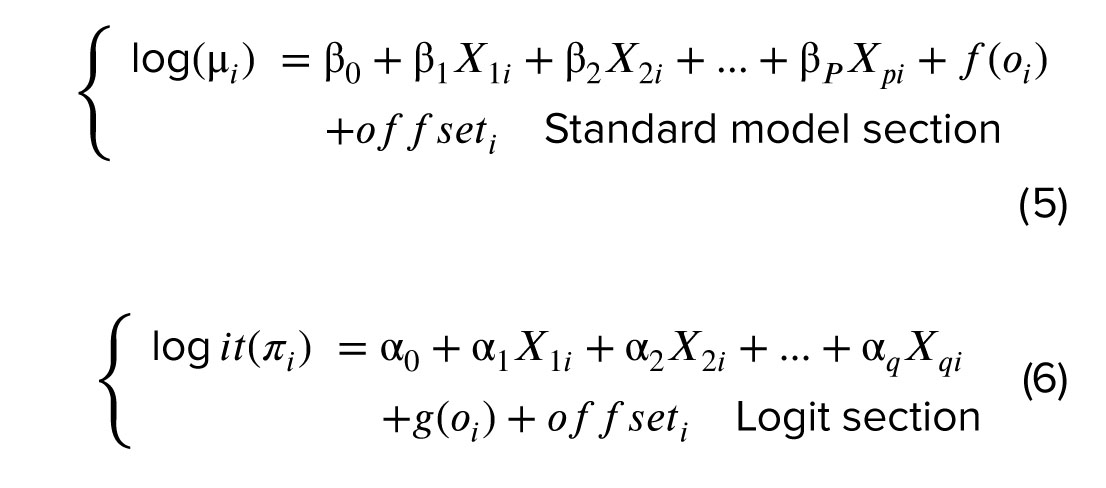
In this model, where i=1, 2, …, n represents the individuals, βj( j= 0, 1, 2,…, p) and αk (k= 0, 1, 2,…, q) are the coefficients for the Poisson or NB and logistic components, respectively. The nature of the relationship between these coefficients in the model is linear. However, the relationship involving oi in the model is nonlinear. It can represent a sum of unspecified functions of multiple variables, with functions f and g serving as the smoothing functions for oi in the Poisson or NB and logistic components, respectively.
2.4. Model evaluation and selection
The average rate of MNO, from women visiting the Royan Institute, Tehran, Iran was modeled as a function of age, BMI, and other explanatory variables using Poisson regression, NB regression, ZIP regression, ZINB regression, and SZIP or SZINB regression models. The same explanatory variables were included in both components (the logit and count components) of the ZI models. The Vuong test, along with the Akaike Information Criterion (AIC) and Bayesian Information Criterion (BIC), was used to compare the models and select the one that offers the better fit to the data, balancing model accuracy and complexity. The Vuong test produces a p-value to compare the fit of 2 non-nested models. At the 5% significance level, the interpretation was as follows: if the p < 0.05, the difference between the models was statistically significant, with a positive test statistic indicating that the first model provides the better fit and a negative test statistic indicating that the second model provides the better fit. For p-value greater than or equal to 0.05, no significant difference was observed between the models, and selection may rely on AIC or BIC for further comparison.
2.5. Ethical Considerations
This study was approved by the Ethics Committee of Kerman University of Medical Sciences, Kerman, Iran (Code: IR.KMU.REC.1402.071). Informed written consent was obtained from all participants at the time of their initial treatment (January 2012-December 2013) at Royan Institute, Tehran, Iran as part of the routine clinical protocol before any ART procedure. Patients were informed that their anonymized clinical and laboratory data might be used for research purposes and published only in aggregate form, with no possibility of individual identification.
2.6. Statistical Analysis
The number of MNOs is a count variable. Poisson and NB regression were used to model the count variables. In this study, the outcome variable of interest was the number of MNO, which was zero in 71.2% of cases. Due to the excess of zeros, ZI models and semiparametric ZI models were used to model these count data. The Vuong test, along with the AIC and BIC, was used for model selection to ensure robust evaluation of model fit, considering both predictive accuracy and model complexity. The Vuong test provides a significant z-test that is utilized for selecting the best linear models. To check for overdispersion in the models in R, the dispersion test function from the AER package was used. This test evaluates the null hypothesis of equidispersion in Poisson models against the alternative hypothesis of overdispersion or underdispersion. In NB modeling, overdispersion was assessed indirectly through model parameters. All models and statistical computations were executed using R version 4.4.1. The significance level of all tests was considered to be 0.05.
3. Results
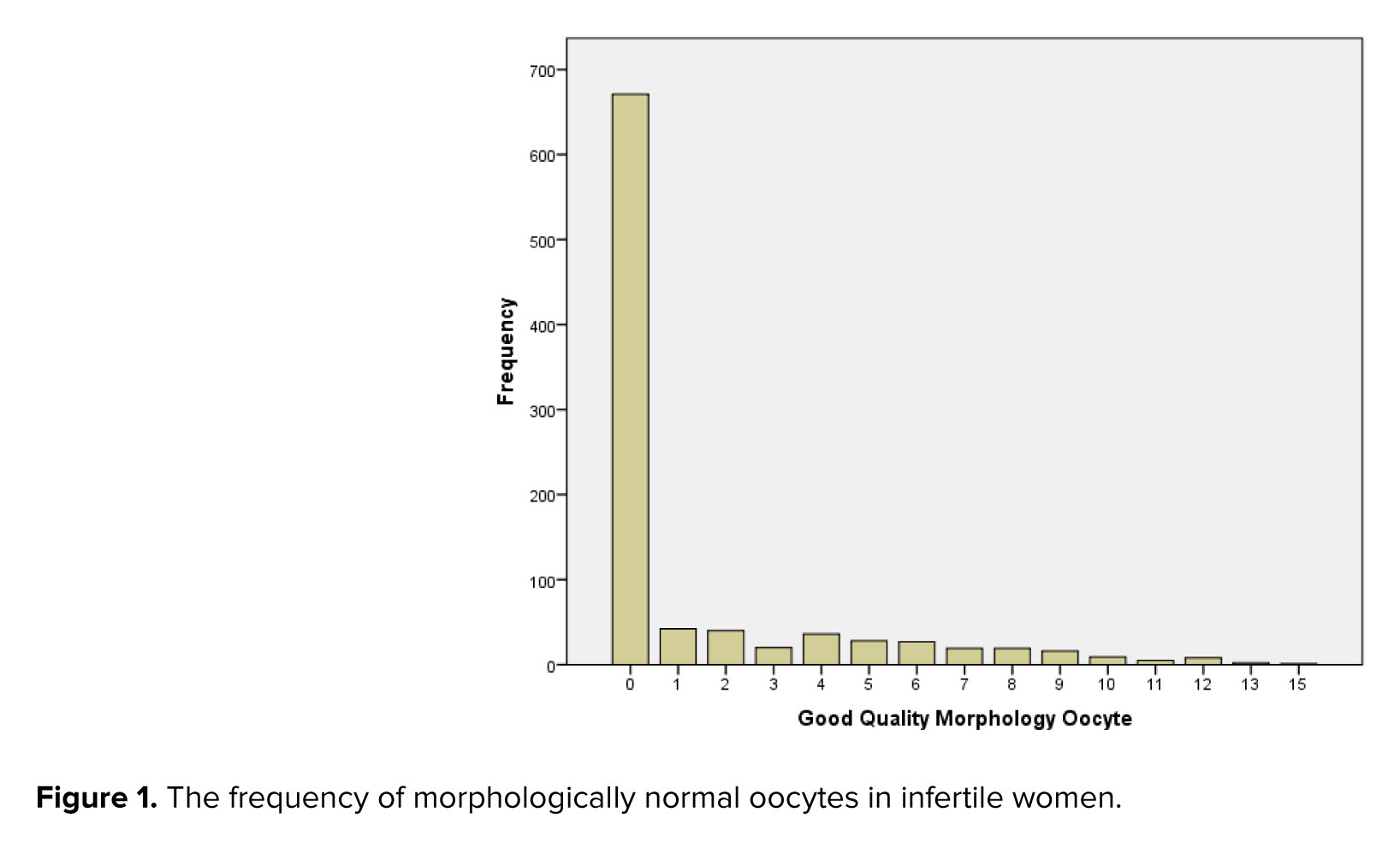
Out of the 950 infertile women whose medical records were initially extracted, 943 had complete oocyte morphology data and were included in the final analysis. Among these 943 women referred to the Royan Institute, Tehran, Iran for ART, MNO were zero in 671 cases (71.2%) and the mean (standard deviation) MNO was 1.42 (2.80), range 0-15, interquartile range = 1 (Figure 1).
The mean age of the women was 34.41 ± 5.30 yr, and the mean BMI was 25.87 ± 3.80 kg/m². Detailed information on other variables, including hormone levels and ovarian conditions, is provided in table I.
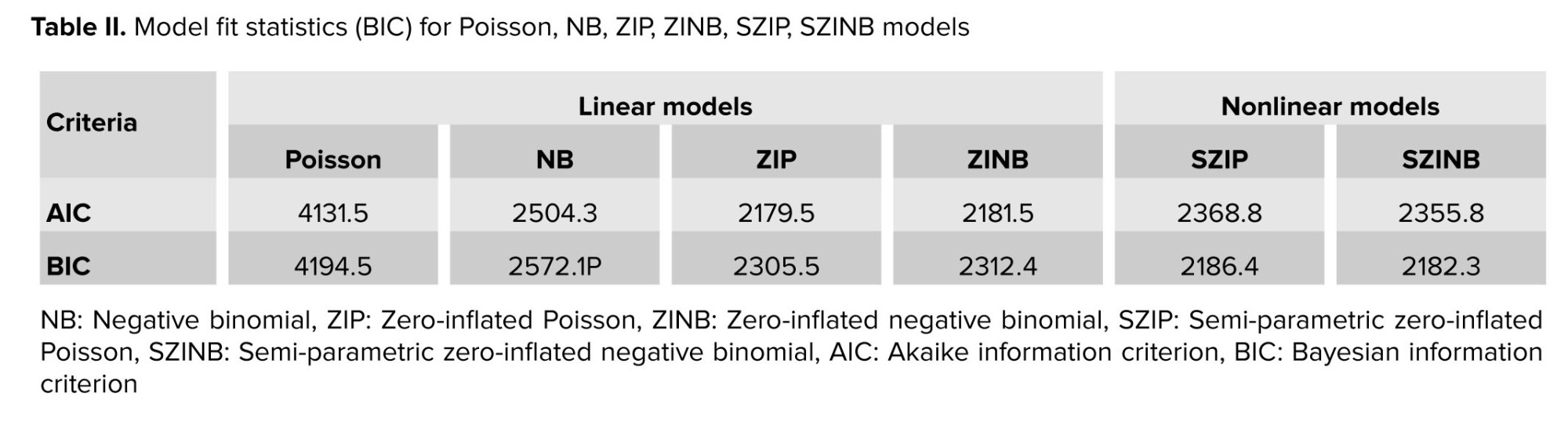
Based on the results of the Vuong test statistics, along with AIC and BIC, the Poisson regression provided the worst fit for the data among all regression models. According to both AIC and BIC criteria, the ZIP model was the best fit among linear models, while among nonlinear models, the SZINB model provided the best fit (Table II). These results confirm the robustness of the ZIP and SZINB models in capturing the complex relationships in the data.
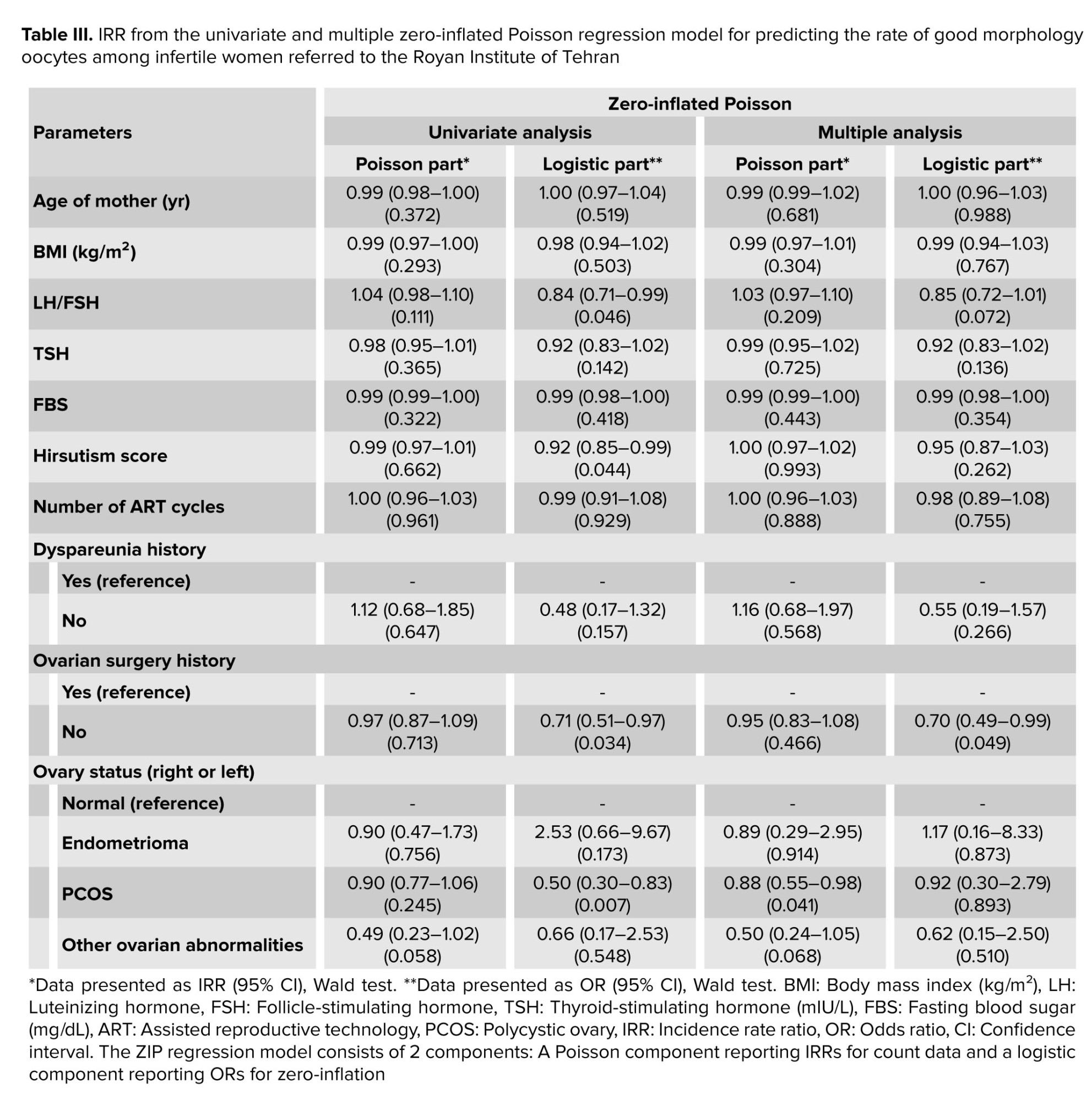
The univariate ZIP model analysis revealed that the LH/FSH ratio, hirsutism score, ovarian surgery history, and the presence of PCOS in ovary status were significantly associated with a decreased likelihood of structural zeros (i.e., cases where no high-quality oocytes are produced due to genetic or unknown disorders) in the logistic component. This suggests that women with these conditions, likely due to targeted medical interventions such as hormonal treatments, have a higher probability of producing at least one MNO, potentially improving fertility outcomes. In the Poisson component of the univariate analysis, no factors were significantly associated with the rate of MNO (Table III). In the multiple ZIP analysis, ovarian surgery history remained significantly associated with a decreased likelihood of structural zeros in the logistic component, implying that women with a history of ovarian surgery, possibly due to enhanced medical management, are more likely to produce at least one MNO even after adjusting for other variables. In contrast, in the Poisson component, the presence of PCOS was significantly associated with a reduced rate of MNO, indicating that PCOS negatively impacts the quantity of high-quality oocytes among women who produce at least some MNO (Table III). These findings underscore the role of medical interventions in mitigating the risk of complete absence of high-quality oocytes and highlight the specific negative impact of PCOS on oocyte quantity.
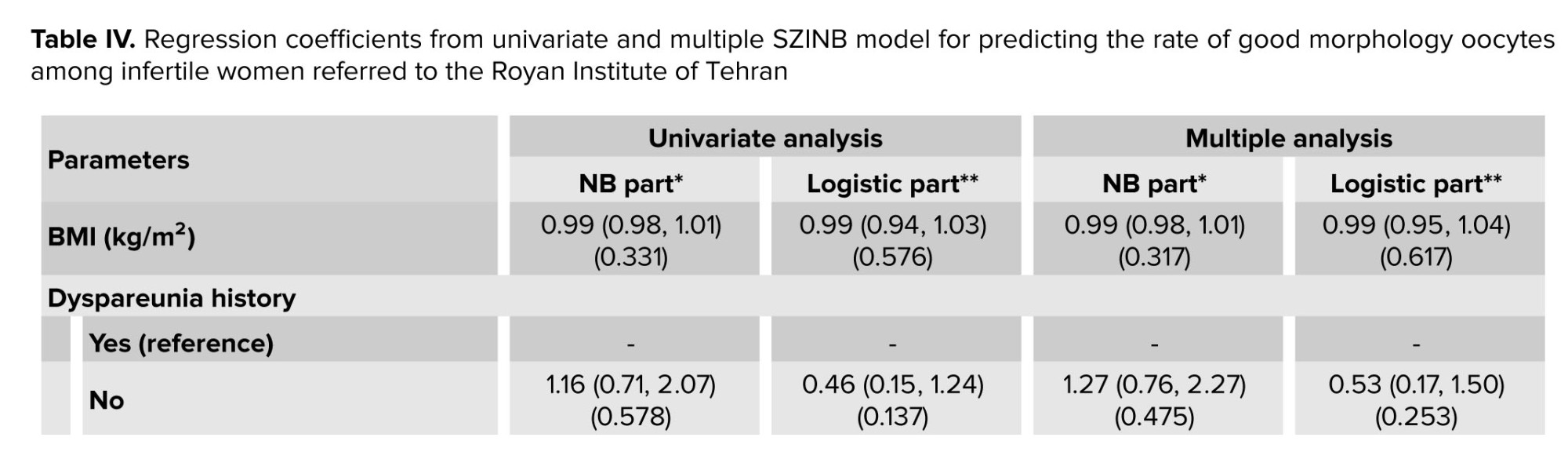
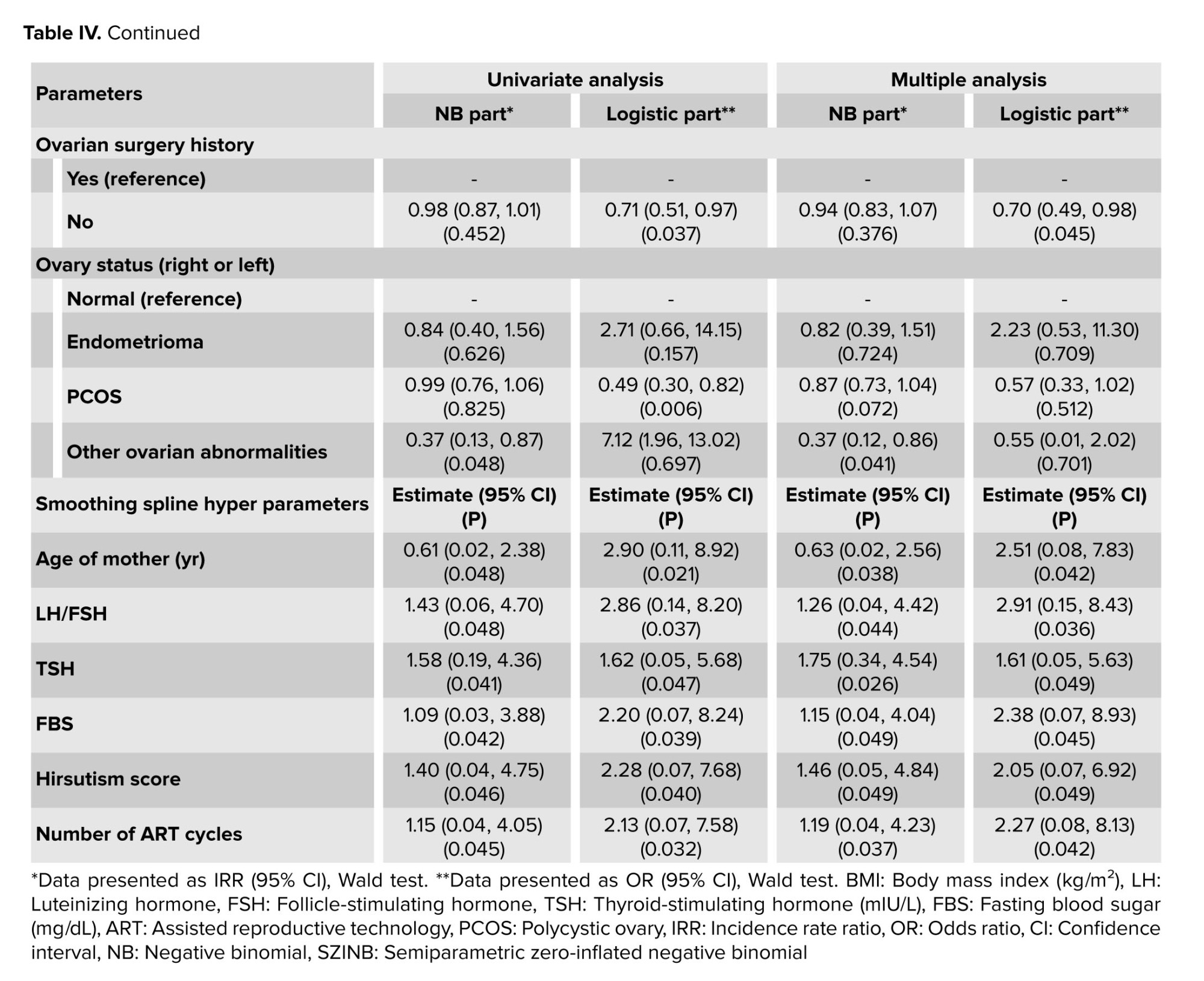
The univariate SZINB model analysis revealed that ovarian surgery history and the presence of PCOS were significantly associated with a decreased likelihood of structural zeros (i.e., cases where no high-quality oocytes are produced due to genetic or unknown disorders) in the logistic component. This suggests that women with these conditions, likely due to targeted medical interventions such as hormonal treatments, have a higher probability of producing at least one MNO, potentially improving fertility outcomes. In contrast, other ovarian abnormalities were significantly associated with a reduced rate of MNO in the NB component, indicating a negative impact on the quantity of MNO (Table IV). In the multiple SZINB analysis, ovarian surgery history continued to show a significant association with a decreased likelihood of structural zeros in the logistic component, implying that women with a history of ovarian surgery, possibly due to enhanced medical management, are more likely to produce at least one MNO even after adjusting for other variables. Meanwhile, other ovarian abnormalities remained significantly associated with a reduced rate of MNO in the NB component, indicating a negative impact on the quantity of MNO (Table IV).
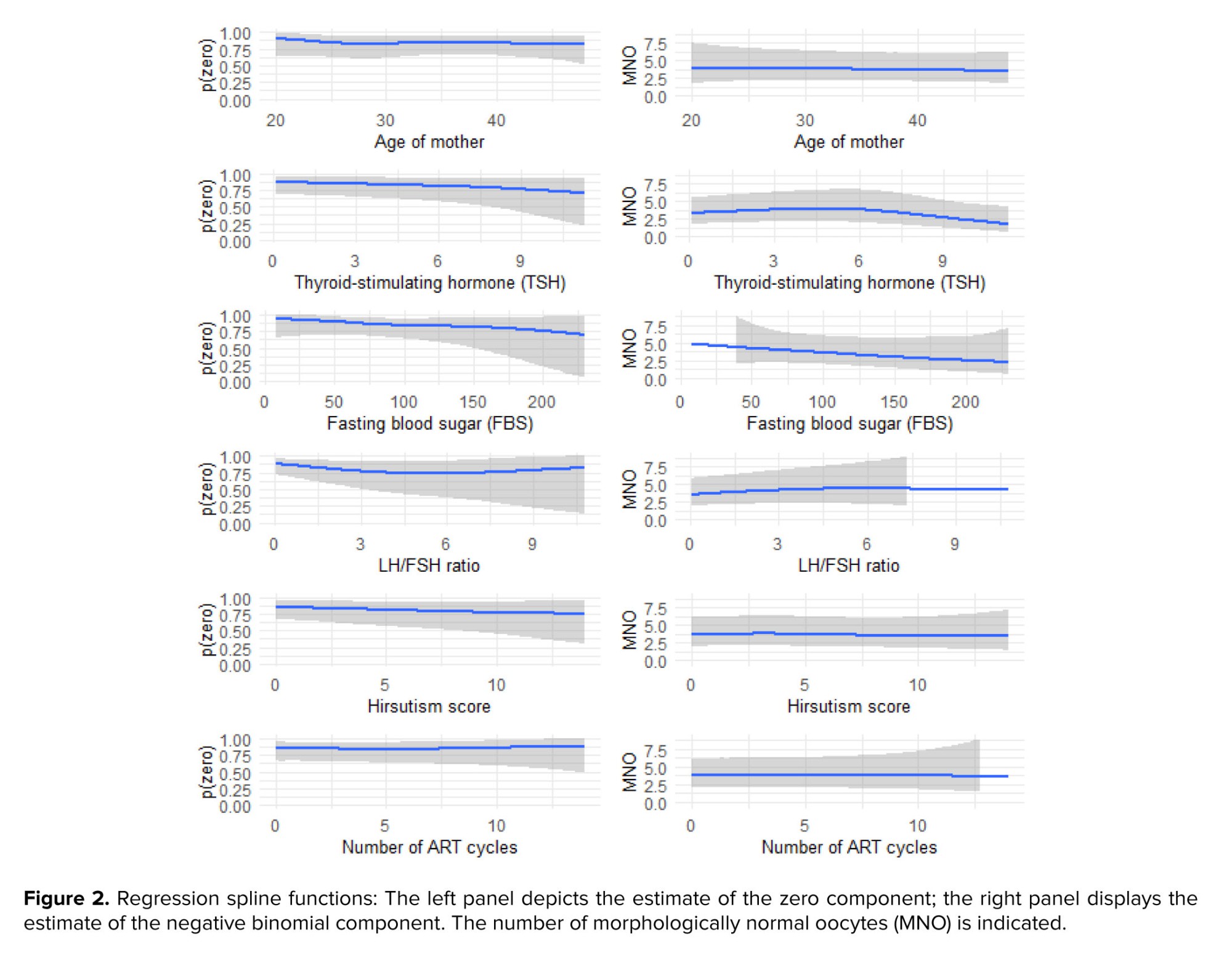
The SZINB regression model consists of 3 components: an NB component reporting incidence rate ratios for count data, a logistic component reporting odd ratios for zero-inflation, and smoothing splines for modeling non-linear relationships. In this model, all quantitative variables that demonstrated a nonlinear relationship with the response in either or both parts of the NB or structural zeros were included as splines in the multiple model (Table IV). The results and graphs of the SZNB model within a multiple context are presented in table IV and figure 2.
From the age of 20 to about 32, the probability of structural zeros (due to genetic and unknown disorders that never lead to the production of high-quality oocytes) gradually decreases. This probability remains stable between ages 32 and 38 and then increases again. As maternal age increases, the number of MNOs decreases, which is particularly noticeable at older ages and affects the quality of the oocytes. The second chart on the left shows that as TSH levels increase from about 0-6, the probability of structural zeros gradually decreases. From levels 6-8, this probability remains relatively stable and then decreases again. As TSH levels increase from 0-3, the number of MNO gradually increases. From levels 3-6, this number remains stable, and after level 6, it gradually decreases.
These results indicate an improvement in oocyte quality at lower TSH levels and a decline in quality at higher levels. Increased FBS reduces the probability of structural zeros, especially at lower blood sugar ranges (below 150), where this reduction is more pronounced. Additionally, increased FBS has a negative impact on the number of MNO. Increasing the LH/FSH ratio up to level 5 reduces the probability of structural zeros. From levels 5-6, this probability remains stable and then increases after level 6. As the LH/FSH ratio increases from 0-5, the number of MNO gradually increases, but after level 5, it decreases with a gentle slope.
The fifth chart on the left shows that as the hirsutism score increases, the probability of structural zeros in individuals decreases. As the hirsutism score increases, the number of high-quality oocytes remains relatively stable with slight variation. However, after a score of 5, the number of MNO gradually decreases, eventually reaching about 2.5. These results indicate the negative impact of the hirsutism score on the number of high-quality oocytes, especially at higher scores. As the number of ART cycles increases up to about 5, the probability of structural zeros decreases. This probability remains stable from 5-6 and then increases after level 6. As the number of ART cycles increases up to about 3, the number of MNO clearly increases. From level 3 to about 6, the number of good oocytes remains relatively stable, and after 6, this number gradually decreases (Figure 2).
4. Discussion
This study explored factors influencing MNO in infertile women visiting the Royan Institute, Tehran, Iran emphasizing the efficacy of nonlinear models like SZINB. Unlike linear models such as ZIP, the SZINB effectively captured nonlinear relationships between MNO and variables like age, FBS, and clinical factors. This suggests that variables traditionally analyzed linearly may exhibit nonlinear patterns, better addressed by SZINB. The study highlights the importance of nonlinear modeling in clinical research, particularly for fertility treatment, where nuanced variable-response relationships are critical. To our knowledge, this is the first study to apply SZINB to MNO analysis, unlike prior studies that used ZIP for count data with excess zeros (16, 17).
ZIP identified a significant link between LH/FSH and reduced probability of structural zeros, while SZINB revealed a nonlinear relationship, indicating an optimal LH/FSH range beyond which the association reverses. This aligns with research linking imbalanced LH/FSH to poor oocyte quality (18, 19).
Hirsutism and PCOS negatively impacted oocyte quality and decreased the likelihood of structural zeros, consistent with studies identifying PCOS as a primary cause of infertility due to hormonal imbalances and elevated androgen levels (9). Increased hirsutism scores correlated with a gradual decline in high-quality oocytes, supported by research on PCOS symptom severity (20). Previous ovarian surgeries, particularly endometrioma-related procedures, also reduced the likelihood of structural zeros, highlighting the need for cautious surgical decisions (21-24).
Both ZIP and SZINB confirmed a significant relationship between PCOS in either ovary and reduced MNO rates. The SZINB further indicated that other ovarian abnormalities significantly lowered the probability of producing high-quality oocytes, emphasizing the critical role of ovarian health in fertility outcomes (25, 26). No linear relationship was observed between age and MNO in the ZIP, in line with studies suggesting oocyte quantity may not always decline with age (27). However, SZINB revealed a nonlinear relationship, with younger ages linked to a lower probability of structural zeros and older ages showing a significant decline in MNO, particularly beyond 35, due to genetic damage accumulation (28, 29). ZI models have been used to investigate oocytes and embryos in relation to quality and age, showing that increasing age may negatively affect MNO (30). Similar nonlinear patterns were observed with TSH, where higher levels reduced oocyte quality, aligning with studies linking elevated TSH to reduced fertility (31, 32). The number of ART cycles showed no linear relationship with MNO in the ZIP, but SZINB identified a nonlinear pattern: MNO initially increased, stabilized, then decreased with more cycles, consistent with studies noting initial improvements followed by declines in oocyte quality (33).
FBS levels were measured at the patients' first visit. For patients requiring blood sugar control, infertility treatment commenced once the FBS reached the normal range. Results indicate an inverse relationship between FBS and the MNO. Medical interventions in individuals with disorders like PCOS, high hirsutism scores, or a history of ovarian surgery may help reduce severe disorders and decrease the likelihood of structural zeros. However, these disorders negatively impact oocyte quality. To our knowledge, no previous studies have reported the results of medical interventions on MNO based on structural zero models.
Several limitations should be considered. First, we could not examine the impact of cigarette and alcohol consumption on MNO, as only 3 participants reported smoking, likely due to low consumption rates among Iranian women and potential under-reporting influenced by cultural sensitivities. The study focused on oocyte quality, a critical but singular component of infertility treatment outcomes, which are also influenced by sperm quality, fertilization success, and embryo development. This targeted approach, while offering valuable insights into oocyte morphology, excludes other factors essential to overall treatment success. Data on anti-müllerian hormone levels were unavailable for most participants (96.6%), preventing inclusion in the analyses. These limitations may have impacted the study’s ability to fully explore all factors related to oocyte quality. Future studies should collect more comprehensive data on AMH and other relevant variables to enhance understanding of oocyte quality determinants.
5. Conclusion
The analysis revealed that ovarian surgery history and the presence of disorders in either the ovary status (right or left), along with PCOS, are significantly correlated with the rate of MNO in both ZIP and SZINB models. Furthermore, maternal age, elevated LH/FSH ratio, increased TSH levels, higher FBS, hirsutism scores, and the number of ART cycles exhibited non-linear associations with MNO. These findings emphasize the critical role of both medical history and hormonal factors in influencing oocyte quality, highlighting the need for targeted interventions in managing infertility.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
Z. Asadollahi, as the principal investigator, designed the study, conducted data analysis, and prepared the manuscript. S. Haji-Maghsoudi collaborated in data analysis and participated in the review and final editing process. A. Haghdoost, as scientific and clinical advisor, assisted in writing and the review and final editing process of the manuscript, while also facilitating communication with Royan Research Institute, Tehran, Iran. M. Ashrafi supervised all study phases and conducted the review and final editing process of the manuscript. P. Eftekhari-Yazdi participated in oocyte quality assessment and performed the review and final editing of the manuscript.
Y. Jahani, as the primary corresponding author and supervisor from Kerman University of Medical Sciences, Kerman, Iran made significant contributions to the study design, data analysis, and manuscript writing. A. Ghaheri, as the secondary corresponding author from Royan Institute, Tehran, Iran contributed to writing the article, facilitated access to data from Royan Institute, Tehran, Iran and participated in critical revision and final editing of the manuscript.
Acknowledgments
We sincerely appreciate the Royan Institute, Tehran, Iran, for providing the essential data for this study. This research was financially supported by Kerman University of Medical Sciences, Kerman, Iran (grant number: 402000084). All aspects of manuscript preparation were performed by the authors. AI-assisted tools (MONICA) were used only for English language polishing in the final revision. We also extend our heartfelt thanks to the women experiencing infertility who participated in this research and shared their experiences with us.
Conflict of Interest
The authors declare that there is no conflict of interest.
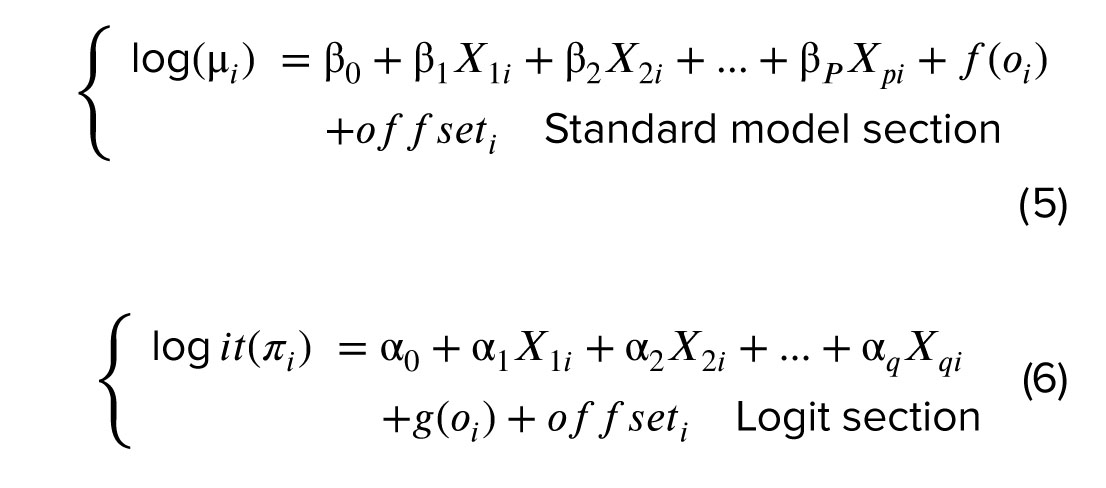
In this model, where i=1, 2, …, n represents the individuals, βj( j= 0, 1, 2,…, p) and αk (k= 0, 1, 2,…, q) are the coefficients for the Poisson or NB and logistic components, respectively. The nature of the relationship between these coefficients in the model is linear. However, the relationship involving oi in the model is nonlinear. It can represent a sum of unspecified functions of multiple variables, with functions f and g serving as the smoothing functions for oi in the Poisson or NB and logistic components, respectively.
2.4. Model evaluation and selection
The average rate of MNO, from women visiting the Royan Institute, Tehran, Iran was modeled as a function of age, BMI, and other explanatory variables using Poisson regression, NB regression, ZIP regression, ZINB regression, and SZIP or SZINB regression models. The same explanatory variables were included in both components (the logit and count components) of the ZI models. The Vuong test, along with the Akaike Information Criterion (AIC) and Bayesian Information Criterion (BIC), was used to compare the models and select the one that offers the better fit to the data, balancing model accuracy and complexity. The Vuong test produces a p-value to compare the fit of 2 non-nested models. At the 5% significance level, the interpretation was as follows: if the p < 0.05, the difference between the models was statistically significant, with a positive test statistic indicating that the first model provides the better fit and a negative test statistic indicating that the second model provides the better fit. For p-value greater than or equal to 0.05, no significant difference was observed between the models, and selection may rely on AIC or BIC for further comparison.
2.5. Ethical Considerations
This study was approved by the Ethics Committee of Kerman University of Medical Sciences, Kerman, Iran (Code: IR.KMU.REC.1402.071). Informed written consent was obtained from all participants at the time of their initial treatment (January 2012-December 2013) at Royan Institute, Tehran, Iran as part of the routine clinical protocol before any ART procedure. Patients were informed that their anonymized clinical and laboratory data might be used for research purposes and published only in aggregate form, with no possibility of individual identification.
2.6. Statistical Analysis
The number of MNOs is a count variable. Poisson and NB regression were used to model the count variables. In this study, the outcome variable of interest was the number of MNO, which was zero in 71.2% of cases. Due to the excess of zeros, ZI models and semiparametric ZI models were used to model these count data. The Vuong test, along with the AIC and BIC, was used for model selection to ensure robust evaluation of model fit, considering both predictive accuracy and model complexity. The Vuong test provides a significant z-test that is utilized for selecting the best linear models. To check for overdispersion in the models in R, the dispersion test function from the AER package was used. This test evaluates the null hypothesis of equidispersion in Poisson models against the alternative hypothesis of overdispersion or underdispersion. In NB modeling, overdispersion was assessed indirectly through model parameters. All models and statistical computations were executed using R version 4.4.1. The significance level of all tests was considered to be 0.05.
3. Results
Out of the 950 infertile women whose medical records were initially extracted, 943 had complete oocyte morphology data and were included in the final analysis. Among these 943 women referred to the Royan Institute, Tehran, Iran for ART, MNO were zero in 671 cases (71.2%) and the mean (standard deviation) MNO was 1.42 (2.80), range 0-15, interquartile range = 1 (Figure 1).
The mean age of the women was 34.41 ± 5.30 yr, and the mean BMI was 25.87 ± 3.80 kg/m². Detailed information on other variables, including hormone levels and ovarian conditions, is provided in table I.
Based on the results of the Vuong test statistics, along with AIC and BIC, the Poisson regression provided the worst fit for the data among all regression models. According to both AIC and BIC criteria, the ZIP model was the best fit among linear models, while among nonlinear models, the SZINB model provided the best fit (Table II). These results confirm the robustness of the ZIP and SZINB models in capturing the complex relationships in the data.
The univariate ZIP model analysis revealed that the LH/FSH ratio, hirsutism score, ovarian surgery history, and the presence of PCOS in ovary status were significantly associated with a decreased likelihood of structural zeros (i.e., cases where no high-quality oocytes are produced due to genetic or unknown disorders) in the logistic component. This suggests that women with these conditions, likely due to targeted medical interventions such as hormonal treatments, have a higher probability of producing at least one MNO, potentially improving fertility outcomes. In the Poisson component of the univariate analysis, no factors were significantly associated with the rate of MNO (Table III). In the multiple ZIP analysis, ovarian surgery history remained significantly associated with a decreased likelihood of structural zeros in the logistic component, implying that women with a history of ovarian surgery, possibly due to enhanced medical management, are more likely to produce at least one MNO even after adjusting for other variables. In contrast, in the Poisson component, the presence of PCOS was significantly associated with a reduced rate of MNO, indicating that PCOS negatively impacts the quantity of high-quality oocytes among women who produce at least some MNO (Table III). These findings underscore the role of medical interventions in mitigating the risk of complete absence of high-quality oocytes and highlight the specific negative impact of PCOS on oocyte quantity.
The univariate SZINB model analysis revealed that ovarian surgery history and the presence of PCOS were significantly associated with a decreased likelihood of structural zeros (i.e., cases where no high-quality oocytes are produced due to genetic or unknown disorders) in the logistic component. This suggests that women with these conditions, likely due to targeted medical interventions such as hormonal treatments, have a higher probability of producing at least one MNO, potentially improving fertility outcomes. In contrast, other ovarian abnormalities were significantly associated with a reduced rate of MNO in the NB component, indicating a negative impact on the quantity of MNO (Table IV). In the multiple SZINB analysis, ovarian surgery history continued to show a significant association with a decreased likelihood of structural zeros in the logistic component, implying that women with a history of ovarian surgery, possibly due to enhanced medical management, are more likely to produce at least one MNO even after adjusting for other variables. Meanwhile, other ovarian abnormalities remained significantly associated with a reduced rate of MNO in the NB component, indicating a negative impact on the quantity of MNO (Table IV).
The SZINB regression model consists of 3 components: an NB component reporting incidence rate ratios for count data, a logistic component reporting odd ratios for zero-inflation, and smoothing splines for modeling non-linear relationships. In this model, all quantitative variables that demonstrated a nonlinear relationship with the response in either or both parts of the NB or structural zeros were included as splines in the multiple model (Table IV). The results and graphs of the SZNB model within a multiple context are presented in table IV and figure 2.
From the age of 20 to about 32, the probability of structural zeros (due to genetic and unknown disorders that never lead to the production of high-quality oocytes) gradually decreases. This probability remains stable between ages 32 and 38 and then increases again. As maternal age increases, the number of MNOs decreases, which is particularly noticeable at older ages and affects the quality of the oocytes. The second chart on the left shows that as TSH levels increase from about 0-6, the probability of structural zeros gradually decreases. From levels 6-8, this probability remains relatively stable and then decreases again. As TSH levels increase from 0-3, the number of MNO gradually increases. From levels 3-6, this number remains stable, and after level 6, it gradually decreases.
These results indicate an improvement in oocyte quality at lower TSH levels and a decline in quality at higher levels. Increased FBS reduces the probability of structural zeros, especially at lower blood sugar ranges (below 150), where this reduction is more pronounced. Additionally, increased FBS has a negative impact on the number of MNO. Increasing the LH/FSH ratio up to level 5 reduces the probability of structural zeros. From levels 5-6, this probability remains stable and then increases after level 6. As the LH/FSH ratio increases from 0-5, the number of MNO gradually increases, but after level 5, it decreases with a gentle slope.
The fifth chart on the left shows that as the hirsutism score increases, the probability of structural zeros in individuals decreases. As the hirsutism score increases, the number of high-quality oocytes remains relatively stable with slight variation. However, after a score of 5, the number of MNO gradually decreases, eventually reaching about 2.5. These results indicate the negative impact of the hirsutism score on the number of high-quality oocytes, especially at higher scores. As the number of ART cycles increases up to about 5, the probability of structural zeros decreases. This probability remains stable from 5-6 and then increases after level 6. As the number of ART cycles increases up to about 3, the number of MNO clearly increases. From level 3 to about 6, the number of good oocytes remains relatively stable, and after 6, this number gradually decreases (Figure 2).







4. Discussion
This study explored factors influencing MNO in infertile women visiting the Royan Institute, Tehran, Iran emphasizing the efficacy of nonlinear models like SZINB. Unlike linear models such as ZIP, the SZINB effectively captured nonlinear relationships between MNO and variables like age, FBS, and clinical factors. This suggests that variables traditionally analyzed linearly may exhibit nonlinear patterns, better addressed by SZINB. The study highlights the importance of nonlinear modeling in clinical research, particularly for fertility treatment, where nuanced variable-response relationships are critical. To our knowledge, this is the first study to apply SZINB to MNO analysis, unlike prior studies that used ZIP for count data with excess zeros (16, 17).
ZIP identified a significant link between LH/FSH and reduced probability of structural zeros, while SZINB revealed a nonlinear relationship, indicating an optimal LH/FSH range beyond which the association reverses. This aligns with research linking imbalanced LH/FSH to poor oocyte quality (18, 19).
Hirsutism and PCOS negatively impacted oocyte quality and decreased the likelihood of structural zeros, consistent with studies identifying PCOS as a primary cause of infertility due to hormonal imbalances and elevated androgen levels (9). Increased hirsutism scores correlated with a gradual decline in high-quality oocytes, supported by research on PCOS symptom severity (20). Previous ovarian surgeries, particularly endometrioma-related procedures, also reduced the likelihood of structural zeros, highlighting the need for cautious surgical decisions (21-24).
Both ZIP and SZINB confirmed a significant relationship between PCOS in either ovary and reduced MNO rates. The SZINB further indicated that other ovarian abnormalities significantly lowered the probability of producing high-quality oocytes, emphasizing the critical role of ovarian health in fertility outcomes (25, 26). No linear relationship was observed between age and MNO in the ZIP, in line with studies suggesting oocyte quantity may not always decline with age (27). However, SZINB revealed a nonlinear relationship, with younger ages linked to a lower probability of structural zeros and older ages showing a significant decline in MNO, particularly beyond 35, due to genetic damage accumulation (28, 29). ZI models have been used to investigate oocytes and embryos in relation to quality and age, showing that increasing age may negatively affect MNO (30). Similar nonlinear patterns were observed with TSH, where higher levels reduced oocyte quality, aligning with studies linking elevated TSH to reduced fertility (31, 32). The number of ART cycles showed no linear relationship with MNO in the ZIP, but SZINB identified a nonlinear pattern: MNO initially increased, stabilized, then decreased with more cycles, consistent with studies noting initial improvements followed by declines in oocyte quality (33).
FBS levels were measured at the patients' first visit. For patients requiring blood sugar control, infertility treatment commenced once the FBS reached the normal range. Results indicate an inverse relationship between FBS and the MNO. Medical interventions in individuals with disorders like PCOS, high hirsutism scores, or a history of ovarian surgery may help reduce severe disorders and decrease the likelihood of structural zeros. However, these disorders negatively impact oocyte quality. To our knowledge, no previous studies have reported the results of medical interventions on MNO based on structural zero models.
Several limitations should be considered. First, we could not examine the impact of cigarette and alcohol consumption on MNO, as only 3 participants reported smoking, likely due to low consumption rates among Iranian women and potential under-reporting influenced by cultural sensitivities. The study focused on oocyte quality, a critical but singular component of infertility treatment outcomes, which are also influenced by sperm quality, fertilization success, and embryo development. This targeted approach, while offering valuable insights into oocyte morphology, excludes other factors essential to overall treatment success. Data on anti-müllerian hormone levels were unavailable for most participants (96.6%), preventing inclusion in the analyses. These limitations may have impacted the study’s ability to fully explore all factors related to oocyte quality. Future studies should collect more comprehensive data on AMH and other relevant variables to enhance understanding of oocyte quality determinants.
5. Conclusion
The analysis revealed that ovarian surgery history and the presence of disorders in either the ovary status (right or left), along with PCOS, are significantly correlated with the rate of MNO in both ZIP and SZINB models. Furthermore, maternal age, elevated LH/FSH ratio, increased TSH levels, higher FBS, hirsutism scores, and the number of ART cycles exhibited non-linear associations with MNO. These findings emphasize the critical role of both medical history and hormonal factors in influencing oocyte quality, highlighting the need for targeted interventions in managing infertility.
Data Availability
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
Author Contributions
Z. Asadollahi, as the principal investigator, designed the study, conducted data analysis, and prepared the manuscript. S. Haji-Maghsoudi collaborated in data analysis and participated in the review and final editing process. A. Haghdoost, as scientific and clinical advisor, assisted in writing and the review and final editing process of the manuscript, while also facilitating communication with Royan Research Institute, Tehran, Iran. M. Ashrafi supervised all study phases and conducted the review and final editing process of the manuscript. P. Eftekhari-Yazdi participated in oocyte quality assessment and performed the review and final editing of the manuscript.
Y. Jahani, as the primary corresponding author and supervisor from Kerman University of Medical Sciences, Kerman, Iran made significant contributions to the study design, data analysis, and manuscript writing. A. Ghaheri, as the secondary corresponding author from Royan Institute, Tehran, Iran contributed to writing the article, facilitated access to data from Royan Institute, Tehran, Iran and participated in critical revision and final editing of the manuscript.
Acknowledgments
We sincerely appreciate the Royan Institute, Tehran, Iran, for providing the essential data for this study. This research was financially supported by Kerman University of Medical Sciences, Kerman, Iran (grant number: 402000084). All aspects of manuscript preparation were performed by the authors. AI-assisted tools (MONICA) were used only for English language polishing in the final revision. We also extend our heartfelt thanks to the women experiencing infertility who participated in this research and shared their experiences with us.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Fertility & Infertility
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |