Thu, May 21, 2026
[Archive]
Volume 24, Issue 1 (January 2026)
IJRM 2026, 24(1): 23-36 |
Back to browse issues page
Ethics code: IR.MODARES.REC.1400.209
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Sadeghi E, Moazzeni S, Salehnia M. Fabrication and characterization of decellularized human endometrial hydrogel as a scaffold for in vitro embryo implantation: An experimental study. IJRM 2026; 24 (1) :23-36
URL: http://ijrm.ir/article-1-3691-en.html
URL: http://ijrm.ir/article-1-3691-en.html
1- Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran. & Department of Anatomical Sciences, School of Medicine, Ilam University of Medical Sciences, Ilam, Iran.
2- Department of Biomedical Engineering, Rutgers, The State University of New Jersey, Piscataway, NJ, USA.
3- Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran. & Discipline of Biomedical Engineering, School of Electrical and Electronic Engineering, The University of Adelaide, Adelaide, Australia. ,salehnim@modares.ac.ir
2- Department of Biomedical Engineering, Rutgers, The State University of New Jersey, Piscataway, NJ, USA.
3- Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran. & Discipline of Biomedical Engineering, School of Electrical and Electronic Engineering, The University of Adelaide, Adelaide, Australia. ,
Keywords: Decellularized extracellular matrix, Endometrium, Hydrogel, Embryo implantation, Mesenchymal stem cells.
Full-Text [PDF 6189 kb]
(269 Downloads)
| Abstract (HTML) (216 Views)
5. Conclusion
Full-Text: (36 Views)
1. Introduction
Endometrial tissue engineering is an emerging field dedicated to creating functional uterine tissues using advanced techniques such as organoid culture, decellularized tissues, and hydrogels (1-4). This field has promising clinical applications, including treatments for uterine diseases and injuries, addressing infertility, and supporting in vitro research in reproductive science, such as developing embryo implantation models (1, 5, 6). Decellularized endometrial tissue (DET) is derived from endometrial tissue to remove cellular components while preserving the extracellular matrix (ECM), which retains the biochemical composition, structure, and mechanical properties of the native tissue (2, 7-12). The decellularization process typically involves physical, chemical, and enzymatic methods, such as using detergents and enzymes, followed by validation to confirm the absence of cellular DNA and the retention of ECM components like collagen and glycosaminoglycans (3, 11, 13, 14). DET is highly biocompatible, reducing the risk of immune rejection, and supports cell function similar to that in the native endometrial environment. It is widely studied in tissue engineering as a scaffold for regenerating endometrial tissue in conditions like Asherman’s syndrome or uterine factor infertility and in disease models for endometrial disorders, including endometriosis and cancer (1, 5). Recently, we have developed a protocol for the decellularization of human endometrial tissue fragments that effectively preserves the ECM components, closely resembling those of intact tissue (10, 15).
Hydrogels derived from the decellularized endometrium are biomaterials designed to mimic the native ECM, providing a supportive, 3-dimensional environment for cell growth, differentiation, and tissue regeneration (16-21). The decellularized ECM was processed into a hydrogel form, often through solubilization and crosslinking techniques, resulting in a biocompatible, injectable material (21). Endometrial-derived hydrogels are rich in ECM proteins like collagen, laminin, and fibronectin, as well as bioactive molecules that promote cell adhesion, proliferation, and angiogenesis (18, 22). These properties make them particularly suitable for applications in reproductive medicine (7, 20, 23, 24). Hydrogel is also compatible with encapsulation of endometrial cells or stem cells, providing a scaffold that supports cellular function and integration (16, 19, 23).
Endometrial stem cells (ESCs) are a population of multipotent stem cells found within the endometrial tissue of the uterus, playing a crucial role in the dynamic regenerative processes of the endometrium during the menstrual cycle and following injury (25-28). These cells are characterized by their ability to self-renew and differentiate into various cell types, including epithelial, stromal, and possibly endothelial cells (29). ESCs are identified by specific markers and are being investigated for their potential in tissue engineering, bioengineering of uterine constructs, and disease modeling (25-27). Research has focused on utilizing ESCs with various scaffolds to reconstruct the endometrium in 3-D culture systems, with some methods successfully developing in vitro embryo implantation models (15, 30, 31).
Embryo implantation models using biomaterials and scaffolds are innovative platforms designed to study the complex process of embryo implantation in vitro, offering insights into reproductive biology and potential solutions for infertility (15, 32, 33). The scaffold is seeded with endometrial cells, including epithelial and stromal cells, or other types of stem cells, to recreate the functional layers of the endometrium in co-culture with the embryo (6, 33).
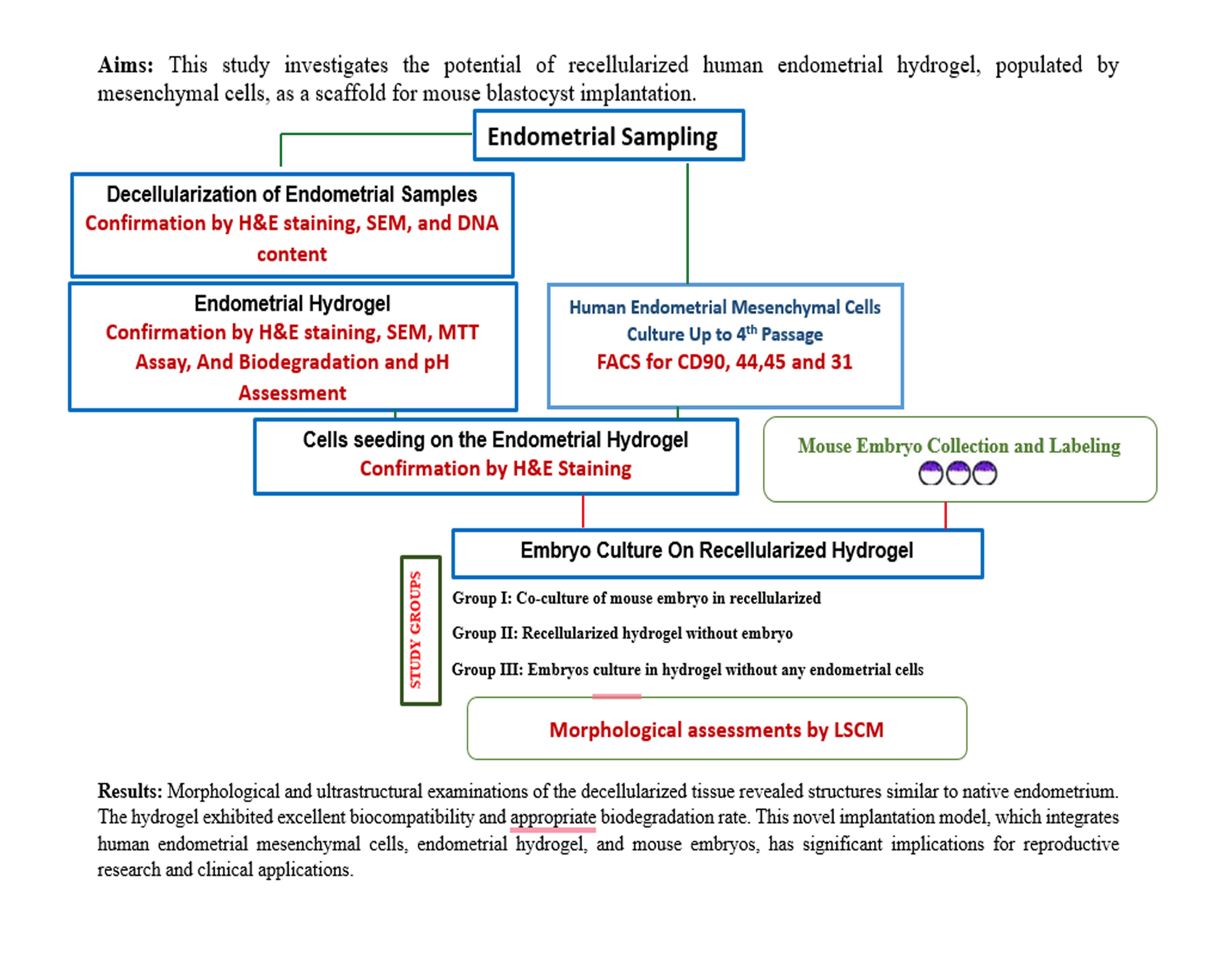
This study aimed to evaluate the potential of recellularized human endometrial hydrogel, populated by endometrial mesenchymal cells, as a scaffold for mouse blastocyst implantation as an in vitro model.
2. Materials and Methods
2.1. Study design
This experimental study was done in 2023 at Anatomical Sciences Department of Tarbiat Modares University, Tehran, Iran and involved the preparation of a hydrogel using decellularized human endometrium, with evaluations of its morphology, ultrastructure, and residual DNA content. The biocompatibility, biodegradation, and pore sizes of the hydrogel were also analyzed. Human ESCs were isolated from fresh tissue samples, cultured up to the fourth passage, and characterized using flow cytometry. The hydrogel was recellularized by seeding human ESCs for 7 days, followed by co-culturing mouse blastocysts on these hydrogels for 48 hr. Blastocyst implantation within the hydrogels was assessed using laser confocal scanning microscopy. All materials were sourced from Sigma Aldrich (London, UK), and experiments were performed in triplicate unless otherwise stated.
2.2. Human endometrial tissue collection
Endometrial tissues were collected from fertile women aged 25-35 yr (n = 6) who had been diagnosed with gynecological conditions unrelated to endometrial disease. None of the participants had received exogenous hormone therapy 3 months before sampling. The tissues were transferred under sterile conditions, dissected into 2-3 cm² fragments, and preserved in Dulbecco’s Modified Eagle’s Medium (DMEM) on ice. A portion of the tissue fragments was assessed for normal morphology, while the remaining fragments were used for decellularization, cell isolation, and in vitro culture (30).
2.3. Tissue decellularization
The endometrial tissues were decellularized using a previously established method (15). Briefly, the tissue samples underwent 3 successive cycles of freezing at -80°C and thawing at 37°C, followed by treatment with 1% Triton X-100 for 15 hr and 1% sodium dodecyl sulfate for 72 hr at room temperature. The samples were then washed 6 times with phosphate-buffered saline (PBS), each wash lasting 8 hr. Following this process, the decellularized tissues were analyzed for DNA content, subjected to hematoxylin and eosin (H&E) staining, and examined using scanning electron microscopy (SEM).
2.3.1. Morphological study
The control tissue samples and decellularized endometrial fragment (n = 3/group) were fixed in 10% formaldehyde, dehydrated using a graded series of ethanol, and subsequently embedded in paraffin wax. Sections with 5 μm thickness were then prepared and stained using the H&E procedure. The stained sections were observed under a light microscope.
2.3.2. SEM
To characterize the ultrastructural properties of the decellularized tissues in comparison with the native control, 3 samples from each group were fixed in 2.5% glutaraldehyde, followed by dehydration using a graded series of ethanol and overnight drying with hexamethyldisilazane. The samples were then coated with gold and examined using SEM (VEGA/TESCAN-XMU, Czech Republic).
2.3.3. DNA content analysis
Total DNA from decellularized endometrial samples and control tissue (n = 3/group) was extracted using a TRIzol® kit (Invitrogen, UK) according to the standardized protocol. The samples were weighed, treated with TRIzol® reagent, and subjected to chloroform extraction. The DNA layer was then isolated by centrifugation at 2000 g for 10 min at 4°C, followed by resuspension in NaOH and further centrifugation at 12,000 g for 10 min at 4°C. The DNA concentration was determined using UV spectrophotometry (Eppendorf, Germany) at 260 nm.
2.4. Endometrial cells isolation, culture, and labeling
Endometrial stromal cells were isolated and cultured according to a previously described method (30). Briefly, endometrial tissue samples (n = 6) were cut into 1 mm fragments after rinsing in PBS. They were placed in DMEM/F-12 medium supplemented with 100 mg/ml Penicillin G sodium, 100 mg/ml Streptomycin sulfate, and 10% fetal bovine serum. The fragments were then treated with collagenase type I (300 μg/ml) and deoxyribonuclease type I (40 μg/ml) for 1-2 hr. The resulting endometrial stromal cells were cultured up to the fourth passage at 37°C in a 5% CO2 incubator. For characterization of cultured endometrial cells, they were suspended (1×10⁵) in 50 μl of PBS and incubated with FITC-conjugated antibodies (anti-human CD90, CD44, and CD45 as mesenchymal and hematopoietic markers, respectively) and phycoerythrin-conjugated antibodies (anti-human CD31 as an endothelial marker) at a 1:50 dilution at 4°C for 45 min. After washing in PBS, the cells were analyzed using a fluorescence-activated cell sorting Calibur apparatus (Becton Dickinson, USA).
The cultured cells were used for the 3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide (MTT) assay and labeling with di-alkyl indocarbocyanine (DiI) according to our previous study (15). Briefly, a suspension of 1×10⁵ cells/ml was incubated with 2 µg/mL DiI for 20 min at 4°C in the dark, followed by washing 3 times in culture media. The labeled cells were then used for cell seeding on the hydrogel.
2.5. Preparation of human endometrial hydrogel
The DET was cut into several fragments (1×1 mm) and washed 4 times in ice-cold PBS to remove residual detergents. The fragments were lyophilized overnight (0.30-0.31 gr/each), milled with dry ice, frozen in liquid nitrogen, ground, and stored at -20°C. The resulting powder (1.45 gr) was solubilized using a modified protocol, suspending 1% (w/v) powder in 0.01 M HCl with 0.1% (w/v) pepsin, followed by digestion for 48 hr, neutralization in 10% (v/v) 0.1 M NaOH, aliquotation, flash freezing, and storage at -80°C. For coating, the hydrogel solution was added to a 24-well culture plate and left overnight at 4°C. Then washed with PBS, and culture medium (DMEM/F-12, Invitrogen, UK) (8).
2.5.1. Endometrial hydrogel morphology and ultrastructure
Endometrial hydrogel samples (n = 3) were fixed and processed for H&E staining, followed by observation under the light microscope, as previously described for decellularized tissue. To examine the ultrastructure of the endometrial hydrogels, the samples were freeze-dried (Alpha 2-4 LDplus, Martin Christ, Germany) for 12 hr. Dried samples were then segmented, coated with a thin layer of gold, and observed under SEM. Multiple micrographs were captured from different regions of the endometrial hydrogels. The average pore size and porosity of the hydrogels were measured from SEM images using ImageJ 1.33U software (National Institutes of Health, USA).
2.5.2. Biocompatibility (MTT assay)
The MTT assay was used to evaluate the biocompatibility of the prepared endometrial hydrogels. The human ESCs were cultured at a density of 1×105 cells in a 96-well plate, with or without pre-loaded endometrial hydrogel. After incubation for 24 and 72 hr, the supernatant was discarded, and the cells were exposed to MTT solution (0.5 mg/mL in PBS) for 4 hr. The resulting formazan product was dissolved in dimethyl sulfoxide for 20 min. Finally, the amount of formazan was quantified by measuring its absorbance at 540 nm using a microplate reader (Biochrom, Germany).
2.5.3. Biodegradation and pH assessment
The endometrial hydrogel samples (n = 3) with 5×5×2 mm³ size were subjected to a 72-hr degradation study. Initially, the hydrogel samples were lyophilized, weighed using a digital analytical balance (Hawach Scientific, China), and placed in a controlled PBS solution with a pH of 7.4, as per the previous study (34). The PBS was replaced every 12 hr to prevent the accumulation of degradation by products. After 24, 48, and 72 hr of incubation, the samples were lyophilized again, and their weight were measured, respectively. The percentage weight loss of the samples over time was calculated using the formula:

Dried hydrogel samples were transferred into 5 mL microtubes, each containing 4 mL of PBS at pH 7.4. The microtubes were incubated at 37°C, and pH measurements were taken at 2, 4, 6, 8, and 24-hr intervals using a calibrated pH meter (APERA, PH850, USA).
2.5.4. Recellularization of endometrial hydrogel
Hydrogels were placed in individual wells of a 24-well plate and incubated with culture medium (DMEM/F-12, as described earlier) for 24 hr before cell seeding. Subsequently, 1×105 labelled cells were added to each well and incubated at 37°C in a 5% CO2 atmosphere for 1 wk. The medium was changed every 2 days. Also, the same hydrogel series without cell seeding was considered the control group. The recellularized and control hydrogels were then used for subsequent experiments.
2.6. Blastocyst collection and labelling
Adult female (n = 10) and male (n = 5) mice were maintained under standard laboratory conditions, based on the laboratory animal care ethics of Tarbiat Modares University, Tehran, Iran. To induce superovulation, female mice were administered a single intraperitoneal dose of 7.5 IU pregnant mare serum gonadotropin (Folligon, Intervet, Australia), followed by a subsequent intraperitoneal dose of 10 IU human chorionic gonadotropin (Pregnyl, Netherlands). Afterward, females were mated with fertile males. Approximately 120 hr post-human chorionic gonadotropin injection, blastocysts were collected by flushing from the uterine horns. To enable visualization and tracking of the blastocysts within the endometrial hydrogel, the blastocysts (n = 60) were labelled with Hoechst 34580 dye solution (1 ng/ml in distilled water) for 20 min, followed by washing with culture medium (DMEM/F-12).
2.6.1. Blastocysts co-culture in recellularized hydrogel
One-week post-recellularization of the hydrogel, we applied 3 study groups. Group I: co-culture of mouse embryo in hydrogels that recellularized with endometrial mesenchymal cells. Group II: the recellularized hydrogel with endometrial mesenchymal cells in the absence of a mouse embryo. Group III: culture of mouse embryos in hydrogel alone without any endometrial cells. In groups I and III, the labelled mouse blastocysts were introduced onto the surface of each endometrial hydrogel-loaded well (n = 2 embryos per well and 30 in total) and cultured for 48 hr at 37°C in 5% CO2. In group II, the recellularized endometrial hydrogels, without blastocysts, were subjected to similar culture conditionsAt least 3 wells from each experimental and control group were observed under laser confocal scanning microscopy (LCSM, Zeiss LSM800, Japan), and embryo penetration into the scaffold was evaluated at excitation and emission wavelengths of 392 and 440 nm for Hoechst-labeled embryonic cells and 546 and 563 nm for DiI-labeled endometrial cells.
2.7. Ethical Considerations
The study adhered to the ethical guidelines of the Tarbiat Modares University Ethics Committee, Tehran, Iran (Code: IR.MODARES.REC.1400.209). An informed consent was obtained from participants for the use of their tissues.
2.8. Statistical Analysis
All experiments were replicated at least 3 times. Data analysis was performed using GraphPad Prism Software 8 (Denver, CO, USA). The normality of the data was assessed using the Shapiro-Wilk test. To compare DNA content between groups, one-way ANOVA was used followed by Tukey's post-hoc test. The independent sample t test was applied to compare MTT assay results between groups. Statistical significance was set at a p < 0.05.
3. Results
3.1. Characterization of decellularized tissue
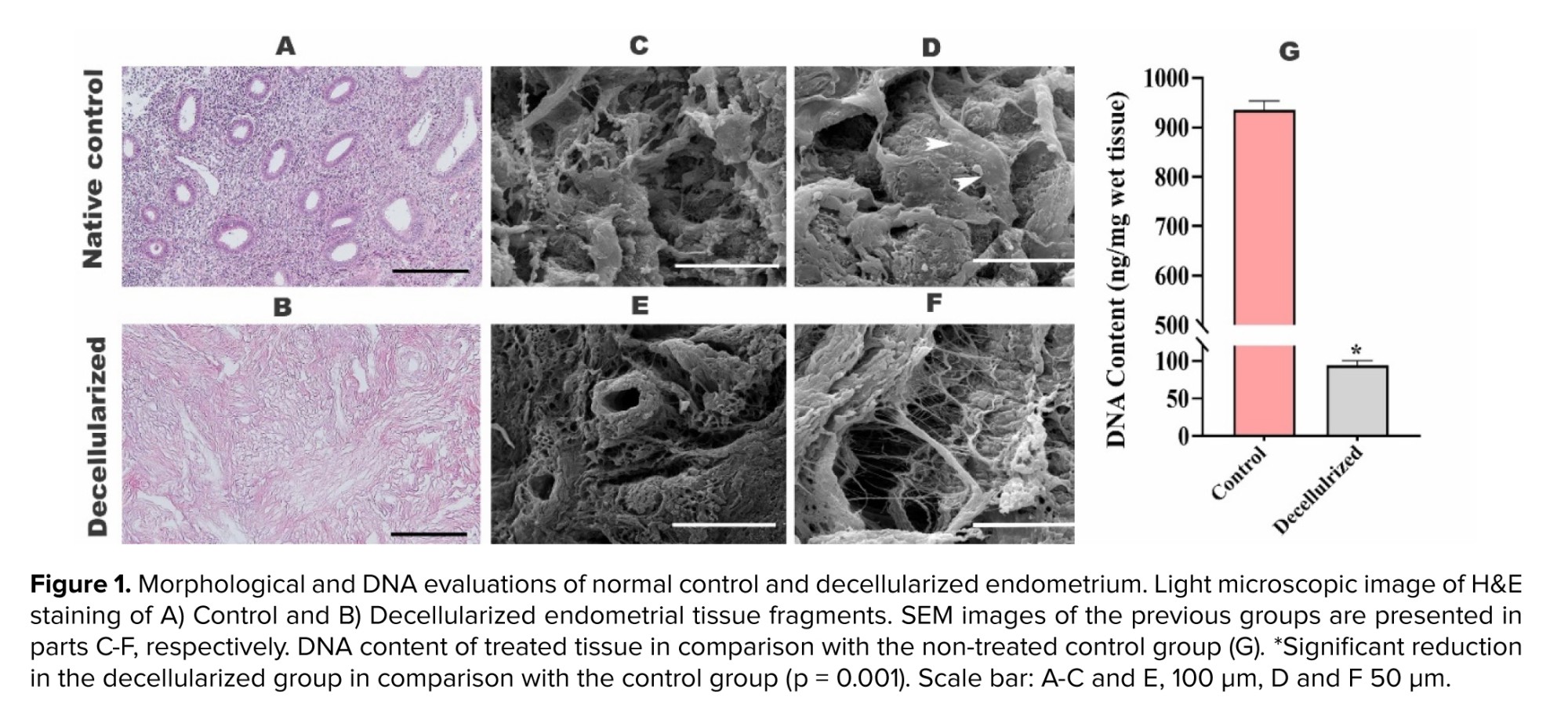
Light microscopic observation of H&E-stained sections revealed complete cell removal in the decellularized tissue (Figure 1), with no detectable cell nuclei and a fibrillary stromal structure similar to that of the native control tissue (Figure 1A and B). However, several gland sections were observed in the normal endometrium. SEM micrographs demonstrated the well-preserved fine structure of the decellularized tissue compared to the control (Figure 1C and D). Additionally, DNA quantification showed a significant reduction (p = 0.001) in DNA content in the decellularized tissue, with more than 89% of the DNA removed (Figure 1E).
3.2. Characterization of endometrial hydrogel
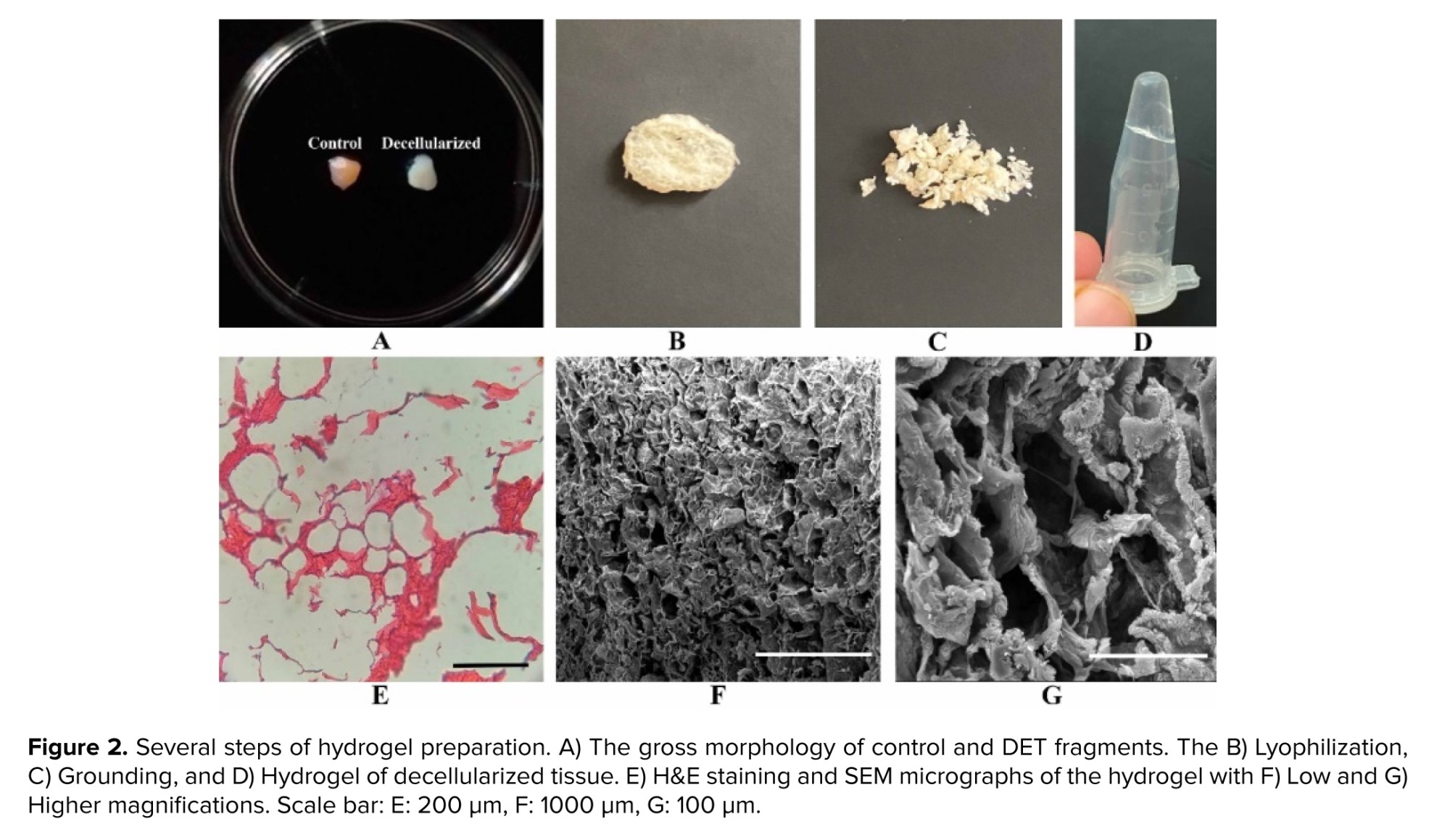
The gross morphology of the decellularized tissue and hydrogel preparation procedure is illustrated in figure 2 A-D. The decellularized endometrium appears white, while the hydrogel is clear and transparent. Under light microscopy, the endometrial hydrogel exhibits an eosinophilic honeycomb morphology (Figure 2E), and SEM micrographs with low and high-power magnifications revealed a spongy structure with good porosity (Figure 2F and G). Analysis of the micrographs showed an average porosity of 32.43 ± 5.34% and an average pore size of 20 ± 4.03 µm.
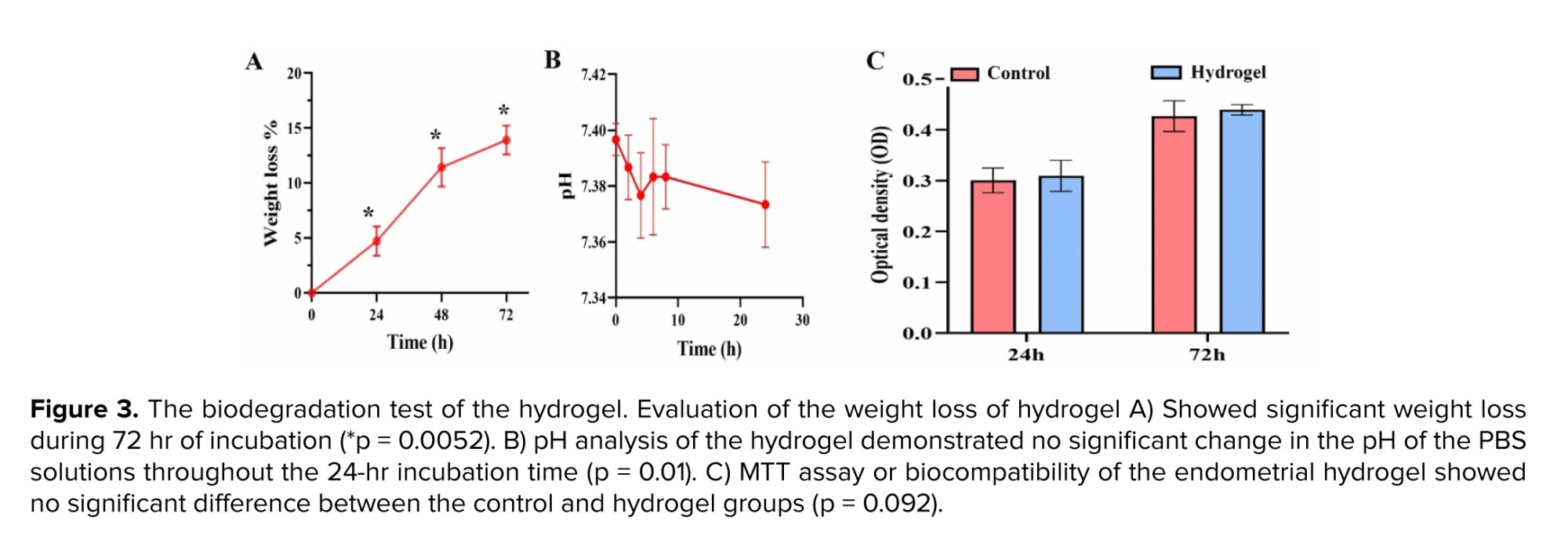
The in vitro biodegradation profile of the hydrogel in PBS at pH 7.4 is shown in figure 3A. After 24, 48, and 72 hr of incubation, weight losses of 4.7%, 11.4%, and 13.4% were observed (p = 0.0052), respectively. Furthermore, the results demonstrated no significant change in the pH of the PBS solutions throughout the 24-hr incubation period (p = 0.01; Figure 3B).
The MTT assay was performed to evaluate cell viability in both the control group and the decellularized hydrogel group at both time points: 24 hr and 72 hr (Figure 3C). Statistical analysis showed no significant differences between the 2 groups at both time points (p = 0.086).
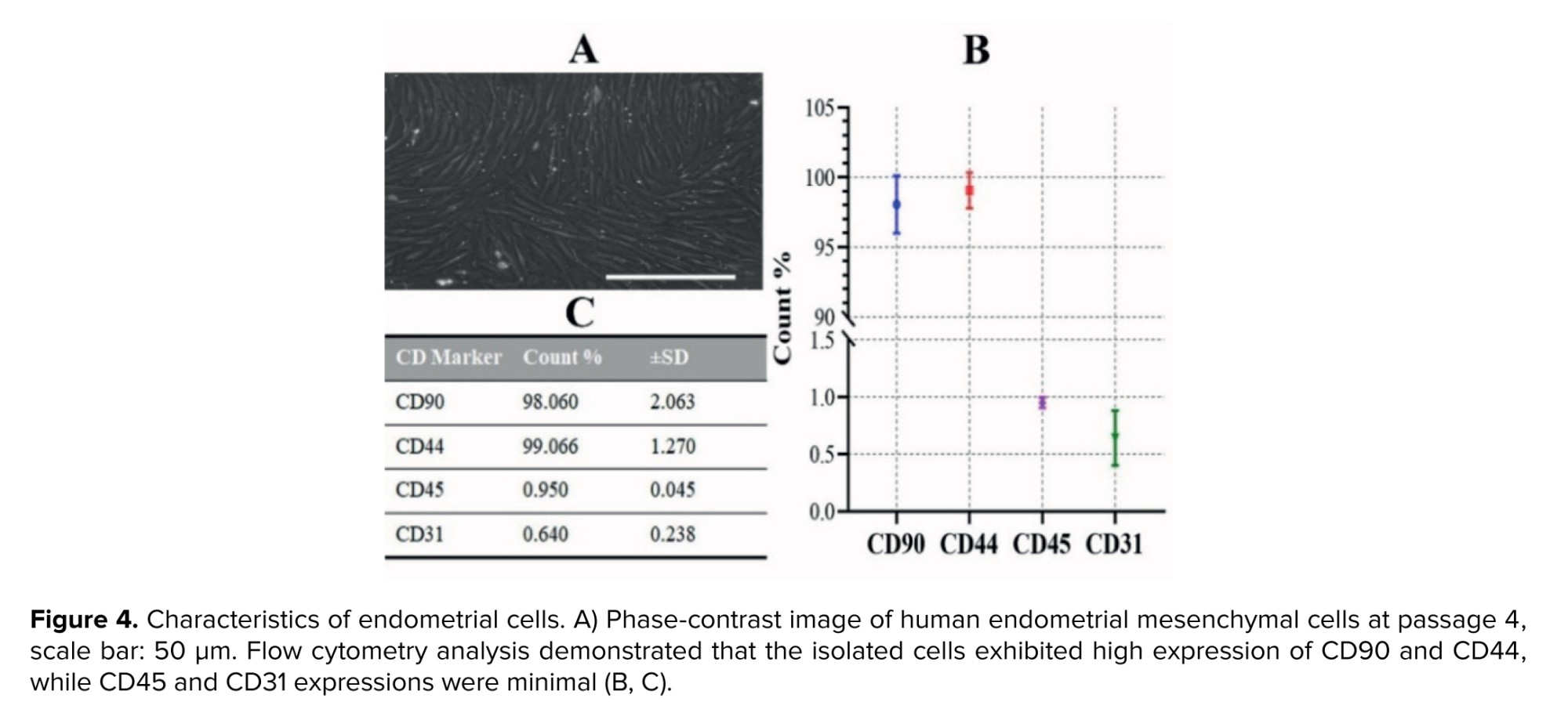
3.3. Phenotypes of human endometrial mesenchymal cells
The isolated stromal cells from human endometrial tissues appeared flattened and spindle-shaped. They were more homogeneous in the fourth passage (Figure 4A). Flow cytometry analysis (Figure 4B and C) revealed that the cultured cells exhibited a characteristic mesenchymal cell surface phenotype (supplementary histograms). Specifically, the cells showed high expression levels of the mesenchymal markers CD90 (98.06% ± 2.06) and CD44 (99.06% ± 1.27). In contrast, they were negative for the hematopoietic marker CD45 (0.95% ± 0.045) and the endothelial cell marker CD31 (0.64% ± 0.238).
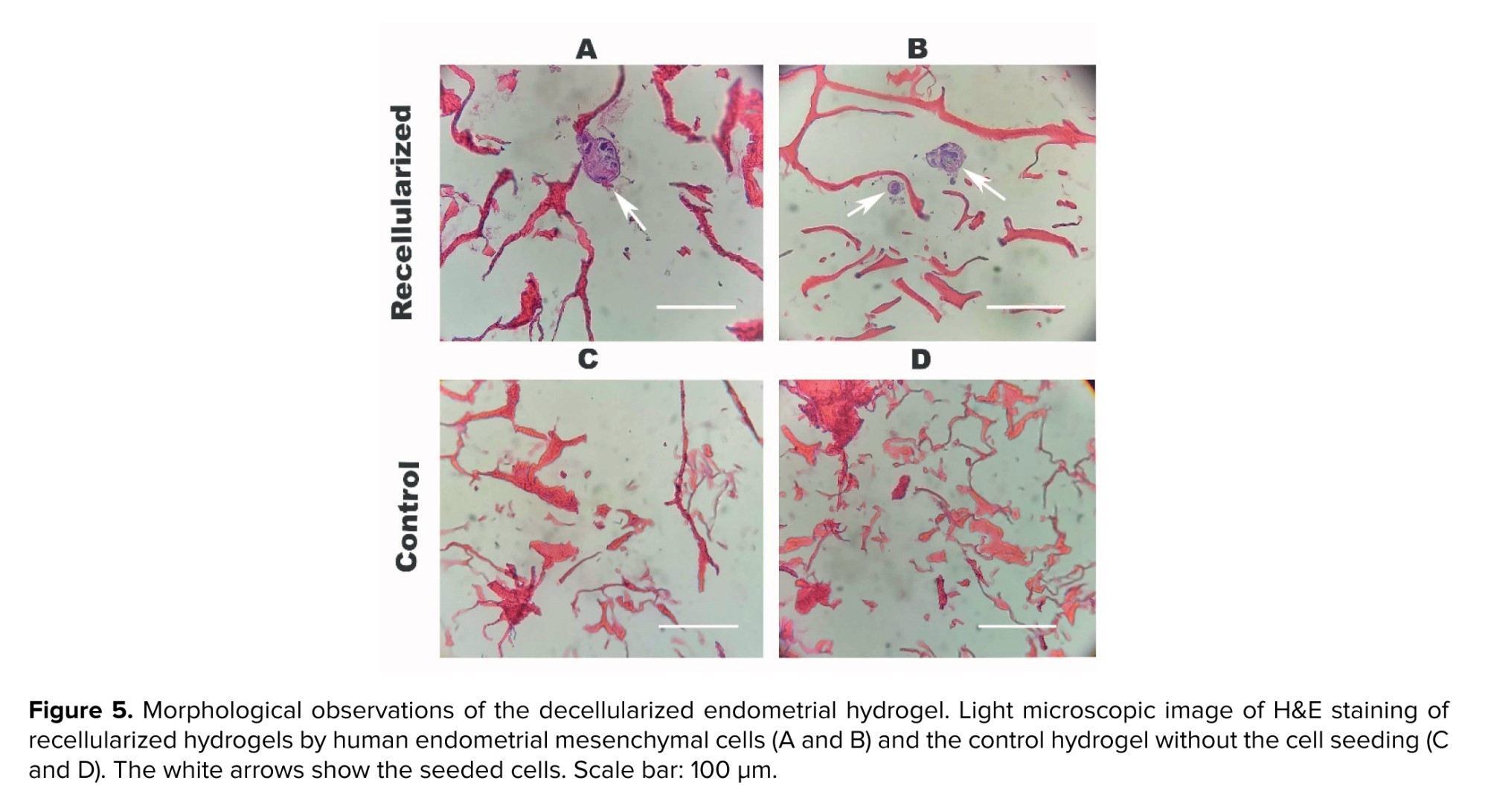
3.4. Light microscopic observation of recellularized hydrogel with human endometrial mesenchymal cells
The morphology of recellularized hydrogel that were seeded by human endometrial mesenchymal cells are illustrated in figure 5A, B. As these micrographs demonstrated that the nucleus of the seeded cells were stained by H&E and these cells were dispersed through the scaffold as an eosinophilic matrix. However, there is suitable porosity for homing the cells. There are no cells in the control group figure 5C, D.
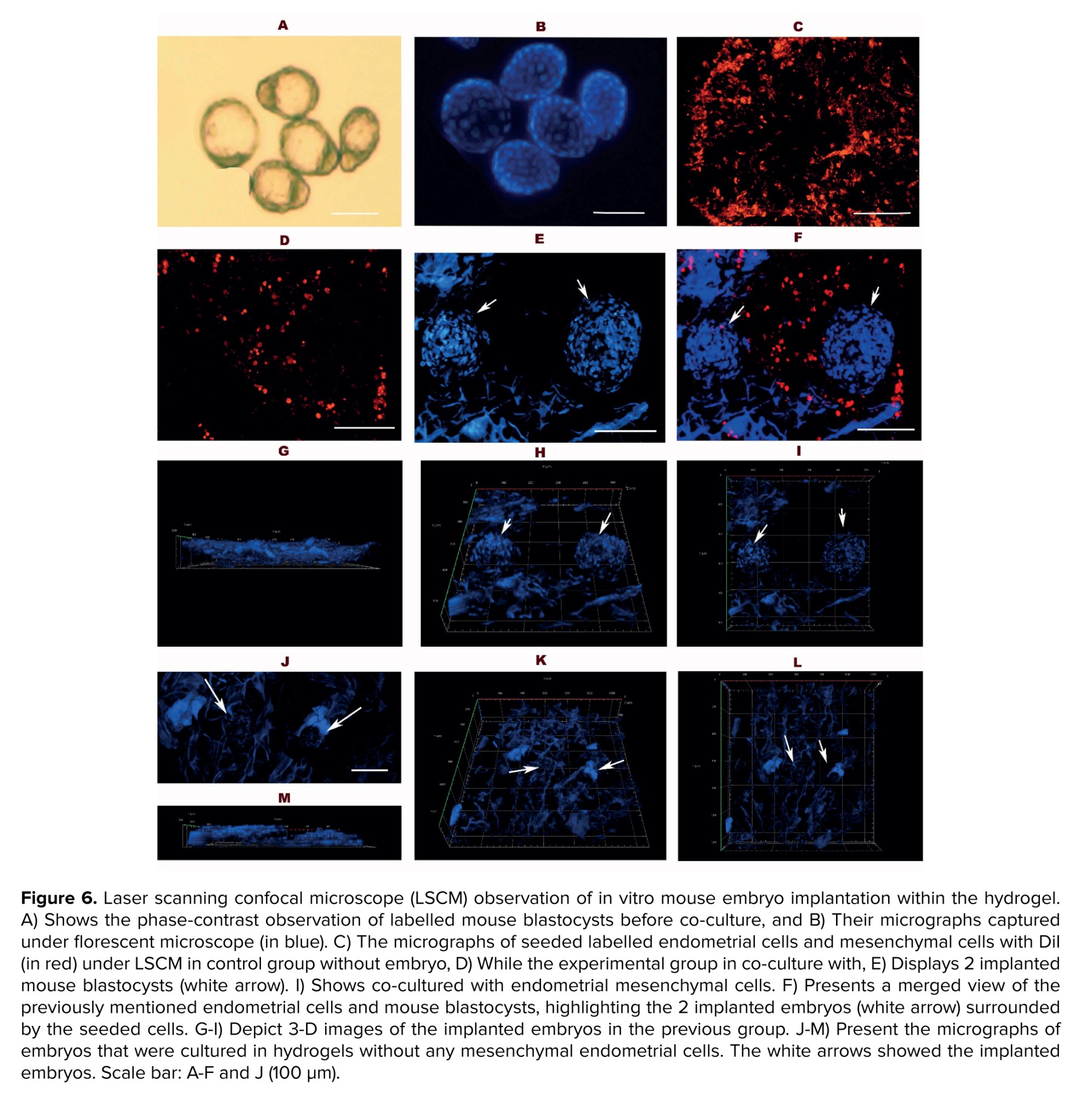
3.5. Evaluations of the recellularized hydrogel co-cultured with mouse blastocyst
The representative micrographs of 3 groups of study using laser scanning confocal microscope are shown in figure 6 (A-M). In these figures, the labelled embryos were seen in blue (6B), and the DiI-labeled endometrial mesenchymal cells are colored red. The endometrial mesenchymal cells are distributed throughout the hydrogel and are in groups I and III (Figure 6C and D). Moreover, these cells are more densely populated around the implanted embryos, which are shown in blue (Figure 6D and F). The depth of embryo penetration was demonstrated through the thickness of the hydrogels by 3-D micrographs (Figure 6G-I and K-M). The embryo expansion was better demonstrated in hydrogels that co-cultured with endometrial mesenchymal cells in comparison with the group where embryos were cultured in hydrogel alone.
4. Discussion
In recent years, the solubilization of tissues and organs into hydrogels has gained significant attention, with applications extending across major organ systems (35, 36). This study aimed to evaluate the potential of the human endometrial hydrogel as a scaffold to support mouse blastocyst implantation. Firstly, the decellularization procedure was evaluated by comparing its characteristics to those of a native sample. In line with our previous research, the protocol used demonstrated a substantial reduction in DNA and cellular materials while effectively preserving ECM components (15). Our approach successfully achieved decellularization without the use of enzymatic treatments, which are known to compromise the stability and integrity of tissues (11). At the initial stage of the decellularization process, we incorporated freezing and thawing cycles to enhance cell membrane permeability, potentially facilitating improved detergent penetration into the tissue.
Results of MTT assay demonstrated that the produced endometrial hydrogel had no impact on the survival of cultured cells. These data confirmed indirectly the successful removal of detergents that were applied during decellularization and gel formation protocols in the present study. In addition, our results demonstrated that the hydrogel was degradable, with the metabolic excretion of its degradation products being non-toxic. Importantly, the hydrogel degradation did not alter environmental pH, as evidenced by the progressive removal of the hydrogel without any significant acidic or alkaline shifts in the solution during incubation. These findings indicate that the endometrial hydrogel produced in this study exhibits excellent pH stability under physiological conditions and does not adversely affect cell viability, confirming its biocompatibility.
In another aspect of this study, recellularization of the hydrogel with human ESCs showed effective cell attachment and survival within the scaffold during cultivation period. This can be attributed to the composition of the endometrial hydrogel, which mimics the natural endometrial microenvironment, playing a crucial role in facilitating cell attachment and localization. This observation is in line with our previous studies that applied these cells for cell seeding in decellularized endometrial fragments (15). These experiments also showed the successful cell attachment and penetration of ESCs within scaffolds (15, 24, 37). Furthermore, the scaffold is believed to stimulate cellular proliferation and differentiation, closely replicating the conditions of the native endometrium. Supporting this, several in vitro culture studies have demonstrated the inductive effects of decellularized and solubilized tissues on stem cell differentiation (24, 38). These properties of the scaffold highlight its potential to provide a biomimetic environment conducive to endometrial tissue regeneration and function.
Laser confocal microscopy revealed that mouse embryonic cells successfully adhered to the hydrogel and interacted with the surrounding human ESCs. Notably, a higher population of human EMCs was observed around the embryo attachment site. These cells demonstrated an enhanced ability to interact both with one another and with the embryonic cells, potentially facilitating the implantation process (39). Successful embryo implantation critically depends on 2-way communication between embryonic and endometrial cells, mediated and facilitated by various adhesion molecules (6, 40). While the mouse blastocysts in the hydrogel group, without any endometrial mesenchymal cells, showed less expansion and growth. This part of study needs additional study to show the efficacy of endometrial hydrogel for the enhancement of embryo implantation and growth.
Additionally, the porosity and pore size of the scaffold play a crucial role in embryo invasion and growth within the scaffold. SEM micrographs showed that the hydrogel developed in this study provided sufficient space and pores to accommodate the expanding embryos. These findings align with recent research on 3-D hydrogel models to study endometrial physiology and embryo implantation (16-20, 23). For instance, gelatin methacryloyl hydrogels have been shown to support the growth of primary human endometrial stromal cells and epithelial gland organoids, offering a platform to study trophoblast invasion, endometrial function, and angiogenesis (41, 42). Furthermore, hyaluronic acid hydrogels combined with endometrial stromal cells have demonstrated synergistic regenerative effects in a murine model of uterine damage, resulting in successful embryo implantation and live births (43).
Endometrial tissue engineering is an emerging field dedicated to creating functional uterine tissues using advanced techniques such as organoid culture, decellularized tissues, and hydrogels (1-4). This field has promising clinical applications, including treatments for uterine diseases and injuries, addressing infertility, and supporting in vitro research in reproductive science, such as developing embryo implantation models (1, 5, 6). Decellularized endometrial tissue (DET) is derived from endometrial tissue to remove cellular components while preserving the extracellular matrix (ECM), which retains the biochemical composition, structure, and mechanical properties of the native tissue (2, 7-12). The decellularization process typically involves physical, chemical, and enzymatic methods, such as using detergents and enzymes, followed by validation to confirm the absence of cellular DNA and the retention of ECM components like collagen and glycosaminoglycans (3, 11, 13, 14). DET is highly biocompatible, reducing the risk of immune rejection, and supports cell function similar to that in the native endometrial environment. It is widely studied in tissue engineering as a scaffold for regenerating endometrial tissue in conditions like Asherman’s syndrome or uterine factor infertility and in disease models for endometrial disorders, including endometriosis and cancer (1, 5). Recently, we have developed a protocol for the decellularization of human endometrial tissue fragments that effectively preserves the ECM components, closely resembling those of intact tissue (10, 15).
Hydrogels derived from the decellularized endometrium are biomaterials designed to mimic the native ECM, providing a supportive, 3-dimensional environment for cell growth, differentiation, and tissue regeneration (16-21). The decellularized ECM was processed into a hydrogel form, often through solubilization and crosslinking techniques, resulting in a biocompatible, injectable material (21). Endometrial-derived hydrogels are rich in ECM proteins like collagen, laminin, and fibronectin, as well as bioactive molecules that promote cell adhesion, proliferation, and angiogenesis (18, 22). These properties make them particularly suitable for applications in reproductive medicine (7, 20, 23, 24). Hydrogel is also compatible with encapsulation of endometrial cells or stem cells, providing a scaffold that supports cellular function and integration (16, 19, 23).
Endometrial stem cells (ESCs) are a population of multipotent stem cells found within the endometrial tissue of the uterus, playing a crucial role in the dynamic regenerative processes of the endometrium during the menstrual cycle and following injury (25-28). These cells are characterized by their ability to self-renew and differentiate into various cell types, including epithelial, stromal, and possibly endothelial cells (29). ESCs are identified by specific markers and are being investigated for their potential in tissue engineering, bioengineering of uterine constructs, and disease modeling (25-27). Research has focused on utilizing ESCs with various scaffolds to reconstruct the endometrium in 3-D culture systems, with some methods successfully developing in vitro embryo implantation models (15, 30, 31).
Embryo implantation models using biomaterials and scaffolds are innovative platforms designed to study the complex process of embryo implantation in vitro, offering insights into reproductive biology and potential solutions for infertility (15, 32, 33). The scaffold is seeded with endometrial cells, including epithelial and stromal cells, or other types of stem cells, to recreate the functional layers of the endometrium in co-culture with the embryo (6, 33).
This study aimed to evaluate the potential of recellularized human endometrial hydrogel, populated by endometrial mesenchymal cells, as a scaffold for mouse blastocyst implantation as an in vitro model.
2. Materials and Methods
2.1. Study design
This experimental study was done in 2023 at Anatomical Sciences Department of Tarbiat Modares University, Tehran, Iran and involved the preparation of a hydrogel using decellularized human endometrium, with evaluations of its morphology, ultrastructure, and residual DNA content. The biocompatibility, biodegradation, and pore sizes of the hydrogel were also analyzed. Human ESCs were isolated from fresh tissue samples, cultured up to the fourth passage, and characterized using flow cytometry. The hydrogel was recellularized by seeding human ESCs for 7 days, followed by co-culturing mouse blastocysts on these hydrogels for 48 hr. Blastocyst implantation within the hydrogels was assessed using laser confocal scanning microscopy. All materials were sourced from Sigma Aldrich (London, UK), and experiments were performed in triplicate unless otherwise stated.
2.2. Human endometrial tissue collection
Endometrial tissues were collected from fertile women aged 25-35 yr (n = 6) who had been diagnosed with gynecological conditions unrelated to endometrial disease. None of the participants had received exogenous hormone therapy 3 months before sampling. The tissues were transferred under sterile conditions, dissected into 2-3 cm² fragments, and preserved in Dulbecco’s Modified Eagle’s Medium (DMEM) on ice. A portion of the tissue fragments was assessed for normal morphology, while the remaining fragments were used for decellularization, cell isolation, and in vitro culture (30).
2.3. Tissue decellularization
The endometrial tissues were decellularized using a previously established method (15). Briefly, the tissue samples underwent 3 successive cycles of freezing at -80°C and thawing at 37°C, followed by treatment with 1% Triton X-100 for 15 hr and 1% sodium dodecyl sulfate for 72 hr at room temperature. The samples were then washed 6 times with phosphate-buffered saline (PBS), each wash lasting 8 hr. Following this process, the decellularized tissues were analyzed for DNA content, subjected to hematoxylin and eosin (H&E) staining, and examined using scanning electron microscopy (SEM).
2.3.1. Morphological study
The control tissue samples and decellularized endometrial fragment (n = 3/group) were fixed in 10% formaldehyde, dehydrated using a graded series of ethanol, and subsequently embedded in paraffin wax. Sections with 5 μm thickness were then prepared and stained using the H&E procedure. The stained sections were observed under a light microscope.
2.3.2. SEM
To characterize the ultrastructural properties of the decellularized tissues in comparison with the native control, 3 samples from each group were fixed in 2.5% glutaraldehyde, followed by dehydration using a graded series of ethanol and overnight drying with hexamethyldisilazane. The samples were then coated with gold and examined using SEM (VEGA/TESCAN-XMU, Czech Republic).
2.3.3. DNA content analysis
Total DNA from decellularized endometrial samples and control tissue (n = 3/group) was extracted using a TRIzol® kit (Invitrogen, UK) according to the standardized protocol. The samples were weighed, treated with TRIzol® reagent, and subjected to chloroform extraction. The DNA layer was then isolated by centrifugation at 2000 g for 10 min at 4°C, followed by resuspension in NaOH and further centrifugation at 12,000 g for 10 min at 4°C. The DNA concentration was determined using UV spectrophotometry (Eppendorf, Germany) at 260 nm.
2.4. Endometrial cells isolation, culture, and labeling
Endometrial stromal cells were isolated and cultured according to a previously described method (30). Briefly, endometrial tissue samples (n = 6) were cut into 1 mm fragments after rinsing in PBS. They were placed in DMEM/F-12 medium supplemented with 100 mg/ml Penicillin G sodium, 100 mg/ml Streptomycin sulfate, and 10% fetal bovine serum. The fragments were then treated with collagenase type I (300 μg/ml) and deoxyribonuclease type I (40 μg/ml) for 1-2 hr. The resulting endometrial stromal cells were cultured up to the fourth passage at 37°C in a 5% CO2 incubator. For characterization of cultured endometrial cells, they were suspended (1×10⁵) in 50 μl of PBS and incubated with FITC-conjugated antibodies (anti-human CD90, CD44, and CD45 as mesenchymal and hematopoietic markers, respectively) and phycoerythrin-conjugated antibodies (anti-human CD31 as an endothelial marker) at a 1:50 dilution at 4°C for 45 min. After washing in PBS, the cells were analyzed using a fluorescence-activated cell sorting Calibur apparatus (Becton Dickinson, USA).
The cultured cells were used for the 3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide (MTT) assay and labeling with di-alkyl indocarbocyanine (DiI) according to our previous study (15). Briefly, a suspension of 1×10⁵ cells/ml was incubated with 2 µg/mL DiI for 20 min at 4°C in the dark, followed by washing 3 times in culture media. The labeled cells were then used for cell seeding on the hydrogel.
2.5. Preparation of human endometrial hydrogel
The DET was cut into several fragments (1×1 mm) and washed 4 times in ice-cold PBS to remove residual detergents. The fragments were lyophilized overnight (0.30-0.31 gr/each), milled with dry ice, frozen in liquid nitrogen, ground, and stored at -20°C. The resulting powder (1.45 gr) was solubilized using a modified protocol, suspending 1% (w/v) powder in 0.01 M HCl with 0.1% (w/v) pepsin, followed by digestion for 48 hr, neutralization in 10% (v/v) 0.1 M NaOH, aliquotation, flash freezing, and storage at -80°C. For coating, the hydrogel solution was added to a 24-well culture plate and left overnight at 4°C. Then washed with PBS, and culture medium (DMEM/F-12, Invitrogen, UK) (8).
2.5.1. Endometrial hydrogel morphology and ultrastructure
Endometrial hydrogel samples (n = 3) were fixed and processed for H&E staining, followed by observation under the light microscope, as previously described for decellularized tissue. To examine the ultrastructure of the endometrial hydrogels, the samples were freeze-dried (Alpha 2-4 LDplus, Martin Christ, Germany) for 12 hr. Dried samples were then segmented, coated with a thin layer of gold, and observed under SEM. Multiple micrographs were captured from different regions of the endometrial hydrogels. The average pore size and porosity of the hydrogels were measured from SEM images using ImageJ 1.33U software (National Institutes of Health, USA).
2.5.2. Biocompatibility (MTT assay)
The MTT assay was used to evaluate the biocompatibility of the prepared endometrial hydrogels. The human ESCs were cultured at a density of 1×105 cells in a 96-well plate, with or without pre-loaded endometrial hydrogel. After incubation for 24 and 72 hr, the supernatant was discarded, and the cells were exposed to MTT solution (0.5 mg/mL in PBS) for 4 hr. The resulting formazan product was dissolved in dimethyl sulfoxide for 20 min. Finally, the amount of formazan was quantified by measuring its absorbance at 540 nm using a microplate reader (Biochrom, Germany).
2.5.3. Biodegradation and pH assessment
The endometrial hydrogel samples (n = 3) with 5×5×2 mm³ size were subjected to a 72-hr degradation study. Initially, the hydrogel samples were lyophilized, weighed using a digital analytical balance (Hawach Scientific, China), and placed in a controlled PBS solution with a pH of 7.4, as per the previous study (34). The PBS was replaced every 12 hr to prevent the accumulation of degradation by products. After 24, 48, and 72 hr of incubation, the samples were lyophilized again, and their weight were measured, respectively. The percentage weight loss of the samples over time was calculated using the formula:

Dried hydrogel samples were transferred into 5 mL microtubes, each containing 4 mL of PBS at pH 7.4. The microtubes were incubated at 37°C, and pH measurements were taken at 2, 4, 6, 8, and 24-hr intervals using a calibrated pH meter (APERA, PH850, USA).
2.5.4. Recellularization of endometrial hydrogel
Hydrogels were placed in individual wells of a 24-well plate and incubated with culture medium (DMEM/F-12, as described earlier) for 24 hr before cell seeding. Subsequently, 1×105 labelled cells were added to each well and incubated at 37°C in a 5% CO2 atmosphere for 1 wk. The medium was changed every 2 days. Also, the same hydrogel series without cell seeding was considered the control group. The recellularized and control hydrogels were then used for subsequent experiments.
2.6. Blastocyst collection and labelling
Adult female (n = 10) and male (n = 5) mice were maintained under standard laboratory conditions, based on the laboratory animal care ethics of Tarbiat Modares University, Tehran, Iran. To induce superovulation, female mice were administered a single intraperitoneal dose of 7.5 IU pregnant mare serum gonadotropin (Folligon, Intervet, Australia), followed by a subsequent intraperitoneal dose of 10 IU human chorionic gonadotropin (Pregnyl, Netherlands). Afterward, females were mated with fertile males. Approximately 120 hr post-human chorionic gonadotropin injection, blastocysts were collected by flushing from the uterine horns. To enable visualization and tracking of the blastocysts within the endometrial hydrogel, the blastocysts (n = 60) were labelled with Hoechst 34580 dye solution (1 ng/ml in distilled water) for 20 min, followed by washing with culture medium (DMEM/F-12).
2.6.1. Blastocysts co-culture in recellularized hydrogel
One-week post-recellularization of the hydrogel, we applied 3 study groups. Group I: co-culture of mouse embryo in hydrogels that recellularized with endometrial mesenchymal cells. Group II: the recellularized hydrogel with endometrial mesenchymal cells in the absence of a mouse embryo. Group III: culture of mouse embryos in hydrogel alone without any endometrial cells. In groups I and III, the labelled mouse blastocysts were introduced onto the surface of each endometrial hydrogel-loaded well (n = 2 embryos per well and 30 in total) and cultured for 48 hr at 37°C in 5% CO2. In group II, the recellularized endometrial hydrogels, without blastocysts, were subjected to similar culture conditionsAt least 3 wells from each experimental and control group were observed under laser confocal scanning microscopy (LCSM, Zeiss LSM800, Japan), and embryo penetration into the scaffold was evaluated at excitation and emission wavelengths of 392 and 440 nm for Hoechst-labeled embryonic cells and 546 and 563 nm for DiI-labeled endometrial cells.
2.7. Ethical Considerations
The study adhered to the ethical guidelines of the Tarbiat Modares University Ethics Committee, Tehran, Iran (Code: IR.MODARES.REC.1400.209). An informed consent was obtained from participants for the use of their tissues.
2.8. Statistical Analysis
All experiments were replicated at least 3 times. Data analysis was performed using GraphPad Prism Software 8 (Denver, CO, USA). The normality of the data was assessed using the Shapiro-Wilk test. To compare DNA content between groups, one-way ANOVA was used followed by Tukey's post-hoc test. The independent sample t test was applied to compare MTT assay results between groups. Statistical significance was set at a p < 0.05.
3. Results
3.1. Characterization of decellularized tissue
Light microscopic observation of H&E-stained sections revealed complete cell removal in the decellularized tissue (Figure 1), with no detectable cell nuclei and a fibrillary stromal structure similar to that of the native control tissue (Figure 1A and B). However, several gland sections were observed in the normal endometrium. SEM micrographs demonstrated the well-preserved fine structure of the decellularized tissue compared to the control (Figure 1C and D). Additionally, DNA quantification showed a significant reduction (p = 0.001) in DNA content in the decellularized tissue, with more than 89% of the DNA removed (Figure 1E).
3.2. Characterization of endometrial hydrogel
The gross morphology of the decellularized tissue and hydrogel preparation procedure is illustrated in figure 2 A-D. The decellularized endometrium appears white, while the hydrogel is clear and transparent. Under light microscopy, the endometrial hydrogel exhibits an eosinophilic honeycomb morphology (Figure 2E), and SEM micrographs with low and high-power magnifications revealed a spongy structure with good porosity (Figure 2F and G). Analysis of the micrographs showed an average porosity of 32.43 ± 5.34% and an average pore size of 20 ± 4.03 µm.
The in vitro biodegradation profile of the hydrogel in PBS at pH 7.4 is shown in figure 3A. After 24, 48, and 72 hr of incubation, weight losses of 4.7%, 11.4%, and 13.4% were observed (p = 0.0052), respectively. Furthermore, the results demonstrated no significant change in the pH of the PBS solutions throughout the 24-hr incubation period (p = 0.01; Figure 3B).
The MTT assay was performed to evaluate cell viability in both the control group and the decellularized hydrogel group at both time points: 24 hr and 72 hr (Figure 3C). Statistical analysis showed no significant differences between the 2 groups at both time points (p = 0.086).
3.3. Phenotypes of human endometrial mesenchymal cells
The isolated stromal cells from human endometrial tissues appeared flattened and spindle-shaped. They were more homogeneous in the fourth passage (Figure 4A). Flow cytometry analysis (Figure 4B and C) revealed that the cultured cells exhibited a characteristic mesenchymal cell surface phenotype (supplementary histograms). Specifically, the cells showed high expression levels of the mesenchymal markers CD90 (98.06% ± 2.06) and CD44 (99.06% ± 1.27). In contrast, they were negative for the hematopoietic marker CD45 (0.95% ± 0.045) and the endothelial cell marker CD31 (0.64% ± 0.238).
3.4. Light microscopic observation of recellularized hydrogel with human endometrial mesenchymal cells
The morphology of recellularized hydrogel that were seeded by human endometrial mesenchymal cells are illustrated in figure 5A, B. As these micrographs demonstrated that the nucleus of the seeded cells were stained by H&E and these cells were dispersed through the scaffold as an eosinophilic matrix. However, there is suitable porosity for homing the cells. There are no cells in the control group figure 5C, D.
3.5. Evaluations of the recellularized hydrogel co-cultured with mouse blastocyst
The representative micrographs of 3 groups of study using laser scanning confocal microscope are shown in figure 6 (A-M). In these figures, the labelled embryos were seen in blue (6B), and the DiI-labeled endometrial mesenchymal cells are colored red. The endometrial mesenchymal cells are distributed throughout the hydrogel and are in groups I and III (Figure 6C and D). Moreover, these cells are more densely populated around the implanted embryos, which are shown in blue (Figure 6D and F). The depth of embryo penetration was demonstrated through the thickness of the hydrogels by 3-D micrographs (Figure 6G-I and K-M). The embryo expansion was better demonstrated in hydrogels that co-cultured with endometrial mesenchymal cells in comparison with the group where embryos were cultured in hydrogel alone.






4. Discussion
In recent years, the solubilization of tissues and organs into hydrogels has gained significant attention, with applications extending across major organ systems (35, 36). This study aimed to evaluate the potential of the human endometrial hydrogel as a scaffold to support mouse blastocyst implantation. Firstly, the decellularization procedure was evaluated by comparing its characteristics to those of a native sample. In line with our previous research, the protocol used demonstrated a substantial reduction in DNA and cellular materials while effectively preserving ECM components (15). Our approach successfully achieved decellularization without the use of enzymatic treatments, which are known to compromise the stability and integrity of tissues (11). At the initial stage of the decellularization process, we incorporated freezing and thawing cycles to enhance cell membrane permeability, potentially facilitating improved detergent penetration into the tissue.
Results of MTT assay demonstrated that the produced endometrial hydrogel had no impact on the survival of cultured cells. These data confirmed indirectly the successful removal of detergents that were applied during decellularization and gel formation protocols in the present study. In addition, our results demonstrated that the hydrogel was degradable, with the metabolic excretion of its degradation products being non-toxic. Importantly, the hydrogel degradation did not alter environmental pH, as evidenced by the progressive removal of the hydrogel without any significant acidic or alkaline shifts in the solution during incubation. These findings indicate that the endometrial hydrogel produced in this study exhibits excellent pH stability under physiological conditions and does not adversely affect cell viability, confirming its biocompatibility.
In another aspect of this study, recellularization of the hydrogel with human ESCs showed effective cell attachment and survival within the scaffold during cultivation period. This can be attributed to the composition of the endometrial hydrogel, which mimics the natural endometrial microenvironment, playing a crucial role in facilitating cell attachment and localization. This observation is in line with our previous studies that applied these cells for cell seeding in decellularized endometrial fragments (15). These experiments also showed the successful cell attachment and penetration of ESCs within scaffolds (15, 24, 37). Furthermore, the scaffold is believed to stimulate cellular proliferation and differentiation, closely replicating the conditions of the native endometrium. Supporting this, several in vitro culture studies have demonstrated the inductive effects of decellularized and solubilized tissues on stem cell differentiation (24, 38). These properties of the scaffold highlight its potential to provide a biomimetic environment conducive to endometrial tissue regeneration and function.
Laser confocal microscopy revealed that mouse embryonic cells successfully adhered to the hydrogel and interacted with the surrounding human ESCs. Notably, a higher population of human EMCs was observed around the embryo attachment site. These cells demonstrated an enhanced ability to interact both with one another and with the embryonic cells, potentially facilitating the implantation process (39). Successful embryo implantation critically depends on 2-way communication between embryonic and endometrial cells, mediated and facilitated by various adhesion molecules (6, 40). While the mouse blastocysts in the hydrogel group, without any endometrial mesenchymal cells, showed less expansion and growth. This part of study needs additional study to show the efficacy of endometrial hydrogel for the enhancement of embryo implantation and growth.
Additionally, the porosity and pore size of the scaffold play a crucial role in embryo invasion and growth within the scaffold. SEM micrographs showed that the hydrogel developed in this study provided sufficient space and pores to accommodate the expanding embryos. These findings align with recent research on 3-D hydrogel models to study endometrial physiology and embryo implantation (16-20, 23). For instance, gelatin methacryloyl hydrogels have been shown to support the growth of primary human endometrial stromal cells and epithelial gland organoids, offering a platform to study trophoblast invasion, endometrial function, and angiogenesis (41, 42). Furthermore, hyaluronic acid hydrogels combined with endometrial stromal cells have demonstrated synergistic regenerative effects in a murine model of uterine damage, resulting in successful embryo implantation and live births (43).
5. Conclusion
This novel implantation model, which integrates human EMCs, endometrial hydrogel, and mouse embryos, presents significant implications for reproductive studies and clinical applications. However, further research is needed to fully explore and refine its potential.
Because of some limitations for the usage of the human embryos, such as their ethical and availability, in the present study, we have used the mouse embryo as a surrogate model. However, this implantation process is not completely similar to the human; therefore, further studies in more physiologically relevant animal models are needed.
Data Availability
Data supporting the findings of this study are available as supplementary files and also upon reasonable request from the corresponding author.
Author Contributions
M. Salehnia: Supervised all procedures of the experiments and designed the study, also wrote, and edited the manuscript and had full access to all of the data. E. Sadeghi: Performed the study, prepared the primary draft of the manuscript, and conducted data analysis. S. Moazzeni: Was the scientific research assistant, performed the statistical analysis, and edited the manuscript. All authors read and approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgements
This paper was extracted from a PhD. thesis at Tarbiat Modares University, Tehran, Iran. The study received no grant for this research. No artificial intelligence was used for the preparation of this manuscript.
Conflict of Interest
The author declares that there is no conflict of interest.
Because of some limitations for the usage of the human embryos, such as their ethical and availability, in the present study, we have used the mouse embryo as a surrogate model. However, this implantation process is not completely similar to the human; therefore, further studies in more physiologically relevant animal models are needed.
Data Availability
Data supporting the findings of this study are available as supplementary files and also upon reasonable request from the corresponding author.
Author Contributions
M. Salehnia: Supervised all procedures of the experiments and designed the study, also wrote, and edited the manuscript and had full access to all of the data. E. Sadeghi: Performed the study, prepared the primary draft of the manuscript, and conducted data analysis. S. Moazzeni: Was the scientific research assistant, performed the statistical analysis, and edited the manuscript. All authors read and approved the final manuscript and take responsibility for the integrity of the data.
Acknowledgements
This paper was extracted from a PhD. thesis at Tarbiat Modares University, Tehran, Iran. The study received no grant for this research. No artificial intelligence was used for the preparation of this manuscript.
Conflict of Interest
The author declares that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Biology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |