Thu, May 21, 2026
[Archive]
Volume 24, Issue 1 (January 2026)
IJRM 2026, 24(1): 23-36 |
Back to browse issues page
Ethics code: IR.MODARES.REC.1400.209
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Sadeghi E, Moazzeni S, Salehnia M. Fabrication and characterization of decellularized human endometrial hydrogel as a scaffold for in vitro embryo implantation: An experimental study. IJRM 2026; 24 (1) :23-36
URL: http://ijrm.ir/article-1-3691-en.html
URL: http://ijrm.ir/article-1-3691-en.html
1- Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran. & Department of Anatomical Sciences, School of Medicine, Ilam University of Medical Sciences, Ilam, Iran.
2- Department of Biomedical Engineering, Rutgers, The State University of New Jersey, Piscataway, NJ, USA.
3- Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran. & Discipline of Biomedical Engineering, School of Electrical and Electronic Engineering, The University of Adelaide, Adelaide, Australia. ,salehnim@modares.ac.ir
2- Department of Biomedical Engineering, Rutgers, The State University of New Jersey, Piscataway, NJ, USA.
3- Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran. & Discipline of Biomedical Engineering, School of Electrical and Electronic Engineering, The University of Adelaide, Adelaide, Australia. ,
Abstract: (215 Views)
Background: Endometrial-derived hydrogels are rich in extracellular matrix proteins as well as bioactive molecules that promote cell adhesion, proliferation, and angiogenesis.
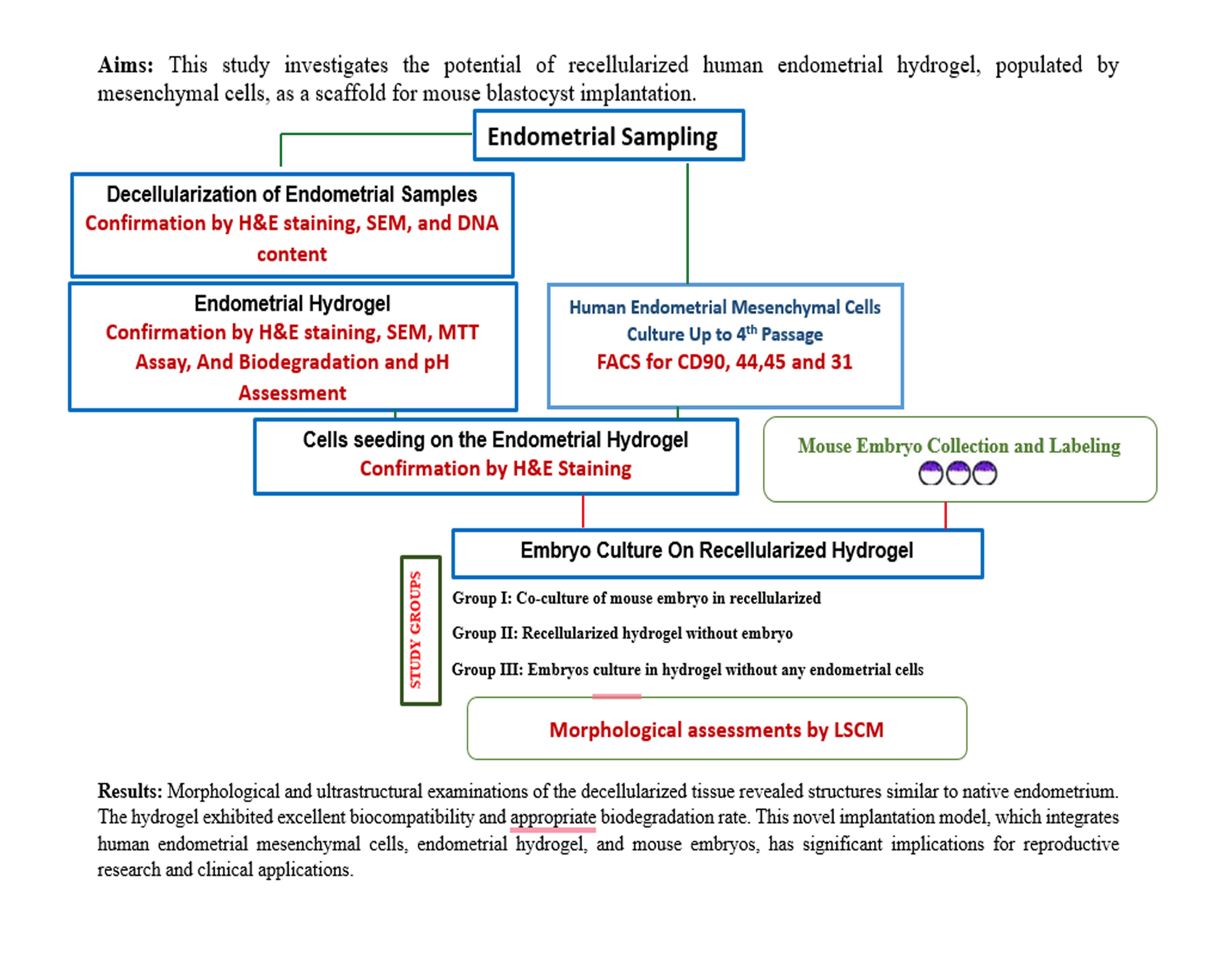
Objective: This study investigates the potential of recellularized human endometrial hydrogel, populated by mesenchymal cells, as a scaffold for mouse blastocyst implantation.
Materials and Methods: In this experimental study, we characterized the decellularization process of human endometrial tissue through morphological analysis, DNA quantification, and scanning electron microscopy. We then evaluated the morphology, ultrastructure, biocompatibility, biodegradation, and pH changes of the hydrogel incubated in phosphate-buffered saline. The hydrogel was recellularized with human endometrial mesenchymal cells, and mouse blastocysts were co-cultured on this recellularized hydrogel for 48 hr, observed via laser confocal scanning microscopy.
Results: Morphological and ultrastructural examinations of the decellularized tissue revealed structures similar to native endometrium, while the absence of cells and nuclei, confirmed through morphological staining and DNA analysis, validated the success of the decellularization process. The hydrogel exhibited excellent biocompatibility according to the 3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide assay results, demonstrated an appropriate biodegradation rate in vitro, and showed no significant pH alteration. Mesenchymal cells successfully attached to the hydrogel scaffold, and mouse embryos adhered to and invaded the recellularized hydrogel.
Conclusion: This novel implantation model, which integrates human endometrial mesenchymal cells, endometrial hydrogel, and mouse embryos, has significant implications for reproductive research and clinical applications. However, further investigation is necessary to fully explore and optimize its potential.
Objective: This study investigates the potential of recellularized human endometrial hydrogel, populated by mesenchymal cells, as a scaffold for mouse blastocyst implantation.
Materials and Methods: In this experimental study, we characterized the decellularization process of human endometrial tissue through morphological analysis, DNA quantification, and scanning electron microscopy. We then evaluated the morphology, ultrastructure, biocompatibility, biodegradation, and pH changes of the hydrogel incubated in phosphate-buffered saline. The hydrogel was recellularized with human endometrial mesenchymal cells, and mouse blastocysts were co-cultured on this recellularized hydrogel for 48 hr, observed via laser confocal scanning microscopy.
Results: Morphological and ultrastructural examinations of the decellularized tissue revealed structures similar to native endometrium, while the absence of cells and nuclei, confirmed through morphological staining and DNA analysis, validated the success of the decellularization process. The hydrogel exhibited excellent biocompatibility according to the 3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide assay results, demonstrated an appropriate biodegradation rate in vitro, and showed no significant pH alteration. Mesenchymal cells successfully attached to the hydrogel scaffold, and mouse embryos adhered to and invaded the recellularized hydrogel.
Conclusion: This novel implantation model, which integrates human endometrial mesenchymal cells, endometrial hydrogel, and mouse embryos, has significant implications for reproductive research and clinical applications. However, further investigation is necessary to fully explore and optimize its potential.
Keywords: Decellularized extracellular matrix, Endometrium, Hydrogel, Embryo implantation, Mesenchymal stem cells.
Type of Study: Original Article |
Subject:
Reproductive Biology
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |