Tue, Jun 9, 2026
[Archive]
Volume 24, Issue 2 (February 2026)
IJRM 2026, 24(2): 101-108 |
Back to browse issues page
Ethics code: IR.SSU.MEDICINE.REC.1402.122
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Namjoo K, Iravani F, Montazeri F, Jafari Najaf Abadi M H, Kalantar S M, Ghasemi N. Meiotic gene expression of STAG3 and DMC1 in blood: A novel non-invasive biomarker approach for diminished ovarian reserve: A case-control study. IJRM 2026; 24 (2) :101-108
URL: http://ijrm.ir/article-1-3728-en.html
URL: http://ijrm.ir/article-1-3728-en.html
Khosro Namjoo1 

 , Farzaneh Iravani2
, Farzaneh Iravani2 

 , Fateme Montazeri3
, Fateme Montazeri3 

 , Mohammad Hassan Jafari Najaf Abadi4
, Mohammad Hassan Jafari Najaf Abadi4 

 , Seyed Mehdi Kalantar2
, Seyed Mehdi Kalantar2 

 , Nasrin Ghasemi *5
, Nasrin Ghasemi *5 




 , Farzaneh Iravani2
, Farzaneh Iravani2 

 , Fateme Montazeri3
, Fateme Montazeri3 

 , Mohammad Hassan Jafari Najaf Abadi4
, Mohammad Hassan Jafari Najaf Abadi4 

 , Seyed Mehdi Kalantar2
, Seyed Mehdi Kalantar2 

 , Nasrin Ghasemi *5
, Nasrin Ghasemi *5 


1- Department of Medical Genetics, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
2- Department of Medical Genetics, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
4- Department of Medical Biotechnology, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
5- Department of Medical Genetics, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,nghasemi479@gmail.com; ghasemi@ssu.ac.ir
2- Department of Medical Genetics, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
4- Department of Medical Biotechnology, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
5- Department of Medical Genetics, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. & Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,
Full-Text [PDF 474 kb]
(335 Downloads)
| Abstract (HTML) (688 Views)
Full-Text: (73 Views)
1. Introduction
Diminished ovarian reserve (DOR) represents a significant cause of female infertility, characterized by reduced oocyte quantity and quality. Diagnosis involves decreased antral follicle count, elevated follicle-stimulating hormone (FSH) levels, and reduced anti-Müllerian hormone (AMH) concentrations (1). While normal FSH may provide false reassurance, AMH decline often precedes FSH elevation, making it a more sensitive early biomarker with superior prognostic accuracy. Research has shown that combining multiple biomarkers provides more effective clinical counseling than single markers alone (2). Consistent with biomarker-oriented genetic studies, recent bioinformatic analyses of pathogenic variants support the exploration of gene-based biomarkers in complex disorders (3).
Premature ovarian insufficiency (POI) and DOR constitute pathophysiologically convergent reproductive disorders that culminate in aberrant ovarian follicular dynamics, compromising reproductive capacity, and it has been suggested that DOR might potentially mitigate the subsequent development of POI (4), which may sometimes need assisted reproductive methods (5).
The etiology of DOR encompasses many factors, such as autoimmune diseases, environmental factors, iatrogenic causes such as ovarian surgery, chemotherapy, radiotherapy, and genetic factors. While some of these genetic causes have been established, research continues on numerous candidate genes (6). Madihi et al. showed that the expression of some key meiotic recombination genes (STAG3, MCM8, DMC1, FANCM, and RBBP8) was significantly downregulated in granulosa cells of women with DOR compared to controls (7). STAG3 is exclusively expressed in gonads and encodes a meiosis-specific cohesin subunit located at chromosomal region 7q22.1. The meiotic cohesin complex is essential for the integrity of the synaptonemal complex and for proper chromosome pairing during meiosis I. STAG3 was first identified as a POI-associated gene in 2014, with homozygous mutations causing primary amenorrhea and infertility in both sexes (8, 9). DMC1, a meiosis-specific gene at 22q13 encoding a 340-amino acid protein, is crucial for homologous recombination and double-strand break repair. Functionally coordinated with RAD51 during meiotic prophase, DMC1 facilitates synaptonemal complex formation and chromosome crossover. Knockout models show meiotic arrest and infertility resembling POI (10).
In the present study, we aim to quantitatively assess the differential expression profiles of the meiosis-specific genes STAG3 and DMC1 to elucidate their potential roles as noninvasive diagnostic biomarkers and regulatory perturbations associated with DOR.
2. Materials and Methods
2.1. Participants
This case-control study was conducted from June 2023 to May 2025 at the Yazd Research Institute of Reproductive Sciences, Yazd, Iran. A total of 50 Iranian women were enrolled: 25 women diagnosed with DOR, comprising the case group, and 25 healthy women serving as controls.
Inclusion criteria for DOR cases, including those women who were aged between 20 and 35 yr, with normal body mass index (BMI) and AMH levels < 1.1 ng/mL, were eligible. Control group criteria included healthy women aged between 20 and 35 yr with normal BMI attending the clinic for male-factor infertility were recruited as controls, ensuring they met the same exclusion criteria as cases but without ovarian dysfunction.
Participants with severe endometriosis (stages III-IV), history of infectious diseases, congenital disorders, including type 1 autoimmune polyendocrinopathy syndrome, premature aging syndromes, pseudohypoparathyroidism type 1a, or Demiran syndrome that could affect ovarian reserve were excluded. Additionally, women with previous ovarian or pelvic surgeries that might compromise ovarian function were not eligible for participation.
2.2. RNA extraction and complementary DNA (cDNA) synthesis
Peripheral blood samples (5 mL) were collected from all participants in anticoagulant complete blood count tubes and centrifuged at 3000 rpm for 5 min to isolate peripheral blood mononuclear cells (PBMCs). Total RNA extraction was performed using a column-based silica method under sterile conditions in a biosafety cabinet maintained at 4°C to prevent RNA degradation. The extraction protocol involved initial cell lysis using TRIzol reagent, followed by phase separation with chloroform (200 μL) and centrifugation at 13,000 rpm for 15 min at 4°C.
The aqueous phase containing RNA was collected and precipitated with isopropanol, then purified using silica spin columns with multiple ethanol-based wash steps. Final elution was performed with 20 μL of RNase-free water. RNA quality and quantity were assessed using NanoDrop spectrophotometry, with absorbance at 260 nm for concentration determination. RNA purity was evaluated through A260/A280 ratios (acceptable range: 1.8-2.0) and A260/A230 ratios (acceptable range: 2.0-2.2) to exclude protein and phenol contamination, respectively. RNA integrity was confirmed by agarose gel electrophoresis, verifying the presence of distinct 18S, 28S, and 5S ribosomal RNA bands. The cDNA synthesis was performed using a commercial reverse transcription kit (Pars Tous, Iran) according to the manufacturer's instructions, with RNA samples stored at -80°C until cDNA synthesis.
2.3. Real-time polymerase chain reaction (PCR)
Quantitative real-time PCR was performed using 2x qPCRBIO Sygreen Mix Hi-ROX to analyze the expression levels of DMC1 and STAG3 genes. Specific primers were designed for target genes and validated for efficiency and specificity. Gene expression levels were normalized to the appropriate housekeeping gene (18srRNA), and relative expression was calculated using the 2^-ΔCt method, comparing DOR women to healthy controls.
2.4. Ethical Considerations
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. The research protocol received approval from the Shahid Sadoughi University of Medical Sciences, Yazd, Iran ethics committee (Code: IR.SSU.MEDICINE.REC.1402.122). Written informed consent was obtained from all participants before enrollment. All biological samples were handled according to institutional biosafety guidelines, and participant confidentiality was maintained throughout the study.
2.5. Statistical Analysis
All statistical analyses were conducted using GraphPad Prism version 10 (GraphPad Software, San Diego, CA, USA). The distribution of the data was initially examined using the Shapiro-Wilk test to assess normality. Since the data did not follow a normal distribution, comparisons of gene expression levels between the 2 groups were performed using the non-parametric Mann-Whitney U test. Graphical illustrations of gene expression differences were produced in GraphPad Prism. A p < 0.05 was considered statistically significant.
3. Results
3.1. Demographic and clinical characteristics of study participants
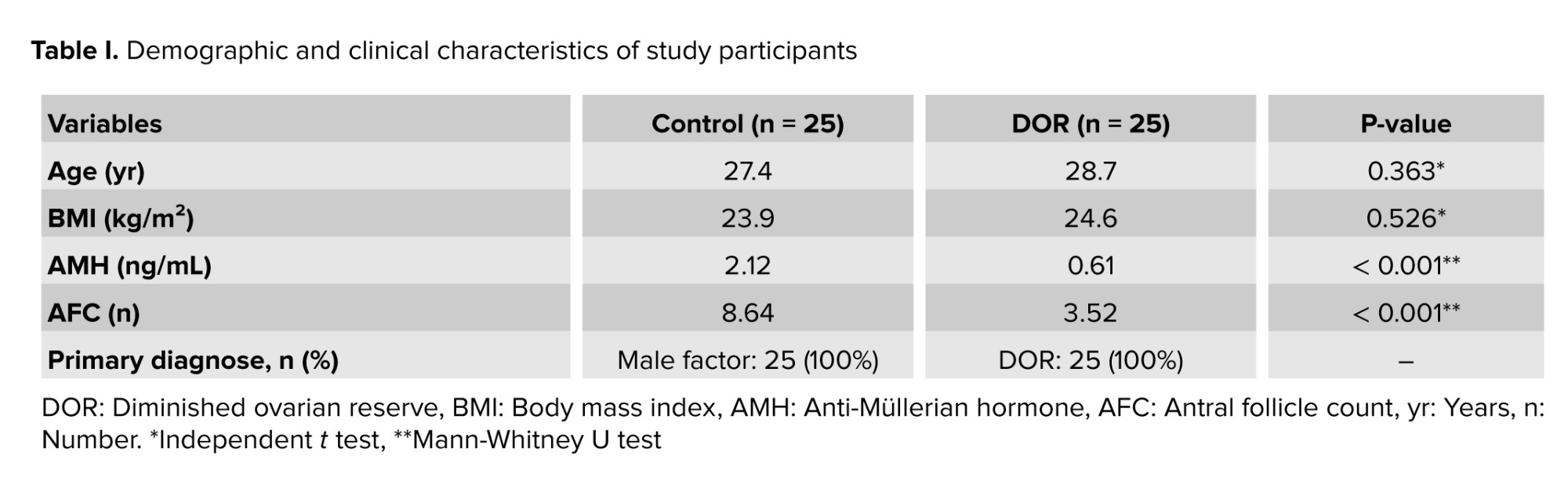
A total of 50 participants were enrolled in this case-control study, comprising 25 women with DOR and 25 healthy controls. The demographic analysis (Table I), revealed that the mean age of DOR women was 28.7 yr, compared with 27.4 yr in the control group, with no statistically significant difference between the groups (p > 0.05). Similarly, BMI values showed no significant variation between DOR women (24.6 kg/m²) and controls (23.9 kg/m²), indicating successful matching of participants for this potential confounding variable.
As expected, significant differences were observed in reproductive parameters between the groups (Table I). AMH levels were markedly reduced in the DOR group (0.61 ng/mL) compared to controls (2.12 ng/mL), confirming the diagnostic criteria for DOR. Antral follicle count was similarly decreased in DOR women (3.52) vs. controls (8.64), further validating the clinical classification of participants.
3.2. Gene expression analysis of STAG3 and DMC1
3.2.1. STAG3 expression in PBMCs
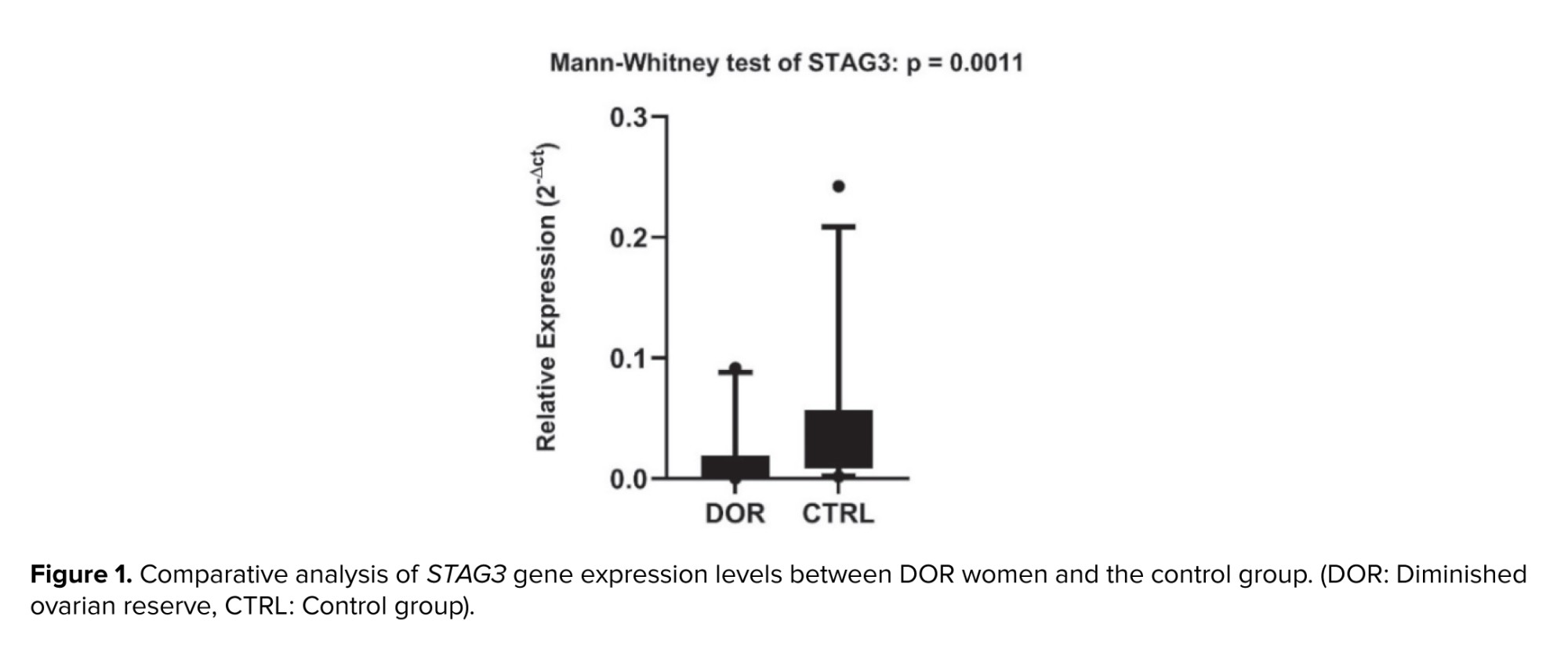
Quantitative real-time PCR analysis revealed significant downregulation of STAG3 gene expression in PBMCs of women with DOR compared to healthy controls. The relative expression analysis demonstrated a statistically significant reduction in STAG3 mRNA levels (p = 0.0011, Figure 1), indicating potential involvement of this meiotic recombination gene in the pathophysiology of DOR.
3.2.2. DMC1 expression analysis in blood cells
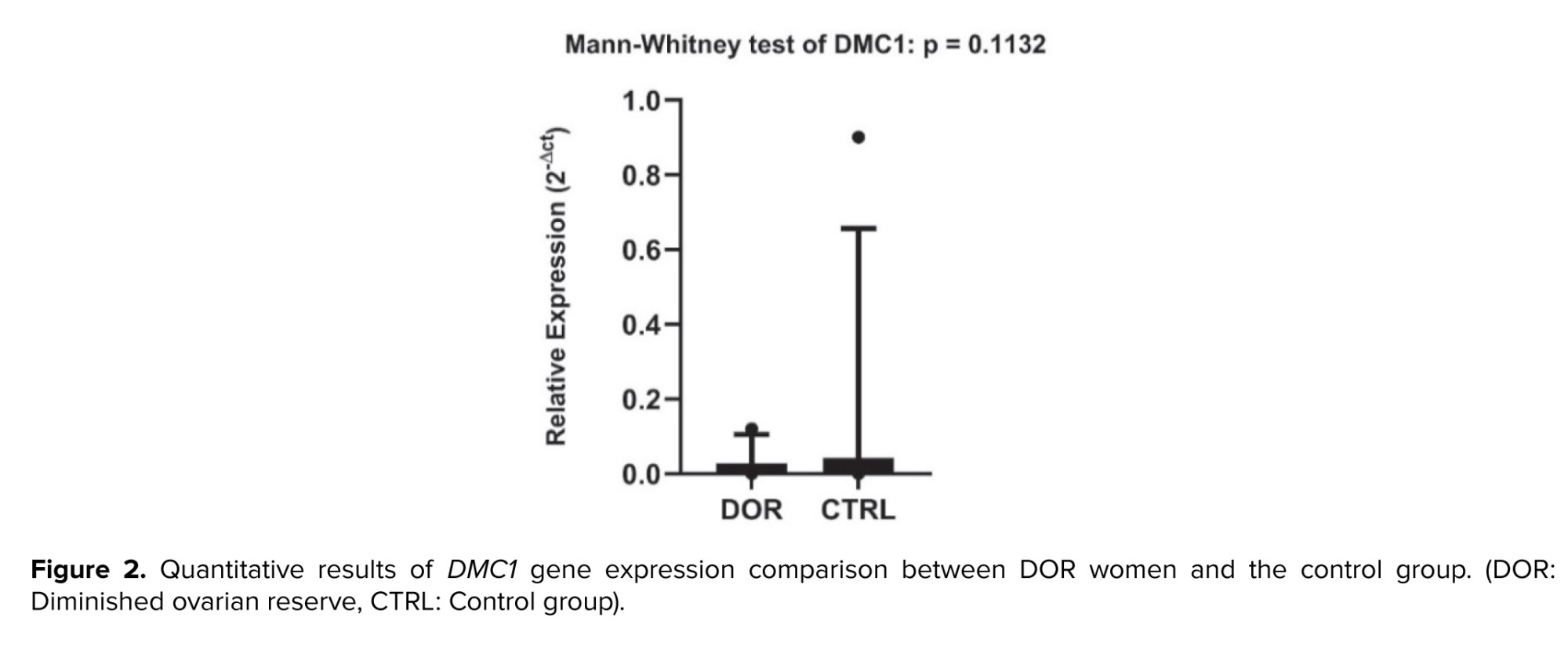
The expression analysis of the DMC1 gene in PBMCs revealed a tendency toward reduced expression in DOR women compared to controls; however, this reduction did not reach statistical significance (p = 0.1132, Figure 2). Although the downregulation pattern was consistent with the STAG3 findings, the lack of statistical significance suggests that DMC1 expression changes may be more subtle or require larger sample sizes for detection.
The differential expression patterns observed between STAG3 and DMC1 suggest that meiotic recombination pathway genes may be variably affected in DOR pathogenesis, with STAG3 potentially serving as a more sensitive biomarker for ovarian reserve dysfunction.
4. Discussion
The present study investigated the expression patterns of key meiotic recombination genes, STAG3 and DMC1, in PBMCs of women with DOR compared to healthy controls. Our findings demonstrate significant downregulation of STAG3 expression (p = 0.0011), aligning with previous studies showing the critical role of STAG3 in ovarian function and a non-significant reduction in DMC1 expression in DOR women, providing novel insights into the molecular mechanisms underlying ovarian reserve dysfunction (11).
This significant reduction of STAG3 expression in DOR women suggests that transcriptional dysregulation of this gene may contribute to the pathophysiology of ovarian reserve depletion even before the complete ovarian failure seen in POI. This supports the hypothesis that DOR represents an early manifestation of the spectrum of STAG3-related ovarian dysfunction (12). Previous work by Madihi et al. (7) similarly demonstrated downregulation of STAG3 in granulosa cells of DOR women, and also aligns with genetic evidence from Demain et al. who reported biallelic STAG3 loss-of-function variants causing primary ovarian insufficiency. This suggests that transcriptional attenuation and disruptive mutations of STAG3 converge on common pathogenic pathways leading to ovarian reserve depletion, corroborating our findings in a different cell type and reinforcing the systemic nature of meiotic gene dysregulation in ovarian reserve disorders (11).
While DMC1 expression showed a downward trend in DOR women, the difference was not statistically significant (p = 0.1132). This finding contrasts with some previous reports but aligns with others showing variable DMC1 defects involving ovarian dysfunction. Another study using next-generation sequencing in 269 POI women of diverse ethnic backgrounds, including Asian origin, identified DMC1 variants (c.449G>A, p.Gly150Asp; c.598A>G, p.Met200Val) in 7% of cases (13), while subsequent studies by Wang et al. found no association between DMC1 mutations and premature ovarian failure in Chinese women (14). More recently, Cao et al. reported a pathogenic DMC1 frameshift mutation causing both non-obstructive azoospermia and DOR, but noted that DMC1 appears dispensable for human oogenesis compared to spermatogenesis (15).
The non-significant reduction in DMC1 expression in our study may reflect the differential requirements for DMC1 function between species and sexes. Mouse studies show complete germ cell loss in DMC1-deficient females, whereas human cases demonstrate more variable phenotypes, suggesting compensatory mechanisms or different threshold effects for DMC1 function in human ovarian development (13). The correlation between peripheral blood gene expression and ovarian pathology has been supported by studies showing that circulating immune cells reflect tissue-specific disease processes (16). This systemic reflection of local pathology may be particularly relevant for genes involved in fundamental cellular processes, such as meiosis, which are expressed across multiple tissue types during development (17).
Our findings indicate that STAG3 expression in PBMCs may serve as a valuable biomarker for early detection of ovarian reserve dysfunction, with its significant downregulation in DOR women supporting a continuum of STAG3-related ovarian pathology linked to POI. Yu et al. demonstrated that peripheral blood immune cells show distinct expression patterns in POI women, with altered B cells, NK cells, and monocyte populations (18). Similarly, our approach of measuring meiotic gene expression in PBMCs provides a non-invasive method for assessing systemic dysregulation of genes critical for ovarian function. This highlights the potential for early identification and timely fertility preservation. The recent transcriptomic analyses of peripheral blood in ovarian insufficiency support this perspective. Wu et al. performed RNA-seq on peripheral blood from POI patients and identified 39 differentially expressed genes (including SLC25A39 and CNIH3) (19), underscoring that blood transcripts mirror ovarian dysfunction. Zhang et al. used single-cell RNA-seq of PBMCs in POI and reported altered immune cell subsets and gene expression patterns (e.g., reduced LYZ and altered IGKC) (20), further linking circulating immune gene signatures to ovarian failure. Additionally, a recent meta-analysis identified blood NSUN4 levels as a mitochondrial biomarker of ovarian aging (21). Together, these studies reinforce that PBMC expression changes can serve as non-invasive indicators of ovarian status. In line with this, our observation of markedly lower STAG3 (but not DMC1) in DOR PBMCs suggests STAG3 a potential early blood biomarker of declining ovarian reserve. The contrasting STAG3 vs.DMC1 patterns highlight differential susceptibility of meiotic components during human ovarian aging.
4.1. Strengths and Limitations
Our study has limitations, including the relatively small sample size and reliance on PBMCs rather than ovarian tissue for gene expression analysis. Future research should validate these findings in larger cohorts and explore the relationship between peripheral blood and ovarian tissue expression. Functional studies on the impact of reduced STAG3 expression on meiotic processes are also needed. Nonetheless, our results highlight detectable meiotic gene dysregulation, particularly STAG3 downregulation, in DOR women’s blood, advancing understanding of DOR pathophysiology and supporting non-invasive biomarker development in reproductive medicine.
5. Conclusion
Reduced expression of STAG3 in women with DOR underscores its promise as a noninvasive molecular biomarker for early ovarian dysfunction detection. These findings lay the foundation for precision and personalized reproductive medicine, where individual molecular profiles can predict susceptibility to POI and inform tailored interventions. Future research should focus on expanding biomarker discovery and integrating molecular diagnostics into clinical practice to enable genotype‑driven management and optimize reproductive health outcomes.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Author Contributions
Kh. Namjoo had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Acquisition, analysis, or interpretation of data: Kh. Namjoo was responsible for patient recruitment, sample collection, and performance of genetic experiments. Kh. Namjoo and F. Iravani contributed molecular genetic analysis and data interpretation. F. Montazeri provided technical training and methodological support during laboratory procedures and contributed to data acquisition. Drafting of the manuscript: Kh. Namjoo and F. Iravani. Statistical analysis: Kh. Namjoo and F. Iravani. Concept and design: MH. Jafari Najaf Abadi, SM. Kalantar, and N. Ghasemi. Supervision: MH. Jafari Najaf Abadi, S.M. Kalantar, and N. Ghasemi. Critical revision of the manuscript for important intellectual content: All authors. N. Ghasemi served as the corresponding author. All authors approved the final manuscript and agree to be accountable for all aspects of the work.
Acknowledgments
The authors would sincerely like to thank the laboratory technical staff of the Yazd Research Institute of Reproductive Sciences,Yazd, Iran. for their valuable assistance with sample processing and molecular analyses. We are also deeply grateful to all the participants who generously volunteered for this study; their cooperation and trust made this research possible. Artificial intelligence (Chat GPT-4) was utilized for translation and grammar-checking assistance.
Conflict of Interest
The authors declare that there is no conflict of interest.
Diminished ovarian reserve (DOR) represents a significant cause of female infertility, characterized by reduced oocyte quantity and quality. Diagnosis involves decreased antral follicle count, elevated follicle-stimulating hormone (FSH) levels, and reduced anti-Müllerian hormone (AMH) concentrations (1). While normal FSH may provide false reassurance, AMH decline often precedes FSH elevation, making it a more sensitive early biomarker with superior prognostic accuracy. Research has shown that combining multiple biomarkers provides more effective clinical counseling than single markers alone (2). Consistent with biomarker-oriented genetic studies, recent bioinformatic analyses of pathogenic variants support the exploration of gene-based biomarkers in complex disorders (3).
Premature ovarian insufficiency (POI) and DOR constitute pathophysiologically convergent reproductive disorders that culminate in aberrant ovarian follicular dynamics, compromising reproductive capacity, and it has been suggested that DOR might potentially mitigate the subsequent development of POI (4), which may sometimes need assisted reproductive methods (5).
The etiology of DOR encompasses many factors, such as autoimmune diseases, environmental factors, iatrogenic causes such as ovarian surgery, chemotherapy, radiotherapy, and genetic factors. While some of these genetic causes have been established, research continues on numerous candidate genes (6). Madihi et al. showed that the expression of some key meiotic recombination genes (STAG3, MCM8, DMC1, FANCM, and RBBP8) was significantly downregulated in granulosa cells of women with DOR compared to controls (7). STAG3 is exclusively expressed in gonads and encodes a meiosis-specific cohesin subunit located at chromosomal region 7q22.1. The meiotic cohesin complex is essential for the integrity of the synaptonemal complex and for proper chromosome pairing during meiosis I. STAG3 was first identified as a POI-associated gene in 2014, with homozygous mutations causing primary amenorrhea and infertility in both sexes (8, 9). DMC1, a meiosis-specific gene at 22q13 encoding a 340-amino acid protein, is crucial for homologous recombination and double-strand break repair. Functionally coordinated with RAD51 during meiotic prophase, DMC1 facilitates synaptonemal complex formation and chromosome crossover. Knockout models show meiotic arrest and infertility resembling POI (10).
In the present study, we aim to quantitatively assess the differential expression profiles of the meiosis-specific genes STAG3 and DMC1 to elucidate their potential roles as noninvasive diagnostic biomarkers and regulatory perturbations associated with DOR.
2. Materials and Methods
2.1. Participants
This case-control study was conducted from June 2023 to May 2025 at the Yazd Research Institute of Reproductive Sciences, Yazd, Iran. A total of 50 Iranian women were enrolled: 25 women diagnosed with DOR, comprising the case group, and 25 healthy women serving as controls.
Inclusion criteria for DOR cases, including those women who were aged between 20 and 35 yr, with normal body mass index (BMI) and AMH levels < 1.1 ng/mL, were eligible. Control group criteria included healthy women aged between 20 and 35 yr with normal BMI attending the clinic for male-factor infertility were recruited as controls, ensuring they met the same exclusion criteria as cases but without ovarian dysfunction.
Participants with severe endometriosis (stages III-IV), history of infectious diseases, congenital disorders, including type 1 autoimmune polyendocrinopathy syndrome, premature aging syndromes, pseudohypoparathyroidism type 1a, or Demiran syndrome that could affect ovarian reserve were excluded. Additionally, women with previous ovarian or pelvic surgeries that might compromise ovarian function were not eligible for participation.
2.2. RNA extraction and complementary DNA (cDNA) synthesis
Peripheral blood samples (5 mL) were collected from all participants in anticoagulant complete blood count tubes and centrifuged at 3000 rpm for 5 min to isolate peripheral blood mononuclear cells (PBMCs). Total RNA extraction was performed using a column-based silica method under sterile conditions in a biosafety cabinet maintained at 4°C to prevent RNA degradation. The extraction protocol involved initial cell lysis using TRIzol reagent, followed by phase separation with chloroform (200 μL) and centrifugation at 13,000 rpm for 15 min at 4°C.
The aqueous phase containing RNA was collected and precipitated with isopropanol, then purified using silica spin columns with multiple ethanol-based wash steps. Final elution was performed with 20 μL of RNase-free water. RNA quality and quantity were assessed using NanoDrop spectrophotometry, with absorbance at 260 nm for concentration determination. RNA purity was evaluated through A260/A280 ratios (acceptable range: 1.8-2.0) and A260/A230 ratios (acceptable range: 2.0-2.2) to exclude protein and phenol contamination, respectively. RNA integrity was confirmed by agarose gel electrophoresis, verifying the presence of distinct 18S, 28S, and 5S ribosomal RNA bands. The cDNA synthesis was performed using a commercial reverse transcription kit (Pars Tous, Iran) according to the manufacturer's instructions, with RNA samples stored at -80°C until cDNA synthesis.
2.3. Real-time polymerase chain reaction (PCR)
Quantitative real-time PCR was performed using 2x qPCRBIO Sygreen Mix Hi-ROX to analyze the expression levels of DMC1 and STAG3 genes. Specific primers were designed for target genes and validated for efficiency and specificity. Gene expression levels were normalized to the appropriate housekeeping gene (18srRNA), and relative expression was calculated using the 2^-ΔCt method, comparing DOR women to healthy controls.
2.4. Ethical Considerations
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. The research protocol received approval from the Shahid Sadoughi University of Medical Sciences, Yazd, Iran ethics committee (Code: IR.SSU.MEDICINE.REC.1402.122). Written informed consent was obtained from all participants before enrollment. All biological samples were handled according to institutional biosafety guidelines, and participant confidentiality was maintained throughout the study.
2.5. Statistical Analysis
All statistical analyses were conducted using GraphPad Prism version 10 (GraphPad Software, San Diego, CA, USA). The distribution of the data was initially examined using the Shapiro-Wilk test to assess normality. Since the data did not follow a normal distribution, comparisons of gene expression levels between the 2 groups were performed using the non-parametric Mann-Whitney U test. Graphical illustrations of gene expression differences were produced in GraphPad Prism. A p < 0.05 was considered statistically significant.
3. Results
3.1. Demographic and clinical characteristics of study participants
A total of 50 participants were enrolled in this case-control study, comprising 25 women with DOR and 25 healthy controls. The demographic analysis (Table I), revealed that the mean age of DOR women was 28.7 yr, compared with 27.4 yr in the control group, with no statistically significant difference between the groups (p > 0.05). Similarly, BMI values showed no significant variation between DOR women (24.6 kg/m²) and controls (23.9 kg/m²), indicating successful matching of participants for this potential confounding variable.
As expected, significant differences were observed in reproductive parameters between the groups (Table I). AMH levels were markedly reduced in the DOR group (0.61 ng/mL) compared to controls (2.12 ng/mL), confirming the diagnostic criteria for DOR. Antral follicle count was similarly decreased in DOR women (3.52) vs. controls (8.64), further validating the clinical classification of participants.
3.2. Gene expression analysis of STAG3 and DMC1
3.2.1. STAG3 expression in PBMCs
Quantitative real-time PCR analysis revealed significant downregulation of STAG3 gene expression in PBMCs of women with DOR compared to healthy controls. The relative expression analysis demonstrated a statistically significant reduction in STAG3 mRNA levels (p = 0.0011, Figure 1), indicating potential involvement of this meiotic recombination gene in the pathophysiology of DOR.
3.2.2. DMC1 expression analysis in blood cells
The expression analysis of the DMC1 gene in PBMCs revealed a tendency toward reduced expression in DOR women compared to controls; however, this reduction did not reach statistical significance (p = 0.1132, Figure 2). Although the downregulation pattern was consistent with the STAG3 findings, the lack of statistical significance suggests that DMC1 expression changes may be more subtle or require larger sample sizes for detection.
The differential expression patterns observed between STAG3 and DMC1 suggest that meiotic recombination pathway genes may be variably affected in DOR pathogenesis, with STAG3 potentially serving as a more sensitive biomarker for ovarian reserve dysfunction.



4. Discussion
The present study investigated the expression patterns of key meiotic recombination genes, STAG3 and DMC1, in PBMCs of women with DOR compared to healthy controls. Our findings demonstrate significant downregulation of STAG3 expression (p = 0.0011), aligning with previous studies showing the critical role of STAG3 in ovarian function and a non-significant reduction in DMC1 expression in DOR women, providing novel insights into the molecular mechanisms underlying ovarian reserve dysfunction (11).
This significant reduction of STAG3 expression in DOR women suggests that transcriptional dysregulation of this gene may contribute to the pathophysiology of ovarian reserve depletion even before the complete ovarian failure seen in POI. This supports the hypothesis that DOR represents an early manifestation of the spectrum of STAG3-related ovarian dysfunction (12). Previous work by Madihi et al. (7) similarly demonstrated downregulation of STAG3 in granulosa cells of DOR women, and also aligns with genetic evidence from Demain et al. who reported biallelic STAG3 loss-of-function variants causing primary ovarian insufficiency. This suggests that transcriptional attenuation and disruptive mutations of STAG3 converge on common pathogenic pathways leading to ovarian reserve depletion, corroborating our findings in a different cell type and reinforcing the systemic nature of meiotic gene dysregulation in ovarian reserve disorders (11).
While DMC1 expression showed a downward trend in DOR women, the difference was not statistically significant (p = 0.1132). This finding contrasts with some previous reports but aligns with others showing variable DMC1 defects involving ovarian dysfunction. Another study using next-generation sequencing in 269 POI women of diverse ethnic backgrounds, including Asian origin, identified DMC1 variants (c.449G>A, p.Gly150Asp; c.598A>G, p.Met200Val) in 7% of cases (13), while subsequent studies by Wang et al. found no association between DMC1 mutations and premature ovarian failure in Chinese women (14). More recently, Cao et al. reported a pathogenic DMC1 frameshift mutation causing both non-obstructive azoospermia and DOR, but noted that DMC1 appears dispensable for human oogenesis compared to spermatogenesis (15).
The non-significant reduction in DMC1 expression in our study may reflect the differential requirements for DMC1 function between species and sexes. Mouse studies show complete germ cell loss in DMC1-deficient females, whereas human cases demonstrate more variable phenotypes, suggesting compensatory mechanisms or different threshold effects for DMC1 function in human ovarian development (13). The correlation between peripheral blood gene expression and ovarian pathology has been supported by studies showing that circulating immune cells reflect tissue-specific disease processes (16). This systemic reflection of local pathology may be particularly relevant for genes involved in fundamental cellular processes, such as meiosis, which are expressed across multiple tissue types during development (17).
Our findings indicate that STAG3 expression in PBMCs may serve as a valuable biomarker for early detection of ovarian reserve dysfunction, with its significant downregulation in DOR women supporting a continuum of STAG3-related ovarian pathology linked to POI. Yu et al. demonstrated that peripheral blood immune cells show distinct expression patterns in POI women, with altered B cells, NK cells, and monocyte populations (18). Similarly, our approach of measuring meiotic gene expression in PBMCs provides a non-invasive method for assessing systemic dysregulation of genes critical for ovarian function. This highlights the potential for early identification and timely fertility preservation. The recent transcriptomic analyses of peripheral blood in ovarian insufficiency support this perspective. Wu et al. performed RNA-seq on peripheral blood from POI patients and identified 39 differentially expressed genes (including SLC25A39 and CNIH3) (19), underscoring that blood transcripts mirror ovarian dysfunction. Zhang et al. used single-cell RNA-seq of PBMCs in POI and reported altered immune cell subsets and gene expression patterns (e.g., reduced LYZ and altered IGKC) (20), further linking circulating immune gene signatures to ovarian failure. Additionally, a recent meta-analysis identified blood NSUN4 levels as a mitochondrial biomarker of ovarian aging (21). Together, these studies reinforce that PBMC expression changes can serve as non-invasive indicators of ovarian status. In line with this, our observation of markedly lower STAG3 (but not DMC1) in DOR PBMCs suggests STAG3 a potential early blood biomarker of declining ovarian reserve. The contrasting STAG3 vs.DMC1 patterns highlight differential susceptibility of meiotic components during human ovarian aging.
4.1. Strengths and Limitations
Our study has limitations, including the relatively small sample size and reliance on PBMCs rather than ovarian tissue for gene expression analysis. Future research should validate these findings in larger cohorts and explore the relationship between peripheral blood and ovarian tissue expression. Functional studies on the impact of reduced STAG3 expression on meiotic processes are also needed. Nonetheless, our results highlight detectable meiotic gene dysregulation, particularly STAG3 downregulation, in DOR women’s blood, advancing understanding of DOR pathophysiology and supporting non-invasive biomarker development in reproductive medicine.
5. Conclusion
Reduced expression of STAG3 in women with DOR underscores its promise as a noninvasive molecular biomarker for early ovarian dysfunction detection. These findings lay the foundation for precision and personalized reproductive medicine, where individual molecular profiles can predict susceptibility to POI and inform tailored interventions. Future research should focus on expanding biomarker discovery and integrating molecular diagnostics into clinical practice to enable genotype‑driven management and optimize reproductive health outcomes.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Author Contributions
Kh. Namjoo had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Acquisition, analysis, or interpretation of data: Kh. Namjoo was responsible for patient recruitment, sample collection, and performance of genetic experiments. Kh. Namjoo and F. Iravani contributed molecular genetic analysis and data interpretation. F. Montazeri provided technical training and methodological support during laboratory procedures and contributed to data acquisition. Drafting of the manuscript: Kh. Namjoo and F. Iravani. Statistical analysis: Kh. Namjoo and F. Iravani. Concept and design: MH. Jafari Najaf Abadi, SM. Kalantar, and N. Ghasemi. Supervision: MH. Jafari Najaf Abadi, S.M. Kalantar, and N. Ghasemi. Critical revision of the manuscript for important intellectual content: All authors. N. Ghasemi served as the corresponding author. All authors approved the final manuscript and agree to be accountable for all aspects of the work.
Acknowledgments
The authors would sincerely like to thank the laboratory technical staff of the Yazd Research Institute of Reproductive Sciences,Yazd, Iran. for their valuable assistance with sample processing and molecular analyses. We are also deeply grateful to all the participants who generously volunteered for this study; their cooperation and trust made this research possible. Artificial intelligence (Chat GPT-4) was utilized for translation and grammar-checking assistance.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Original Article |
Subject:
Reproductive Genetics
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |



