Wed, May 13, 2026
[Archive]
Volume 24, Issue 3 (March 2026)
IJRM 2026, 24(3): 193-208 |
Back to browse issues page
Ethics code: IR.SBMU.MSP.REC.1403.331
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Babakhanzadeh E, Khodadadian A, Nazari M, Mozhdeh M, Fazeli J, Vahidi S, et al . A novel hsa_circ_0070963/miR-223-3p/CCDC96–CCDC112 regulatory axis as a non-invasive biomarker for predicting sperm retrieval outcomes in non-obstructive azoospermia: A case-control study. IJRM 2026; 24 (3) :193-208
URL: http://ijrm.ir/article-1-3726-en.html
URL: http://ijrm.ir/article-1-3726-en.html
Emad Babakhanzadeh1 

 , Ali Khodadadian2
, Ali Khodadadian2 

 , Majid Nazari2
, Majid Nazari2 

 , Mahya Mozhdeh3
, Mahya Mozhdeh3 

 , Javad Fazeli2
, Javad Fazeli2 

 , Serajoddin Vahidi4
, Serajoddin Vahidi4 

 , Mohammad Hassan Jafari5
, Mohammad Hassan Jafari5 

 , Mohammadreza Dehghani2
, Mohammadreza Dehghani2 

 , Soudeh Ghafouri-Fard *6
, Soudeh Ghafouri-Fard *6 

 , Nasrin Ghasemi7
, Nasrin Ghasemi7 




 , Ali Khodadadian2
, Ali Khodadadian2 

 , Majid Nazari2
, Majid Nazari2 

 , Mahya Mozhdeh3
, Mahya Mozhdeh3 

 , Javad Fazeli2
, Javad Fazeli2 

 , Serajoddin Vahidi4
, Serajoddin Vahidi4 

 , Mohammad Hassan Jafari5
, Mohammad Hassan Jafari5 

 , Mohammadreza Dehghani2
, Mohammadreza Dehghani2 

 , Soudeh Ghafouri-Fard *6
, Soudeh Ghafouri-Fard *6 

 , Nasrin Ghasemi7
, Nasrin Ghasemi7 


1- Department of Medical Genetics, Shahid Beheshti University of Medical Sciences, Tehran, Iran. & Department of Medical Genetics, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
2- Department of Medical Genetics, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Department of Biology, Science and Research Branch, Islamic Azad University, Tehran, Iran.
4- Andrology Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
5- Department of Medical Biotechnology, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
6- Department of Medical Genetics, Shahid Beheshti University of Medical Sciences, Tehran, Iran. ,s.ghafourifard@sbmu.ac.ir
7- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
2- Department of Medical Genetics, School of Medicine, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
3- Department of Biology, Science and Research Branch, Islamic Azad University, Tehran, Iran.
4- Andrology Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
5- Department of Medical Biotechnology, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
6- Department of Medical Genetics, Shahid Beheshti University of Medical Sciences, Tehran, Iran. ,
7- Abortion Research Center, Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
Abstract: (2 Views)
Background: Non-obstructive azoospermia (NOA) is a severe cause of male infertility. Testicular sperm extraction has limited success, largely due to the absence of reliable, non-invasive biomarkers to predict outcomes. Emerging evidence highlights the role of non-coding RNAs in spermatogenesis, providing promising targets for biomarker development.
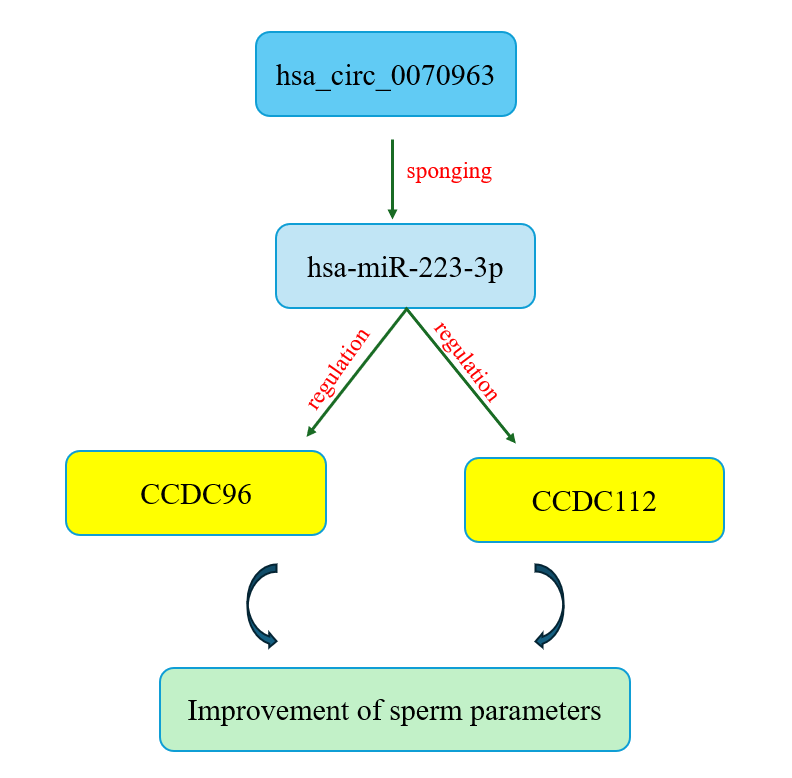
Objective: This study investigated the competing endogenous RNA regulatory axis hsa_circ_0070963/miR-223-3p/CCDC96–CCDC112 as a potential biomarker for predicting microsurgical testicular sperm extraction (micro-TESE) outcomes in NOA individuals.
Materials and Methods: In this case-control study, 60 men with NOA undergoing micro-TESE and 40 fertile controls were recruited. Testicular histology categorized individuals into Sertoli cell-only syndrome, hypospermatogenesis, or maturation arrest. Individuals were further stratified into NOA⁺ (successful retrieval) and NOA⁻ (failed retrieval). Plasma RNA was extracted, and expression levels of candidate genes were measured by quantitative reverse transcription polymerase chain reaction.
Results: Findings demonstrated significant downregulation of CCDC96 and CCDC112 in NOA plasma, particularly in NOA⁻ individuals, while hsa-miR-223-3p was upregulated, with the highest expression in NOA⁻ cases. Conversely, hsa_circ_0070963 was markedly reduced in NOA, especially in NOA⁻ individuals. Dysregulation was more pronounced in hypospermatogenesis and maturation arrest compared to Sertoli cell-only syndrome. Receiver operating characteristic analysis revealed strong predictive accuracy, with hsa_circ_0070963 (area under the receiver operating characteristic curve = 0.983) and hsa-miR-223-3p (area under the curve = 0.970) outperforming CCDC96 and CCDC112.
Conclusion: The hsa_circ_0070963/miR-223-3p/CCDC96–CCDC112 axis represents a novel competing endogenous RNAs regulatory network linked to impaired spermatogenesis. Its expression profile provides mechanistic insights and serves as a robust, non-invasive biomarker for predicting micro-TESE outcomes, supporting improved stratification and individualized treatment strategies in male infertility.
Objective: This study investigated the competing endogenous RNA regulatory axis hsa_circ_0070963/miR-223-3p/CCDC96–CCDC112 as a potential biomarker for predicting microsurgical testicular sperm extraction (micro-TESE) outcomes in NOA individuals.
Materials and Methods: In this case-control study, 60 men with NOA undergoing micro-TESE and 40 fertile controls were recruited. Testicular histology categorized individuals into Sertoli cell-only syndrome, hypospermatogenesis, or maturation arrest. Individuals were further stratified into NOA⁺ (successful retrieval) and NOA⁻ (failed retrieval). Plasma RNA was extracted, and expression levels of candidate genes were measured by quantitative reverse transcription polymerase chain reaction.
Results: Findings demonstrated significant downregulation of CCDC96 and CCDC112 in NOA plasma, particularly in NOA⁻ individuals, while hsa-miR-223-3p was upregulated, with the highest expression in NOA⁻ cases. Conversely, hsa_circ_0070963 was markedly reduced in NOA, especially in NOA⁻ individuals. Dysregulation was more pronounced in hypospermatogenesis and maturation arrest compared to Sertoli cell-only syndrome. Receiver operating characteristic analysis revealed strong predictive accuracy, with hsa_circ_0070963 (area under the receiver operating characteristic curve = 0.983) and hsa-miR-223-3p (area under the curve = 0.970) outperforming CCDC96 and CCDC112.
Conclusion: The hsa_circ_0070963/miR-223-3p/CCDC96–CCDC112 axis represents a novel competing endogenous RNAs regulatory network linked to impaired spermatogenesis. Its expression profile provides mechanistic insights and serves as a robust, non-invasive biomarker for predicting micro-TESE outcomes, supporting improved stratification and individualized treatment strategies in male infertility.
Type of Study: Original Article |
Subject:
Reproductive Genetics
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |