Wed, Jun 10, 2026
[Archive]
Volume 23, Issue 12 (December 2025)
IJRM 2025, 23(12): 971-984 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Vasiee M, Agha-Rahimi A. Complex in vitro fertilization laboratory factors affecting sperm DNA integrity: A narrative review. IJRM 2025; 23 (12) :971-984
URL: http://ijrm.ir/article-1-3743-en.html
URL: http://ijrm.ir/article-1-3743-en.html
1- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
2- Research and Clinical Center for Infertility, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,a.agharahimi@ssu.ac.ir; 63rahimi@gmail.com
2- Research and Clinical Center for Infertility, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran. ,
Keywords: Sperm, DNA fragmentation, Male infertility, Oxidative stress, Assisted reproductive technologies.
Full-Text [PDF 684 kb]
(434 Downloads)
| Abstract (HTML) (427 Views)
6.3. Glove materials in ART lab
While gloves are indispensable in ART laboratories for maintaining aseptic conditions and ensuring operator safety, certain glove materials and surface treatments may inadvertently compromise gamete integrity. Specifically, VOCs, powder particles, and chemical residues released from gloves have been linked to an increase in SDF. Composed of natural rubber proteins and chemical accelerators such as thiurams, carbamates, and benzothiazoles, latex gloves pose a risk of leaching toxic substances into culture media or directly onto gametes. Powdered latex gloves, though largely phased out in ART settings can release fine particles that contaminate sperm dishes and media, contributing to DNA damage. Despite the phase-out of powdered gloves in ART settings, residues from gloves still pose a risk to gamete integrity, emphasizing the critical need to use powder-free and carefully tested gloves in ART laboratories to avoid compromising sperm and embryo quality (77). Considered safer than latex, nitrile gloves still carry risks. Residual sulfur-based compounds and zinc may leach from the glove surface, and lower-quality nitrile variants have been shown to emit VOCs that can impair both sperm and oocyte quality (73). Polyvinyl chloride gloves are known to release phthalates, plasticizers with endocrine-disrupting properties. These compounds have been associated with compromised chromatin structure and elevated SDF levels. The study emphasizes that variability in phthalate content exists between glove brands and even batches, complicating the assurance of phthalate-free gloves. Due to their hormone-disrupting properties, phthalates from polyvinyl chloride gloves can negatively affect sperm quality and reproductive outcomes in ART settings (78).
Thus, selecting gloves in ART laboratories requires a careful assessment of chemical hazards, exposure levels, and task sensitivity. Powder-free, high-quality gloves that minimize leaching of toxic chemicals such as phthalates, VOCs, and sulfur compounds are essential to protect sperm DNA integrity and avoid contamination. Nitrile gloves are generally preferred due to their chemical resistance and low allergy risk. Whereas latex and vinyl gloves pose higher risks from chemical residues and plasticizers. Proper practices include consulting safety data sheets, inspecting gloves for damage, and timely replacement to maintain a safe environment for gametes and personnel, thereby supporting optimal ART outcomes.
7. Conclusion
Sperm DNA integrity is a critical determinant of male fertility and ART success. Complex factors, spanning medical, surgical, and laboratory practices significantly compromise the sperm genomic stability through OS, mechanical injury, and chemical exposure. Among laboratory procedures, excessive centrifugation, prolonged incubation, inappropriate temperature or pH, and exposure to ROS are major contributors to DNA fragmentation. Similarly, the choice of materials, such as culture media, cryoprotectants, and even gloves or plasticware, can influence oxidative balance and DNA quality. To ensure optimal outcomes, laboratories must implement evidence-based protocols emphasizing gentle sperm handling, antioxidant supplementation, strict air-quality control, and validation of all consumables under sperm-safe conditions. Training embryologists in minimizing mechanical and OS is equally vital. Future research should focus on developing standardized assessment methods for sperm DNA integrity and on exploring novel microfluidic and non-invasive sperm selection techniques to minimize damage.
Acknowledgments
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The authors used ChatGPT (GPT-5, OpenAI) only for improving the grammar and language clarity of the manuscript. The authors reviewed and take full responsibility for the content.
Conflict of Interest
The authors declare that there is no conflict of interest.
Full-Text: (85 Views)
1. Introduction
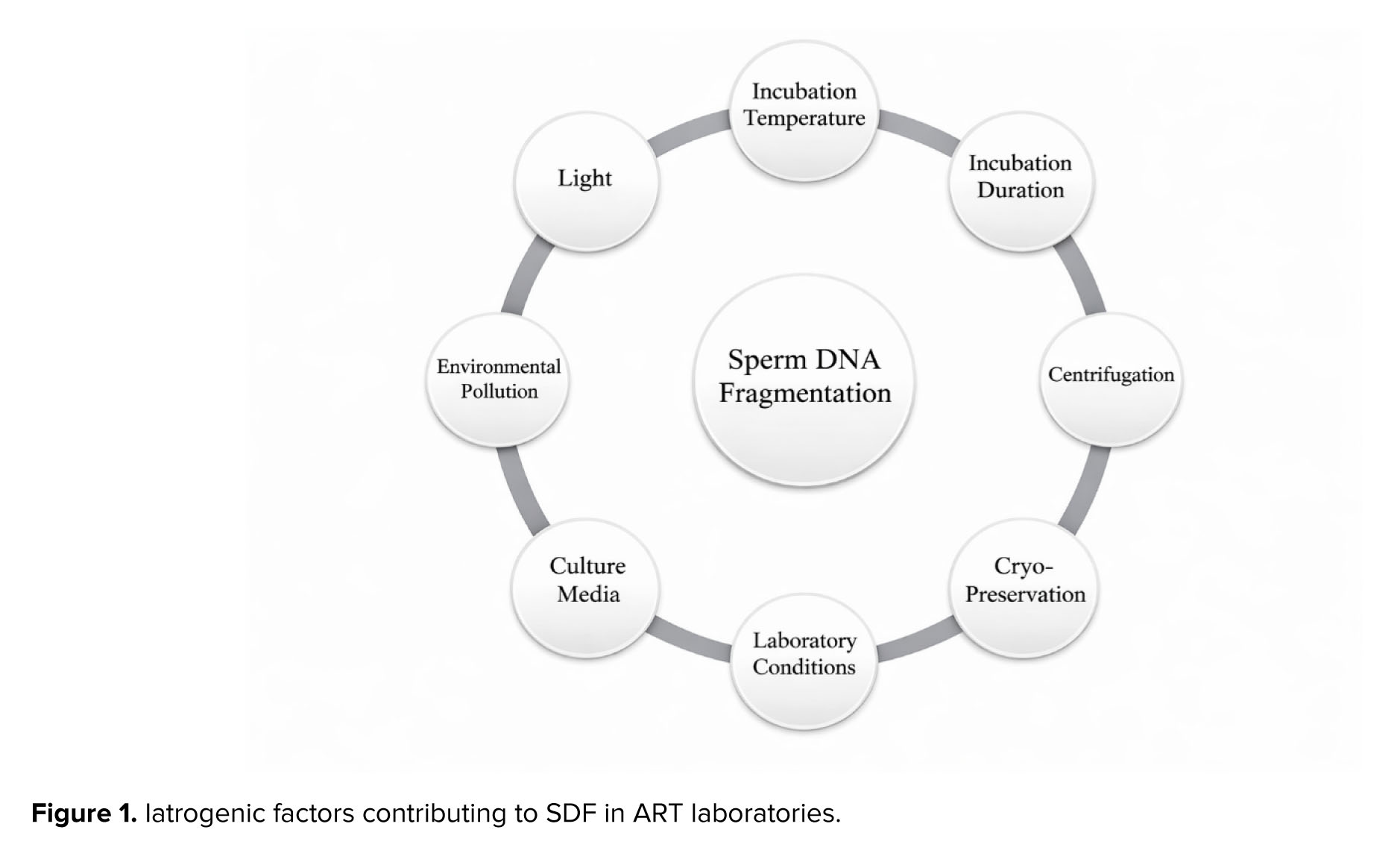
Conventional semen analysis does not assess the DNA integrity of spermatozoa. Thus, sperm DNA fragmentation (SDF) testing provides a more direct measure of genetic quality and fertility potential (1-3). Sperm DNA is normally compacted by protamines and disulfide bonds yet it remains vulnerable to iatrogenic damage during assisted reproductive technologies (ART) procedures. 2 main mechanisms contribute to SDF: abortive apoptosis, where incomplete cell death leaves DNA strand breaks, and oxidative stress (OS), in which reactive oxygen species (ROS) attack DNA bases and induce strand breaks (4, 5). Factors, such as temperature fluctuations, light exposure, and chemical agents (Figure 1) can elevate ROS levels, leading to lipid peroxidation, protein oxidation, chromatin relaxation, and ultimately DNA fragmentation. Therefore, minimizing oxidative triggers during sperm handling is essential.
SDF has emerged as a critical biomarker of male fertility, reflecting both chromatin integrity and the genetic quality of sperm. Elevated SDF has been consistently associated with infertility, impaired outcomes in ART, and increased risk of pregnancy loss (6-8). In men with mild male factor infertility, low pregnancy success rates are often linked to increased DNA fragmentation, in addition to reduced motility and higher levels of apoptosis (8, 9). Multiple factors influence SDF, including age, lifestyle, and infections. However, iatrogenic contributors, those arising from medical or laboratory interventions, are increasingly recognized as significant determinants (10, 11). The integrity of sperm DNA is essential for male fertility and for the proper development of the embryo. Conventional semen analysis, including sperm count, motility, and morphology does not provide information about DNA quality.
Since mature spermatozoa have minimal DNA repair capacity, any exogenous insult can accumulate. Iatrogenic interventions often elevate ROS or directly damage DNA (e.g., ionizing radiation), overwhelming antioxidant defenses. Removal of seminal plasma during sperm processing eliminates natural antioxidants such as superoxide dismutase, catalase, and glutathione, increasing susceptibility to oxidative injury. When ROS exceeds scavenging capacity, the DNA bases, particularly guanine, undergo oxidative modification (e.g., 8-oxoguanine), leading to strand breaks (12-14).
While many studies have investigated the impact of semen SDF on infertility outcomes, there has been comparatively less focus on preserving the integrity of seminal DNA within laboratory environments. Iatrogenic damage during ART through improper centrifugation, prolonged incubation, or OS from cryopreservation can compromise sperm genomic integrity, highlighting the need for standardized laboratory protocols (15). As illustrated in figure 1, various laboratory factors, including temperature, incubation duration, media composition, and even glove materials, may induce oxidative or mechanical stress on sperm chromatin. Controlling these parameters is essential to minimize DNA fragmentation and optimize ART outcomes. The aim of this review was to summarize complex factors and laboratory practices that may compromise sperm DNA integrity during ART procedures. It also highlights the underlying mechanisms of damage and proposes preventive laboratory strategies to minimize SDF and improve clinical outcomes.
2. Search strategy and selection criteria
This narrative review was conducted through a comprehensive search of the PubMed, Scopus, and Google Scholar databases for articles published between 2000 and 2025. The following keywords and their combinations were used: SDF, OS, iatrogenic factors, laboratory practices, and ART. Only English-language articles were included. Original research, clinical studies, and relevant review papers focusing on human sperm DNA integrity and ART-related laboratory interventions were considered. Studies involving animal models, non-reproductive systems, or unrelated oxidative mechanisms were excluded. Data were extracted based on methodological relevance, clarity of results, and contribution to understanding iatrogenic factors affecting sperm DNA integrity.
3. Physical factors and methods
3.1. Incubation temperature
The temperature at which semen or sperm suspensions are maintained in vitro has a strong impact on DNA integrity. At higher temperatures, sperm metabolism and ROS production increase, leading to accelerated DNA damage. In clinical studies, incubation at 37°C has been associated with a significant rise in SDF after only 2-3 hr, as measured by the sperm chromatin dispersion test (16). By contrast, routine handling at room temperature (RT) appears to be less harmful. Several guidelines recommend working at RT for short periods and avoiding unnecessary warming to 37°C, except when physiologically required (e.g., capacitation) (17). Supporting this, a study reported that although the DNA fragmentation index increased over time at both RT and 37°C, it was significantly lower after 24 h at RT than at 37°C (18).
Further studies confirm that incubation at RT maintains better motility and morphology, while reducing apoptosis and necrosis, compared with incubation at 35°C (19, 20). Even in asthenozoospermic samples, short-term incubation (2-4 hr) at RT or 37°C negatively affected DNA integrity and overall sperm quality (21). In summary, minimizing exposure to elevated temperatures and using gentle cooling strategies for prolonged storage are important measures to preserve DNA integrity during in vitro handling.
3.2. Sperm incubation duration
The duration of in vitro incubation or storage has a direct impact on SDF. Sperm naturally accumulate DNA damage after ejaculation if left unprotected, and experimental studies confirm that even a few hour of incubation can increase the DNA fragmentation index (16, 22). One investigation showed that SDF rose significantly after 2 hr of incubation at RT and after 4 hr at 37°C, underlining the importance of minimizing incubation time (23).
Testicular sperm retrieved from azoospermic men have been shown to display markedly higher DNA fragmentation when cryopreserved and subsequently incubated for 4-24 hr post-thaw, with damage continuing to increase over time. Furthermore, incubation at 37°C for as little as 1.5 hr has been associated with reduced progressive motility, abnormal morphology, and elevated DNA fragmentation (24). A progressive increase in DNA breaks has been observed in swim-up-selected sperm during long-term incubation, even in the absence of additional stressors (25). Taken together, these findings emphasize that sperm should be used for insemination or analysis as soon as possible after ejaculation. In clinical practice, most embryology laboratories begin sperm preparation within 30-60 min of collection and complete testing or intra cytoplasmic sperm injection (ICSI) procedures promptly. Prolonged storage at any temperature increases oxidative metabolism and endonuclease activity, thereby raising the risk of DNA fragmentation (26). As long as feasible, both the duration and the centrifugal force should be minimized.
3.3. Sperm processing using centrifugation
Conventional semen analysis does not assess the DNA integrity of spermatozoa. Thus, sperm DNA fragmentation (SDF) testing provides a more direct measure of genetic quality and fertility potential (1-3). Sperm DNA is normally compacted by protamines and disulfide bonds yet it remains vulnerable to iatrogenic damage during assisted reproductive technologies (ART) procedures. 2 main mechanisms contribute to SDF: abortive apoptosis, where incomplete cell death leaves DNA strand breaks, and oxidative stress (OS), in which reactive oxygen species (ROS) attack DNA bases and induce strand breaks (4, 5). Factors, such as temperature fluctuations, light exposure, and chemical agents (Figure 1) can elevate ROS levels, leading to lipid peroxidation, protein oxidation, chromatin relaxation, and ultimately DNA fragmentation. Therefore, minimizing oxidative triggers during sperm handling is essential.
SDF has emerged as a critical biomarker of male fertility, reflecting both chromatin integrity and the genetic quality of sperm. Elevated SDF has been consistently associated with infertility, impaired outcomes in ART, and increased risk of pregnancy loss (6-8). In men with mild male factor infertility, low pregnancy success rates are often linked to increased DNA fragmentation, in addition to reduced motility and higher levels of apoptosis (8, 9). Multiple factors influence SDF, including age, lifestyle, and infections. However, iatrogenic contributors, those arising from medical or laboratory interventions, are increasingly recognized as significant determinants (10, 11). The integrity of sperm DNA is essential for male fertility and for the proper development of the embryo. Conventional semen analysis, including sperm count, motility, and morphology does not provide information about DNA quality.
Since mature spermatozoa have minimal DNA repair capacity, any exogenous insult can accumulate. Iatrogenic interventions often elevate ROS or directly damage DNA (e.g., ionizing radiation), overwhelming antioxidant defenses. Removal of seminal plasma during sperm processing eliminates natural antioxidants such as superoxide dismutase, catalase, and glutathione, increasing susceptibility to oxidative injury. When ROS exceeds scavenging capacity, the DNA bases, particularly guanine, undergo oxidative modification (e.g., 8-oxoguanine), leading to strand breaks (12-14).
While many studies have investigated the impact of semen SDF on infertility outcomes, there has been comparatively less focus on preserving the integrity of seminal DNA within laboratory environments. Iatrogenic damage during ART through improper centrifugation, prolonged incubation, or OS from cryopreservation can compromise sperm genomic integrity, highlighting the need for standardized laboratory protocols (15). As illustrated in figure 1, various laboratory factors, including temperature, incubation duration, media composition, and even glove materials, may induce oxidative or mechanical stress on sperm chromatin. Controlling these parameters is essential to minimize DNA fragmentation and optimize ART outcomes. The aim of this review was to summarize complex factors and laboratory practices that may compromise sperm DNA integrity during ART procedures. It also highlights the underlying mechanisms of damage and proposes preventive laboratory strategies to minimize SDF and improve clinical outcomes.

2. Search strategy and selection criteria
This narrative review was conducted through a comprehensive search of the PubMed, Scopus, and Google Scholar databases for articles published between 2000 and 2025. The following keywords and their combinations were used: SDF, OS, iatrogenic factors, laboratory practices, and ART. Only English-language articles were included. Original research, clinical studies, and relevant review papers focusing on human sperm DNA integrity and ART-related laboratory interventions were considered. Studies involving animal models, non-reproductive systems, or unrelated oxidative mechanisms were excluded. Data were extracted based on methodological relevance, clarity of results, and contribution to understanding iatrogenic factors affecting sperm DNA integrity.
3. Physical factors and methods
3.1. Incubation temperature
The temperature at which semen or sperm suspensions are maintained in vitro has a strong impact on DNA integrity. At higher temperatures, sperm metabolism and ROS production increase, leading to accelerated DNA damage. In clinical studies, incubation at 37°C has been associated with a significant rise in SDF after only 2-3 hr, as measured by the sperm chromatin dispersion test (16). By contrast, routine handling at room temperature (RT) appears to be less harmful. Several guidelines recommend working at RT for short periods and avoiding unnecessary warming to 37°C, except when physiologically required (e.g., capacitation) (17). Supporting this, a study reported that although the DNA fragmentation index increased over time at both RT and 37°C, it was significantly lower after 24 h at RT than at 37°C (18).
Further studies confirm that incubation at RT maintains better motility and morphology, while reducing apoptosis and necrosis, compared with incubation at 35°C (19, 20). Even in asthenozoospermic samples, short-term incubation (2-4 hr) at RT or 37°C negatively affected DNA integrity and overall sperm quality (21). In summary, minimizing exposure to elevated temperatures and using gentle cooling strategies for prolonged storage are important measures to preserve DNA integrity during in vitro handling.
3.2. Sperm incubation duration
The duration of in vitro incubation or storage has a direct impact on SDF. Sperm naturally accumulate DNA damage after ejaculation if left unprotected, and experimental studies confirm that even a few hour of incubation can increase the DNA fragmentation index (16, 22). One investigation showed that SDF rose significantly after 2 hr of incubation at RT and after 4 hr at 37°C, underlining the importance of minimizing incubation time (23).
Testicular sperm retrieved from azoospermic men have been shown to display markedly higher DNA fragmentation when cryopreserved and subsequently incubated for 4-24 hr post-thaw, with damage continuing to increase over time. Furthermore, incubation at 37°C for as little as 1.5 hr has been associated with reduced progressive motility, abnormal morphology, and elevated DNA fragmentation (24). A progressive increase in DNA breaks has been observed in swim-up-selected sperm during long-term incubation, even in the absence of additional stressors (25). Taken together, these findings emphasize that sperm should be used for insemination or analysis as soon as possible after ejaculation. In clinical practice, most embryology laboratories begin sperm preparation within 30-60 min of collection and complete testing or intra cytoplasmic sperm injection (ICSI) procedures promptly. Prolonged storage at any temperature increases oxidative metabolism and endonuclease activity, thereby raising the risk of DNA fragmentation (26). As long as feasible, both the duration and the centrifugal force should be minimized.
3.3. Sperm processing using centrifugation
Centrifugation is a routine step in semen preparation protocols, commonly employed for washing, concentrating, and isolating motile sperm via techniques such as swim-up or density gradient centrifugation (DGC). Despite its utility, this mechanical process can inadvertently induce DNA damage through oxidative and physical stress. The shear forces generated during centrifugation elevate ROS levels and exert pressure on spermatozoa, compromising chromatin integrity (27, 28). To mitigate these risks, clinical protocols typically employ moderate centrifugal forces (300-600 × g) and limit spin durations to 5-10 min. Although DGC may enhance motility, its efficacy in selecting sperm with intact DNA remains questionable. A study involving 35 men undergoing ART found no significant reduction in apoptotic DNA fragmentation post DGC (29). This suggests that DGC may not be effective in selecting sperm with intact DNA.
In summary, centrifugation-based processing is necessary for sperm washing; however, care must be taken to minimize force, duration, and the number of spins, thus reducing the iatrogenic rise in DNA fragmentation (30). Proper sperm preparation prior to cryopreservation is crucial to minimize complications associated with centrifugation, which some spermatozoa may not tolerate well. Both centrifugation and freezing can independently increase the DNA fragmentation index, and their combination during thawing may further exacerbate DNA damage. Therefore, minimizing mechanical stress throughout sperm preparation and cryopreservation is essential for preserving sperm quality and improving ART outcomes (31). To overcome centrifugation-related drawbacks, alternative selection techniques have been developed. Microfluidic sperm-sorting devices significantly reduce SDF by mimicking the natural selection process within the female reproductive tract, enriching motile and morphologically normal sperm. This approach has been associated with improved fertilization rates and embryo development in ART (32-34).
3.4. Pipetting
Mechanical shear stress caused by pipetting viscous semen or sperm suspensions can physically disrupt sperm membranes and potentially lead to DNA damage. Vigorous pipetting, especially with narrow-bore tips, may result in tail detachment or even head shearing in some sperm cells. To mitigate this risk, embryologists typically use wide-bore or cut pipette tips when handling thick samples, thereby reducing mechanical stress and minimizing shear-induced damage. Gentle and brief aspiration-expulsion cycles are recommended to preserve cell integrity (35, 36).
Although there are no specific studies quantifying DNA fragmentation resulting directly from pipetting-induced shear, it is widely accepted that excessive mechanical manipulation can compromise sperm viability. For instance, when liquefying highly viscous semen, a 1:1 dilution with culture medium followed by gentle pipetting, often using origio tips is advised. During ICSI, suction pressure is carefully adjusted to the lowest feasible level to avoid mechanical trauma. By minimizing the number and intensity of pipetting steps and using appropriate sized pipette tips, are considered essential practices to prevent iatrogenic damage (37, 38).
3.5. Sperm cryopreservation
Cryopreservation is a widely used technique in assisted reproduction. But it can inadvertently lead to SDF due to osmotic shock and OS. During the freezing process, ice crystal formation and cellular dehydration impose both mechanical and chemical stress on sperm cells. Numerous studies have explored whether the freeze-thaw cycle itself contributes to increasing SDF. Although, findings vary, several studies have demonstrated that conventional slow freezing results in a measurable rise in DNA damage post-thaw. Specifically, a significant increase in DNA strand breaks has been observed in thawed sperm compared with fresh samples (39). Additional research has shown that thawing may activate caspases and mitochondria-mediated apoptotic pathways, further contributing to DNA fragmentation (40).
A study evaluated sperm quality after vitrification using 2 approaches: direct submersion in liquid nitrogen and cryopreservation with liquid nitrogen vapor. Normozoospermic semen samples were prepared by the swim-up method and divided into 3 groups: fresh control, direct submersion, and vapor. The results showed that direct submersion in liquid nitrogen yielded significantly better outcomes for small sperm quantities, including higher motility, viability, and chromatin integrity, compared with the vapor-based method (41).
Also, Agha-Rahimi et al. observed that both rapid freezing and vitrification significantly reduced sperm motility, viability, and normal morphology, with rapid freezing increasing DNA fragmentation. Vitrification yielded similar recovery rates and reduced cryoprotectant toxicity risks, though neither technique improved recovery outcomes over the other (42). The cryotop vial device (CVD) was an effective method for providing superior preservation of motility, viability, and DNA integrity compared with rapid freezing. CVD was more effective than other cryopreservation methods in maintaining sperm motility and viability, showing less DNA fragmentation than rapid freezing. Therefore, CVD is a promising choice for preserving small quantities of sperm (43). The study concluded that although both conventional cryopreservation and vitrification increase SDF due to low-temperature stress, vitrification causes significantly less DNA damage along with better long-term preservation of sperm genomic integrity. Therefore, vitrification is recommended as the preferred method for sperm freezing, particularly in reproductive techniques that require extended sperm longevity, such as artificial insemination (44).
3.6. Type of storage tube
The choice of sample containers can significantly influence sperm DNA outcomes. Semen is typically collected and stored in medical-grade polypropylene tubes or sterile glass containers. A major concern with certain plastics is their potential to leach harmful chemicals into biological fluids. For instance, bisphenol A, a known endocrine disruptor, has been associated with reduced sperm quality and increased DNA damage in animal studies, underscoring the importance of selecting safe materials in reproductive health contexts (45-47). Fortunately, most laboratory plastics in current use are certified as biologically inert and bisphenol A-free. To ensure sample purity, labs routinely use new, sterile tubes for each specimen. In studies involving microplastics or trace contaminants, some facilities even wash glassware at high temperatures to eliminate residual toxins.
Proper sealing of tubes is also essential, as evaporation or air exchange can alter the osmolarity and pH of the sample, potentially compromising sperm viability and DNA integrity. Although switching between plastic and glass containers mid-procedure is uncommon, laboratories typically standardize their materials to minimize variability. While direct data on the relationship between tube material and SDF is limited, the consensus is clear: using non-leaching, high-purity labware is considered best practice. Ultimately, careful selection of storage containers helps prevent inadvertent chemical exposure and supports the preservation of sperm DNA integrity (48-50). The Histone H2AX assay test was used to evaluate DNA damage and repair mechanisms in the context of ethylene oxide (EtO) exposure. Such testing is important for characterizing the genotoxic effects of EtO and the cellular responses activated to repair DNA damage in sperm and other cell types. These assays provide valuable insights into the mutagenic and carcinogenic risks associated with EtO exposure (51). Gamma ray sterilization offers an effective alternative to EO by using high-energy radiation to eliminate micro-organisms without the toxic residues and extensive aeration required by EO, while effectively penetrating materials to sterilize complex products. For example, most syringes are sterilized with EO and should not be used for holding serum.
4. Chemical factors
4.1. Media selection
The composition of media and chemical additives used during sperm handling plays a critical role in maintaining DNA stability. Many commercially available sperm preparation media are formulated with protective agents, such as antioxidants like taurine, EDTA, and citrate to mitigate OS, a major contributor to SDF (52-54). For instance, taurine and EDTA function by chelating metal ions and neutralizing free radicals, thereby preserving DNA integrity during processing (55). However, improperly prepared or contaminated media can adversely affect sperm quality. Factors such as suboptimal pH or osmolarity, as well as toxins leached from plasticware, may exacerbate cellular stress. Additional concerns include chemical exposures from antibiotics (e.g., gentamicin, which has been implicated in inducing OS), residual detergents, or heavy metal contaminants originating from glassware or environmental sources (56).
To minimize these risks, laboratories typically employ validated medical-grade reagents and operate under sterile, clean-air conditions. In cases where OS is suspected, such as elevated leukocyte counts in semen media enriched with antioxidants or supplemented with compounds like glutathione may help reduce DNA damage (57). The effects of 3 sperm preparation media (one commercial medium from Origio® [Denmark], Ham’s F10 [Biochrome, Germany], and VitaSperm™ [Innovative Biotech, Iran]) on sperm motility, viability, and DNA fragmentation were investigated at intervals of 0, 1, 2, and 24 h post swim-up. No significant differences in total motility were observed across the media at any time point. However, progressive motility was notably higher in spermatozoa prepared with Origio compared with VitaSperm, with no significant difference relative to Ham’s F10. Sperm viability remained consistent across all media, while DNA fragmentation levels 1 hr after swim-up were lower in the Origio medium compared with VitaSperm (58).
4.2. Ambient oxygen
The integrity of sperm DNA is significantly affected by storage conditions, particularly the presence of oxygen. Ambient oxygen contributes to the production of ROS, which can cause oxidative damage to sperm DNA. This damage leads to DNA strand breaks and modifications, such as the formation of 8-hydroxy-2’-deoxyguanosine, a marker of OS. Elevated levels of ROS and consequent DNA damage can negatively impact sperm motility, viability, and fertilization ability, ultimately affecting reproductive outcomes (13, 59, 60). Both environmental and pathological hypoxia adversely affect male reproductive health. The evidence demonstrates that hypoxia negatively impacts sperm quality, testicular function, hormone secretion, and pregnancy outcomes. The underlying mechanisms include excessive OS driven by ROS, apoptosis of germ cells mediated by hypoxia-inducible factor 1-alpha, inhibition of cellular proliferation, systemic inflammation, and epigenetic modifications. These factors collectively contribute to the detrimental effects of hypoxia on male fertility (61, 62). At present, the storage conditions for sperm, eggs, and embryos in laboratories are similar, and a scarcity of research in this area. Considering the distinct physiological characteristics of these cells, it is clear that further research and optimization of incubation conditions for sperm are necessary.
5. Chemical factors
5.1. Polyvinylpyrrolidone (PVP)
Despite its widespread use, particularly in procedures like ICSI, the combined influence of media composition and chemical additives like PVP on sperm DNA integrity remains insufficiently studied. PVP has been implicated in progressive chromatin damage during sperm handling, primarily through mechanisms, such as OS induction and alterations to the cellular microenvironment. These disruptions can destabilize sperm DNA, compromise chromatin integrity and ultimately affect fertilization potential. Accumulated chromatin damage may also negatively impact embryonic development and overall reproductive outcomes (63).
One investigation assessed the effects of varying incubation durations in PVP-containing media on SDF and viability. The findings revealed a significant decline in both parameters after just 10 min of exposure, highlighting the detrimental impact of prolonged contact with PVP (64). The study concludes that using a lower concentration of 5% PVP in the ICSI process is more beneficial than a higher concentration (10% PVP) for male factor infertility, as it leads to reduced SDF, improved expression of protective apoptotic genes, and higher rates of fertilization and high-quality embryo formation, while also resulting in fewer negative cytokinetic outcomes (65). Given these concerns, careful consideration of exposure time and concentration is essential when using PVP in assisted reproduction protocols to safeguard sperm quality and genetic integrity.
5.2. Antibiotics in semen and culture media
Antibiotics are routinely incorporated into semen and culture media to prevent microbial contamination during ART. However, emerging evidence highlights potential adverse effects on sperm DNA integrity at certain concentrations. Reviews of semen handling practices report widespread use of antibiotics such as penicillin-streptomycin and gentamicin in ART media. Studies indicate that various antibiotic supplements, including gentamicin, streptomycin-penicillin, ciprofloxacin, and doxycycline, can impair sperm motility, compromise membrane integrity, and induce DNA fragmentation, underscoring the need for cautious application. To mitigate these risks, unnecessary exposure to antibiotics during sperm preparation should be avoided. When contamination control is essential, exposure should be limited to brief durations and adhere strictly to manufacturer-recommended concentrations.
Furthermore, thorough post-treatment sperm selection techniques, such as DGC, swim-up, or microfluidic sorting, are recommended to effectively eliminate residual antibiotics and preserve sperm quality (66, 67).
6. Environmental factors
6.1. Light exposure
Exposure to light, particularly ultraviolet (UV) radiation can compromise sperm DNA by generating ROS (68). In laboratory settings, ambient fluorescent lighting and microscope illumination are potential sources of concern. Studies have demonstrated that intense UV exposure, such as from UV-C germicidal lamps, can induce DNA damage under specific conditions. For example, Da Costa et al. found that UV-induced DNA fragmentation was detectable when sperm were dried, whereas no significant damage occurred when sperm remained suspended in aqueous media (69). This suggests that routine bright visible light, such as during semen analysis, does not substantially increase DNA fragmentation if hydration is maintained.
Nonetheless, strong light sources, especially UV or high-intensity blue light, should be avoided in direct contact with sperm samples. While routine laboratory lighting is generally considered safe, technicians often minimize exposure to microscope illumination during prolonged procedures to reduce photodamage (70). For fluorescence-based DNA assays (e.g., terminal deoxynucleotidyl transferase dUTP nick end labeling under UV illumination), rapid imaging techniques are employed to minimize photodamage. Overall, moderate laboratory lighting has minimal impact on sperm DNA integrity. However, excessive UV exposure used, occasionally, for sterilization or prolonged direct illumination can increase SDF. To mitigate this risk, light-sensitive handling practices such as using covered tubes and limiting UV exposure are recommended (69).
Interestingly, one study investigated the effects of coherent red laser light at 633 nm on sperm motility and DNA safety. Using a novel wavelet-based method to assess sperm swimming speed, researchers found that red light significantly enhanced motility. Crucially, no notable increase in DNA damage, including double-strand breaks or oxidative lesions, was observed following red light exposure (71). To minimize OS and evaporation, semen samples are typically stored in capped tubes or incubators. Exposure to ambient air or light can increase ROS, which in turn may damage DNA. Therefore, some laboratories recommend shielding samples from light, such as wrapping tubes in aluminum foil to reduce light-induced ROS generation (13).
6.2. Laboratory conditions and environmental pollution
Laboratory environmental factors, such as air quality, temperature regulation, and equipment maintenance can indirectly affect sperm DNA integrity. In vitro fertiliation laboratories are typically maintained under controlled conditions, including stable temperatures (20-22°C), low endotoxin levels, and HEPA-filtered air to minimize contamination. Fluctuations in ambient conditions, such as heat stress, elevated humidity, or exposure to urban air pollutants may theoretically impose stress on sperm samples. Although, some studies suggest that airborne toxins like volatile organic compounds (VOCs) and endocrine-disrupting chemicals from plastics are present in laboratory air, their concentrations are generally negligible compared to broader environmental exposures.
Nonetheless, maintaining strict cleanliness is essential to prevent microbial contamination, which can generate ROS and compromise DNA integrity. Mechanical vibrations from nearby equipment (e.g., centrifuges or HVAC systems) are unlikely to cause DNA fragmentation. However, excessive physical agitation, such as transporting samples in a jostling vehicle should be avoided. While direct research on the impact of laboratory environments on sperm DNA is limited, adherence to good manufacturing practice standards helps minimize extraneous stressors (72). VOCs released from laboratory materials, incubators, and consumables can accumulate in culture media, triggering OS and DNA damage in sperm. Studies have demonstrated that IVF laboratories equipped with advanced air purification systems, including HEPA filters, activated carbon, and VOC absorbers exhibit significantly lower SDF levels and improved fertilization and embryo development outcomes compared to labs lacking such systems (73).
Once, sperm are removed from the protective environment of seminal plasma, they become highly susceptible to environmental pollutants. Exposure to airborne contaminants, plastic-derived chemicals, and media additives can artificially elevate SDF, compromising reproductive success. Therefore, maintaining optimal ART laboratory conditions, including VOC-free plastics, antioxidant-enriched media, and controlled air handling systems is critical for preserving sperm chromatin integrity (74). Clinical outcomes such as fertilization rates, blastocyst formation, and miscarriage incidence are indirectly influenced by elevated SDF resulting from environmental and laboratory pollution. To mitigate these risks, ART facilities must implement protective measures:
Mechanical shear stress caused by pipetting viscous semen or sperm suspensions can physically disrupt sperm membranes and potentially lead to DNA damage. Vigorous pipetting, especially with narrow-bore tips, may result in tail detachment or even head shearing in some sperm cells. To mitigate this risk, embryologists typically use wide-bore or cut pipette tips when handling thick samples, thereby reducing mechanical stress and minimizing shear-induced damage. Gentle and brief aspiration-expulsion cycles are recommended to preserve cell integrity (35, 36).
Although there are no specific studies quantifying DNA fragmentation resulting directly from pipetting-induced shear, it is widely accepted that excessive mechanical manipulation can compromise sperm viability. For instance, when liquefying highly viscous semen, a 1:1 dilution with culture medium followed by gentle pipetting, often using origio tips is advised. During ICSI, suction pressure is carefully adjusted to the lowest feasible level to avoid mechanical trauma. By minimizing the number and intensity of pipetting steps and using appropriate sized pipette tips, are considered essential practices to prevent iatrogenic damage (37, 38).
3.5. Sperm cryopreservation
Cryopreservation is a widely used technique in assisted reproduction. But it can inadvertently lead to SDF due to osmotic shock and OS. During the freezing process, ice crystal formation and cellular dehydration impose both mechanical and chemical stress on sperm cells. Numerous studies have explored whether the freeze-thaw cycle itself contributes to increasing SDF. Although, findings vary, several studies have demonstrated that conventional slow freezing results in a measurable rise in DNA damage post-thaw. Specifically, a significant increase in DNA strand breaks has been observed in thawed sperm compared with fresh samples (39). Additional research has shown that thawing may activate caspases and mitochondria-mediated apoptotic pathways, further contributing to DNA fragmentation (40).
A study evaluated sperm quality after vitrification using 2 approaches: direct submersion in liquid nitrogen and cryopreservation with liquid nitrogen vapor. Normozoospermic semen samples were prepared by the swim-up method and divided into 3 groups: fresh control, direct submersion, and vapor. The results showed that direct submersion in liquid nitrogen yielded significantly better outcomes for small sperm quantities, including higher motility, viability, and chromatin integrity, compared with the vapor-based method (41).
Also, Agha-Rahimi et al. observed that both rapid freezing and vitrification significantly reduced sperm motility, viability, and normal morphology, with rapid freezing increasing DNA fragmentation. Vitrification yielded similar recovery rates and reduced cryoprotectant toxicity risks, though neither technique improved recovery outcomes over the other (42). The cryotop vial device (CVD) was an effective method for providing superior preservation of motility, viability, and DNA integrity compared with rapid freezing. CVD was more effective than other cryopreservation methods in maintaining sperm motility and viability, showing less DNA fragmentation than rapid freezing. Therefore, CVD is a promising choice for preserving small quantities of sperm (43). The study concluded that although both conventional cryopreservation and vitrification increase SDF due to low-temperature stress, vitrification causes significantly less DNA damage along with better long-term preservation of sperm genomic integrity. Therefore, vitrification is recommended as the preferred method for sperm freezing, particularly in reproductive techniques that require extended sperm longevity, such as artificial insemination (44).
3.6. Type of storage tube
The choice of sample containers can significantly influence sperm DNA outcomes. Semen is typically collected and stored in medical-grade polypropylene tubes or sterile glass containers. A major concern with certain plastics is their potential to leach harmful chemicals into biological fluids. For instance, bisphenol A, a known endocrine disruptor, has been associated with reduced sperm quality and increased DNA damage in animal studies, underscoring the importance of selecting safe materials in reproductive health contexts (45-47). Fortunately, most laboratory plastics in current use are certified as biologically inert and bisphenol A-free. To ensure sample purity, labs routinely use new, sterile tubes for each specimen. In studies involving microplastics or trace contaminants, some facilities even wash glassware at high temperatures to eliminate residual toxins.
Proper sealing of tubes is also essential, as evaporation or air exchange can alter the osmolarity and pH of the sample, potentially compromising sperm viability and DNA integrity. Although switching between plastic and glass containers mid-procedure is uncommon, laboratories typically standardize their materials to minimize variability. While direct data on the relationship between tube material and SDF is limited, the consensus is clear: using non-leaching, high-purity labware is considered best practice. Ultimately, careful selection of storage containers helps prevent inadvertent chemical exposure and supports the preservation of sperm DNA integrity (48-50). The Histone H2AX assay test was used to evaluate DNA damage and repair mechanisms in the context of ethylene oxide (EtO) exposure. Such testing is important for characterizing the genotoxic effects of EtO and the cellular responses activated to repair DNA damage in sperm and other cell types. These assays provide valuable insights into the mutagenic and carcinogenic risks associated with EtO exposure (51). Gamma ray sterilization offers an effective alternative to EO by using high-energy radiation to eliminate micro-organisms without the toxic residues and extensive aeration required by EO, while effectively penetrating materials to sterilize complex products. For example, most syringes are sterilized with EO and should not be used for holding serum.
4. Chemical factors
4.1. Media selection
The composition of media and chemical additives used during sperm handling plays a critical role in maintaining DNA stability. Many commercially available sperm preparation media are formulated with protective agents, such as antioxidants like taurine, EDTA, and citrate to mitigate OS, a major contributor to SDF (52-54). For instance, taurine and EDTA function by chelating metal ions and neutralizing free radicals, thereby preserving DNA integrity during processing (55). However, improperly prepared or contaminated media can adversely affect sperm quality. Factors such as suboptimal pH or osmolarity, as well as toxins leached from plasticware, may exacerbate cellular stress. Additional concerns include chemical exposures from antibiotics (e.g., gentamicin, which has been implicated in inducing OS), residual detergents, or heavy metal contaminants originating from glassware or environmental sources (56).
To minimize these risks, laboratories typically employ validated medical-grade reagents and operate under sterile, clean-air conditions. In cases where OS is suspected, such as elevated leukocyte counts in semen media enriched with antioxidants or supplemented with compounds like glutathione may help reduce DNA damage (57). The effects of 3 sperm preparation media (one commercial medium from Origio® [Denmark], Ham’s F10 [Biochrome, Germany], and VitaSperm™ [Innovative Biotech, Iran]) on sperm motility, viability, and DNA fragmentation were investigated at intervals of 0, 1, 2, and 24 h post swim-up. No significant differences in total motility were observed across the media at any time point. However, progressive motility was notably higher in spermatozoa prepared with Origio compared with VitaSperm, with no significant difference relative to Ham’s F10. Sperm viability remained consistent across all media, while DNA fragmentation levels 1 hr after swim-up were lower in the Origio medium compared with VitaSperm (58).
4.2. Ambient oxygen
The integrity of sperm DNA is significantly affected by storage conditions, particularly the presence of oxygen. Ambient oxygen contributes to the production of ROS, which can cause oxidative damage to sperm DNA. This damage leads to DNA strand breaks and modifications, such as the formation of 8-hydroxy-2’-deoxyguanosine, a marker of OS. Elevated levels of ROS and consequent DNA damage can negatively impact sperm motility, viability, and fertilization ability, ultimately affecting reproductive outcomes (13, 59, 60). Both environmental and pathological hypoxia adversely affect male reproductive health. The evidence demonstrates that hypoxia negatively impacts sperm quality, testicular function, hormone secretion, and pregnancy outcomes. The underlying mechanisms include excessive OS driven by ROS, apoptosis of germ cells mediated by hypoxia-inducible factor 1-alpha, inhibition of cellular proliferation, systemic inflammation, and epigenetic modifications. These factors collectively contribute to the detrimental effects of hypoxia on male fertility (61, 62). At present, the storage conditions for sperm, eggs, and embryos in laboratories are similar, and a scarcity of research in this area. Considering the distinct physiological characteristics of these cells, it is clear that further research and optimization of incubation conditions for sperm are necessary.
5. Chemical factors
5.1. Polyvinylpyrrolidone (PVP)
Despite its widespread use, particularly in procedures like ICSI, the combined influence of media composition and chemical additives like PVP on sperm DNA integrity remains insufficiently studied. PVP has been implicated in progressive chromatin damage during sperm handling, primarily through mechanisms, such as OS induction and alterations to the cellular microenvironment. These disruptions can destabilize sperm DNA, compromise chromatin integrity and ultimately affect fertilization potential. Accumulated chromatin damage may also negatively impact embryonic development and overall reproductive outcomes (63).
One investigation assessed the effects of varying incubation durations in PVP-containing media on SDF and viability. The findings revealed a significant decline in both parameters after just 10 min of exposure, highlighting the detrimental impact of prolonged contact with PVP (64). The study concludes that using a lower concentration of 5% PVP in the ICSI process is more beneficial than a higher concentration (10% PVP) for male factor infertility, as it leads to reduced SDF, improved expression of protective apoptotic genes, and higher rates of fertilization and high-quality embryo formation, while also resulting in fewer negative cytokinetic outcomes (65). Given these concerns, careful consideration of exposure time and concentration is essential when using PVP in assisted reproduction protocols to safeguard sperm quality and genetic integrity.
5.2. Antibiotics in semen and culture media
Antibiotics are routinely incorporated into semen and culture media to prevent microbial contamination during ART. However, emerging evidence highlights potential adverse effects on sperm DNA integrity at certain concentrations. Reviews of semen handling practices report widespread use of antibiotics such as penicillin-streptomycin and gentamicin in ART media. Studies indicate that various antibiotic supplements, including gentamicin, streptomycin-penicillin, ciprofloxacin, and doxycycline, can impair sperm motility, compromise membrane integrity, and induce DNA fragmentation, underscoring the need for cautious application. To mitigate these risks, unnecessary exposure to antibiotics during sperm preparation should be avoided. When contamination control is essential, exposure should be limited to brief durations and adhere strictly to manufacturer-recommended concentrations.
Furthermore, thorough post-treatment sperm selection techniques, such as DGC, swim-up, or microfluidic sorting, are recommended to effectively eliminate residual antibiotics and preserve sperm quality (66, 67).
6. Environmental factors
6.1. Light exposure
Exposure to light, particularly ultraviolet (UV) radiation can compromise sperm DNA by generating ROS (68). In laboratory settings, ambient fluorescent lighting and microscope illumination are potential sources of concern. Studies have demonstrated that intense UV exposure, such as from UV-C germicidal lamps, can induce DNA damage under specific conditions. For example, Da Costa et al. found that UV-induced DNA fragmentation was detectable when sperm were dried, whereas no significant damage occurred when sperm remained suspended in aqueous media (69). This suggests that routine bright visible light, such as during semen analysis, does not substantially increase DNA fragmentation if hydration is maintained.
Nonetheless, strong light sources, especially UV or high-intensity blue light, should be avoided in direct contact with sperm samples. While routine laboratory lighting is generally considered safe, technicians often minimize exposure to microscope illumination during prolonged procedures to reduce photodamage (70). For fluorescence-based DNA assays (e.g., terminal deoxynucleotidyl transferase dUTP nick end labeling under UV illumination), rapid imaging techniques are employed to minimize photodamage. Overall, moderate laboratory lighting has minimal impact on sperm DNA integrity. However, excessive UV exposure used, occasionally, for sterilization or prolonged direct illumination can increase SDF. To mitigate this risk, light-sensitive handling practices such as using covered tubes and limiting UV exposure are recommended (69).
Interestingly, one study investigated the effects of coherent red laser light at 633 nm on sperm motility and DNA safety. Using a novel wavelet-based method to assess sperm swimming speed, researchers found that red light significantly enhanced motility. Crucially, no notable increase in DNA damage, including double-strand breaks or oxidative lesions, was observed following red light exposure (71). To minimize OS and evaporation, semen samples are typically stored in capped tubes or incubators. Exposure to ambient air or light can increase ROS, which in turn may damage DNA. Therefore, some laboratories recommend shielding samples from light, such as wrapping tubes in aluminum foil to reduce light-induced ROS generation (13).
6.2. Laboratory conditions and environmental pollution
Laboratory environmental factors, such as air quality, temperature regulation, and equipment maintenance can indirectly affect sperm DNA integrity. In vitro fertiliation laboratories are typically maintained under controlled conditions, including stable temperatures (20-22°C), low endotoxin levels, and HEPA-filtered air to minimize contamination. Fluctuations in ambient conditions, such as heat stress, elevated humidity, or exposure to urban air pollutants may theoretically impose stress on sperm samples. Although, some studies suggest that airborne toxins like volatile organic compounds (VOCs) and endocrine-disrupting chemicals from plastics are present in laboratory air, their concentrations are generally negligible compared to broader environmental exposures.
Nonetheless, maintaining strict cleanliness is essential to prevent microbial contamination, which can generate ROS and compromise DNA integrity. Mechanical vibrations from nearby equipment (e.g., centrifuges or HVAC systems) are unlikely to cause DNA fragmentation. However, excessive physical agitation, such as transporting samples in a jostling vehicle should be avoided. While direct research on the impact of laboratory environments on sperm DNA is limited, adherence to good manufacturing practice standards helps minimize extraneous stressors (72). VOCs released from laboratory materials, incubators, and consumables can accumulate in culture media, triggering OS and DNA damage in sperm. Studies have demonstrated that IVF laboratories equipped with advanced air purification systems, including HEPA filters, activated carbon, and VOC absorbers exhibit significantly lower SDF levels and improved fertilization and embryo development outcomes compared to labs lacking such systems (73).
Once, sperm are removed from the protective environment of seminal plasma, they become highly susceptible to environmental pollutants. Exposure to airborne contaminants, plastic-derived chemicals, and media additives can artificially elevate SDF, compromising reproductive success. Therefore, maintaining optimal ART laboratory conditions, including VOC-free plastics, antioxidant-enriched media, and controlled air handling systems is critical for preserving sperm chromatin integrity (74). Clinical outcomes such as fertilization rates, blastocyst formation, and miscarriage incidence are indirectly influenced by elevated SDF resulting from environmental and laboratory pollution. To mitigate these risks, ART facilities must implement protective measures:
- Utilize HEPA filtration, VOC absorbers, and maintain positive pressure environments
- Ensure all media, plastics, and cleaning agents are certified toxin free
- Incorporate antioxidant support during sperm handling
- Minimize aerosol exposure throughout preparation protocols (75, 76).
6.3. Glove materials in ART lab
While gloves are indispensable in ART laboratories for maintaining aseptic conditions and ensuring operator safety, certain glove materials and surface treatments may inadvertently compromise gamete integrity. Specifically, VOCs, powder particles, and chemical residues released from gloves have been linked to an increase in SDF. Composed of natural rubber proteins and chemical accelerators such as thiurams, carbamates, and benzothiazoles, latex gloves pose a risk of leaching toxic substances into culture media or directly onto gametes. Powdered latex gloves, though largely phased out in ART settings can release fine particles that contaminate sperm dishes and media, contributing to DNA damage. Despite the phase-out of powdered gloves in ART settings, residues from gloves still pose a risk to gamete integrity, emphasizing the critical need to use powder-free and carefully tested gloves in ART laboratories to avoid compromising sperm and embryo quality (77). Considered safer than latex, nitrile gloves still carry risks. Residual sulfur-based compounds and zinc may leach from the glove surface, and lower-quality nitrile variants have been shown to emit VOCs that can impair both sperm and oocyte quality (73). Polyvinyl chloride gloves are known to release phthalates, plasticizers with endocrine-disrupting properties. These compounds have been associated with compromised chromatin structure and elevated SDF levels. The study emphasizes that variability in phthalate content exists between glove brands and even batches, complicating the assurance of phthalate-free gloves. Due to their hormone-disrupting properties, phthalates from polyvinyl chloride gloves can negatively affect sperm quality and reproductive outcomes in ART settings (78).
Thus, selecting gloves in ART laboratories requires a careful assessment of chemical hazards, exposure levels, and task sensitivity. Powder-free, high-quality gloves that minimize leaching of toxic chemicals such as phthalates, VOCs, and sulfur compounds are essential to protect sperm DNA integrity and avoid contamination. Nitrile gloves are generally preferred due to their chemical resistance and low allergy risk. Whereas latex and vinyl gloves pose higher risks from chemical residues and plasticizers. Proper practices include consulting safety data sheets, inspecting gloves for damage, and timely replacement to maintain a safe environment for gametes and personnel, thereby supporting optimal ART outcomes.
7. Conclusion
Sperm DNA integrity is a critical determinant of male fertility and ART success. Complex factors, spanning medical, surgical, and laboratory practices significantly compromise the sperm genomic stability through OS, mechanical injury, and chemical exposure. Among laboratory procedures, excessive centrifugation, prolonged incubation, inappropriate temperature or pH, and exposure to ROS are major contributors to DNA fragmentation. Similarly, the choice of materials, such as culture media, cryoprotectants, and even gloves or plasticware, can influence oxidative balance and DNA quality. To ensure optimal outcomes, laboratories must implement evidence-based protocols emphasizing gentle sperm handling, antioxidant supplementation, strict air-quality control, and validation of all consumables under sperm-safe conditions. Training embryologists in minimizing mechanical and OS is equally vital. Future research should focus on developing standardized assessment methods for sperm DNA integrity and on exploring novel microfluidic and non-invasive sperm selection techniques to minimize damage.
Acknowledgments
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The authors used ChatGPT (GPT-5, OpenAI) only for improving the grammar and language clarity of the manuscript. The authors reviewed and take full responsibility for the content.
Conflict of Interest
The authors declare that there is no conflict of interest.
Type of Study: Review Article |
Subject:
Assisted Reproductive Technologies
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |